Submitted:
01 August 2025
Posted:
04 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
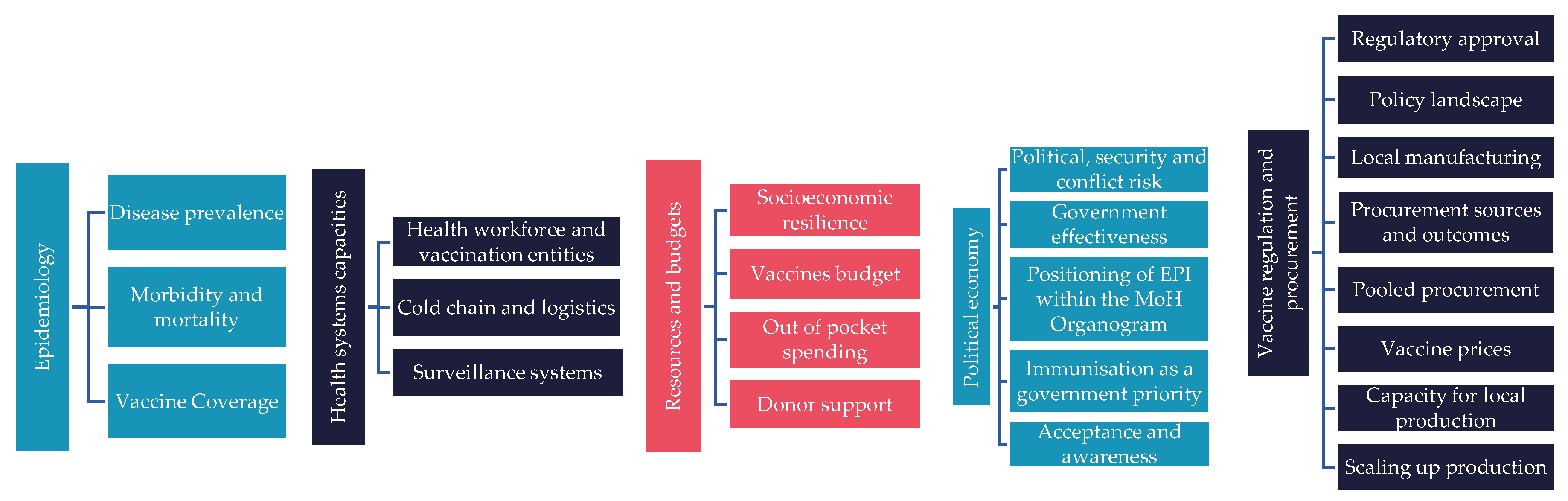
2. Materials and Methods
2.1. Ready Materials (R)
2.2. Extract Data (E)
2.3. Expert Consultation
- UNICEF MENARO and UNICEF country office consultations: We engaged with UNICEF Middle East and North Africa Regional Office (MENARO) and country offices to gather region-specific insights and data at the country level, supplementing our findings.
- Expert consultations: Consultations with experts in immunisation, public health, and vaccine policy were conducted to identify barriers and facilitators to vaccine introduction. These included experts from the MENA region, UNICEF regional office, and UNICEF country offices. In addition, we also gathered inputs and validated findings from an expert Steering Committee, consisting of members from the World Health Organisation, UNICEF Supply Division (SD), and Gavi, the Vaccine Alliance. These consultations provided contextual insights into challenges such as political will, financing, and public awareness that may not have been fully captured in the documents.
2.4. Analyse Data (A)
2.5. Distil Data (D)
3. Results
3.1. Epidemiology of Vaccine Preventable Diseases
| Algeria | Egypt | Iran | Jordan | Lebanon | Morocco | Tunisia | |
| Prevalence of rotavirus-attributable gastroenteritis/severe, various years diarrhoea in children under 5 | Unknown | 24% [17] | ~60% [18] | 6% [12] |
66% in children 0-12 years [19] | 44% [20] |
30% [21] |
| Incidence of pneumonia (severe) in children under 5, 2015 [15] | 2% (0.8%) | 1% (0.4%) | 2% (0.9%) | 2% (0.9%) | 1% (0.4%) | 2% (0.8%) | 2% (0.8%) |
| Incidence of cervical cancer in women, 2022 [16] | 0.008% | 0.003% | 0.002% | 0.003% | 0.003% | 0.01% | 0.005% |
3.2. The State of New Vaccine Introductions (NVI)
3.2.1. Rotavirus (RV) Vaccine
| RV | Indicator | Algeria | Egypt | Iran | Jordan | Lebanon | Morocco | Tunisia |
| Vaccine supply | Vaccine in NIP [22] | No | No | Yes | Yes | Yes | Yes | No |
| Year of introduction in NIP [22] | N.A. | N.A. | 2024 (in select provinces) | 2015 | 2022 | 2010 | N.A. | |
| Vaccine in private channels | No | Yes [30] | Yes [31] | Yes [32] | Yes [32] | Yes [28] | Yes [32] | |
| Vaccine / valence [33] | N.A. | Unknown | Rotasiil | Rotarix; Rotateq | Rotarix | Rotasiil | Unknown | |
| Procurement and financing | Source of procurement [28,31] | N.A. | Imported [28,31] | UNICEF | Self-procured | UNICEF | Self-procured | Imported |
| Source of financing [28,31,34] | N.A. | Out-of-pocket for the individual | Government funds | Government funds | Government funds and Gavi support | Government funds | Out-of-pocket for the individual | |
| Gavi MICs support | Eligibility, Status of application [35] | Not eligible | Eligible, Not applied | Eligible, applied | Not eligible, (vaccine introduced before MICs strategy) | Not eligible (vaccine introduced before MICs strategy) | Not eligible (vaccine introduced before MICs strategy) | Eligible, not applied. |
| Vaccine coverage | Coverage % (2023, WUENIC) [27] | N.A. | N.A. | Recently introduced | 96% | 40% | 98% | N.A. |
3.2.2. Pneumococcal Conjugate Vaccine (PCV)
| PCV | Indicator | Algeria | Egypt | Iran | Jordan | Lebanon | Morocco | Tunisia |
| Vaccine supply | Vaccine in NIP [10] | Yes | No | Yes | No, (Recommended by NITAG in 2015) [26] | Yes | Yes | Yes |
| Year of introduction in NIP [10] | 2016 | N.A. | 2024 (in select provinces) | N.A. | 2010 | 2010 | 2019 | |
| Vaccine in private channels | No [28] | Yes [40] | Yes [28] | Yes [28] | Yes [28] | Yes [28] | No [28] | |
| Vaccine name/valence | PCV 13 [41] | Unknown | PCV 10 (NIP) [41] | PCV 10 (private channels) | PCV 10 (in NIP, transitioned from PCV 13) [28,41] | PCV 10 and PCV 13 [41] | PCV 10 [41] | |
| Procurement and financing | Source of procurement | International market from Pfizer via the national procurement agency, Pharmacie Centrale des Hopitaux [28] | Imported for distribution in the private market [28] | UNICEF [38] | Imported for private distribution [28] | UNICEF SD [28] | Self procured [28,39] | Self-procured (via Pharmacie Centrale de Tunisie) [28,39] |
| Source of financing [28,31] | Government funds | Out of pocket for the individual or private insurance | Government funds, | Out of pocket for the individual or private insurance | Gavi + Government funds | Government funds | Government funds | |
| GAVI MICs support | Eligibility, Status of application [35] | Not eligible | Eligible, Not applied | Eligible, Applied | Eligible, Applied | Not eligible (vaccine introduced before MICs support) | Not eligible (vaccine introduced before MICs support) | Not eligible (vaccine introduced before MICs support) |
| Vaccine coverage | Coverage % (2023, WUENIC) [7] | 89% (2023, WUENIC) | N.A. |
NA - Recently introduced |
N.A. | 65% (2023, WUENIC) | 98% (2023, WUENIC) | 97% (2023, WUENIC) |
3.2.3. HPV Vaccine
| HPV | Indicator | Algeria | Egypt | Iran | Jordan | Lebanon | Morocco | Tunisia |
| Vaccine supply | Vaccine in NIP [23] | No | No | No | No | No | Yes | No, planned introduction in 2025 [43,54] |
| Year of introduction in NIP [23] | N.A. | N.A. | N.A. | N.A. | N.A. | 2022 | Planned for 2025 | |
| Vaccine in private channels | No [28] | Yes [45] | Yes [55] | Yes [51] |
Yes [50,56] | Yes [57] | No, (available for several years but not any more) [42] | |
| Vaccine name/ valence | N.A. | Gardasil 4, Cervarix | Gardasil 4 [58] | Gardasil 4 [32] | Gardasil, Cervarix [59] | Gardasil 4 | Bivalent (planned) [42] | |
| Target group (if introduced in NIP) | N.A. | N.A. | N.A. | N.A. | N.A. | Girls aged 11 [52] | Girls aged 12 who are currently in school (out of school girls missed) [43] | |
| Procurement and financing | Source of procurement | N.A. | Imported for distribution through private channels [28] | Imported for distribution through private channels [28] | Imported for distribution through private channels [28] | Imported for distribution through private channels [28] | UNICEF SD [47] | UNICEF SD [42] |
| Source of financing | N.A. | Out-of-pocket for the individual [31] | Out-of-pocket for the individual [55] | Out-of-pocket for the individual [31] | Out-of-pocket for the individual [31] | Government funds [47] | Government funds (+ Gavi for half of the first birth cohort) [42] | |
| Gavi MICs support | Eligibility/status of application [35] | Not eligible | Eligible, Not applied | Eligible, Not applied | Eligible, Not applied | Eligible, Not applied | Eligible, Not applied | Eligble, Applied |
| Vaccine coverage | Coverage % | N.A. | N.A. | N.A. | N.A. | N.A. | 55% (MoH estimate, 2022) [36] | N.A. |
3.3. Supplementary Findings
3.3.1. Immunisation Financing
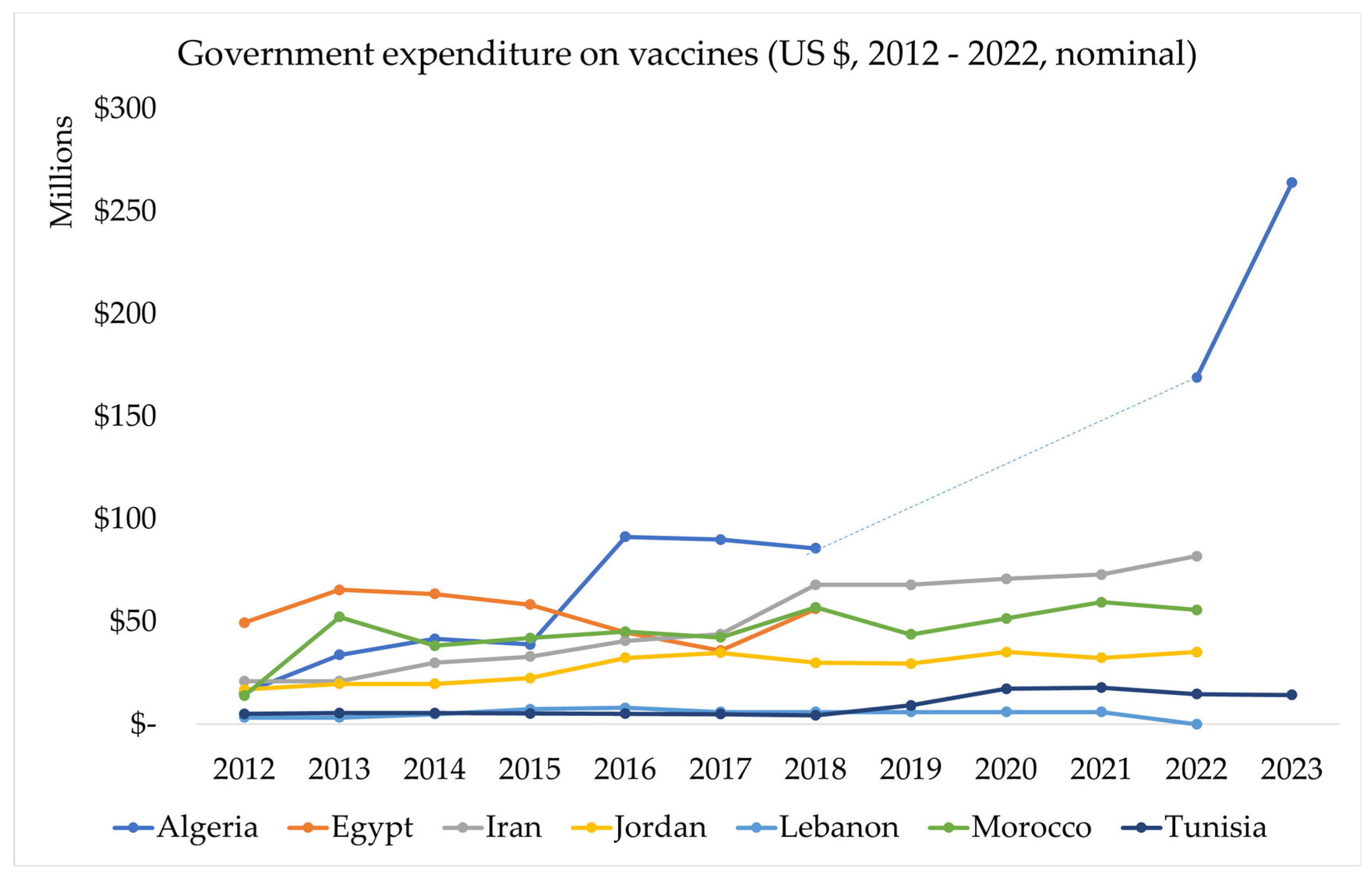
3.3.1.1. Government Expenditure on Vaccines Used in Routine Immunisation
3.3.1.2. Private Sector Involvement in Immunisation
3.3.2. Local Vaccine Manufacturing Capabilities
| Country | Spend per capita [61] |
| Algeria | US $2.07 / capita (2018) |
| Egypt | US $0.58 / capita (2018) |
| Iran | US $0.82 / capita (2019) |
| Jordan | US $2.93 / capita (2019) |
| Lebanon | Not available |
| Morocco | US $1.20 / capita (2019) |
| Tunisia | US $0.45 / capita (2017) |
| LMIC medians | US$0.55 (2011) and US$1.01 (2014) for LMICs |
3.3.3. Political and Economic Context
| Country | Major vaccine manufacturers | Vaccines manufactured | Existing capabilities |
| Algeria | Institut Pasteur d’Algérie (Founded in 1894) Saidal (Founded in 1982) |
Anti-rabies |
DS, F&F |
| Egypt | Egy Vac (Vacsera, founded in 1897) Minapharm (Founded in 1958) Biogeneric pharma |
F&F – Cholera, Covid-19, DT, DTP, Tetanus, Typhoid Import for distribution - Hib, Hepatitis A/B, HPV, Influenza, BCG, IPV, Meningitis, measles-mumps-rubella (MMR), OPV, Penta, PCV, Rabies, Rotavirus, Varicella, Yellow fever |
F&F BGM signed an MoU with Sanofi |
| Iran | Razi Institute Institute Pasteur of Iran Shifa Pharmed Industrial Co. CinnaGen Co. |
BCG, Cholera, recombinant hepatitis B, measles, polio, MMR, trivalent vaccines, and divalent | DS |
| Jordan [28] | No information available | No information available | No information available |
| Lebanon [28] | No information available | No information available | No information available |
| Morocco | Institut Pasteur du Maroc (Founded in 1967) | Import for distribution – BCG, Influenza, Rabies, Tetanus, Typhoid, yellow fever | Importing for distribution |
| Tunisia | Institut Pasteur de Tunis (Founded in 1893) | BCG | DS, F&F |
4. Discussion
4.1. Barriers to New Vaccine Introduction
4.2. Facilitators of New Vaccine Introduction
| Theme | Barriers | Facilitators |
| Epidemiology | Lack of recent and comparable data on disease prevalence. Outdated data limits informed decision-making on NVI | Evidence from high-burden studies (where available) can support NVI; Potential for enhanced surveillance and data collection initiatives |
| Healthcare infrastructure | Impact of conflict and displacement on healthcare infrastructure in the region disrupts vaccine delivery | Successful infrastructure, like Morocco's NVI model, offers a scalable approach for strengthening vaccine rollout. |
| Financial resources | Fiscal constraints due to economic vulnerability and public debt | Gavi MICs support with funding applications |
| Political economy | Political and economic uncertainty; High rates of displacement within and across borders driven by war and conflict | Political commitment to facilitate NVI |
| Local manufacturing | Limited local production in the region and high dependency on imports | Investing in local manufacturers may reduce dependency on imports and enhance local vaccine accessibility in the future |
| Awareness and trust | Low public awareness and acceptability of the HPV vaccine; Gender-related barriers | Potential to boost public confidence and enhance uptake through targeted interventions |
4.3. Evidence Gaps and Study Limitations
4.4. Practical Recommendations and Areas for Further Research
- Strengthen NITAGs: Establishing and empowering NITAGs in each country could improve evidence-based decision-making and streamline vaccine introduction processes.
- Enhance local vaccine production: Prioritising investments in local manufacturing capabilities may reduce dependency on imports and improve vaccine accessibility. Collaborations with international partners such as UNICEF and WHO can provide technical support and funding to support these initiatives.
- Address vaccine hesitancy: Developing culturally sensitive awareness campaigns targeting communities and healthcare providers could combat misinformation and build public trust, particularly regarding HPV vaccines.
- Optimise use of Gavi MICs support: Eligible countries are encouraged to apply for Gavi MICs funding to bridge initial financial gaps or for technical support, particularly with regard to the introduction of HPV vaccines.
- Expand coverage to vulnerable populations: Immunisation programmes can explicitly target displaced populations, out-of-school children, and marginalised groups.
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| NVI | New Vaccine Introduction |
| MICs | Middle-Income Countries |
| MENA | Middle East and North Africa |
| HPV | Human Papillomavirus Vaccine |
| PCV | Pneumococcal Conjugate Vaccine |
| RV | Rotavirus Vaccine |
References
- Zhu J, Cole CB, Fihman J, Adjagba A, Dasic M, Cernuschi T. Opportunities to accelerate immunization progress in middle-income countries. Vaccine. 2024 Apr 8;42:S98–106.
- UNFPA. Regional Report Sexual and Reproductive Health Laws and Policies in Selected Arab Countries July 2016 [Internet]. 2018 [cited 2024 Nov 26]. Available from: https://arabstates.unfpa.org/sites/default/files/pub-pdf/Sexual.pdf.
- Gavi’s approach to engaging with middle-income countries [Internet]. [cited 2024 Nov 26]. Available from: https://www.gavi.org/types-support/sustainability/gavi-mics-approach.
- Gavi. Gavi’s approach to engagement with middle-income countries. 2022.
- Gavi. Gavi MICs. 2023 [cited 2024 Nov 27]. List of countries and economies eligible for support under the MICs Approach as of 1 July 20231,2 (46) . Available from: https://www.gavi.org/sites/default/files/programmes-impact/support/Countries-and-economies-eligible-for-support-under-Gavi-MICs-Approach.pdf.
- Marangu D, Zar HJ. Childhood pneumonia in low-and-middle-income countries: An update. Paediatr Respir Rev [Internet]. 2019 Nov 1 [cited 2024 Nov 27];32:3. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC6990397/.
- Reiner RC, Wiens KE, Deshpande A, Baumann MM, Lindstedt PA, Blacker BF, et al. Mapping geographical inequalities in childhood diarrhoeal morbidity and mortality in low-income and middle-income countries, 2000–17: analysis for the Global Burden of Disease Study 2017. The Lancet. 2020 Jun 6;395(10239):1779–801.
- Mavundza EJ, Jaca A, Cooper S, Ndwandwe DE, Wiysonge CS. Factors associated with HPV vaccine acceptance and hesitancy in Africa: a systematic review protocol. BMJ Open [Internet]. 2024 Nov 14 [cited 2024 Nov 27];14(11):e082592. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC11575253/.
- Karami M, Berangi Z. The Need for Rotavirus Vaccine Introduction in the National Immunization Program of More Than 100 Countries around the World. Infect Control Hosp Epidemiol [Internet]. 2018 Jan 1 [cited 2024 Nov 27];39(1):124–5. Available from: https://www.cambridge.org/core/journals/infection-control-and-hospital-epidemiology/article/need-for-rotavirus-vaccine-introduction-in-the-national-immunization-program-of-more-than-100-countries-around-the-world/2560BDEA0B7F56F5DCCDE367A46E6F5C.
- Introduction of PCV (Pneumococcal conjugate vaccine) [Internet]. [cited 2024 Nov 27]. Available from: https://immunizationdata.who.int/global/wiise-detail-page/introduction-of-pcv-(pneumococcal-conjugate-vaccine)?ISO_3_CODE=&CODE=Middle%20East%20and%20North%20Africa&YEAR=.
- Dalglish SL, Khalid H, McMahon SA. Document analysis in health policy research: the READ approach. Health Policy Plan [Internet]. 2021 Feb 16 [cited 2024 Nov 26];35(10):1424–31. Available from: https://dx.doi.org/10.1093/heapol/czaa064.
- Rawashdeh M, Al-Zoubi B, Aliwat MB, Burayzat S, Alhindawi E, Al-Matti AA, et al. National Consensus for the Management of Acute Gastroenteritis in Jordanian Children: Consensus Recommendations Endorsed by the Jordanian Paediatric Society. Int J Pediatr [Internet]. 2022 [cited 2024 Nov 26];2022. Available from: https://pubmed.ncbi.nlm.nih.gov/36082204/.
- Zaraket H, Charide R, Kreidieh K, Dbaibo G, Melhem NM. Update on the epidemiology of rotavirus in the Middle East and North Africa. Vaccine. 2017 Oct 27;35(45):6047–58.
- WHO IB-VPD and Rotavirus Surveillance Bulletin - February 2020 [Internet]. [cited 2024 Nov 26]. Available from: https://mailchi.mp/d61958d8bbd1/who-ib-vpd-and-rotavirus-surveillance-bulletin-june-4779233?e.
- Wahl B, O’Brien KL, Greenbaum A, Majumder A, Liu L, Chu Y, et al. Burden of Streptococcus pneumoniae and Haemophilus influenzae type b disease in children in the era of conjugate vaccines: global, regional, and national estimates for 2000–15. Lancet Glob Health [Internet]. 2018 Jul 1 [cited 2024 Nov 26];6(7):e744–57. Available from: http://www.thelancet.com/article/S2214109X1830247X/fulltext.
- HPV INFORMATION CENTRE [Internet]. [cited 2024 Nov 26]. Available from: https://hpvcentre.net/datastatistics.php.
- El-Senousy WM, Abu Senna ASM, Mohsen NA, Hasan SF, Sidkey NM. Clinical and Environmental Surveillance of Rotavirus Common Genotypes Showed High Prevalence of Common P Genotypes in Egypt. Food Environ Virol [Internet]. 2020 Jun 1 [cited 2024 Nov 26];12(2):99–117. Available from: https://link.springer.com/article/10.1007/s12560-020-09426-0.
- Moradi-Lakeh M, Esteghamati A. National Immunization Program in Iran: whys and why nots. Hum Vaccin Immunother [Internet]. 2013 Jan [cited 2024 Nov 26];9(1):112–4. Available from: https://pubmed.ncbi.nlm.nih.gov/23442584/.
- Zaraket R, Salami A, Bahmad M, El Roz A, Khalaf B, Ghssein G, et al. Prevalence, risk factors, and clinical characteristics of rotavirus and adenovirus among Lebanese hospitalized children with acute gastroenteritis. Heliyon. 2020 Jun 1;6(6):e04248.
- Benhafid M, Elomari N, Azzouzi Idrissi M, Rguig A, Gentsch JR, Parashar U, et al. Effect of Monovalent Rotavirus Vaccine on Rotavirus Disease Burden and Circulating Rotavirus Strains Among Children in Morocco. J Med Virol [Internet]. 2015 Jun 1 [cited 2024 Nov 26];87(6):944. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC7934338/.
- Soltani M, Bouanene I, Trabelsi A, Harbi A, Hachicha M, Amri F, et al. [Epidemiology of rotavirus gastroenteritis among children under 5 years of age in Tunisia - results of sentinel hospital surveillance 2009 to 2011]. Rev Epidemiol Sante Publique [Internet]. 2012 Dec [cited 2024 Nov 26];60(6):473–80. Available from: https://pubmed.ncbi.nlm.nih.gov/23141818/.
- Introduction of Rotavirus vaccine [Internet]. [cited 2024 Nov 27]. Available from: https://immunizationdata.who.int/global/wiise-detail-page/introduction-of-rotavirus-vaccine?ISO_3_CODE=&YEAR=.
- Introduction of HPV Vaccine [Internet]. 2024 [cited 2024 Nov 27]. Immunization, Vaccines and Biologicals. Available from: https://www.who.int/teams/immunization-vaccines-and-biologicals/diseases/human-papillomavirus-vaccines-(HPV)/hpv-clearing-house/hpv-dashboard.
- Anne L. Buffardi and Susan Njambi-Szlapka. The role of National Immunisation Technical Advisory Groups in evidence-informed decision-making Enablers, constraints and future support options [Internet]. 2019 [cited 2024 Nov 27]. Available from: https://www.nitag-resource.org/sites/default/files/2b9ccfd309e13fa4c76aa6d9e99653bc94dedbff_1.pdf.
- Home | NITAG RESOURCE CENTER [Internet]. [cited 2024 Nov 27]. Available from: https://www.nitag-resource.org/.
- Harnessing the Power of Vaccines in Jordan: Steps in introducing the pneumococcal conjugate vaccine (PCV) - Your Say [Internet]. [cited 2024 Nov 26]. Available from: https://yoursay.plos.org/2023/04/harnessing-the-power-of-vaccines-in-jordan-steps-in-introducing-the-pneumococcal-conjugate-vaccine-pcv/.
- WHO Immunization Data portal - Global [Internet]. [cited 2024 Nov 26]. Available from: https://immunizationdata.who.int/.
- Triangulate and UNICEF MENARO. Interviews with Experts (UNICEF MENARO). Online; 2024.
- Iran Press. Iran Kicks Off Rotavirus Vaccination Nationwide [Internet]. 2024 [cited 2025 Mar 14]. Available from: https://iranpress.com/iran-kicks-off-rotavirus-vaccination-nationwide.
- Azzazy EA, Amer RM, Abdellatif GM, Abd-Elmoneim HA, Abo-Alella DA. Frequency and genotyping of group A rotavirus among Egyptian children with acute gastroenteritis: a hospital-based cross-sectional study. Virol J [Internet]. 2024 Dec 1 [cited 2024 Nov 27];21(1):238. Available from: https://virologyj.biomedcentral.com/articles/10.1186/s12985-024-02495-8.
- Triangulate Health and UNICEF MENARO. Interview with experts (UNICEF Country Office). Online; 2024.
- UNICEF. PRIVATE SECTOR ENGAGEMENT IN IMMUNIZATION: Findings and recommendations from the Middle East and North Africa Region [Internet]. 2020 [cited 2024 Nov 26]. Available from: https://immunizationeconomics.org/wp-content/uploads/2024/05/Immunization-Private-Sector-Study-MENA_Final.pdf.
- Vaccination schedule for Rotavirus [Internet]. [cited 2024 Nov 27]. Available from: https://immunizationdata.who.int/global/wiise-detail-page/vaccination-schedule-for-rotavirus?ISO_3_CODE=&TARGETPOP_GENERAL=.
- Saxenian H, Sadr-Azodi N, Kaddar M, Senouci K. Immunisation financing and programme performance in the Middle East and North Africa, 2010 to 2017. BMJ Glob Health [Internet]. 2019 Mar 1 [cited 2024 Nov 26];4(2):e001248. Available from: https://gh.bmj.com/content/4/2/e001248.
- Jan-Christopher Castilhos França, Senior Country Manager G. 2024. [cited 2024 Nov 26]. MICs support mechanisms and Gavi 6.0. Available from: https://www.linkedimmunisation.org/wp-content/uploads/2024/07/MICs-support-mechanisms-and-Gavi-6.0_01072024.pdf.
- Vaccination and Immunization Statistics - UNICEF DATA [Internet]. [cited 2024 Nov 27]. Available from: https://data.unicef.org/topic/child-health/immunization/.
- Walvax announces the receipt of the purchase order from Morocco for the 13-valent pneumococcal polysaccharide conjugate vaccine [Internet]. [cited 2024 Nov 26]. Available from: https://en.walvax.com/news/157.html.
- Iran receives 564,000 doses of Pneumococcal Conjugate Vaccine (PCV) with UNICEF support [Internet]. [cited 2024 Nov 26]. Available from: https://www.unicef.org/iran/en/press-releases/iran-receives-564000-doses-pneumococcal-conjugate-vaccine-pcv-unicef-support.
- Kaddar M, Saxenian H, Senouci K, Mohsni E, Sadr-Azodi N. Vaccine procurement in the Middle East and North Africa region: Challenges and ways of improving program efficiency and fiscal space. Vaccine. 2019 Jun 12;37(27):3520–8.
- Sibak M, Moussa I, El-Tantawy N, Badr S, Chaudhri I, Allam E, et al. Cost-effectiveness analysis of the introduction of the pneumococcal conjugate vaccine (PCV-13) in the Egyptian national immunization program, 2013. Vaccine. 2015 May 7;33(S1):A182–91.
- Vaccination schedule for Pneumococcal disease [Internet]. [cited 2024 Nov 27]. Available from: https://immunizationdata.who.int/global/wiise-detail-page/vaccination-schedule-for-pneumococcal-disease?ISO_3_CODE=&TARGETPOP_GENERAL=.
- Interview with Experts - UNICEF Country Office, Tunisia. 2024.
- Introduction du vaccin contre le HPV dans le calendrier national [Internet]. [cited 2024 Nov 26]. Available from: https://www.tuniscope.com/article/389195/vie/conseils/introduction-du-vaccin-contre-le-hpv-dans-le-calendrier-national-113715.
- WHO Immunization Data portal - Eastern Mediterranean Region [Internet]. [cited 2024 Nov 26]. Available from: https://immunizationdata.who.int/dashboard/regions/eastern-mediterranean-region/LBN.
- From stigma to strategy: Egypt’s journey in combating cervical cancer | Cancerworld Magazine [Internet]. [cited 2024 Nov 26]. Available from: https://cancerworld.net/from-stigma-to-strategy-egypts-cervical-cancer/.
- Prem K, Cernuschi T, Malvolti S, Brisson M, Jit M. Optimal human papillomavirus vaccination strategies in the context of vaccine supply constraints in 100 countries. EClinicalMedicine [Internet]. 2024 Aug 1 [cited 2024 Nov 26];74. Available from: http://www.thelancet.com/article/S2589537024003146/fulltext.
- UNICEF SD. Vaccine Industry Consultation. 2023 [cited 2024 Nov 27]. SESSION 21 VACCINE MARKET UPDATE Human Papillomavirus (HPV) Vaccine. Available from: https://www.unicef.org/supply/media/18931/file/UNICEF-VIC2023-Session21-HPVupdate-UNICEF-2023.pdf.
- Bencherit D, Kidar R, Otmani S, Sallam M, Samara K, Barqawi HJ, et al. Knowledge and Awareness of Algerian Students about Cervical Cancer, HPV and HPV Vaccines: A Cross-Sectional Study. Vaccines (Basel) [Internet]. 2022 Sep 1 [cited 2024 Nov 26];10(9):1420. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC9505646/.
- Mohamed ML, Tawfik AM, Mohammed GF, Elotla SF. Knowledge, Attitude, and Practice of Cervical Cancer Screening, and HPV Vaccination: A Cross-Sectional Study Among Obstetricians and Gynecologists in Egypt. Matern Child Health J [Internet]. 2022 Mar 1 [cited 2024 Nov 26];26(3):565–74. Available from: https://pubmed.ncbi.nlm.nih.gov/35060068/.
- Haddad SF, Kerbage A, Eid R, Kourie HR. Awareness about the human papillomavirus (HPV) and HPV vaccine among medical students in Lebanon. J Med Virol [Internet]. 2022 Jun 1 [cited 2024 Nov 27];94(6):2796–801. Available from: https://pubmed.ncbi.nlm.nih.gov/34877678/.
- Sallam M, Al-Mahzoum K, Eid H, Assaf AM, Abdaljaleel M, Al-Abbadi M, et al. Attitude towards HPV Vaccination and the Intention to Get Vaccinated among Female University Students in Health Schools in Jordan. Vaccines (Basel) [Internet]. 2021 Dec 1 [cited 2024 Nov 26];9(12):1432. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC8707789/.
- Elhaddadi H, Hamami A, Elouali A, Babakhouya A, Rkain M. Acceptance, Hesitancy, and Refusal Among Parents of Young Girls in Relation to Human Papillomavirus Vaccination: A Study From the Mohammed VI University Hospital Center in Oujda, Morocco. Cureus [Internet]. 2024 Apr 11 [cited 2024 Nov 26];16(4):e57992. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC11088456/.
- Vincent SC, Yaquobi S Al, Hashmi A Al. A Systematic Review of Knowledge, Attitudes, and Factors Influencing HPV Vaccine Acceptance Among Adolescents, Parents, Teachers, and Healthcare Professionals in the Middle East and North Africa (MENA) Region. Cureus [Internet]. 2024 May 14 [cited 2024 Nov 26];16(5):e60293. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC11175019/.
- Lahdhiri A, Benzina B, Ennaifer E, Gzara A, Rammeh-Rommani S, Laraj O, et al. Cost-Effectiveness Analysis of HPV Screening Strategies in Tunisia: A modeling study. medRxiv [Internet]. 2024 Oct 15 [cited 2024 Nov 26];2024.10.14.24315454. Available from: https://www.medrxiv.org/content/10.1101/2024.10.14.24315454v1.
- Mohammadpour F, Mansouri A, Hadjibabaie M. Utilization Evaluation of Human Papilloma Virus Vaccine (GARDASIL®) in Iran; A Cross-Sectional Study. Iran J Pharm Res [Internet]. 2020 [cited 2024 Nov 26];19(1):68. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC7462484/.
- Cervical Cancer Awareness – Mount Lebanon Hospital University Medical Center [Internet]. [cited 2024 Nov 27]. Available from: https://www.mlh.com.lb/cervical-cancer-awareness/.
- Belglaiaa E, Mougin C. Cervical cancer: Current situation and management in Morocco. Bull Cancer. 2019 Nov 1;106(11):1008–22.
- Vitality Plus HPV vaccine (cancer prevention vaccine) | Abdali Hospital [Internet]. [cited 2024 Nov 26]. Available from: https://www.abdalihospital.com/vitality-plus-hpv-vaccine-cancer-prevention-vaccine.
- Abou El-Ola MJ, Rajab MA, Abdallah DI, Fawaz IA, Awad LS, Tamim HM, et al. Low rate of human papillomavirus vaccination among schoolgirls in Lebanon: Barriers to vaccination with a focus on mothers’ knowledge about available vaccines. Ther Clin Risk Manag. 2018 Mar 27;14:617–26.
- Immunization expenditure [Internet]. [cited 2024 Nov 26]. Available from: https://immunizationdata.who.int/global/wiise-detail-page/immunization-expenditure?ISO_3_CODE=TUN+DZA+MAR+LBN+EGY+JOR+IRN&YEAR=.
- Catharina de Beer J, Goto D, Miller-Janson H, Holl R, Bencina G. Vaccine financing in the Middle East and Africa: An overview from 2010 to 2019. Vaccine. 2022 Sep 16;40(39):5691–700.
- Mihigo R, Okeibunor J, Cernuschi T, Petu A, Satoulou A, Zawaira F. Improving access to affordable vaccines for middle-income countries in the african region. Vaccine [Internet]. 2019 May 9 [cited 2025 Mar 14];37(21):2838. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC6510904/.
- World Health Organization, United Nations Children’s Fund. Tunisia: WHO and UNICEF estimates of immunization coverage: 2022 revision [Internet]. 2022 [cited 2024 Dec 13]. Available from: https://data.unicef.org/wp-content/uploads/cp/immunisation/tun.pdf.
- Partnerships for African Vaccine Manufacturing (PAVM) Framework for Action – Africa CDC [Internet]. [cited 2024 Nov 26]. Available from: https://africacdc.org/download/partnerships-for-african-vaccine-manufacturing-pavm-framework-for-action/.
- Wellcome. Scaling Up African Vaccine Manufacturing Capacity [Internet]. 2023 [cited 2024 Nov 26]. Available from: https://cms.wellcome.org/sites/default/files/2023-01/Wellcome-Biovac-BCG-Scaling-up-African-vaccine-manufacturing-capacity-report-2023_0.pdf.
- List of National Regulatory Authorities (NRAs) operating at maturity level 3 (ML3)1 and maturity level 4 (ML4)2 (as benchmarked against WHO Global Benchmarking Tool (GBT) (in alphabetical order) - As of June 2024 [Internet]. 2024 [cited 2024 Nov 26]. Available from: https://cdn.who.int/media/docs/default-source/medicines/regulatory-systems/wla/list-of-nras-operating-at-ml3-and-ml4.pdf?sfvrsn=ee93064f_18&download=true.
- IQVIA ME Consulting. Localization of Pharmaceutical Manufacturing in Middle East and North Africa Region [Internet]. 2022 [cited 2024 Nov 26]. Available from: https://www.iqvia.com/-/media/iqvia/pdfs/mea/white-paper/localization-of-pharmaceutical-manufacturing-in-middle-east-and-north-africa-region.pdf.
- Ghanei M, Mohabattalab A, Fartash K, Kolahchi N, Khakdaman A, Kaghazian H, et al. Exploring the experience of developing COVID-19 vaccines in Iran. Clin Exp Vaccine Res [Internet]. 2023 Jan 1 [cited 2024 Nov 26];12(1):1. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC9950226/.
- Ghanei M, Mohabattalab A, Fartash K, Kolahchi N, Khakdaman A, Kaghazian H, et al. Exploring the experience of developing COVID-19 vaccines in Iran. Clin Exp Vaccine Res [Internet]. 2023 Jan 1 [cited 2024 Dec 9];12(1):1. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC9950226/.
- Unraveling Egypt’s political economy puzzle: It’s more than just economics - Atlantic Council [Internet]. [cited 2024 Nov 26]. Available from: https://www.atlanticcouncil.org/blogs/menasource/egypt-economy-debt-fdi-nasser/.
- Egypt faces mounting socio-economic strain from the Gaza war, says UNDP | United Nations Development Programme [Internet]. [cited 2024 Nov 26]. Available from: https://www.undp.org/arab-states/press-releases/egypt-faces-mounting-socio-economic-strain-gaza-war-says-undp.
- What are the big economic challenges facing the government in Iran? - Economics Observatory [Internet]. [cited 2024 Nov 26]. Available from: https://www.economicsobservatory.com/what-are-the-big-economic-challenges-facing-the-government-in-iran.
- Algeria Economic Outlook | African Development Bank Group [Internet]. [cited 2024 Nov 26]. Available from: https://www.afdb.org/en/countries-north-africa-peoples-democratic-republic-algeria/algeria-economic-outlook.
- Lebanon’s Fragile Economy Pulled Back into Recession [Internet]. [cited 2024 Nov 26]. Available from: https://www.worldbank.org/en/news/press-release/2023/12/21/lebanon-s-fragile-economy-pulled-back-into-recession.
- Morocco Economic Outlook | African Development Bank Group [Internet]. [cited 2024 Nov 26]. Available from: https://www.afdb.org/en/countries/north-africa/morocco/morocco-economic-outlook.
- Harnessing the Power of Vaccines in Jordan: Steps in introducing the pneumococcal conjugate vaccine (PCV) - Your Say [Internet]. [cited 2025 Mar 13]. Available from: https://yoursay.plos.org/2023/04/harnessing-the-power-of-vaccines-in-jordan-steps-in-introducing-the-pneumococcal-conjugate-vaccine-pcv/.
- Casey RM, Akaba H, Hyde TB, Bloem P. Covid-19 pandemic and equity of global human papillomavirus vaccination: descriptive study of World Health Organization-Unicef vaccination coverage estimates. BMJ medicine [Internet]. 2024 Jan [cited 2025 Mar 13];3(1):e000726. Available from: https://pubmed.ncbi.nlm.nih.gov/38293682/.
- UNHCR TURA. Middle East and North Africa | Global Focus [Internet]. [cited 2024 Dec 13]. Available from: https://reporting.unhcr.org/global-report-22/middle-east-and-north-africa.
- Middle East and North Africa Product Series | Displacement Tracking Matrix [Internet]. [cited 2025 Jun 2]. Available from: https://dtm.iom.int/report-product-series/middle-east-and-north-africa-product-series.
- UNHCR helps nearly one million refugees in Iran, mostly from Afghanistan and Iraq. [Internet]. [cited 2025 Jun 2]. Available from: https://www.unhcr.org/ir/refugees-iran.
- Lebanon | UNHCR [Internet]. [cited 2025 Jun 2]. Available from: https://www.unhcr.org/where-we-work/countries/lebanon.
- Jordan | UNHCR [Internet]. [cited 2025 Jun 2]. Available from: https://www.unhcr.org/where-we-work/countries/jordan.
- Bouaddi O, Seedat F, Hasaan Mohammed HE, Evangelidou S, Deal A, Requena-Méndez A, et al. Vaccination coverage and access among children and adult migrants and refugees in the Middle East and North African region: a systematic review and meta-analysis. EClinicalMedicine [Internet]. 2024 Dec 1 [cited 2024 Dec 9];78. Available from: http://www.thelancet.com/article/S2589537024005297/fulltext.
- Mansour Z, Hamadeh R, Rady A, Danovaro-Holliday MC, Fahmy K, Said R, et al. Vaccination coverage in Lebanon following the Syrian crisis: Results from the district-based immunization coverage evaluation survey 2016. BMC Public Health [Internet]. 2019 Jan 14 [cited 2024 Dec 13];19(1):1–11. Available from: https://bmcpublichealth.biomedcentral.com/articles/10.1186/s12889-019-6418-9.
- Farag NH, Wannemuehler K, Weldon W, Arbaji A, Belbaisi A, Khuri-Bulos N, et al. Estimating population immunity to poliovirus in Jordan’s high-risk areas. Hum Vaccin Immunother [Internet]. 2020 Mar 3 [cited 2024 Dec 13];16(3):548–53. Available from: https://www.tandfonline.com/doi/abs/10.1080/21645515.2019.1667727.
- Dept. IMFundME and CA. Jordan: 2022 Article IV Consultation and Fourth Review Under the Extended Arrangement Under the Extended Fund Facility, Request for Augmentation and Rephasing of Access, and Modification of Performance Criteria-Press Release; Staff Report; Staff Statement; and Statement by the Executive Director for Jordan. IMF Staff Country Reports [Internet]. 2022 Jul 18 [cited 2024 Nov 26];2022(221). Available from: https://www.elibrary.imf.org/view/journals/002/2022/221/article-A001-en.xml.
- Yacouti A, Elkhoudri N, El got A, Benider A, Hadrya F, Baddou R, et al. Awareness, attitudes and acceptability of the HPV vaccine among female university students in Morocco. PLoS One [Internet]. 2022 Apr 1 [cited 2024 Nov 26];17(4):e0266081. Available from: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0266081.
- UNICEF. Gender barriers and Immunization in East Asia [Internet]. [cited 2025 Jun 2]. Available from: https://www.unicef.org/eap/media/17756/file/Gender%20Barriers%20and%20Immunization%20in%20East%20Asia.pdf.pdf.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




