Submitted:
22 July 2025
Posted:
24 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. The Gastrointestinal Tract (GIT), Cannabinoids and Pharmaceutical Drugs
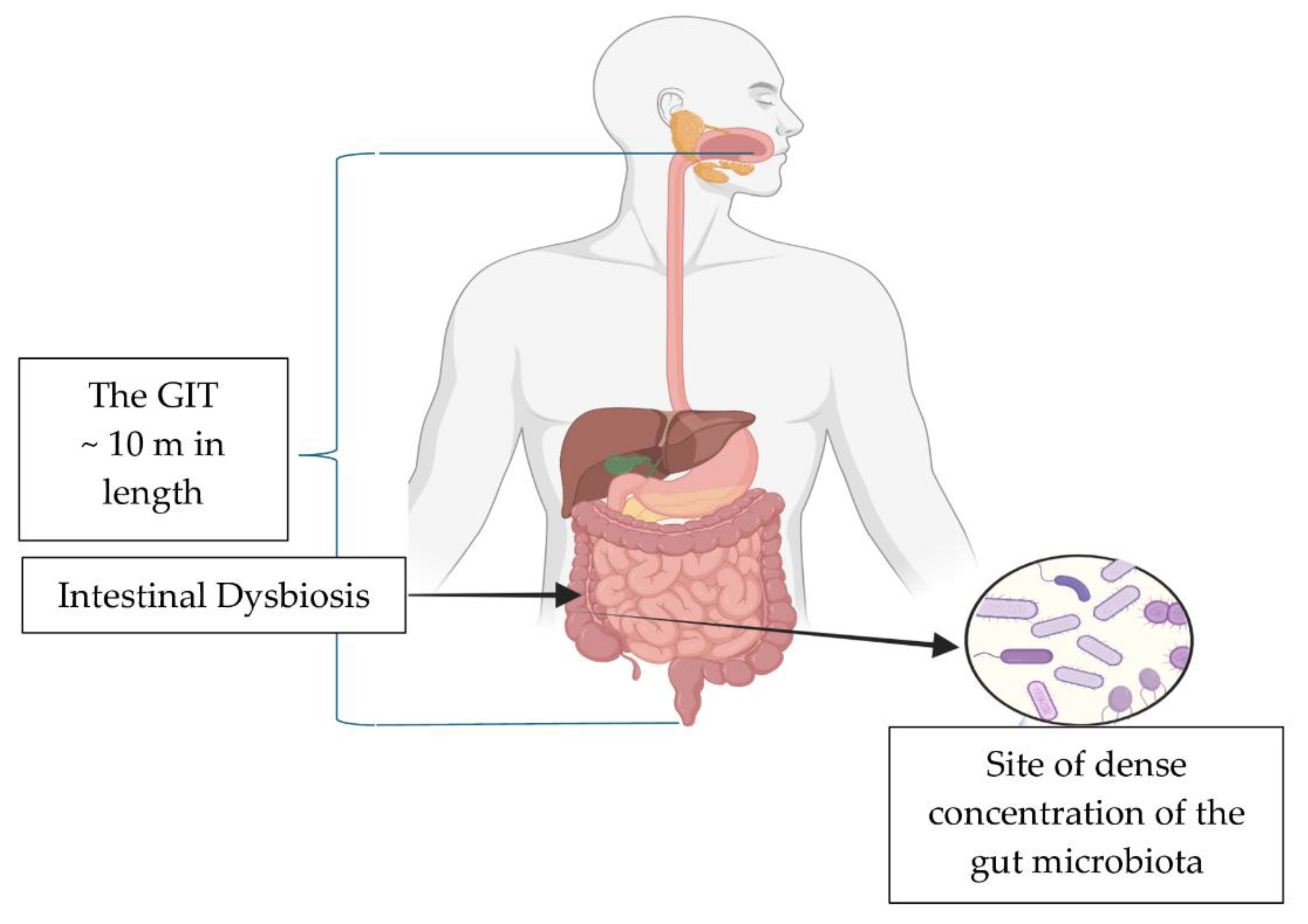
2.1. The Intestinal Microbiome / Microbiota
2.2. GIT Dysbiosis
3. Medicinal Cannabis
3.1. Medicinal Cannabis for Nausea
3.2. Medicinal Cannabis for Pain
4. Nanotechnology for Effective Cannabinoid and Pharmaceutical Drug Delivery
4.1. Micelles
4.2. Liposomes
4.3. Dendrimers
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sharma VK, and Agrawal MK. A Historical Perspective of Liposomes-a Bio Nanomaterial. Materials Today: Proceedings 2021, 45, 2963–66. [Google Scholar] [CrossRef]
- Lombardo, D. , and M. A. Kiselev. Methods of Liposomes Preparation: Formation and Control Factors of Versatile Nanocarriers for Biomedical and Nanomedicine Application. Pharmaceutics 2022, 14. [Google Scholar] [CrossRef]
- Yusuf, A., A. R. Z. Almotairy, H. Henidi, O. Y. Alshehri, and M. S. Aldughaim. Nanoparticles as Drug Delivery Systems: A Review of the Implication of Nanoparticles' Physicochemical Properties on Responses in Biological Systems. Polymers (Basel).
- Mitchell, M. J., M. M. Billingsley, R. M. Haley, M. E. Wechsler, N. A. Peppas, and R. Langer. Engineering Precision Nanoparticles for Drug Delivery. Nat Rev Drug Discov 2021, 20, 101–24. [Google Scholar] [CrossRef]
- Parsons, R. L. Drug Absorption in Gastrointestinal Disease with Particular Reference to Malabsorption Syndromes. Clin Pharmacokinet 1977, 2, 45–60. [Google Scholar] [CrossRef]
- Barua, S. , and S. Mitragotri. Challenges Associated with Penetration of Nanoparticles across Cell and Tissue Barriers: A Review of Current Status and Future Prospects. Nano Today 2014, 9, 223–43. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, S. , and F. Zuhair. Advancing Nanomedicine through Electron Microscopy: Insights into Nanoparticle Cellular Interactions and Biomedical Applications. Int J Nanomedicine 2025, 20, 2847–78. [Google Scholar] [CrossRef] [PubMed]
- Homayun, B., X. Lin, and H. J. Choi. Challenges and Recent Progress in Oral Drug Delivery Systems for Biopharmaceuticals. Pharmaceutics 2019, 11. [Google Scholar]
- Rodger, M. A. , and L. King. Drawing up and Administering Intramuscular Injections: A Review of the Literature. J Adv Nurs 2000, 31, 574–82. [Google Scholar] [CrossRef]
- Alqahtani, M. S., M. Kazi, M. A. Alsenaidy, and M. Z. Ahmad. Advances in Oral Drug Delivery. Front Pharmacol 2021, 12, 618411. [Google Scholar] [CrossRef]
- Bashiardes, S. , and C. Christodoulou. Orally Administered Drugs and Their Complicated Relationship with Our Gastrointestinal Tract. Microorganisms 2024, 12. [Google Scholar]
- Vinarov, Z., M. Abdallah, J. A. G. Agundez, K. Allegaert, A. W. Basit, M. Braeckmans, J. Ceulemans, M. Corsetti, B. T. Griffin, M. Grimm, D. Keszthelyi, M. Koziolek, C. M. Madla, C. Matthys, L. E. McCoubrey, A. Mitra, C. Reppas, J. Stappaerts, N. Steenackers, N. L. Trevaskis, T. Vanuytsel, M. Vertzoni, W. Weitschies, C. Wilson, and P. Augustijns. Impact of Gastrointestinal Tract Variability on Oral Drug Absorption and Pharmacokinetics: An Ungap Review. Eur J Pharm Sci 2021, 162, 105812. [Google Scholar]
- Li, J., H. Jia, X. Cai, H. Zhong, Q. Feng, S. Sunagawa, M. Arumugam, J. R. Kultima, E. Prifti, T. Nielsen, A. S. Juncker, C. Manichanh, B. Chen, W. Zhang, F. Levenez, J. Wang, X. Xu, L. Xiao, S. Liang, D. Zhang, Z. Zhang, W. Chen, H. Zhao, J. Y. Al-Aama, S. Edris, H. Yang, J. Wang, T. Hansen, H. B. Nielsen, S. Brunak, K. Kristiansen, F. Guarner, O. Pedersen, J. Doré, S. D. Ehrlich, P. Bork, and J. Wang. An Integrated Catalog of Reference Genes in the Human Gut Microbiome. Nat Biotechnol 2014, 32, 834–41. [Google Scholar]
- Azzouz LL, and Sharma S. Physiology, Large Intestine. In StatPearls Publishing. StatPearls [Internet] Treasure Island (FL), 2025.
- Bäckhed, F., R. E. Ley, J. L. Sonnenburg, D. A. Peterson, and J. I. Gordon. Host-Bacterial Mutualism in the Human Intestine. Science 2005, 307, 1915–20. [Google Scholar] [CrossRef]
- Gasaly, N., P. de Vos, and M. A. Hermoso. Impact of Bacterial Metabolites on Gut Barrier Function and Host Immunity: A Focus on Bacterial Metabolism and Its Relevance for Intestinal Inflammation. Front Immunol 2021, 12, 658354. [Google Scholar] [CrossRef] [PubMed]
- Chen, K., H. Geng, C. Ye, and J. Liu. Dysbiotic Alteration in the Fecal Microbiota of Patients with Polycystic Ovary Syndrome. Microbiol Spectr 2024, 12, e0429123. [Google Scholar] [CrossRef]
- Stecher, B. The Roles of Inflammation, Nutrient Availability and the Commensal Microbiota in Enteric Pathogen Infection. Microbiol Spectr 2015, 3. [Google Scholar] [CrossRef]
- de Vos, W. M., H. Tilg, M. Van Hul, and P. D. Cani. Gut Microbiome and Health: Mechanistic Insights. Gut 2022, 71, 1020–32. [Google Scholar] [CrossRef]
- Cani, P. D. , and C. Knauf. How Gut Microbes Talk to Organs: The Role of Endocrine and Nervous Routes. Mol Metab 2016, 5, 743–52. [Google Scholar] [CrossRef]
- Dicks, L. M. T. How Important Are Fatty Acids in Human Health and Can They Be Used in Treating Diseases? Gut Microbes 2024, 16, 2420765. [Google Scholar] [CrossRef]
- Enright, E. F., C. G. Gahan, S. A. Joyce, and B. T. Griffin. The Impact of the Gut Microbiota on Drug Metabolism and Clinical Outcome. Yale J Biol Med 2016, 89, 375–82. [Google Scholar]
- Li, H., J. He, and W. Jia. The Influence of Gut Microbiota on Drug Metabolism and Toxicity. Expert Opin Drug Metab Toxicol 2016, 12, 31–40. [Google Scholar] [CrossRef]
- Chen, J., L. Vitetta, J. D. Henson, and S. Hall. Intestinal Dysbiosis, the Tryptophan Pathway and Nonalcoholic Steatohepatitis. Int J Tryptophan Res 2022, 15, 11786469211070533. [Google Scholar] [CrossRef]
- Li, S. Modulation of Immunity by Tryptophan Microbial Metabolites. Front Nutr 2023, 10, 1209613. [Google Scholar] [CrossRef] [PubMed]
- Bhattarai, Y., S. Jie, D. R. Linden, S. Ghatak, R. A. T. Mars, B. B. Williams, M. Pu, J. L. Sonnenburg, M. A. Fischbach, G. Farrugia, L. Sha, and P. C. Kashyap. Bacterially Derived Tryptamine Increases Mucus Release by Activating a Host Receptor in a Mouse Model of Inflammatory Bowel Disease. iScience 2020, 23, 101798. [Google Scholar]
- Bortolotti, P., B. Hennart, C. Thieffry, G. Jausions, E. Faure, T. Grandjean, M. Thepaut, R. Dessein, D. Allorge, B. P. Guery, K. Faure, E. Kipnis, B. Toussaint, and A. Le Gouellec. Tryptophan Catabolism in Pseudomonas Aeruginosa and Potential for Inter-Kingdom Relationship. BMC Microbiol 2016, 16, 137. [Google Scholar]
- Dodd, D., M. H. Spitzer, W. Van Treuren, B. D. Merrill, A. J. Hryckowian, S. K. Higginbottom, A. Le, T. M. Cowan, G. P. Nolan, M. A. Fischbach, and J. L. Sonnenburg. A Gut Bacterial Pathway Metabolizes Aromatic Amino Acids into Nine Circulating Metabolites. Nature 2017, 551, 648–52. [Google Scholar] [CrossRef]
- Malik, Z., D. Baik, and R. Schey. The Role of Cannabinoids in Regulation of Nausea and Vomiting, and Visceral Pain. Curr Gastroenterol Rep 2015, 17, 429. [Google Scholar] [CrossRef]
- Weiss, G. A. , and T. Hennet. Mechanisms and Consequences of Intestinal Dysbiosis. Cell Mol Life Sci 2017, 74, 2959–77. [Google Scholar] [CrossRef]
- Fu, Y., J. Lyu, and S. Wang. The Role of Intestinal Microbes on Intestinal Barrier Function and Host Immunity from a Metabolite Perspective. Front Immunol 2023, 14, 1277102. [Google Scholar] [CrossRef]
- DeGruttola, A. K., D. Low, A. Mizoguchi, and E. Mizoguchi. Current Understanding of Dysbiosis in Disease in Human and Animal Models. Inflamm Bowel Dis 2016, 22, 1137–50. [Google Scholar] [CrossRef]
- Banaszak, M. Górna, D. Woźniak, J. Przysławski, and S. Drzymała-Czyż. Association between Gut Dysbiosis and the Occurrence of Sibo, Libo, Sifo and Imo. Microorganisms 2023, 11. [Google Scholar] [CrossRef]
- Castonguay-Paradis, S., S. Lacroix, G. Rochefort, L. Parent, J. Perron, C. Martin, B. Lamarche, F. Raymond, N. Flamand, V. Di Marzo, and A. Veilleux. Dietary Fatty Acid Intake and Gut Microbiota Determine Circulating Endocannabinoidome Signaling Beyond the Effect of Body Fat. Sci Rep 2020, 10, 15975. [Google Scholar] [CrossRef]
- Procházková, N., G. Falony, L. O. Dragsted, T. R. Licht, J. Raes, and H. M. Roager. Advancing Human Gut Microbiota Research by Considering Gut Transit Time. Gut 2023, 72, 180–91. [Google Scholar] [CrossRef]
- Sugihara, M., S. Takeuchi, M. Sugita, K. Higaki, M. Kataoka, and S. Yamashita. Analysis of Intra- and Intersubject Variability in Oral Drug Absorption in Human Bioequivalence Studies of 113 Generic Products. Mol Pharm 2015, 12, 4405–13. [Google Scholar] [CrossRef]
- Lavanya, D., V. Prasanna, A. Firdous, and S. Thakur. A Systemic Review on Chemotherapy Induced Nausea and Vomiting- Risk and Clinical Management with Alternative Therapies. Cancer Treat Res Commun 2025, 44, 100938. [Google Scholar]
- Braun, I. M., K. Bohlke, D. I. Abrams, H. Anderson, L. G. Balneaves, G. Bar-Sela, D. W. Bowles, P. R. Chai, A. Damani, A. Gupta, S. Hallmeyer, I. M. Subbiah, C. Twelves, M. S. Wallace, and E. J. Roeland. Cannabis and Cannabinoids in Adults with Cancer: Asco Guideline. J Clin Oncol 2024, 42, 1575–93. [Google Scholar] [CrossRef]
- Machado Rocha, F. C., S. C. Stéfano, R. De Cássia Haiek, L. M. Rosa Oliveira, and D. X. Da Silveira. Therapeutic Use of Cannabis Sativa on Chemotherapy-Induced Nausea and Vomiting among Cancer Patients: Systematic Review and Meta-Analysis. Eur J Cancer Care (Engl) 2008, 17, 431–43. [Google Scholar]
- Duran, M., E. Pérez, S. Abanades, X. Vidal, C. Saura, M. Majem, E. Arriola, M. Rabanal, A. Pastor, M. Farré, N. Rams, J. R. Laporte, and D. Capellà. Preliminary Efficacy and Safety of an Oromucosal Standardized Cannabis Extract in Chemotherapy-Induced Nausea and Vomiting. Br J Clin Pharmacol 2010, 70, 656–63. [Google Scholar]
- Venkatesan, T., D. J. Levinthal, B. U. K. Li, S. E. Tarbell, K. A. Adams, R. M. Issenman, I. Sarosiek, S. S. Jaradeh, R. N. Sharaf, S. Sultan, C. D. Stave, A. A. Monte, and W. L. Hasler. Role of Chronic Cannabis Use: Cyclic Vomiting Syndrome Vs Cannabinoid Hyperemesis Syndrome. Neurogastroenterol Motil 31 Suppl 2, no. Suppl 2019, 2, e13606. [Google Scholar]
- Boehnke, K. F., S. Gangopadhyay, D. J. Clauw, and R. L. Haffajee. Qualifying Conditions of Medical Cannabis License Holders in the United States. Health Aff (Millwood) 2019, 38, 295–302. [Google Scholar] [CrossRef]
- Mucke, M., T. Phillips, L. Radbruch, F. Petzke, and W. Hauser. Cannabis-Based Medicines for Chronic Neuropathic Pain in Adults. Cochrane Database Syst Rev 2018, 3, Cd012182. [Google Scholar] [PubMed]
- Aviram, J. , and G. Samuelly-Leichtag. Efficacy of Cannabis-Based Medicines for Pain Management: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Pain Physician 2017, 20, E755–e96. [Google Scholar] [CrossRef]
- Yanes, J. A., Z. E. McKinnell, M. A. Reid, J. N. Busler, J. S. Michel, M. M. Pangelinan, M. T. Sutherland, J. W. Younger, R. Gonzalez, and J. L. Robinson. Effects of Cannabinoid Administration for Pain: A Meta-Analysis and Meta-Regression. Exp Clin Psychopharmacol 2019, 27, 370–82. [Google Scholar] [CrossRef]
- Wilsey, B., T. Marcotte, R. Deutsch, B. Gouaux, S. Sakai, and H. Donaghe. Low-Dose Vaporized Cannabis Significantly Improves Neuropathic Pain. J Pain 2013, 14, 136–48. [Google Scholar] [CrossRef]
- Ware, M. A., T. Wang, S. Shapiro, A. Robinson, T. Ducruet, T. Huynh, A. Gamsa, G. J. Bennett, and J. P. Collet. Smoked Cannabis for Chronic Neuropathic Pain: A Randomized Controlled Trial. Cmaj 2010, 182, E694–701. [Google Scholar] [CrossRef]
- Ellis, R. J., W. Toperoff, F. Vaida, G. van den Brande, J. Gonzales, B. Gouaux, H. Bentley, and J. H. Atkinson. Smoked Medicinal Cannabis for Neuropathic Pain in Hiv: A Randomized, Crossover Clinical Trial. Neuropsychopharmacology 2009, 34, 672–80. [Google Scholar] [CrossRef]
- Sohler, N. L., J. L. Starrels, L. Khalid, M. A. Bachhuber, J. H. Arnsten, S. Nahvi, J. Jost, and C. O. Cunningham. Cannabis Use Is Associated with Lower Odds of Prescription Opioid Analgesic Use among Hiv-Infected Individuals with Chronic Pain. Subst Use Misuse 2018, 53, 1602–07. [Google Scholar] [CrossRef]
- Abrams, D. I., C. A. Jay, S. B. Shade, H. Vizoso, H. Reda, S. Press, M. E. Kelly, M. C. Rowbotham, and K. L. Petersen. Cannabis in Painful Hiv-Associated Sensory Neuropathy: A Randomized Placebo-Controlled Trial. Neurology 2007, 68, 515–21. [Google Scholar] [CrossRef]
- National Academies of Sciences, Engineering, Medicine, Health, Division Medicine, Health Board on Population, Practice Public Health, Review Committee on the Health Effects of Marijuana: An Evidence, and Agenda Research. The National Academies Collection: Reports Funded by National Institutes of Health. In The Health Effects of Cannabis and Cannabinoids: The Current State of Evidence and Recommendations for Research. Washington (DC): National Academies Press (US).
- 2017 by the National Academy of Sciences. All rights reserved.
- Boland, E. G., M. I. Bennett, V. Allgar, and J. W. Boland. Cannabinoids for Adult Cancer-Related Pain: Systematic Review and Meta-Analysis. BMJ Support Palliat Care 2020, 10, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Russo, E. B. Cannabis and Pain. Pain Med 2019, 20, 2083–85. [Google Scholar] [CrossRef] [PubMed]
- Huestis, M. A., J. E. Henningfield, and E. J. Cone. Blood Cannabinoids. I. Absorption of Thc and Formation of 11-Oh-Thc and Thccooh during and after Smoking Marijuana. J Anal Toxicol 1992, 16, 276–82. [Google Scholar] [CrossRef]
- Brown, J. D. , and A. G. Winterstein. Potential Adverse Drug Events and Drug-Drug Interactions with Medical and Consumer Cannabidiol (Cbd) Use. J Clin Med 2019, 8. [Google Scholar] [CrossRef]
- Bruni, N. Della Pepa, S. Oliaro-Bosso, E. Pessione, D. Gastaldi, and F. Dosio. Cannabinoid Delivery Systems for Pain and Inflammation Treatment. Molecules 2018, 23. [Google Scholar] [CrossRef] [PubMed]
- Lombardo D, Kiselev MA, and Caccamo MT. Smart Nanoparticles for Drug Delivery Application: Development of Versatile Nanocarrier Platforms in Biotechnology and Nanomedicine. J Nanomater 2019, 12, 1–26. [Google Scholar]
- Sim, S. , and N. K. Wong. Nanotechnology and Its Use in Imaging and Drug Delivery (Review). Biomed Rep 2021, 14, 42. [Google Scholar] [CrossRef]
- Singh, S. , and H. Sharma. Emerging Applications of Nanotechnology in Drug Delivery and Medical Imaging: Review. Curr Radiopharm 2023, 16, 269–83. [Google Scholar] [CrossRef]
- Ventola, C. L. Progress in Nanomedicine: Approved and Investigational Nanodrugs. P t 2017, 42, 742–55. [Google Scholar]
- Bramlett, K., E. Onel, E. R. Viscusi, and K. Jones. A Randomized, Double-Blind, Dose-Ranging Study Comparing Wound Infiltration of Depofoam Bupivacaine, an Extended-Release Liposomal Bupivacaine, to Bupivacaine Hcl for Postsurgical Analgesia in Total Knee Arthroplasty. Knee 2012, 19, 530–6. [Google Scholar] [CrossRef]
- Liu, L., Q. Ye, M. Lu, S. T. Chen, H. W. Tseng, Y. C. Lo, and C. Ho. A New Approach to Deliver Anti-Cancer Nanodrugs with Reduced Off-Target Toxicities and Improved Efficiency by Temporarily Blunting the Reticuloendothelial System with Intralipid. Sci Rep 2017, 7, 16106. [Google Scholar] [CrossRef]
- Passero, F. C., Jr. Grapsa, K. N. Syrigos, and M. W. Saif. The Safety and Efficacy of Onivyde (Irinotecan Liposome Injection) for the Treatment of Metastatic Pancreatic Cancer Following Gemcitabine-Based Therapy. Expert Rev Anticancer Ther 2016, 16, 697–703. [Google Scholar] [CrossRef]
- Alfayez, M., H. Kantarjian, T. Kadia, F. Ravandi-Kashani, and N. Daver. Cpx-351 (Vyxeos) in Aml. Leuk Lymphoma 2020, 61, 288–97. [Google Scholar] [CrossRef]
- Ohlmann, C. H. , and M. Gross-Langenhoff. Efficacy and Tolerability of Leuprorelin Acetate (Eligard®) in Daily Practice in Germany: Pooled Data from 2 Prospective, Non-Interventional Studies with 3- or 6-Month Depot Formulations in Patients with Advanced Prostate Cancer. Urol Int 2018, 100, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Fishburn, C. S. The Pharmacology of Pegylation: Balancing Pd with Pk to Generate Novel Therapeutics. J Pharm Sci 2008, 97, 4167–83. [Google Scholar] [CrossRef]
- Goel, N. , and S. Stephens. Certolizumab Pegol. MAbs 2010, 2, 137–47. [Google Scholar] [CrossRef]
- Freeman, T. P., C. Hindocha, S. F. Green, and M. A. P. Bloomfield. Medicinal Use of Cannabis Based Products and Cannabinoids. Bmj 2019, 365, l1141. [Google Scholar] [CrossRef]
- Pacher, P., N. M. Kogan, and R. Mechoulam. Beyond Thc and Endocannabinoids. Annu Rev Pharmacol Toxicol 2020, 60, 637–59. [Google Scholar] [CrossRef] [PubMed]
- Grotenhermen, F. Pharmacokinetics and Pharmacodynamics of Cannabinoids. Clin Pharmacokinet 2003, 42, 327–60. [Google Scholar] [CrossRef]
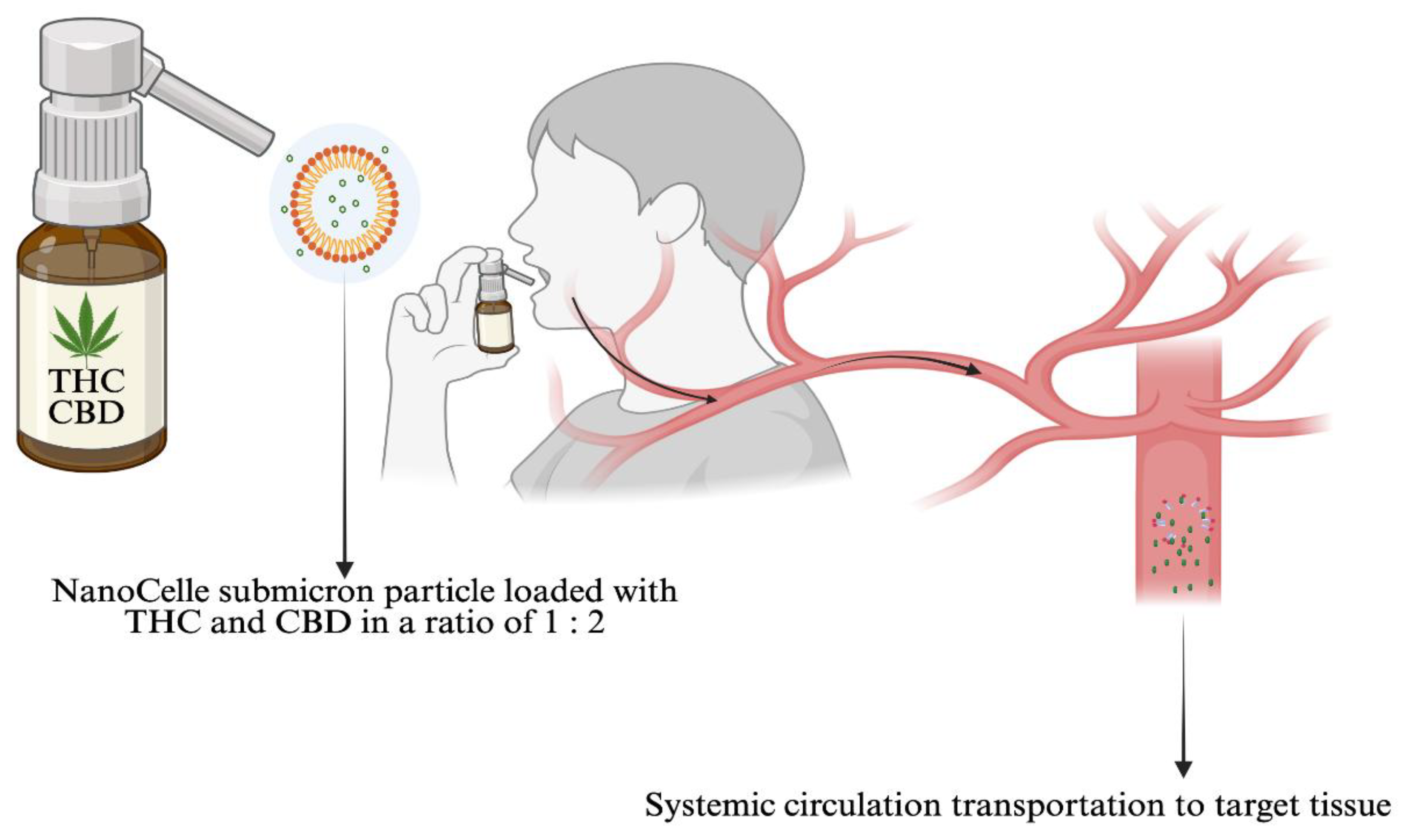
- Reuter, S. E. B. Schultz, A. J. McLachlan, J. D. Henson, and L. Vitetta. Pharmacokinetics and Bioavailability of Cannabinoids Administered Via a Novel Orobuccal Nanoparticle Formulation (Nanocelle™) in Patients with Advanced Cancer. Cannabis Cannabinoid Res 2025. [Google Scholar] [CrossRef]
- Vitetta, L. Zhou, R. Manuel, S. Dal Forno, S. Hall, and D. Rutolo. Route and Type of Formulation Administered Influences the Absorption and Disposition of Vitamin B(12) Levels in Serum. J Funct Biomater 2018, 9. [Google Scholar] [CrossRef]
- Clarke, S., B. E. Butcher, A. J. McLachlan, J. D. Henson, D. Rutolo, S. Hall, and L. Vitetta. Pilot Clinical and Pharmacokinetic Study of Δ9-Tetrahydrocannabinol (Thc)/Cannabidiol (Cbd) Nanoparticle Oro-Buccal Spray in Patients with Advanced Cancer Experiencing Uncontrolled Pain. PLoS One 2022, 17, e0270543. [Google Scholar]
- Vitetta, L., B. Butcher, J. D. Henson, D. Rutolo, and S. Hall. A Pilot Safety, Tolerability and Pharmacokinetic Study of an Oro-Buccal Administered Cannabidiol-Dominant Anti-Inflammatory Formulation in Healthy Individuals: A Randomized Placebo-Controlled Single-Blinded Study. Inflammopharmacology 2021, 29, 1361–70. [Google Scholar] [PubMed]
- Tang, C., K. Zhou, D. Wu, and H. Zhu. Nanoparticles as a Novel Platform for Cardiovascular Disease Diagnosis and Therapy. Int J Nanomedicine 2024, 19, 8831–46. [Google Scholar] [CrossRef] [PubMed]
- Hari Priya, V. M., A. A. Ganapathy, M. G. Veeran, M. S. Raphael, and A. Kumaran. Nanotechnology-Based Drug Delivery Platforms for Erectile Dysfunction: Addressing Efficacy, Safety, and Bioavailability Concerns. Pharm Dev Technol 2024, 29, 996–1015. [Google Scholar] [CrossRef]
- Szebeni, J., P. Bedocs, Z. Rozsnyay, Z. Weiszhár, R. Urbanics, L. Rosivall, R. Cohen, O. Garbuzenko, G. Báthori, M. Tóth, R. Bünger, and Y. Barenholz. Liposome-Induced Complement Activation and Related Cardiopulmonary Distress in Pigs: Factors Promoting Reactogenicity of Doxil and Ambisome. Nanomedicine 2012, 8, 176–84. [Google Scholar]
- Crisafulli, S., P. M. Cutroneo, N. Luxi, A. Fontana, C. Ferrajolo, P. Marchione, L. Sottosanti, G. Zanoni, U. Moretti, S. Franzè, P. Minghetti, and G. Trifirò. Is Pegylation of Drugs Associated with Hypersensitivity Reactions? An Analysis of the Italian National Spontaneous Adverse Drug Reaction Reporting System. Drug Saf 2023, 46, 343–55. [Google Scholar]
- Berman, J. D. U. S Food and Drug Administration Approval of Ambisome (Liposomal Amphotericin B) for Treatment of Visceral Leishmaniasis. Clin Infect Dis 1999, 28, 49–51. [Google Scholar] [CrossRef]
- Alzahrani, A. M., M. A. Alnuhait, and T. Alqahtani. The Clinical Safety and Efficacy of Cytarabine and Daunorubicin Liposome (Cpx-351) in Acute Myeloid Leukemia Patients: A Systematic Review. Cancer Rep (Hoboken) 2025, 8, e70199. [Google Scholar]
- Tiriveedhi, V., K. M. Kitchens, K. J. Nevels, H. Ghandehari, and P. Butko. Kinetic Analysis of the Interaction between Poly(Amidoamine) Dendrimers and Model Lipid Membranes. Biochim Biophys Acta 2011, 1808, 209–18. [Google Scholar] [CrossRef]
- Wang, J., B. Li, L. Qiu, X. Qiao, and H. Yang. Dendrimer-Based Drug Delivery Systems: History, Challenges, and Latest Developments. J Biol Eng 2022, 16, 18. [Google Scholar] [CrossRef]
- Henson, J. D. Vitetta, M. Quezada, and S. Hall. Enhancing Endocannabinoid Control of Stress with Cannabidiol. J Clin Med 2021, 10. [Google Scholar] [CrossRef]
- Fasinu, P. S., S. Phillips, M. A. ElSohly, and L. A. Walker. Current Status and Prospects for Cannabidiol Preparations as New Therapeutic Agents. Pharmacotherapy 2016, 36, 781–96. [Google Scholar] [CrossRef]
- Taylor, L., B. Gidal, G. Blakey, B. Tayo, and G. Morrison. A Phase I, Randomized, Double-Blind, Placebo-Controlled, Single Ascending Dose, Multiple Dose, and Food Effect Trial of the Safety, Tolerability and Pharmacokinetics of Highly Purified Cannabidiol in Healthy Subjects. CNS Drugs 2018, 32, 1053–67. [Google Scholar]
- Guy GW, and Flint ME. A Single Centre, Placebo-Controlled, Four Period, Crossover, Tolerability Study Assessing, Pharmacodynamic Effects, Pharmacokinetic Characteristics and Cognitive Profiles of a Single Dose of Three Formulations of Cannabis Based Medicine Extracts (Cbmes) (Gwpd9901), Plus a Two Period Tolerability Study Comparing Pharmacodynamic Effects and Pharmacokinetic Characteristics of a Single Dose of a Cannabis Based Medicine Extract Given Via Two Administration Routes (Gwpd9901 Ext). Journal of Cannabis Therapeutics 2004, 3, 35–77. [Google Scholar] [CrossRef]
- Ohlsson, A., J. E. Lindgren, S. Andersson, S. Agurell, H. Gillespie, and L. E. Hollister. Single-Dose Kinetics of Deuterium-Labelled Cannabidiol in Man after Smoking and Intravenous Administration. Biomed Environ Mass Spectrom 1986, 13, 77–83. [Google Scholar] [CrossRef]
| Drug Manufacturer Brand / Trade Name [Ref] |
Pharmaceutical | Method of Delivery |
Indication |
|---|---|---|---|
| MICELLES | |||
| Celgene, Summit USA Abraxane [60] |
Paclitaxel | Intravenous Administration |
—Breast cancer —Non-Small Lung cancer —Pancreatic cancer —Ovarian cancer |
| LIPOSOMES | |||
| Pacira Pharmaceuticals, San Diego, CA USA DepoDur [61] |
Morphine injectable (slow release) |
Epidural Administration |
—Postoperative pain |
| Marqibo (Spectrum Pharmaceuticals Boston, MA, USA Oncovin | Vincasar PFS | Vincrex [62] |
Vincristine | Intravenous Infusion |
—Acute Lymphoblastic Leukemia |
| Ipsen Biopharmaceuticals, France Onivyde [63] |
Irinotecan | Intravenous Injection |
—Metastatic Adenocarcinoma of the Pancreas |
| Jazz Pharmaceuticals, Dublin Ireland Vyxeos [64] |
Daunorubicin and Cytarabine |
—Intramuscular —Intrathecal —Subcutaneous Injection |
—Acute Myeloid Leukemia |
| DENDRIMERS (polymer nanoparticles) | |||
| Tolmar Chicago, IL, USA Eligard [65] |
Leuprolide acetate and polymer |
Subcutaneous Injections |
—Prostate cancer |
| Pfizer New York NY, USA Somavert [66] |
Pegvisomant | Subcutaneous Injections |
—Acromegaly |
| UCB Brussels Belgium Cimzia [67] |
Certolizumab | Tablets or Intravenous Injections |
—Rheumatoid Arthritis — Crohn’s Disease —Psoriatic Arthritis, —Ankylosing Spondylitis |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




