Submitted:
18 July 2025
Posted:
22 July 2025
You are already at the latest version
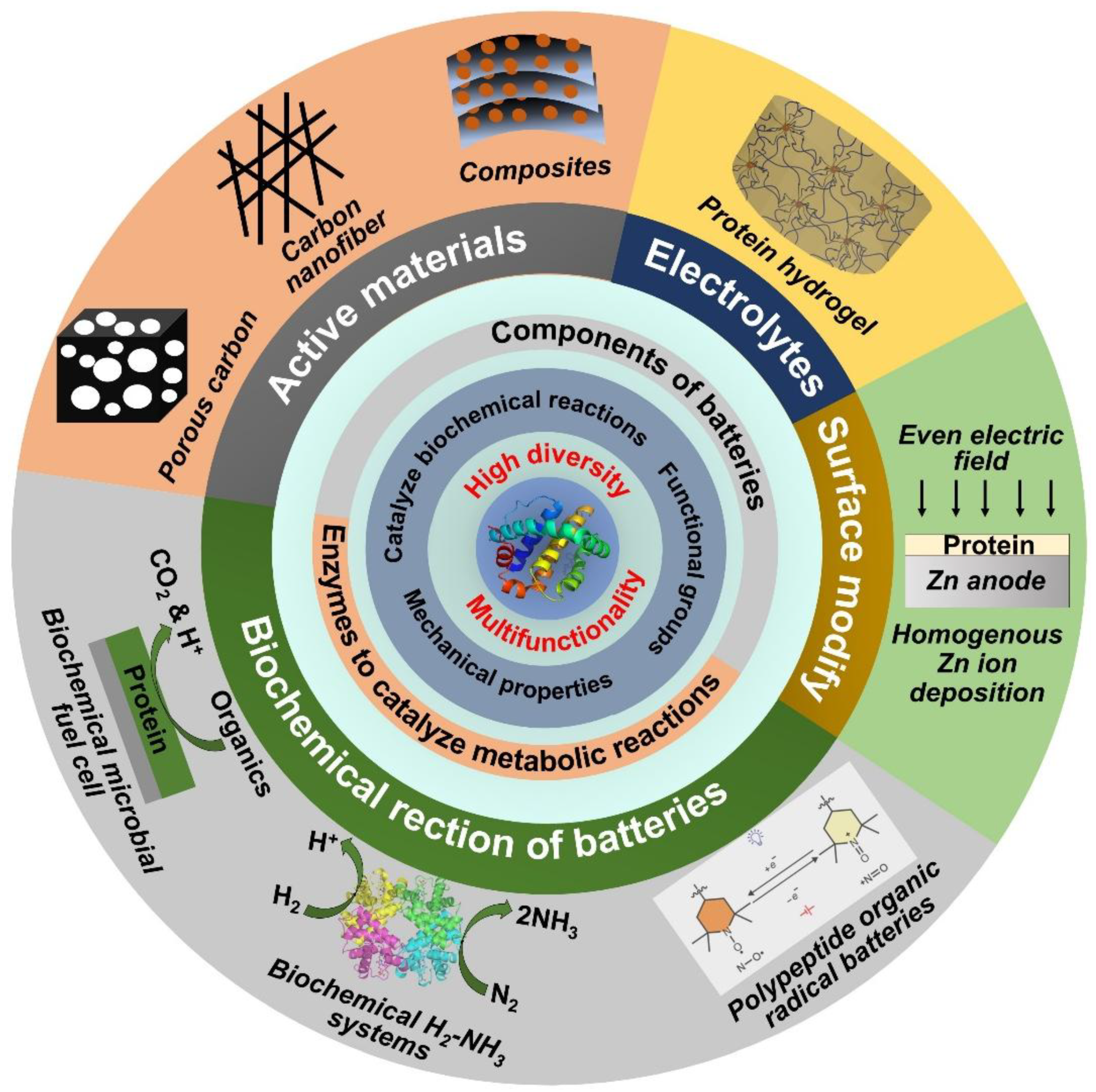
Abstract
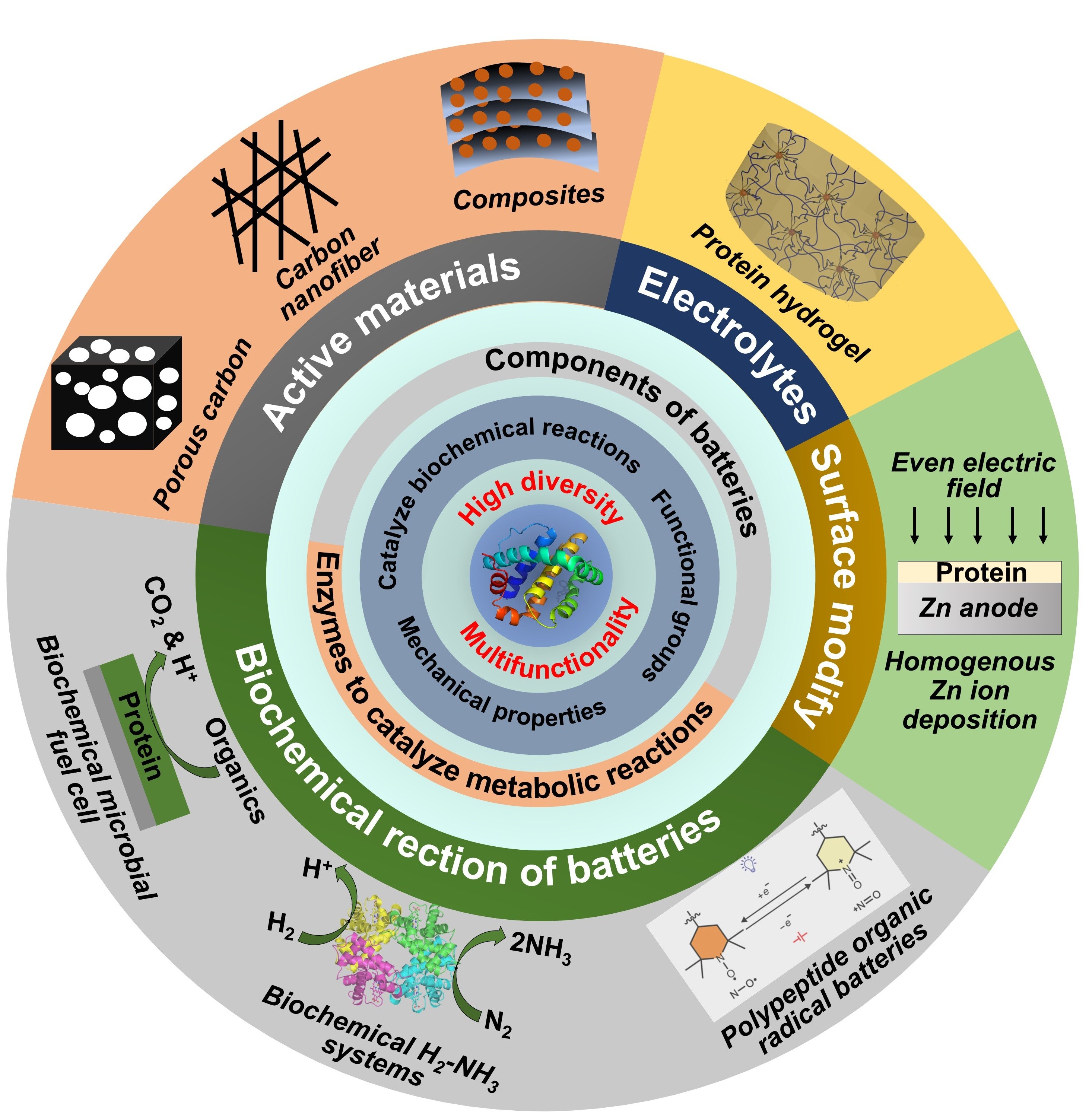
Keywords:
Contents
| Abstract……………………………………………………………………………….……….. | 1 |
| 1. Introduction……………………………………………………………………….………… | 2 |
| 2. Protein-derived materials for electrodes……………………………………….…………. | 3 |
| 2.1. Protein-derived carbon………………………………………………………...………. | 3 |
| 2.1.1 Protein-derived carbon for electrodes of Zn-air batteries………………………. | 4 |
| 2.1.2 Protein-derived carbon for electrodes of vanadium redox flow batteries…….. | 6 |
| 2.2. Protein-modified active materials…………………………………………….……….. | 7 |
| 3. Protective films for Zn metal anodes………………………………………………………. | 9 |
| 4. Protein-based gel state electrolytes for Zn-ion batteries………………………………….. | 11 |
| 5. Protein-derived catalysts…………………………………………………………………….. | 12 |
| 5.1 Biochemical microbial fuel cells………………………………………………………… | 12 |
| 5.2 Biochemical H2-NH3 systems…………………………………………………………... | 13 |
| 5.3 Bio-nanobatteries…………………………………………………………………………. | 14 |
| 6. Polypeptide organic radical batteries………………………………………………………. | 15 |
| 7. Conclusions and Perspectives……………………………………………………………….. | 16 |
| References………………………………………………………………………………………… | 17 |
1. Introduction
2. Protein-Derived Materials for Electrodes
2.1. Protein-Derived Carbon
2.1.1. Protein-Derived Carbon for Electrodes of Zn-Air Batteries
2.1.2. Protein-Derived Carbon for Electrodes of Vanadium Redox Flow Batteries
2.2. Protein-Modified Active Materials
3. Protective Films for Zn Metal Anodes
4. Protein-Based Gel State Electrolytes for Zn-Ion Batteries
5. Protein-Derived Catalysts
5.1. Biochemical Microbial Fuel Cells
5.2. Biochemical H2-NH3 Systems
5.3. Bio-Nanobatteries
6. Polypeptide Organic Radical Batteries
7. Conclusions and Perspectives
Acknowledgments
References
- Hassan, Q.; Viktor, P.; J. Al-Musawi, T.; Mahmood Ali, B.; Algburi, S.; Alzoubi, H.M.; Khudhair Al-Jiboory, A.; Zuhair Sameen, A.; Salman, H.M.; Jaszczur, M. The Renewable Energy Role in the Global Energy Transformations. Renew. Energy Focus 2024, 48, 100545. [Google Scholar] [CrossRef]
- Elalfy, D.A.; Gouda, E.; Kotb, M.F.; Bureš, V.; Sedhom, B.E. Comprehensive Review of Energy Storage Systems Technologies, Objectives, Challenges, and Future Trends. Energy Strateg. Rev. 2024, 54. [Google Scholar] [CrossRef]
- Li, M.; Lu, J.; Chen, Z.; Amine, K. 30 Years of Lithium-Ion Batteries. Adv. Mater. 2018, 30, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Zampardi, G.; La Mantia, F. Open Challenges and Good Experimental Practices in the Research Field of Aqueous Zn-Ion Batteries. Nat. Commun. 2022, 13, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Leong, K.W.; Wang, Y.; Ni, M.; Pan, W.; Luo, S.; Leung, D.Y.C. Rechargeable Zn-Air Batteries: Recent Trends and Future Perspectives. Renew. Sustain. Energy Rev. 2022, 154, 111771. [Google Scholar] [CrossRef]
- Yao, Y.; Lei, J.; Shi, Y.; Ai, F.; Lu, Y.C. Assessment Methods and Performance Metrics for Redox Flow Batteries. Nat. Energy 2021, 6, 582–588. [Google Scholar] [CrossRef]
- Liu, L.; Solin, N.; Inganäs, O. Bio Based Batteries. Adv. Energy Mater. 2021, 11, 1–12. [Google Scholar] [CrossRef]
- Tang, L.; Peng, H.; Kang, J.; Chen, H.; Zhang, M.; Liu, Y.; Kim, D.H.; Liu, Y.; Lin, Z. Zn-Based Batteries for Sustainable Energy Storage: Strategies and Mechanisms. Chem. Soc. Rev. 2024, 53, 4877–4925. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Díez, E.; Ventosa, E.; Guarnieri, M.; Trovò, A.; Flox, C.; Marcilla, R.; Soavi, F.; Mazur, P.; Aranzabe, E.; Ferret, R. Redox Flow Batteries: Status and Perspective towards Sustainable Stationary Energy Storage. J. Power Sources 2021, 481. [Google Scholar] [CrossRef]
- Wang, C.; Fu, X.; Ying, C.; Liu, J.; Zhong, W. Natural Protein as Novel Additive of a Commercial Electrolyte for Long-Cycling Lithium Metal Batteries. Chem. Eng. J. 2022, 437(P1), 135283. [Google Scholar] [CrossRef]
- Wang, C.; Fu, X.; Lin, S.; Liu, J.; Zhong, W.H. A Protein-Enabled Protective Film with Functions of Self-Adapting and Anion-Anchoring for Stabilizing Lithium-Metal Batteries. J. Energy Chem. 2021, 64, 485–495. [Google Scholar] [CrossRef]
- Wang, C.; Odstrcil, R.; Liu, J.; Zhong, W.H. Protein-Modified SEI Formation and Evolution in Li Metal Batteries. J. Energy Chem. 2022, 73, 248–258. [Google Scholar] [CrossRef]
- Wang, C.; Zhong, W.-H. Introduction. In Natural Protein-based Strategies for Batteries; WORLD SCIENTIFIC, 2023; pp. 1–21. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, Y.; Zhou, J.; Wang, C.; Zhang, W.; Li, Z.; Guo, F.; Chen, H.; Zhang, H. Highly Dispersed SnO2 Nanoparticles Confined on Xylem Fiber-Derived Carbon Frameworks as Anodes for Lithium-Ion Batteries. 2020, 879, 114753. [Google Scholar] [CrossRef]
- Wang, C.; Zhong, W. Promising Sustainable Technology for Energy Storage Devices : Natural Protein-Derived Active Materials. Electrochim. Acta 2023, 441, 141860. [Google Scholar] [CrossRef]
- Wang, C.; Ren, L.; Ying, C.; Shang, J.; Mccloy, J.; Liu, J. Soy Protein as a Dual-Functional Bridge Enabling High Performance Solid Electrolyte for Li Metal Batteries. J. Power Sources 2024, 620, 235260. [Google Scholar] [CrossRef]
- Wang, C.; Ren, L.; Ying, C.; Liu, J.; Zhong, W.H. An Amino Acid-Enabled Separator for Effective Stabilization of Li Anodes. ACS Appl. Mater. Interfaces 2024, 16, 15632–15639. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Yang, X.; Zheng, M.; Xu, Y. Synthesis of β-FeOOH Nanorods Adhered to Pine-Biomass Carbon as a Low-Cost Anode Material for Li-Ion Batteries. J. Alloys Compd. 2019, 794, 569–575. [Google Scholar] [CrossRef]
- Sun, Y.; Shi, X.L.; Yang, Y.L.; Suo, G.; Zhang, L.; Lu, S.; Chen, Z.G. Biomass-Derived Carbon for High-Performance Batteries: From Structure to Properties. Adv. Funct. Mater. 2022, 32. [Google Scholar] [CrossRef]
- Wang, C.; Xie, N.H.; Zhang, Y.; Huang, Z.; Xia, K.; Wang, H.; Guo, S.; Xu, B.Q.; Zhang, Y. Silk-Derived Highly Active Oxygen Electrocatalysts for Flexible and Rechargeable Zn-Air Batteries. Chem. Mater. 2019, 31, 1023–1029. [Google Scholar] [CrossRef]
- Hao, M.; Dun, R.; Su, Y.; He, L.; Ning, F.; Zhou, X.; Li, W. In Situ Self-Doped Biomass-Derived Porous Carbon as an Excellent Oxygen Reduction Electrocatalyst for Fuel Cells and Metal-Air Batteries. J. Mater. Chem. A 2021, 9, 14331–14343. [Google Scholar] [CrossRef]
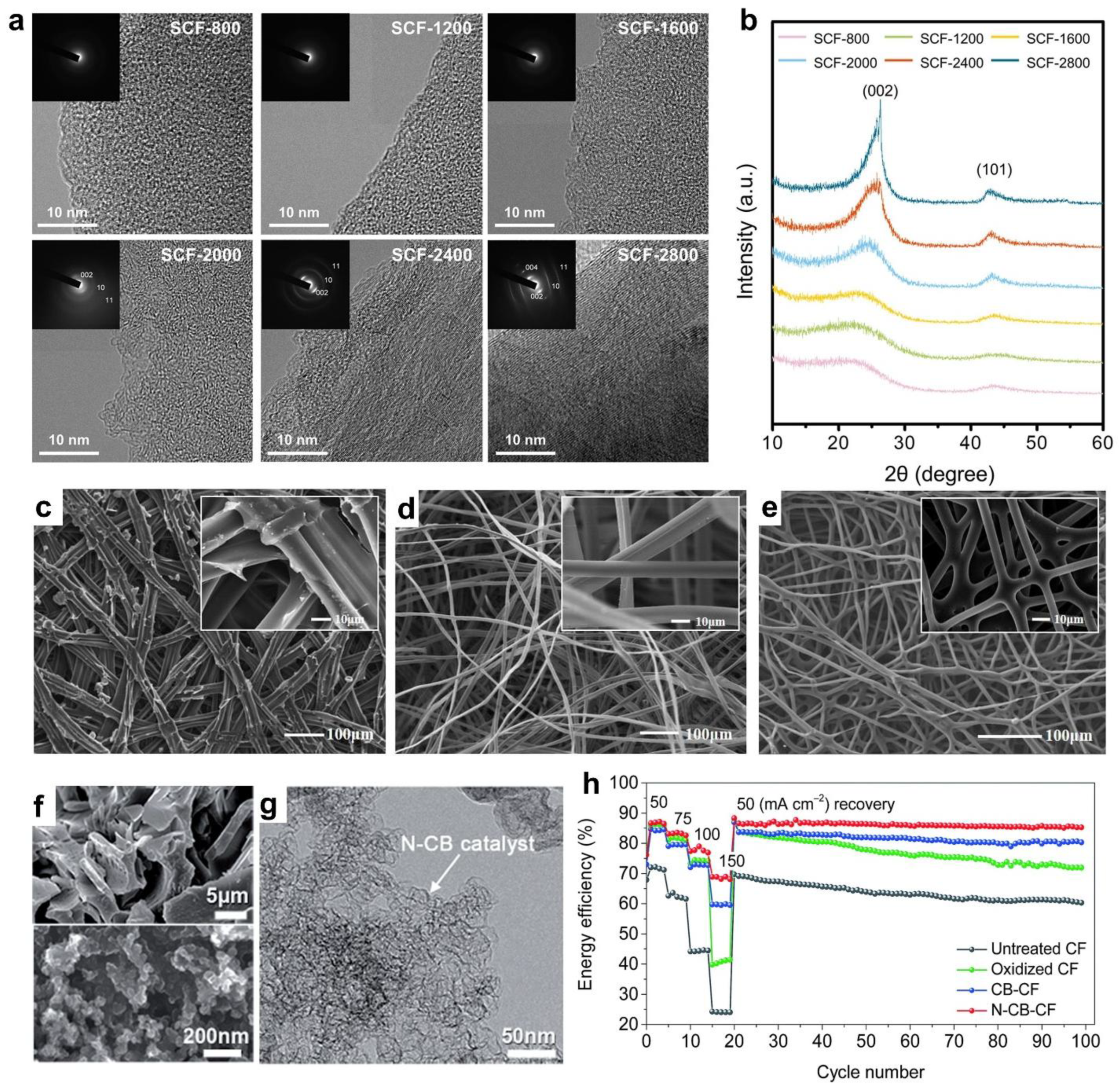
- Lee, M.E.; Jang, D.; Lee, S.; Yoo, J.; Choi, J.; Jin, H.J.; Lee, S.; Cho, S.Y. Silk Protein-Derived Carbon Fabric as an Electrode with High Electro-Catalytic Activity for All-Vanadium Redox Flow Batteries. Appl. Surf. Sci. 2021, 567, 150810. [Google Scholar] [CrossRef]
- Wang, R.; Li, Y. Twin-Cocoon-Derived Self-Standing Nitrogen-Oxygen-Rich Monolithic Carbon Material as the Cost-Effective Electrode for Redox Flow Batteries. J. Power Sources 2019, 421, 139–146. [Google Scholar] [CrossRef]
- Lee, M.E.; Jin, H.J.; Yun, Y.S. Synergistic Catalytic Effects of Oxygen and Nitrogen Functional Groups on Active Carbon Electrodes for All-Vanadium Redox Flow Batteries. RSC Adv. 2017, 7, 43227–43232. [Google Scholar] [CrossRef]
- Park, M.; Ryu, J.; Kim, Y.; Cho, J. Corn Protein-Derived Nitrogen-Doped Carbon Materials with Oxygen-Rich Functional Groups: A Highly Efficient Electrocatalyst for All-Vanadium Redox Flow Batteries. Energy Environ. Sci. 2014, 7, 3727–3735. [Google Scholar] [CrossRef]
- Kim, H.S.; Lee, J.; Jang, J.H.; Jin, H.; Paidi, V.K.; Lee, S.H.; Lee, K.S.; Kim, P.; Yoo, S.J. Waste Pig Blood-Derived 2D Fe Single-Atom Porous Carbon as an Efficient Electrocatalyst for Zinc–Air Batteries and AEMFCs. Appl. Surf. Sci. 2021, 563, 150208. [Google Scholar] [CrossRef]
- Puglia, M.K.; Malhotra, M.; Chivukula, A.; Kumar, C.V. “simple-Stir” Heterolayered MoS2/Graphene Nanosheets for Zn-Air Batteries. ACS Appl. Nano Mater. 2021, 4, 10389–10398. [Google Scholar] [CrossRef]
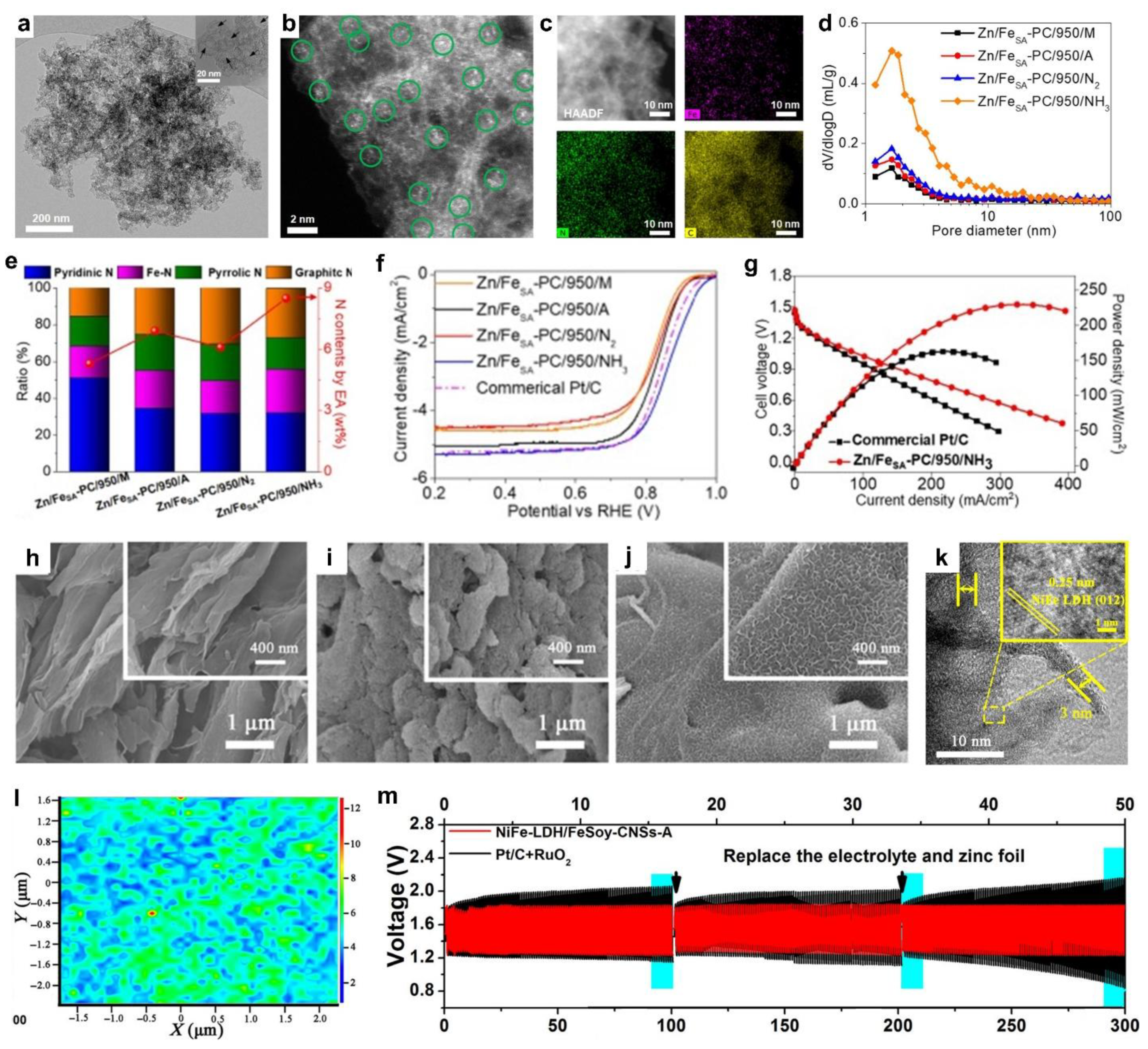
- Zhang, M.; Zhang, J.; Ran, S.; Qiu, L.; Sun, W.; Yu, Y.; Chen, J.; Zhu, Z. A Robust Bifunctional Catalyst for Rechargeable Zn-Air Batteries: Ultrathin NiFe-LDH Nanowalls Vertically Anchored on Soybean-Derived Fe-N-C Matrix. Nano Res. 2021, 14, 1175–1186. [Google Scholar] [CrossRef]
- Shen, F.; Pankratov, D.; Pankratova, G.; Toscano, M.D.; Zhang, J.; Ulstrup, J.; Chi, Q.; Gorton, L. Supercapacitor/Biofuel Cell Hybrid Device Employing Biomolecules for Energy Conversion and Charge Storage. Bioelectrochemistry 2019, 128, 94–99. [Google Scholar] [CrossRef] [PubMed]
- Zhi, J.; Li, S.; Han, M.; Chen, P. Biomolecule-Guided Cation Regulation for Dendrite-Free Metal Anodes. Sci. Adv. 2020, 6, 1–15. [Google Scholar] [CrossRef] [PubMed]
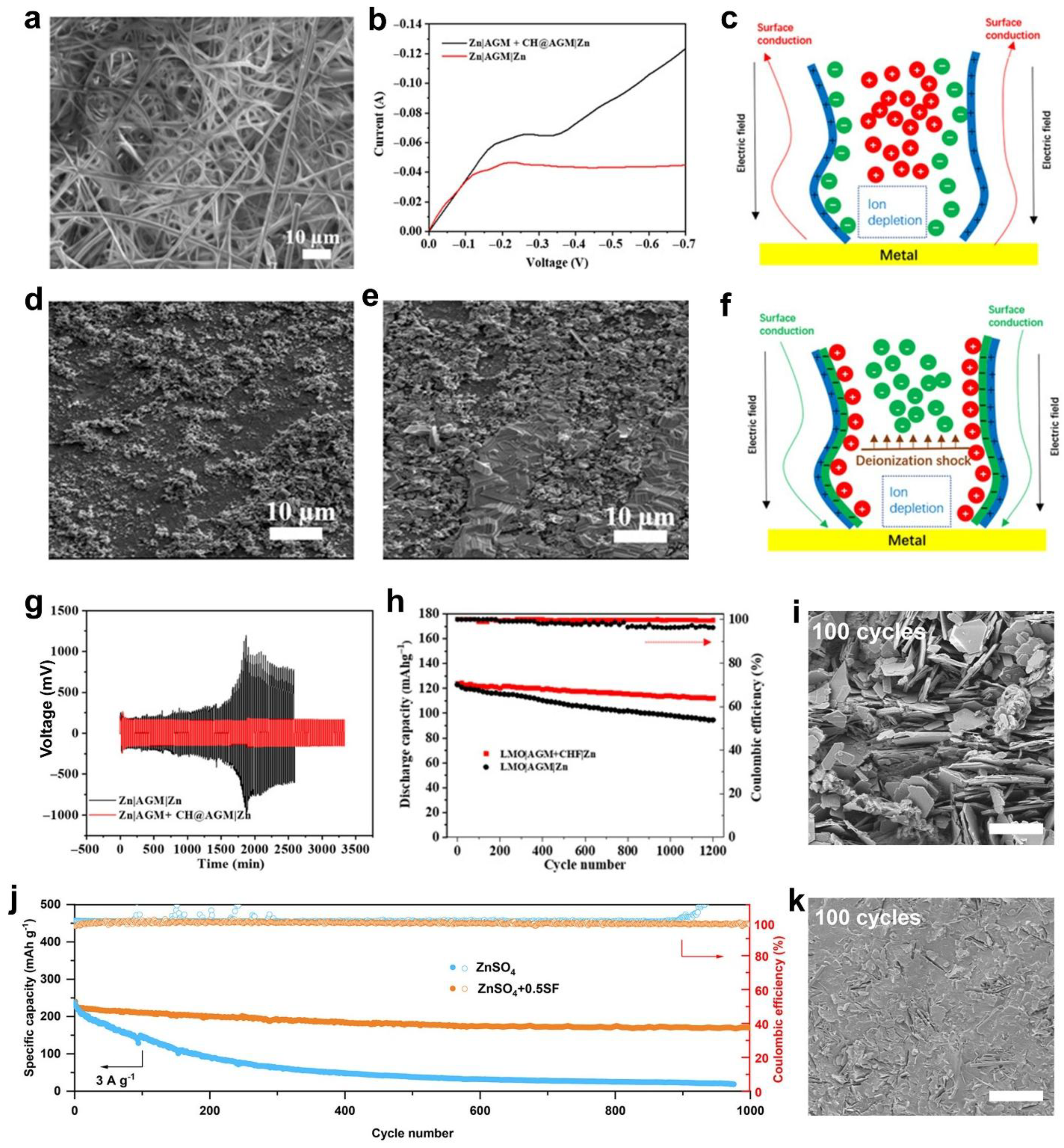
- Xu, J.; Lv, W.; Yang, W.; Jin, Y.; Jin, Q.; Sun, B.; Zhang, Z.; Wang, T.; Zheng, L.; Shi, X.; Sun, B.; Wang, G. In Situ Construction of Protective Films on Zn Metal Anodes via Natural Protein Additives Enabling High-Performance Zinc Ion Batteries. ACS Nano 2022, 16, 11392–11404. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Ran, Q.; Huang, H.; Xie, Y.; Zhong, M.; Lu, G.; Bai, F.-Q.; Lang, X.-Y.; Jia, X.; Chao, D. Interface Reversible Electric Field Regulated by Amphoteric Charged Protein-Based Coating Toward High-Rate and Robust Zn Anode. Nano-Micro Lett. 2022, 14, 1–14. [Google Scholar] [CrossRef] [PubMed]
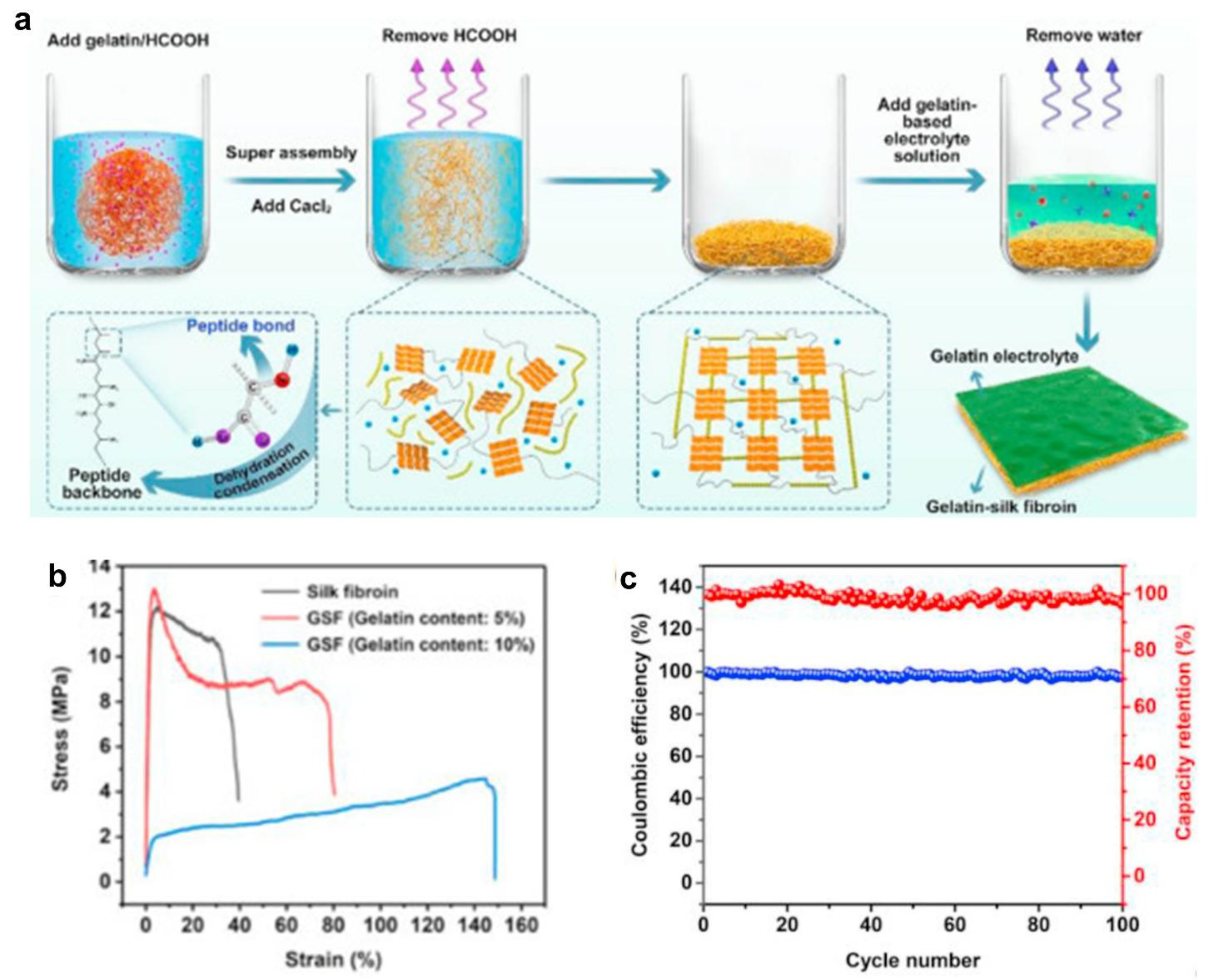
- Zhou, J.; Li, Y.; Xie, L.; Xu, R.; Zhang, R.; Gao, M.; Tian, W.; Li, D.; Qiao, L.; Wang, T.; Cao, J.; Wang, D.; Hou, Y.; Fu, W.; Yang, B.; Zeng, J.; Chen, P.; Liang, K.; Kong, B. Humidity-Sensitive, Shape-Controllable, and Transient Zinc-Ion Batteries Based on Plasticizing Gelatin-Silk Protein Electrolytes. Mater. Today Energy 2021, 21. [Google Scholar] [CrossRef]
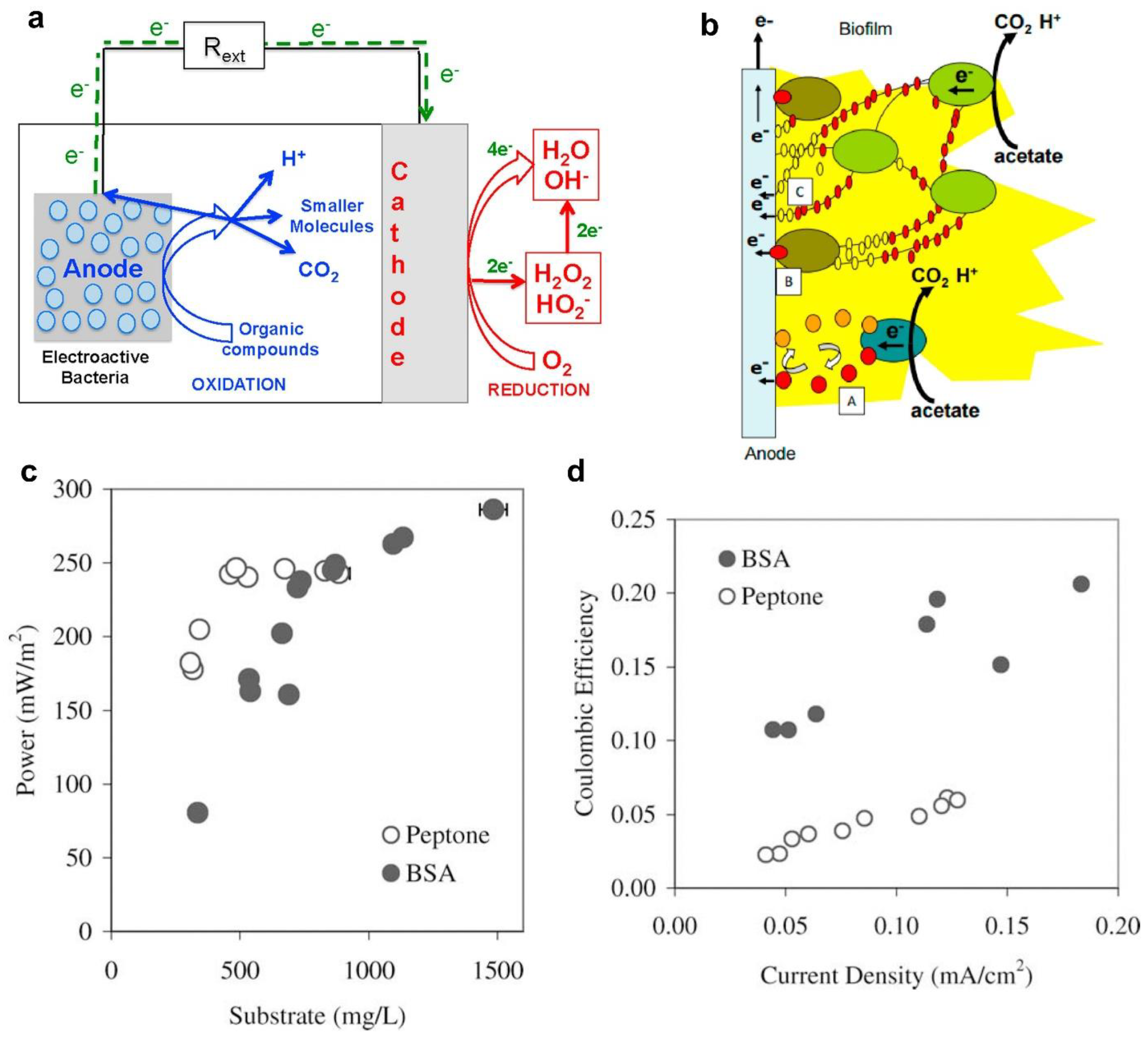
- Santoro, C.; Arbizzani, C.; Erable, B.; Ieropoulos, I. Microbial Fuel Cells: From Fundamentals to Applications. A Review. J. Power Sources 2017, 356, 225–244. [Google Scholar] [CrossRef] [PubMed]
- Heilmann, J.; Logan, B.E. Production of Electricity from Proteins Using a Microbial Fuel Cell. Water Environ. Res. 2006, 78, 531–537. [Google Scholar] [CrossRef] [PubMed]
- Kannan, A.M.; Renugopalakrishnan, V.; Filipek, S.; Li, P.; Audette, G.F.; Munukutla, L. Bio-Batteries and Bio-Fuel Cells: Leveraging on Electronic Charge Transfer Proteins. J. Nanosci. Nanotechnol. 2009, 9, 1665–1678. [Google Scholar] [CrossRef] [PubMed]
- Venkatesha, B.; Guna, V.; Bhuvaneswari, H.B.; Reddy, N. A Study on the Potential of Using Plant Proteins as Electrolytes in a Biochemical Cell. Environ. Prog. Sustain. Energy 2018, 37, 961–967. [Google Scholar] [CrossRef]
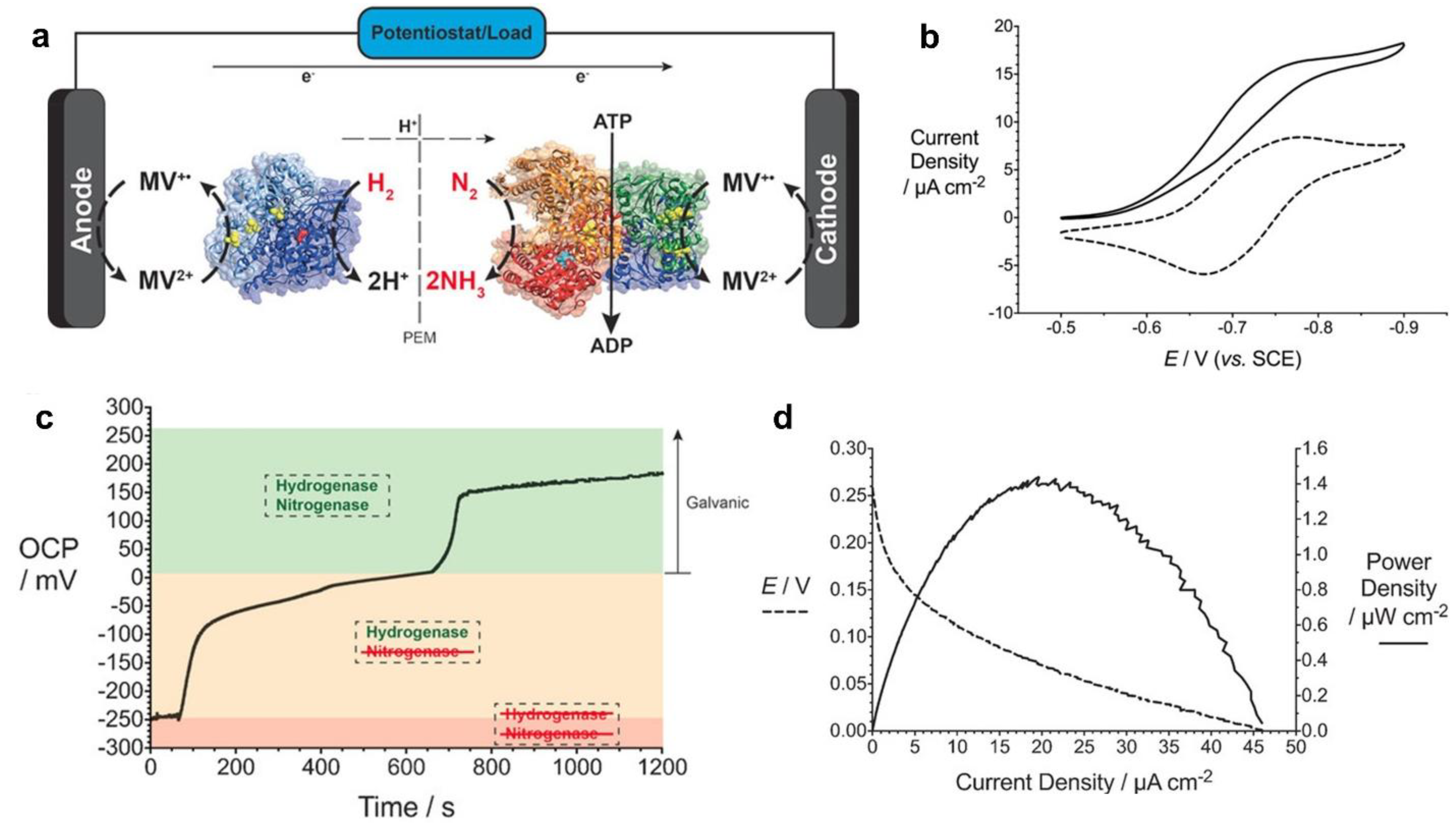
- Milton, R.D.; Cai, R.; Abdellaoui, S.; Leech, D.; De Lacey, A.L.; Pita, M.; Minteer, S.D. Bioelectrochemical Haber–Bosch Process: An Ammonia-Producing H2/N2Fuel Cell. Angew. Chemie - Int. Ed. 2017, 56, 2680–2683. [Google Scholar] [CrossRef] [PubMed]
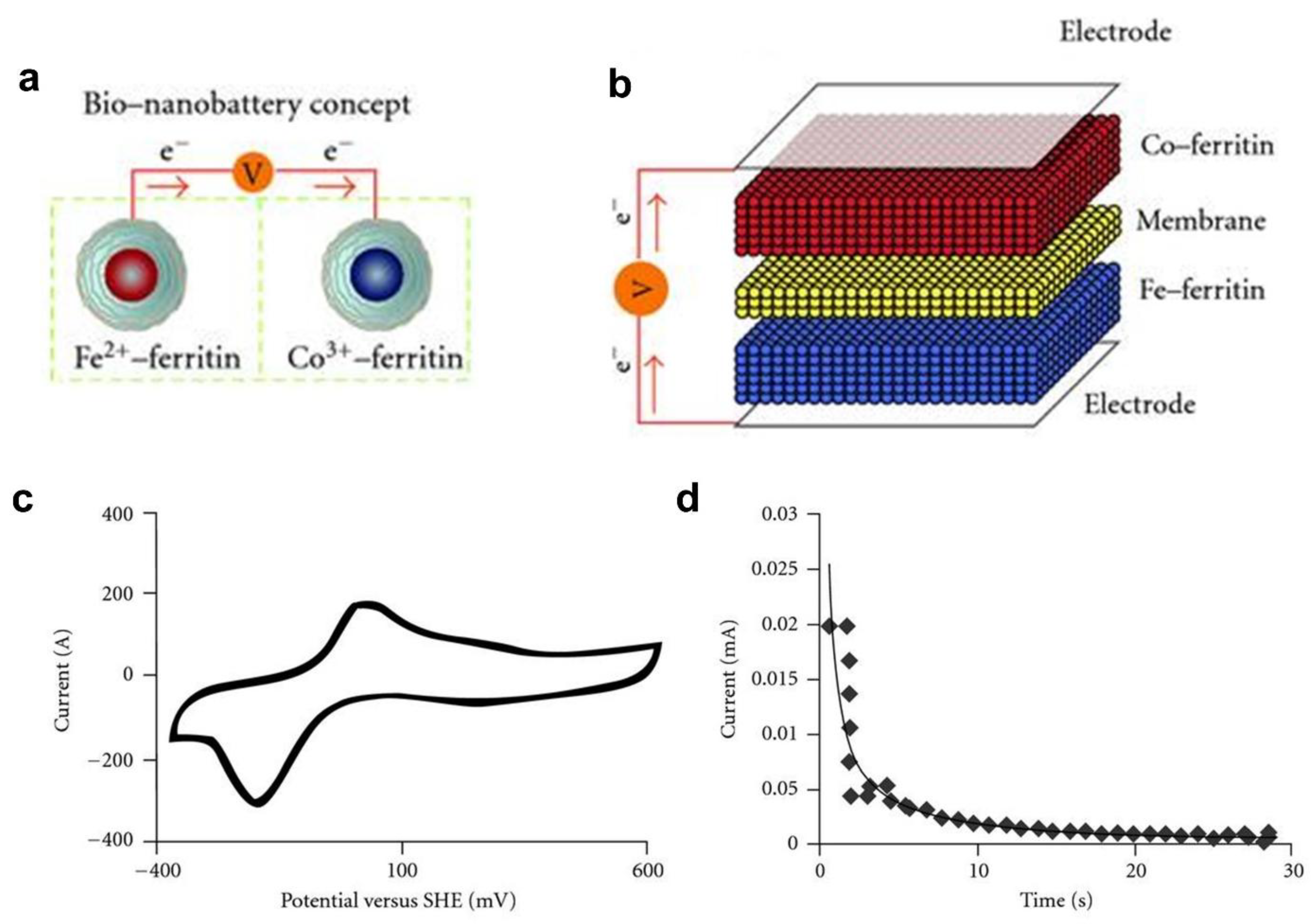
- Watt, G.D.; Kim, J.W.; Zhang, B.; Miller, T.; Harb, J.N.; Davis, R.C.; Choi, S.H. A Protein-Based Ferritin Bio-Nanobattery. J. Nanotechnol. 2012, 2012. [Google Scholar] [CrossRef]
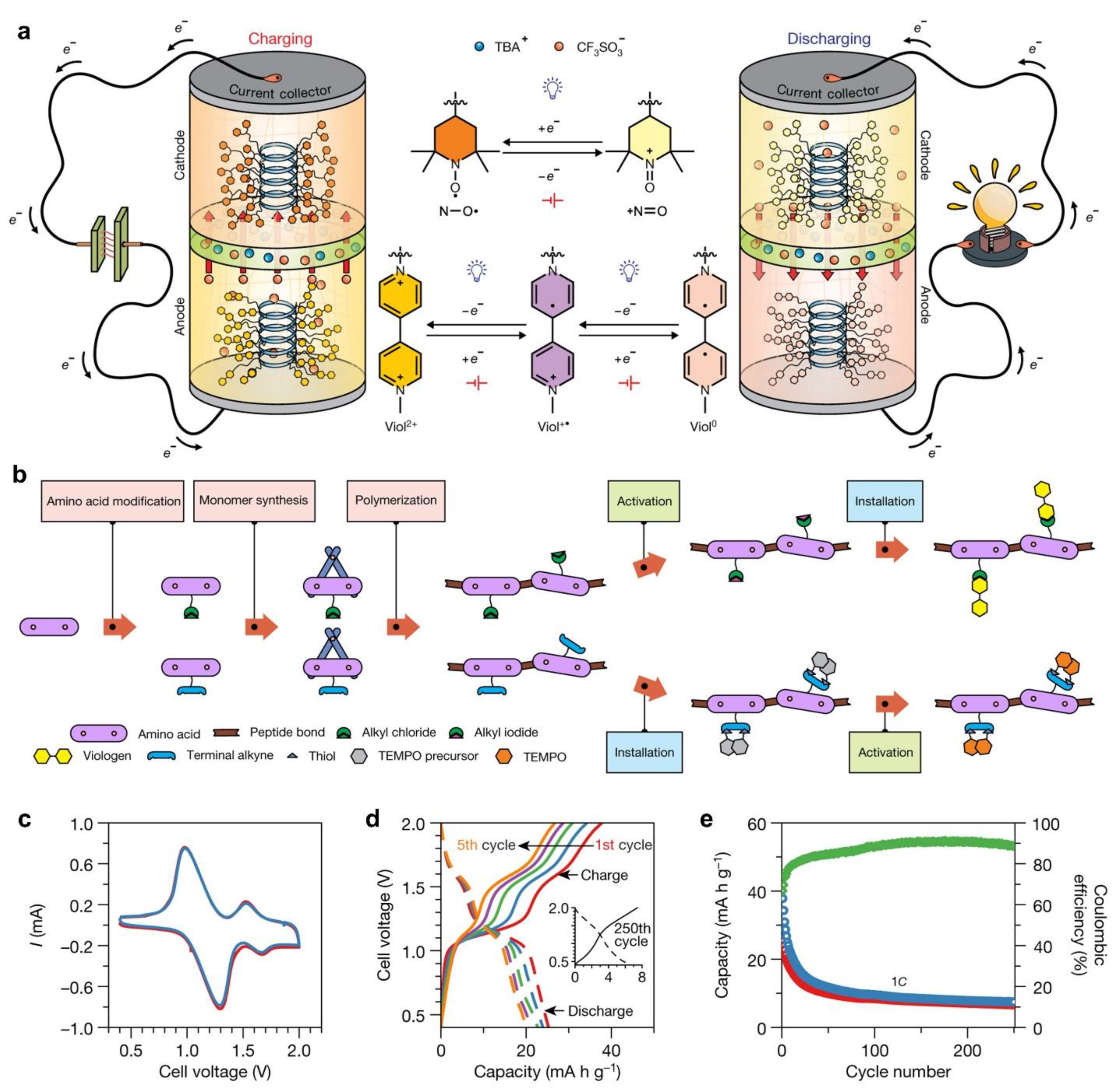
- Nguyen, T.P.; Easley, A.D.; Kang, N.; Khan, S.; Lim, S.M.; Rezenom, Y.H.; Wang, S.; Tran, D.K.; Fan, J.; Letteri, R.A.; He, X.; Su, L.; Yu, C.H.; Lutkenhaus, J.L.; Wooley, K.L. Polypeptide Organic Radical Batteries. Nature 2021, 593, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Janoschka, T.; Hager, M.D.; Schubert, U.S. Powering up the Future: Radical Polymers for Battery Applications. Adv. Mater. 2012, 24, 6397–6409. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Monteiro, M.J.; Jia, Z. Stable Organic Radical Polymers: Synthesis and Applications. Polymer Chemistry The Royal Society of Chemistry September 13. 2016, 5589–5614. [Google Scholar] [CrossRef]
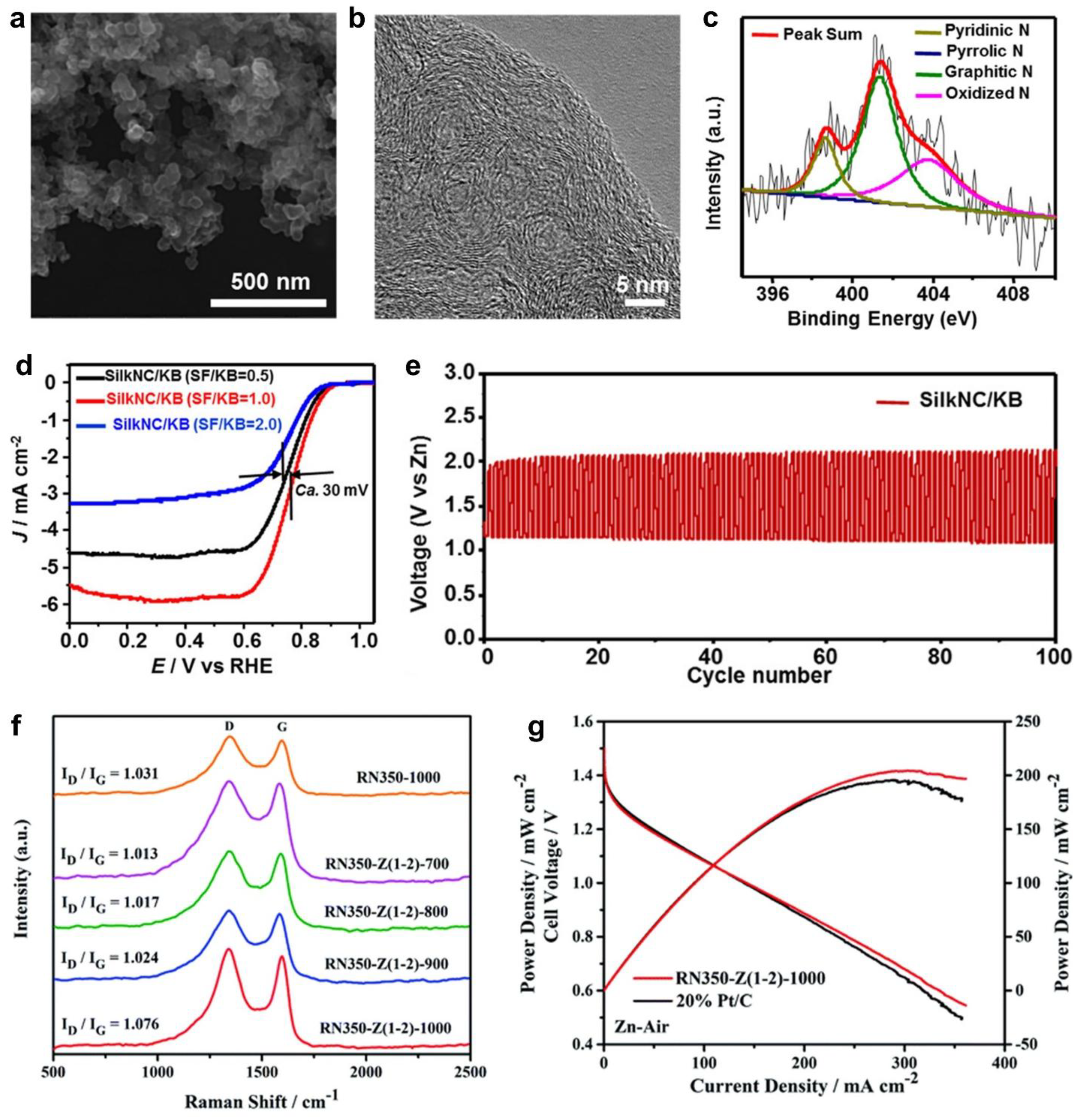
| Silk | Defect-rich and N-doped carbon | Zn-air | Voltage gap = 1.39 V, voltaic efficiency = 41.3% after 100 cycles | 6.0 M KOH + 0.2 M ZnAC, PVA aqueous gel | Zn | [20] |
| Root nodule | Fe, Mo, S, N self-doped porous carbon | Zn-air | Half-wave potentials are 0.723 V (vs. RHE) in 0.1 M HClO4 and 0.868 V and 0.1 M KOH solution | 6 M KOH + 0.2 M Zn(CH3CO2)2 | Zn | [21] |
| Fuel cell | Flow rate of H2 was 10 mL min-1, 17.6 mW cm-2 with an open-circuit voltage of 0.966 V | H2 | air | Graphite | |||
| Silk | Carbon fabrics | All-vanadium redox flow batteries | Energy efficiency = 86.8% | 1.6 M VOSO4 in 4 M H2SO4 solution | Symmetrical | [22] |
| Twin cocoon | Self-standing monolithic carbon | All-vanadium redox flow batteries | 50% redox potential decrease & 192% diffusion slope increase | 1.0 M VOSO4 + 3.0 M H2SO4 | Pt, Ag/AgCl | [23] |
| Pyroprotein | Carbon felts@ pyroprotein | All-vanadium redox flow batteries | ΔEp = 0.17 V, energy efficiency = 90% at 40 mA cm-2 | 0.1 M VOSO4 in 2 M H2SO4 | Three electrodes | [24] |
| Zein | Zein-coated carbon black | All-vanadium redox flow batteries | Energy efficiency =85.2% after 100th cycles | Positive: 2 M VOSO4 in 3 M H2SO4 Negative: 2 M VOSO4 in 3 M H2SO4 |
Symmetrical | [25] |
| Protein type | Carbon type | system type | Electrochemical performance | Electrolyte | Counter Electrode | Reference |
| Pig blood | 2D Zn-Fe single-atom porous carbon catalyst | Zn-air | 220 mW cm-2 | / | 6.0 M KOH | Zn | [26] |
| AEMFCs | 352 mW cm-2 | FAA-3-20 (coating toward cathode) | H2/O2 | Symmetrical gas diffusion layer | |||
| Bovine serum albumin | Protein-coated MoS2/Gr nanosheet | Zn-air | 130 W h kg−1, OCV = 1.4 V |
Whatman filter paper | 4 M KOH | Zn | [27] |
| Soybean | NiFe-LDH nanowalls anchored on Fe-N-C matrix | Zn-air | OER (Ej=10 = 1.53 V vs. RHE) and ORR (E1/2 = 0.91 V vs. RHE) | / | 6.0 M KOH + 0.2 M ZnAC | Zn | [28] |
| Cyt c | bi-protein PQQ-GDH/cyt c signal chain | Supercapacitor/biofuel cell hybrid device | 4.5 μW cm−2, 80% residual activity after 50 pulses | / | Air-saturated 10 mM NaH2PO4 | Bioanode (PQQ-GDH-cyt c-GP), biocathode (BOx-GP-cyt c) | [29] |
| Protein type | Electrode | Systems | Electrochemical performance | Separator | Electrolyte | Counter Electrode | Reference |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




