Submitted:
15 July 2025
Posted:
16 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Role of Antibodies in Emerging and Re-Emerging Infections
2.1. Protective Role of Antibodies in Infectious Diseases
2.1.1. Neutralization
2.1.2. Activation of the Complement System
2.1.3. Antibody-Mediated Phagocytosis
2.1.4. Antibody-Dependent Cellular Cytoxicity (ADCC)
2.2. Pathogenic Role of Antibodies in Infectious Diseases
2.2.1. Autoimmunity and Autoantibodies
2.2.2. Antibody-Dependent Enhancement (ADE)
2.2.3. Non-Neutralizing Antibodies (NNAbs)
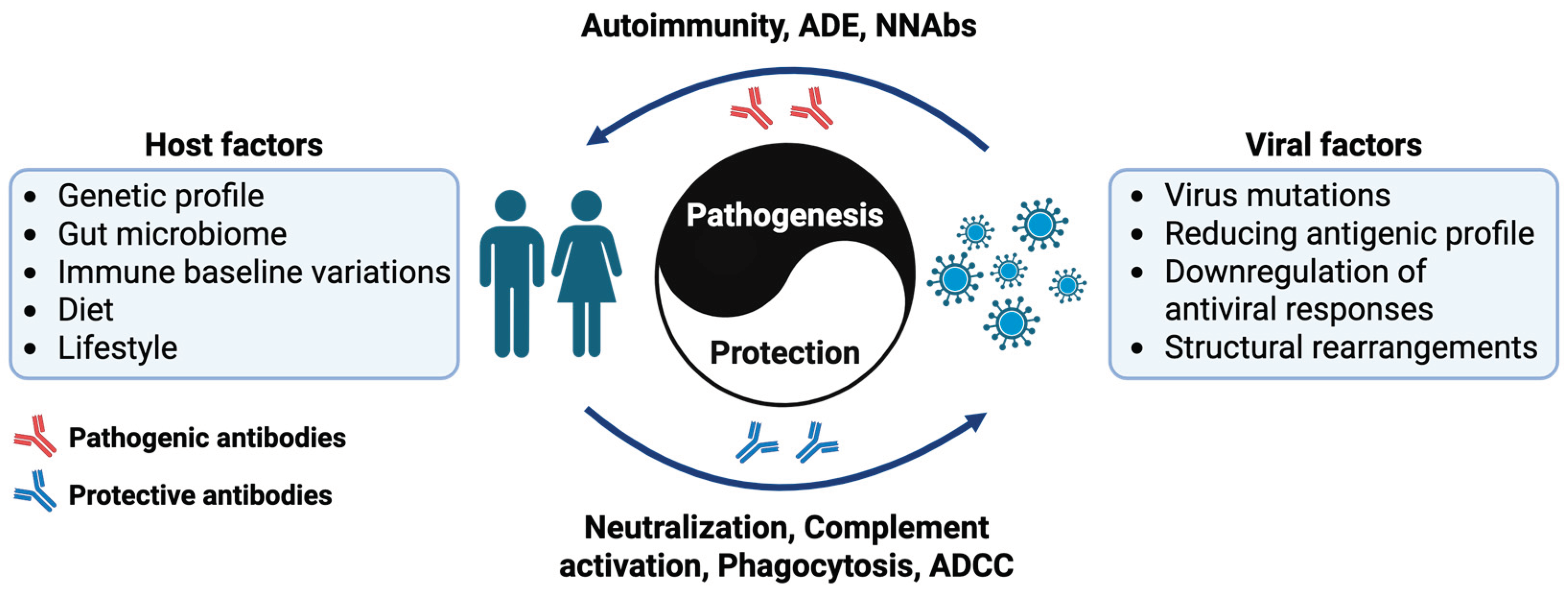
3. Factors Influencing Antibody-Mediated Immunity
3.1. Viral-Based Factors
3.2. Host-Based Factors
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADCC | Antibody-Dependent Cellular Cytotoxicity |
| ADCP | Antibody-Dependent Cellular Phagocytosis |
| ADE | Antibody-Dependent Enhancement |
| ANO2 | Anoctamin-2 |
| APTT | Activated Partial Thromboplastin Time |
| BGLF5 | EBV Ribonuclease Protein |
| CD107 | Lysosomal-Associated Membrane Protein 1 |
| CD16a | FcγRIIIa |
| CD16b | FcγRIIIb |
| CD32a | FcγRIIa |
| CD32b | FcγRIIb |
| CD32c | FcγRIIc |
| CD64 | FcγRI |
| CMV | Cytomegalovirus |
| CRP | C-Reactive Protein |
| CRYAB | Alpha-B Crystallin |
| CSR | Class Switching Recombination |
| CXCR3 | C-X-C Chemokine Receptor Type 3 |
| DENV | Dengue Virus |
| dsDNA | Double-Stranded DNA |
| EBNA1 | Epstein-Barr Nuclear Antigen 1 |
| EBV | Epstein-Barr Virus |
| FcγR | Fc Gamma Receptor |
| FcγRI | Fc Gamma Receptor I |
| FcγRIIa | Fc Gamma Receptor IIa |
| FcγRIIb | Fc Gamma Receptor IIb |
| FcγRIIc | Fc Gamma Receptor IIc |
| FcγRIIIa | Fc Gamma Receptor IIIa |
| FcγRIIIb | Fc Gamma Receptor IIIb |
| FcRs | Fc Receptors |
| GBS | Guillain-Barré Syndrome |
| GlialCAM | Glial Cell Adhesion Molecule |
| gp120 | Glycoprotein 120 |
| HA | Hemagglutinin |
| HA2 | Hemagglutinin Subunit 2 |
| HCV | Hepatitis C Virus |
| HCMV | Human Cytomegalovirus |
| HIV | Human Immunodeficiency Virus |
| HLA | Human Leukocyte Antigen |
| HSV | Herpes Simplex Virus |
| IFN-γ | Interferon-Gamma |
| IgA | Immunoglobulin A |
| IgD | Immunoglobulin D |
| IgE | Immunoglobulin E |
| IgG | Immunoglobulin G |
| IgM | Immunoglobulin M |
| ITAM | Immunoreceptor Tyrosine-Based Activation Motif |
| ITIM | Immunoreceptor Tyrosine-Based Inhibitory Motif |
| JEV | Japanese Encephalitis Virus |
| MBP | Myelin Basic Protein |
| MBCs | Memory B Cells |
| MIS-C | Multisystem Inflammatory Syndrome in Children |
| MS | Multiple Sclerosis |
| NNAbs | Non-Neutralizing Antibodies |
| NK | Natural Killer |
| PD-1 | Programmed Cell Death Protein 1 |
| PT | Prothrombin Time |
| RA | Rheumatoid Arthritis |
| RSV | Respiratory Syncytial Virus |
| SARS-CoV-2 | Severe Acute Respiratory Syndrome Coronavirus 2 |
| SHM | Somatic Hypermutation |
| SLE | Systemic Lupus Erythematosus |
| SNX8 | Sorting Nexin 8 |
| STAT1 | Signal Transducer and Activator of Transcription 1 |
| TIGIT | T Cell Immunoreceptor with Ig and ITIM Domains |
| WNV | West Nile Virus |
| ZIKV | Zika Virus |
References
- Bonilla, F.A.; Oettgen, H.C. Adaptive immunity. J Allergy Clin Immunol 2010, 125, S33–40. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.K. B Cells. In Basics of Hematopoietic Stem Cell Transplant; Springer: 2023; pp. 87-120.
- Stavnezer, J.; Schrader, C.E. IgH chain class switch recombination: mechanism and regulation. J Immunol 2014, 193, 5370–5378. [Google Scholar] [CrossRef] [PubMed]
- Nothelfer, K.; Sansonetti, P.J.; Phalipon, A. Pathogen manipulation of B cells: the best defence is a good offence. Nat Rev Microbiol 2015, 13, 173–184. [Google Scholar] [CrossRef] [PubMed]
- Doria-Rose, N.A.; Joyce, M.G. Strategies to guide the antibody affinity maturation process. Curr Opin Virol 2015, 11, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Pollard, A.J.; Bijker, E.M. A guide to vaccinology: from basic principles to new developments. Nat Rev Immunol 2021, 21, 83–100. [Google Scholar] [CrossRef] [PubMed]
- Dempsey, L.A. Pathogenic antibodies. Nat Immunol 2019, 20, 1414. [Google Scholar] [CrossRef]
- Lu, R.M.; Hwang, Y.C.; Liu, I.J.; Lee, C.C.; Tsai, H.Z.; Li, H.J.; Wu, H.C. Development of therapeutic antibodies for the treatment of diseases. J Biomed Sci 2020, 27, 1. [Google Scholar] [CrossRef] [PubMed]
- Slifka, M.K.; Amanna, I.J. Role of Multivalency and Antigenic Threshold in Generating Protective Antibody Responses. Frontiers in Immunology 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Klasse, P.J.; Sattentau, Q.J. Occupancy and mechanism in antibody-mediated neutralization of animal viruses. J Gen Virol 2002, 83, 2091–2108. [Google Scholar] [CrossRef] [PubMed]
- Pantaleo, G.; Correia, B.; Fenwick, C.; Joo, V.S.; Perez, L. Antibodies to combat viral infections: development strategies and progress. Nature Reviews Drug Discovery 2022, 21, 676–696. [Google Scholar] [CrossRef] [PubMed]
- Burton, D.R. Antiviral neutralizing antibodies: from in vitro to in vivo activity. Nat Rev Immunol 2023, 23, 720–734. [Google Scholar] [CrossRef] [PubMed]
- Klasse, P.J. Neutralization of Virus Infectivity by Antibodies: Old Problems in New Perspectives. Advances in Biology 2014, 2014, 157895. [Google Scholar] [CrossRef] [PubMed]
- Boero, E.; Gorham, R.D., Jr.; Francis, E.A.; Brand, J.; Teng, L.H.; Doorduijn, D.J.; Ruyken, M.; Muts, R.M.; Lehmann, C.; Verschoor, A.; et al. Purified complement C3b triggers phagocytosis and activation of human neutrophils via complement receptor 1. Sci Rep 2023, 13, 274. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.; Smatti, M.K.; Ouhtit, A.; Cyprian, F.S.; Almaslamani, M.A.; Thani, A.A.; Yassine, H.M. Antibody-Dependent Enhancement (ADE) and the role of complement system in disease pathogenesis. Mol Immunol 2022, 152, 172–182. [Google Scholar] [CrossRef] [PubMed]
- Ricklin, D.; Hajishengallis, G.; Yang, K.; Lambris, J.D. Complement: a key system for immune surveillance and homeostasis. Nature Immunology 2010, 11, 785–797. [Google Scholar] [CrossRef] [PubMed]
- Byrne, A.B.; Bonnin, F.A.; López, E.L.; Polack, F.P.; Talarico, L.B. C1q modulation of antibody-dependent enhancement of dengue virus infection in human myeloid cell lines is dependent on cell type and antibody specificity. Microbes and Infection 2024, 26, 105378. [Google Scholar] [CrossRef] [PubMed]
- Byrne, A.B.; Talarico, L.B. Role of the complement system in antibody-dependent enhancement of flavivirus infections. Int J Infect Dis 2021, 103, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Castro-Dopico, T.; Clatworthy, M.R. IgG and Fcγ Receptors in Intestinal Immunity and Inflammation. Front Immunol 2019, 10, 805. [Google Scholar] [CrossRef] [PubMed]
- Lamptey, H.; Bonney, E.Y.; Adu, B.; Kyei, G.B. Are Fc Gamma Receptor Polymorphisms Important in HIV-1 Infection Outcomes and Latent Reservoir Size? Front Immunol 2021, 12, 656894. [Google Scholar] [CrossRef] [PubMed]
- Ravetch, J.V.; Bolland, S. IgG fc receptors. Annual review of immunology 2001, 19, 275–290. [Google Scholar] [CrossRef] [PubMed]
- van der Poel, C.E.; Spaapen, R.M.; van de Winkel, J.G.J.; Leusen, J.H.W. Functional Characteristics of the High Affinity IgG Receptor, FcγRI. The Journal of Immunology 2011, 186, 2699–2704. [Google Scholar] [CrossRef] [PubMed]
- Hargraves, M.M. Discovery of the LE cell and its morphology. Mayo Clin Proc 1969, 44, 579–599. [Google Scholar] [PubMed]
- Waaler, E. On the occurrence of a factor in human serum activating the specific agglutintion of sheep blood corpuscles. 1939. Apmis 2007, 115, 422–438. [Google Scholar] [CrossRef] [PubMed]
- Davies, D.J.; Moran, J.E.; Niall, J.F.; Ryan, G.B. Segmental necrotising glomerulonephritis with antineutrophil antibody: possible arbovirus aetiology? Br Med J (Clin Res Ed) 1982, 285, 606. [Google Scholar] [CrossRef] [PubMed]
- Seeling, M.; Pöhnl, M.; Kara, S.; Horstmann, N.; Riemer, C.; Wöhner, M.; Liang, C.; Brückner, C.; Eiring, P.; Werner, A.; et al. Immunoglobulin G-dependent inhibition of inflammatory bone remodeling requires pattern recognition receptor Dectin-1. Immunity 2023, 56, 1046–1063.e1047. [Google Scholar] [CrossRef] [PubMed]
- Vattepu, R.; Sneed, S.L.; Anthony, R.M. Sialylation as an Important Regulator of Antibody Function. Front Immunol 2022, 13, 818736. [Google Scholar] [CrossRef] [PubMed]
- Irvine, E.B.; Alter, G. Understanding the role of antibody glycosylation through the lens of severe viral and bacterial diseases. Glycobiology 2020, 30, 241–253. [Google Scholar] [CrossRef] [PubMed]
- Gómez Román, V.R.; Murray, J.C.; Weiner, L.M. Chapter 1 - Antibody-Dependent Cellular Cytotoxicity (ADCC). In Antibody Fc, Ackerman, M.E., Nimmerjahn, F., Eds.; Academic Press: Boston, 2014; pp. 1–27. [Google Scholar]
- Niu, Y.-X.; Xu, Z.-X.; Yu, L.-F.; Lu, Y.-P.; Wang, Y.; Wu, C.; Hou, Y.-B.; Li, J.-N.; Huang, S.; Song, X.; et al. Advances of research of Fc-fusion protein that activate NK cells for tumor immunotherapy. International Immunopharmacology 2022, 109, 108783. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, U.; Chakrabarti, A.K.; Kanungo, S.; Dutta, S. Evolutionary dynamics of influenza A/H1N1 virus circulating in India from 2011 to 2021. Infection, Genetics and Evolution 2023, 110, 105424. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Tan, G.S.; Mullarkey, C.E.; Lee, A.J.; Lam, M.M.W.; Krammer, F.; Henry, C.; Wilson, P.C.; Ashkar, A.A.; Palese, P.; et al. Epitope specificity plays a critical role in regulating antibody-dependent cell-mediated cytotoxicity against influenza A virus. Proceedings of the National Academy of Sciences 2016, 113, 11931–11936. [Google Scholar] [CrossRef] [PubMed]
- Muralidharan, A.; Gravel, C.; Harris, G.; Hashem, A.M.; Zhang, W.; Safronetz, D.; Van Domselaar, G.; Krammer, F.; Sauve, S.; Rosu-Myles, M.; et al. Universal antibody targeting the highly conserved fusion peptide provides cross-protection in mice. Hum Vaccin Immunother 2022, 18, 2083428. [Google Scholar] [CrossRef] [PubMed]
- de Taeye, S.W.; Schriek, A.I.; Umotoy, J.C.; Grobben, M.; Burger, J.A.; Sanders, R.W.; Vidarsson, G.; Wuhrer, M.; Falck, D.; Kootstra, N.A.; et al. Afucosylated broadly neutralizing antibodies enhance clearance of HIV-1 infected cells through cell-mediated killing. Communications Biology 2024, 7, 964. [Google Scholar] [CrossRef] [PubMed]
- Nimmerjahn, F. Role of Antibody Glycosylation in Health, Disease, and Therapy. Handb Exp Pharmacol 2025. [Google Scholar] [CrossRef]
- Sundaresan, B.; Shirafkan, F.; Ripperger, K.; Rattay, K. The Role of Viral Infections in the Onset of Autoimmune Diseases. Viruses 2023, 15. [Google Scholar] [CrossRef] [PubMed]
- Bastard, P.; Rosen, L.B.; Zhang, Q.; Michailidis, E.; Hoffmann, H.-H.; Zhang, Y.; Dorgham, K.; Philippot, Q.; Rosain, J.; Béziat, V.; et al. Autoantibodies against type I IFNs in patients with life-threatening COVID-19. Science 2020, 370, eabd4585. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.-K.; Yeo, K.-J.; Chang, S.-H.; Liao, T.-L.; Chou, C.-H.; Lan, J.-L.; Chang, C.-K.; Chen, D.-Y. The detectable anti-interferon-γ autoantibodies in COVID-19 patients may be associated with disease severity. Virology Journal 2023, 20, 33. [Google Scholar] [CrossRef] [PubMed]
- Lanz, T.V.; Brewer, R.C.; Ho, P.P.; Moon, J.S.; Jude, K.M.; Fernandez, D.; Fernandes, R.A.; Gomez, A.M.; Nadj, G.S.; Bartley, C.M.; et al. Clonally expanded B cells in multiple sclerosis bind EBV EBNA1 and GlialCAM. Nature 2022, 603, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Tengvall, K.; Huang, J.; Hellström, C.; Kammer, P.; Biström, M.; Ayoglu, B.; Lima Bomfim, I.; Stridh, P.; Butt, J.; Brenner, N.; et al. Molecular mimicry between Anoctamin 2 and Epstein-Barr virus nuclear antigen 1 associates with multiple sclerosis risk. Proc Natl Acad Sci U S A 2019, 116, 16955–16960. [Google Scholar] [CrossRef] [PubMed]
- Thomas, O.G.; Bronge, M.; Tengvall, K.; Akpinar, B.; Nilsson, O.B.; Holmgren, E.; Hessa, T.; Gafvelin, G.; Khademi, M.; Alfredsson, L.; et al. Cross-reactive EBNA1 immunity targets alpha-crystallin B and is associated with multiple sclerosis. Sci Adv 2023, 9, eadg3032. [Google Scholar] [CrossRef] [PubMed]
- Jog, N.R.; McClain, M.T.; Heinlen, L.D.; Gross, T.; Towner, R.; Guthridge, J.M.; Axtell, R.C.; Pardo, G.; Harley, J.B.; James, J.A. Epstein Barr virus nuclear antigen 1 (EBNA-1) peptides recognized by adult multiple sclerosis patient sera induce neurologic symptoms in a murine model. J Autoimmun 2020, 106, 102332. [Google Scholar] [CrossRef] [PubMed]
- Bodansky, A.; Mettelman, R.C.; Sabatino, J.J., Jr.; Vazquez, S.E.; Chou, J.; Novak, T.; Moffitt, K.L.; Miller, H.S.; Kung, A.F.; Rackaityte, E.; et al. Molecular mimicry in multisystem inflammatory syndrome in children. Nature 2024, 632, 622–629. [Google Scholar] [CrossRef] [PubMed]
- Zhou, F.; Xu, Y.; Liu, X.; Xu, Y.; Wang, Y.; Jiang, D.; Du, P. Zika and Dengue Virus Autoimmunity: An Overview of Related Disorders and Their Potential Mechanisms. Rev Med Virol 2025, 35, e70014. [Google Scholar] [CrossRef] [PubMed]
- Cao-Lormeau, V.M.; Blake, A.; Mons, S.; Lastère, S.; Roche, C.; Vanhomwegen, J.; Dub, T.; Baudouin, L.; Teissier, A.; Larre, P.; et al. Guillain-Barré Syndrome outbreak associated with Zika virus infection in French Polynesia: a case-control study. Lancet 2016, 387, 1531–1539. [Google Scholar] [CrossRef] [PubMed]
- Pisetsky, D.S. Pathogenesis of autoimmune disease. Nature Reviews Nephrology 2023, 19, 509–524. [Google Scholar] [CrossRef]
- Jiang, W.; Johnson, D.; Adekunle, R.; Heather, H.; Xu, W.; Cong, X.; Wu, X.; Fan, H.; Andersson, L.M.; Robertson, J.; et al. COVID-19 is associated with bystander polyclonal autoreactive B cell activation as reflected by a broad autoantibody production, but none is linked to disease severity. J Med Virol 2023, 95, e28134. [Google Scholar] [CrossRef] [PubMed]
- Gregoire, C.; Spinelli, L.; Villazala-Merino, S.; Gil, L.; Holgado, M.P.; Moussa, M.; Dong, C.; Zarubica, A.; Fallet, M.; Navarro, J.M.; et al. Viral infection engenders bona fide and bystander subsets of lung-resident memory B cells through a permissive mechanism. Immunity 2022, 55, 1216–1233.e1219. [Google Scholar] [CrossRef] [PubMed]
- Priora, M.; Borrelli, R.; Parisi, S.; Ditto, M.C.; Realmuto, C.; Laganà, A.; Centanaro Di Vittorio, C.; Degiovanni, R.; Peroni, C.L.; Fusaro, E. Autoantibodies and rheumatologic manifestations in hepatitis C virus infection. Biology 2021, 10, 1071. [Google Scholar] [CrossRef] [PubMed]
- Sattarnezhad, N.; Kockum, I.; Thomas, O.G.; Liu, Y.; Ho, P.P.; Barrett, A.K.; Comanescu, A.I.; Wijeratne, T.U.; Utz, P.J.; Alfredsson, L.; et al. Antibody reactivity against EBNA1 and GlialCAM differentiates multiple sclerosis patients from healthy controls. Proc Natl Acad Sci U S A 2025, 122, e2424986122. [Google Scholar] [CrossRef] [PubMed]
- Sundar, K.; Jacques, S.; Gottlieb, P.; Villars, R.; Benito, M.-E.; Taylor, D.K.; Spatz, L.A. Expression of the Epstein-Barr virus nuclear antigen-1 (EBNA-1) in the mouse can elicit the production of anti-dsDNA and anti-Sm antibodies. Journal of Autoimmunity 2004, 23, 127–140. [Google Scholar] [CrossRef] [PubMed]
- Halstead, S.B.; O'Rourke, E.J.; Allison, A.C. Dengue viruses and mononuclear phagocytes. II. Identity of blood and tissue leukocytes supporting in vitro infection. J Exp Med 1977, 146, 218–229. [Google Scholar] [CrossRef] [PubMed]
- Narayan, R.; Tripathi, S. Intrinsic ADE: The Dark Side of Antibody Dependent Enhancement During Dengue Infection. Front Cell Infect Microbiol 2020, 10, 580096. [Google Scholar] [CrossRef] [PubMed]
- Edgar, J.E.; Bournazos, S. Fc-FcγR interactions during infections: From neutralizing antibodies to antibody-dependent enhancement. Immunological Reviews 2024. [Google Scholar] [CrossRef] [PubMed]
- Marisa, S.F.; Afsar, N.S.; Sami, C.A.; Hasan, M.I.; Ali, N.; Rahman, M.M.; Mohona, S.Q.; Ahmed, R.U.; Ehsan, A.; Baset, F. Dengue Myocarditis: A Retrospective Study From 2019 to 2023 in a Tertiary Care Hospital in Bangladesh. Cureus 2025, 17, e84371. [Google Scholar] [CrossRef] [PubMed]
- Anderson, K.B.; Gibbons, R.V.; Thomas, S.J.; Rothman, A.L.; Nisalak, A.; Berkelman, R.L.; Libraty, D.H.; Endy, T.P. Preexisting Japanese encephalitis virus neutralizing antibodies and increased symptomatic dengue illness in a school-based cohort in Thailand. PLoS Negl Trop Dis 2011, 5, e1311. [Google Scholar] [CrossRef] [PubMed]
- Saito, Y.; Moi, M.L.; Takeshita, N.; Lim, C.K.; Shiba, H.; Hosono, K.; Saijo, M.; Kurane, I.; Takasaki, T. Japanese encephalitis vaccine-facilitated dengue virus infection-enhancement antibody in adults. BMC Infect Dis 2016, 16, 578. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Xu, Y.; Zhao, F.; Tarbe, M.; Zhou, S.; Wang, W.; Zhang, S.; Zhang, W.; Xu, Q.; Shi, L.; et al. The pre-existing cellular immunity to Japanese encephalitis virus heterotypically protects mice from Zika virus infection. Sci Bull (Beijing) 2020, 65, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Shukla, R.; Beesetti, H.; Brown, J.A.; Ahuja, R.; Ramasamy, V.; Shanmugam, R.K.; Poddar, A.; Batra, G.; Krammer, F.; Lim, J.K.; et al. Dengue and Zika virus infections are enhanced by live attenuated dengue vaccine but not by recombinant DSV4 vaccine candidate in mouse models. eBioMedicine 2020, 60. [Google Scholar] [CrossRef] [PubMed]
- Garg, H.; Yeh, R.; Watts, D.M.; Mehmetoglu-Gurbuz, T.; Resendes, R.; Parsons, B.; Gonzales, F.; Joshi, A. Enhancement of Zika virus infection by antibodies from West Nile virus seropositive individuals with no history of clinical infection. BMC Immunol 2021, 22, 5. [Google Scholar] [CrossRef] [PubMed]
- Wen, J.; Cheng, Y.; Ling, R.; Dai, Y.; Huang, B.; Huang, W.; Zhang, S.; Jiang, Y. Antibody-dependent enhancement of coronavirus. Int J Infect Dis 2020, 100, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Krilov, L.R.; Anderson, L.J.; Marcoux, L.; Bonagura, V.R.; Wedgwood, J.F. Antibody-mediated enhancement of respiratory syncytial virus infection in two monocyte/macrophage cell lines. J Infect Dis 1989, 160, 777–782. [Google Scholar] [CrossRef] [PubMed]
- Polack, F.P. Atypical measles and enhanced respiratory syncytial virus disease (ERD) made simple. Pediatr Res 2007, 62, 111–115. [Google Scholar] [CrossRef] [PubMed]
- Kuzmina, N.A.; Younan, P.; Gilchuk, P.; Santos, R.I.; Flyak, A.I.; Ilinykh, P.A.; Huang, K.; Lubaki, N.M.; Ramanathan, P.; Crowe, J.E., Jr.; et al. Antibody-Dependent Enhancement of Ebola Virus Infection by Human Antibodies Isolated from Survivors. Cell Rep 2018, 24, 1802–1815.e1805. [Google Scholar] [CrossRef] [PubMed]
- Lum, F.M.; Couderc, T.; Chia, B.S.; Ong, R.Y.; Her, Z.; Chow, A.; Leo, Y.S.; Kam, Y.W.; Rénia, L.; Lecuit, M.; et al. Antibody-mediated enhancement aggravates chikungunya virus infection and disease severity. Sci Rep 2018, 8, 1860. [Google Scholar] [CrossRef] [PubMed]
- Lidbury, B.A.; Mahalingam, S. Specific ablation of antiviral gene expression in macrophages by antibody-dependent enhancement of Ross River virus infection. J Virol 2000, 74, 8376–8381. [Google Scholar] [CrossRef] [PubMed]
- Saboowala, H. Exploring the recent findings on Virally induced Antibody Dependent Enhancement (ADE) and potential Mechanisms leading to this condition. 2022.
- Kotaki, T.; Nagai, Y.; Yamanaka, A.; Konishi, E.; Kameoka, M. Japanese Encephalitis DNA Vaccines with Epitope Modification Reduce the Induction of Cross-Reactive Antibodies against Dengue Virus and Antibody-Dependent Enhancement of Dengue Virus Infection. Vaccines (Basel) 2022, 10. [Google Scholar] [CrossRef] [PubMed]
- Tay, M.Z.; Wiehe, K.; Pollara, J. Antibody-Dependent Cellular Phagocytosis in Antiviral Immune Responses. Front Immunol 2019, 10, 332. [Google Scholar] [CrossRef] [PubMed]
- Mayr, L.M.; Su, B.; Moog, C. Non-Neutralizing Antibodies Directed against HIV and Their Functions. Front Immunol 2017, 8, 1590. [Google Scholar] [CrossRef] [PubMed]
- Zanella, I.; Degli Antoni, M.; Marchese, V.; Castelli, F.; Quiros-Roldan, E. Non-neutralizing antibodies: Deleterious or propitious during SARS-CoV-2 infection? Int Immunopharmacol 2022, 110, 108943. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Kam, Y.-W. Insights from Avian Influenza: A Review of Its Multifaceted Nature and Future Pandemic Preparedness. Viruses 2024, 16, 458. [Google Scholar] [CrossRef] [PubMed]
- Burton, D.R.; Poignard, P.; Stanfield, R.L.; Wilson, I.A. Broadly Neutralizing Antibodies Present New Prospects to Counter Highly Antigenically Diverse Viruses. Science 2012, 337, 183–186. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Kong, W.; Yang, L.; Ding, Y.; Cui, H. Immunity and Immune Evasion Mechanisms of Epstein-Barr Virus. Viral Immunol 2023, 36, 303–317. [Google Scholar] [CrossRef] [PubMed]
- Jenks, J.A.; Goodwin, M.L.; Permar, S.R. The roles of host and viral antibody fc receptors in herpes simplex virus (HSV) and human cytomegalovirus (HCMV) infections and immunity. Frontiers in immunology 2019, 10, 2110. [Google Scholar] [CrossRef] [PubMed]
- Crispin, M.; Ward, A.B.; Wilson, I.A. Structure and immune recognition of the HIV glycan shield. Annual review of biophysics 2018, 47, 499–523. [Google Scholar] [CrossRef] [PubMed]
- Walls, A.C.; Park, Y.-J.; Tortorici, M.A.; Wall, A.; McGuire, A.T.; Veesler, D. Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell 2020, 181, 281–292.e286. [Google Scholar] [CrossRef] [PubMed]
- Engelbrecht, E.; Rodriguez, O.L.; Lees, W.; Vanwinkle, Z.; Shields, K.; Schultze, S.; Gibson, W.S.; Smith, D.R.; Jana, U.; Saha, S.; et al. Germline polymorphism in the immunoglobulin kappa and lambda loci explain variation in the expressed light chain antibody repertoire. bioRxiv 2025. [Google Scholar] [CrossRef] [PubMed]
- Posteraro, B.; Pastorino, R.; Di Giannantonio, P.; Ianuale, C.; Amore, R.; Ricciardi, W.; Boccia, S. The link between genetic variation and variability in vaccine responses: systematic review and meta-analyses. Vaccine 2014, 32, 1661–1669. [Google Scholar] [CrossRef] [PubMed]
- Garretson, T.A.; Liu, J.; Li, S.H.; Scher, G.; Santos, J.J.S.; Hogan, G.; Vieira, M.C.; Furey, C.; Atkinson, R.K.; Ye, N.; et al. Immune history shapes human antibody responses to H5N1 influenza viruses. Nat Med 2025, 31, 1454–1458. [Google Scholar] [CrossRef] [PubMed]
- Auladell, M.; Phuong, H.V.M.; Mai, L.T.Q.; Tseng, Y.Y.; Carolan, L.; Wilks, S.; Thai, P.Q.; Price, D.; Duong, N.T.; Hang, N.L.K.; et al. Influenza virus infection history shapes antibody responses to influenza vaccination. Nat Med 2022, 28, 363–372. [Google Scholar] [CrossRef] [PubMed]
- Lv, H.; Teo, Q.W.; Lee, C.C.; Liang, W.; Choi, D.; Mao, K.J.; Ardagh, M.R.; Gopal, A.B.; Mehta, A.; Szlembarski, M.; et al. Differential antigenic imprinting effects between influenza H1N1 hemagglutinin and neuraminidase in a mouse model. J Virol 2025, 99, e0169524. [Google Scholar] [CrossRef] [PubMed]
- Bigdelou, B.; Sepand, M.R.; Najafikhoshnoo, S.; Negrete, J.A.T.; Sharaf, M.; Ho, J.Q.; Sullivan, I.; Chauhan, P.; Etter, M.; Shekarian, T.; et al. COVID-19 and Preexisting Comorbidities: Risks, Synergies, and Clinical Outcomes. Frontiers in Immunology 2022, 13. [Google Scholar] [CrossRef] [PubMed]
- Nimmerjahn, F.; Ravetch, J.V. Fcγ receptors as regulators of immune responses. Nature Reviews Immunology 2008, 8, 34–47. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.; Lin, F.; Jiang, Z.; Tan, X.; Lin, X.; Liang, Z.; Xiao, C.; Xia, Y.; Guan, W.; Yang, Z.; et al. The impact of pre-existing influenza antibodies and inflammatory status on the influenza vaccine responses in older adults. Influenza Other Respir Viruses 2023, 17, e13172. [Google Scholar] [CrossRef] [PubMed]
- Rossouw, C.; Ryan, F.J.; Lynn, D.J. The role of the gut microbiota in regulating responses to vaccination: current knowledge and future directions. Febs j 2025, 292, 1480–1499. [Google Scholar] [CrossRef] [PubMed]
- Levy, M.; Kolodziejczyk, A.A.; Thaiss, C.A.; Elinav, E. Dysbiosis and the immune system. Nature Reviews Immunology 2017, 17, 219–232. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, K.A.; Lim, A.I. Maternal-driven immune education in offspring. Immunological Reviews 2024, 323, 288–302. [Google Scholar] [CrossRef] [PubMed]
- Visalli, G.; Laganà, A.; Lo Giudice, D.; Calimeri, S.; Caccamo, D.; Trainito, A.; Di Pietro, A.; Facciolà, A. Towards a Future of Personalized Vaccinology: Study on Individual Variables Influencing the Antibody Response to the COVID-19 Vaccine. Vaccines (Basel) 2023, 11. [Google Scholar] [CrossRef] [PubMed]
- Wrynla, X.H.; Bates, T.A.; Trank-Greene, M.; Wahedi, M.; Hinchliff, A.; Curlin, M.E.; Tafesse, F.G. Immune imprinting and vaccine interval determine antibody responses to monovalent XBB.1.5 COVID-19 vaccination. Commun Med (Lond) 2025, 5, 182. [Google Scholar] [CrossRef] [PubMed]
- Aranow, C. Vitamin D and the Immune System. Journal of Investigative Medicine 2011, 59, 881–886. [Google Scholar] [CrossRef] [PubMed]
- McSorley, H.J.; Maizels, R.M. Helminth infections and host immune regulation. Clin Microbiol Rev 2012, 25, 585–608. [Google Scholar] [CrossRef] [PubMed]
- Li, X.X.; Chen, J.X.; Wang, L.X.; Sun, J.; Chen, S.H.; Chen, J.H.; Zhang, X.Y.; Zhou, X.N. Profiling B and T cell immune responses to co-infection of Mycobacterium tuberculosis and hookworm in humans. Infect Dis Poverty 2015, 4, 20. [Google Scholar] [CrossRef] [PubMed]
- Rizk, S.S.; Moustafa, D.M.; ElBanna, S.A.; Nour El-Din, H.T.; Attia, A.S. Nanobodies in the fight against infectious diseases: repurposing nature's tiny weapons. World J Microbiol Biotechnol 2024, 40, 209. [Google Scholar] [CrossRef] [PubMed]
- Pirkalkhoran, S.; Grabowska, W.R.; Kashkoli, H.H.; Mirhassani, R.; Guiliano, D.; Dolphin, C.; Khalili, H. Bioengineering of Antibody Fragments: Challenges and Opportunities. Bioengineering (Basel) 2023, 10. [Google Scholar] [CrossRef] [PubMed]
- Wang, E.; Cohen, A.A.; Caldera, L.F.; Keeffe, J.R.; Rorick, A.V.; Adia, Y.M.; Gnanapragasam, P.N.P.; Bjorkman, P.J.; Chakraborty, A.K. Designed mosaic nanoparticles enhance cross-reactive immune responses in mice. Cell 2025, 188, 1036–1050.e1011. [Google Scholar] [CrossRef] [PubMed]
- Musnier, A.; Dumet, C.; Mitra, S.; Verdier, A.; Keskes, R.; Chassine, A.; Jullian, Y.; Cortes, M.; Corde, Y.; Omahdi, Z. Applying artificial intelligence to accelerate and de-risk antibody discovery. Frontiers in Drug Discovery 2024, 4, 1339697. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



