Submitted:
15 July 2025
Posted:
16 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
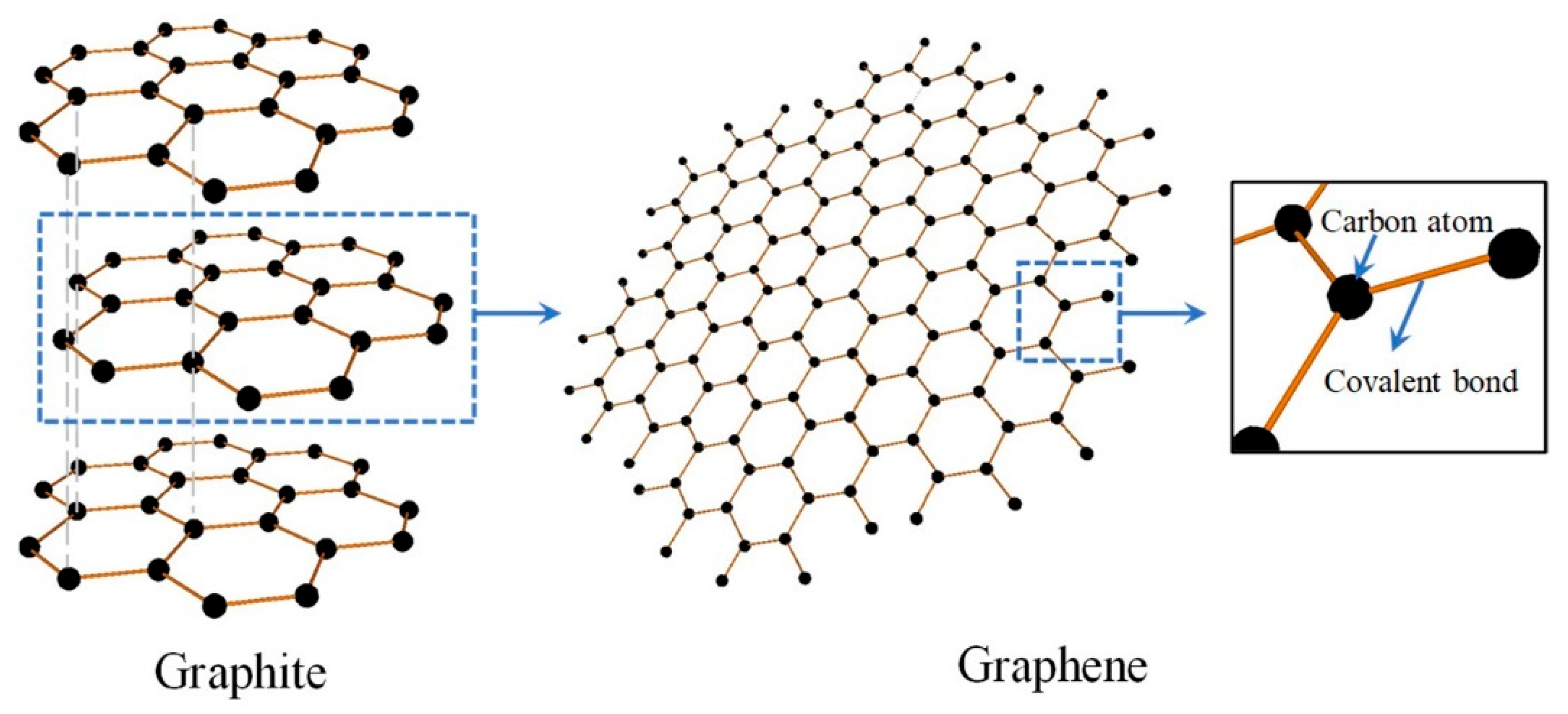
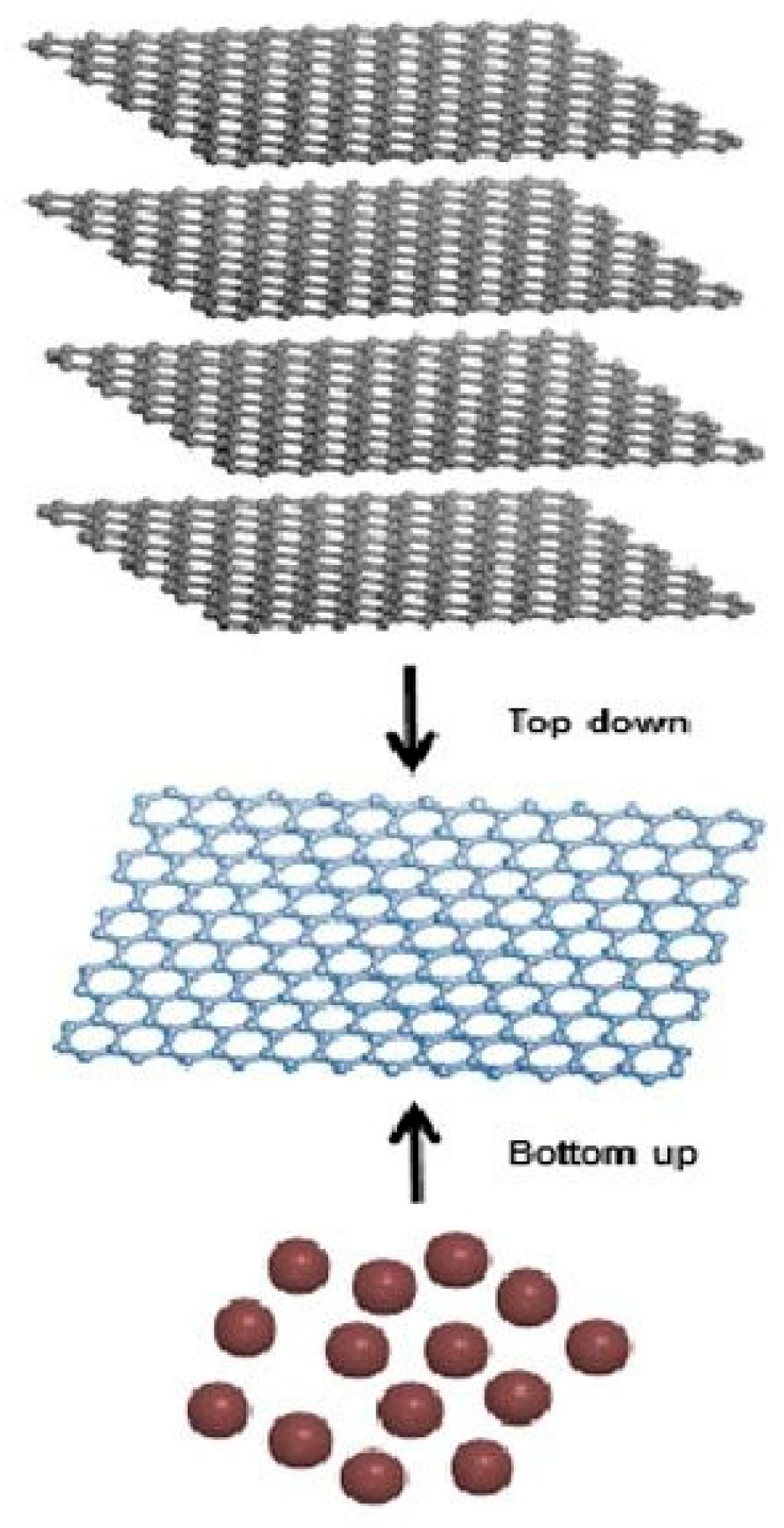
2. Graphene Synthesis
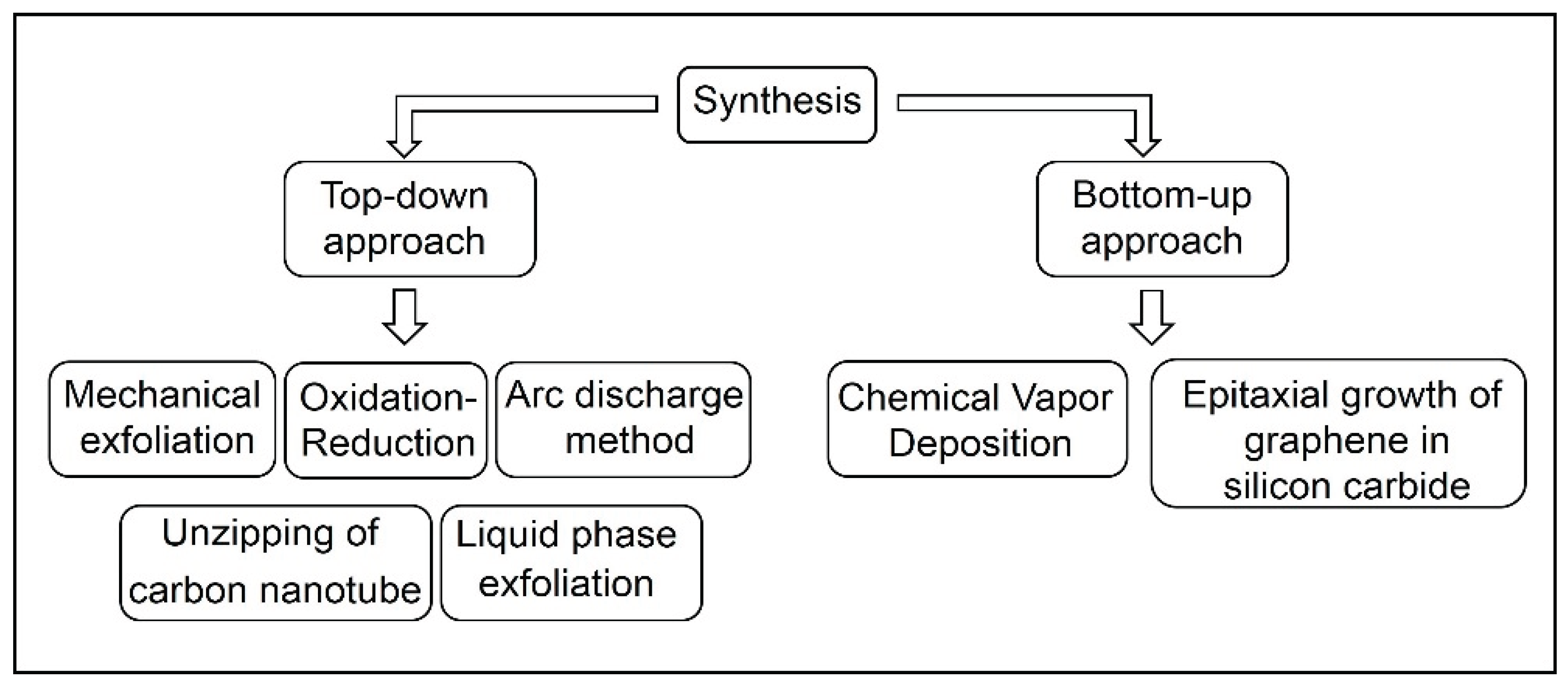
2.1. Top Down
2.1.1. Mechanical Exfoliation
Micromechanical Cleavage
Sonication
Ball Milling
Fluid Dynamics
Supercritical Fluids
Detonation Technique
2.2. Oxidation-Reduction
2.2.1. Oxidation Method
2.2.2. Chemical Oxidation
Thermal Oxidation
Electrochemical Oxidation
Reduction Method
Chemical Reduction
Thermal Reduction
Electrochemical Reduction
2.2.3. Arc Discharge Method
Effect of Buffer Gas Type
Effect of Current Type
Effects of Pressure
Effect of Reaction Temperature
Effect of Reaction Time
Effect of Chamber Type
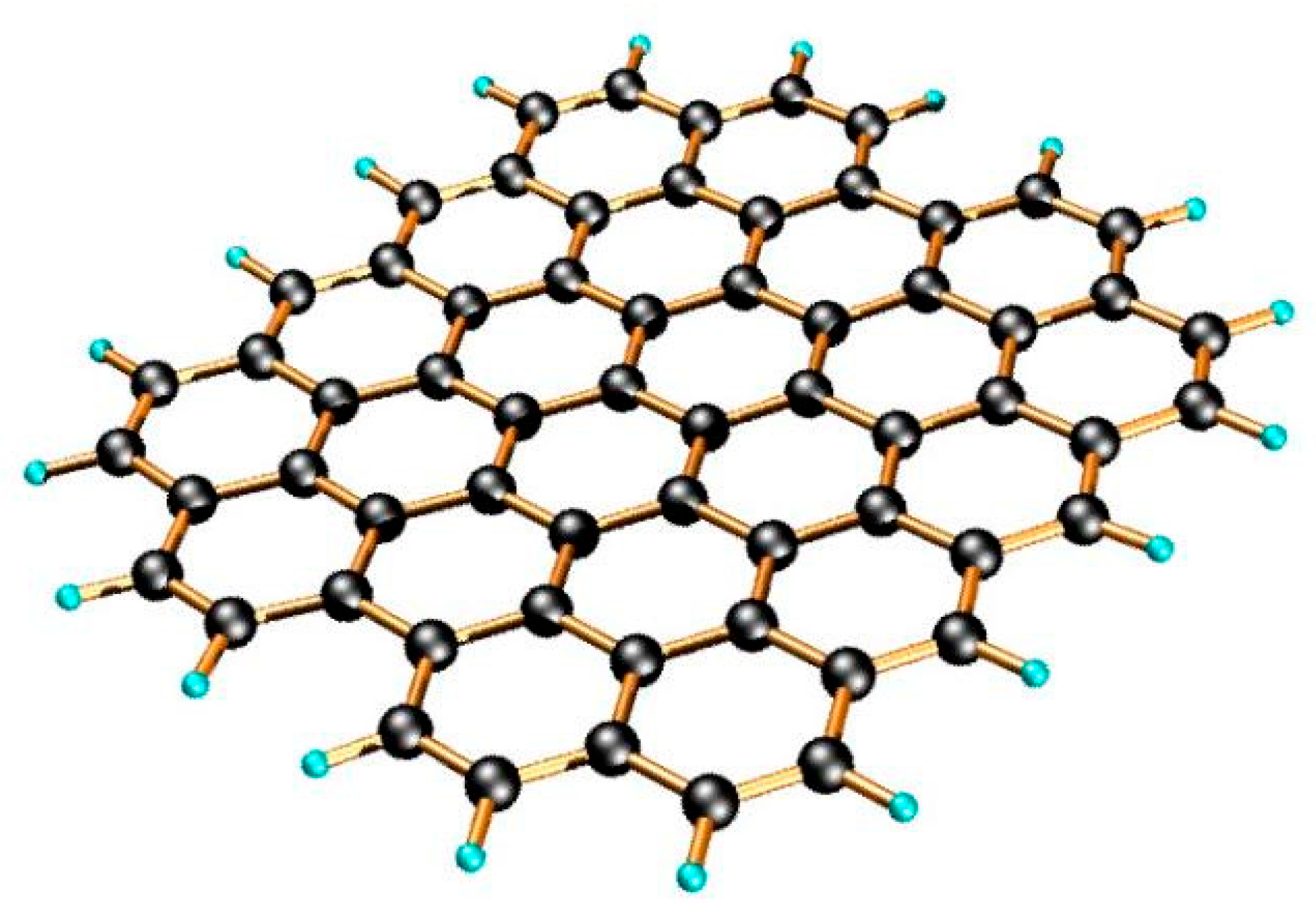
2.2.4. Unzipping of Carbon Nanotube
Oxidative Zipper Retraction
Catalytic Zipper Opening
Electrochemical Zipper Opening
Sonochemical Zipper Opening
2.2.5. Liquid Phase Exfoliation
Graphite Intercalation Compounds (GIC)
Chemical Exfoliation with Organic Solvents
Chemical Exfoliation with Ionic Liquid Exfoliation
Chemical Exfoliation with Surfactants
Exfoliate with Low Boiling Point Solvent
Exfoliation with Electrochemistry
2.3. Bottom up
2.3.1. Chemical Vapor Deposition (CVD)
Temperature
Pressure
Wall/Substrate
Deposition Time
Gas Flow State
Activated Manner
2.3.2. Epitaxial Growth of Graphene Silicon Carbide
Surface
Temperature
Pressure
Catalyst
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| PMMA | Polymethyl methacrylate |
| PDMS | Polydimethylsiloxane |
| FTIR | Fourier Transform Infrared |
| SEM | Scanning Electron Microscopy |
| XRD | X-ray Diffraction |
| AFM | Atomic Force Microscopy |
| TEM | Transmission Electron Microscopy |
| DMF | Dimethylformamide |
| NMP | N-Methyl-2-pyrrolidone |
| SC | Supercritical |
| RDX | Research Department Explosive |
| ER-GO | Explosively Reduced Graphene Oxide |
| SEG | Solvent-Exfoliated Graphene |
| SDOC | Sodium Deoxycholate |
| SDBS | Sodium Dodecylbenzenesulfonate |
| SDS | Sodium Dodecyl Sulfate |
| HTAB | Hexadecyltrimethylammonium Bromide |
| CG | Concentration of Graphene |
| Csur | Concentration of Surfactant |
| CMC | Critical Micelle Concentration |
| APCVD | Atmospheric Pressure Chemical Vapor Deposition |
| LPCVD | Low Pressure Chemical Vapor Deposition |
| PECVD | Plasma Enhanced Chemical Vapor Deposition |
| TCVD | Thermal Chemical Vapor Deposition |
References
- Ares, P.; Novoselov, K.S. Recent Advances in Graphene and Other 2D Materials. Nano Mater. Sci. 2022, 4, 3–9. [Google Scholar] [CrossRef]
- Sur, U.K. Graphene: A Rising Star on the Horizon of Materials Science. Int. J. Electrochem. 2012, 2012, 1–12. [Google Scholar] [CrossRef]
- Hancock, Y. The 2010 Nobel Prize in Physics - Ground-Breaking Experiments on Graphene. J. Phys. D. Appl. Phys. 2011, 44. [Google Scholar] [CrossRef]
- Radadiya, T. An Properties of Graphene. Int. J. Mech. Eng. Inf. Technol. 2015, 3, 983–992. [Google Scholar] [CrossRef]
- Ali Tahir, A.; Ullah, H.; Sudhagar, P.; Asri Mat Teridi, M.; Devadoss, A.; Sundaram, S. The Application of Graphene and Its Derivatives to Energy Conversion, Storage, and Environmental and Biosensing Devices. Chem. Rec. 2016, 16, 1591–1634. [Google Scholar] [CrossRef] [PubMed]
- Luan, D. Applications of Graphene in Different Fields. MATEC Web Conf. 2023, 386, 03015. [Google Scholar] [CrossRef]
- Li, S. Analysis of Large-Scale High-Quality Graphene Production and Applications. Appl. Comput. Eng. 2024, 63, 84–89. [Google Scholar] [CrossRef]
- Yan, Y.; Nashath, F.Z.; Chen, S.; Manickam, S.; Lim, S.S.; Zhao, H.; Lester, E.; Wu, T.; Pang, C.H. Synthesis of Graphene: Potential Carbon Precursors and Approaches. Nanotechnol. Rev. 2020, 9, 1284–1314. [Google Scholar] [CrossRef]
- Grayfer, E.D.; Makotchenko, V.G.; Nazarov, A.S.; Kim, S.J.; Fedorov, V.E. Graphene: Chemical Approaches to the Synthesis and Modification. Russ. Chem. Rev. 2011, 80, 751–770. [Google Scholar] [CrossRef]
- Li, H.; Zhao, G.; Zhang, H. Recent Progress of Cement-Based Materials Modified by Graphene and Its Derivatives. Materials (Basel). 2023, 16. [Google Scholar] [CrossRef] [PubMed]
- Edwards, R.S.; Coleman, K.S. Graphene Synthesis: Relationship to Applications. Nanoscale 2013, 5, 38–51. [Google Scholar] [CrossRef] [PubMed]
- Buzaglo, M.; Bar, I.P.; Varenik, M.; Shunak, L.; Pevzner, S.; Regev, O. Graphite-to-Graphene: Total Conversion. Adv. Mater. 2017, 29. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, M.D.; Kim, H.; Kim, G. Various Defects in Graphene: A Review. RSC Adv. 2022, 12, 21520–21547. [Google Scholar] [CrossRef] [PubMed]
- Biliak, R. Methods of Obtaining Graphene. Comput. Probl. Electr. Eng. 2023, 13, 1–8. [Google Scholar] [CrossRef]
- Shams, S.S.; Zhang, R.; Zhu, J. Graphene Synthesis: A Review. Mater. Sci. Pol. 2015, 33, 566–578. [Google Scholar] [CrossRef]
- Patel, R. V.; Patel, R.H.; Chaki, S.H. Synthesis and Characterization of 2D Graphene Sheets from Graphite Powder. AIP Conf. Proc. 2018, 1961. [Google Scholar] [CrossRef]
- Tour, J.M. Top-down versus Bottom-up Fabrication of Graphene-Based Electronics. Chem. Mater. 2014, 26, 163–171. [Google Scholar] [CrossRef]
- Moreno, C.; Vilas-Varela, M.; Kretz, B.; Garcia-Lekue, A.; Costache, M. V.; Paradinas, M.; Panighel, M.; Ceballos, G.; Valenzuela, S.O.; Peña, D.; et al. Bottom-up Synthesis of Multifunctional Nanoporous Graphene. Science (80-. ). 2018, 360, 199–203. [Google Scholar] [CrossRef] [PubMed]
- Santhiran, A.; Iyngaran, P.; Abiman, P.; Kuganathan, N. Graphene Synthesis and Its Recent Advances in Applications—A Review. C 2021, 7, 76. [Google Scholar] [CrossRef]
- Yi, M.; Shen, Z. A Review on Mechanical Exfoliation for the Scalable Production of Graphene. J. Mater. Chem. A 2015, 3, 11700–11715. [Google Scholar] [CrossRef]
- Mbayachi, V.B.; Ndayiragije, E.; Sammani, T.; Taj, S.; Mbuta, E.R.; khan, A. ullah Graphene Synthesis, Characterization and Its Applications: A Review. Results Chem. 2021, 3, 100163. [Google Scholar] [CrossRef]
- Sinclair, R.C.; Suter, J.L.; Coveney, P. V. Micromechanical Exfoliation of Graphene on the Atomistic Scale. Phys. Chem. Chem. Phys. 2019, 21, 5716–5722. [Google Scholar] [CrossRef] [PubMed]
- Jayasena, B.; Subbiah, S. A Novel Mechanical Cleavage Method for Synthesizing Few-Layer Graphenes. Nanoscale Res. Lett. 2011, 6, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, Y.H.; Mai, P.T.; Nguyen, N.P.T.; Tran, H. Van; Nguyen, H.T.M.; Nguyen, A.T. Van; Nguyen, D.V.; Doan, P.D.; Phan, M.N.; Bui, T.H. Fabrication of Graphene from Graphite Using High-Powered Ultrasonic Vibrators. Mater. Res. Express 2024, 11, 1–16. [Google Scholar] [CrossRef]
- Lavin-Lopez, M.P.; Valverde, J.L.; Sanchez-Silva, L.; Romero, A. Solvent-Based Exfoliation via Sonication of Graphitic Materials for Graphene Manufacture. Ind. Eng. Chem. Res. 2016, 55, 845–855. [Google Scholar] [CrossRef]
- Liyanage, C.D.; Kumar, H.; Perera, I.; Abeykoon, P.G.; Chen, F.; Joya, J.S.; Suib, S.L.; Adamson, D.H. Synthesis of Graphene Oxide: Effect of Sonicating during Oxidation. Carbon N. Y. 2024, 223. [Google Scholar] [CrossRef]
- Nguyen, Y.H.; Mai, P.T.; Nguyen, N.P.T.; Van Tran, H.; Nguyen, H.T.M.; Van Nguyen, A.T.; Nguyen, D.V.; Doan, P.D.; Phan, M.N.; Bui, T.H. Fabrication of Graphene from Graphite Using High-Powered Ultrasonic Vibrators. Mater. Res. Express 2024, 11. [Google Scholar] [CrossRef]
- Sargın, F.; Ak Azem, F.; Kanbur, K.; Birlik, I.; Türk, A. Evaluating the Impact of Sonication Process on Graphene Oxide Structural Properties. Ömer Halisdemir Üniversitesi Mühendislik Bilim. Derg. 2024, 13, 1139–1149. [Google Scholar] [CrossRef]
- Htwe, Y.Z.N.; Mariatti, M.; Chow, W.S.; Suda, Y.; Thant, A.A. Effect of Sonication Time on the Production of Graphene by Electrochemical Exfoliation Method. J. Phys. Conf. Ser. 2018, 1082. [Google Scholar] [CrossRef]
- Azimi, Z.; Alimohammadian, M.; Sohrabi, B. Graphene Quantum Dots Based on Mechanical Exfoliation Methods: A Simple and Eco-Friendly Technique. ACS Omega 2024, 9, 31427–31437. [Google Scholar] [CrossRef] [PubMed]
- Gaurav, A.; Paul, G. Synthesis and Characterization of Graphene Oxide Nanosheets by Mechanical Exfoliation Using Ball Milling. J. Phys. Conf. Ser. 2024, 2818. [Google Scholar] [CrossRef]
- Awan, Z.; Naqvi, A.A.; Shahid, Z.; Butt, F.A.; Raza, F. Synthesis and Characterization of Graphene Sheets from Graphite Powder by Using Ball Milling. Rev. UIS Ing. 2022, 21, 71–76. [Google Scholar] [CrossRef]
- Ahn, J.H.; Kim, Y.J.; Hwang, S.J.; Chung, H.S. High Energy Ball Milling of Catalytically Synthesized Carbon Nanotubes. Mater. Sci. Forum 2007, 534–536, 193–196. [Google Scholar] [CrossRef]
- Zhao, W.; Fang, M.; Wu, F.; Wu, H.; Wang, L.; Chen, G. Preparation of Graphene by Exfoliation of Graphite Using Wet Ball Milling. J. Mater. Chem. 2010, 20, 5817–5819. [Google Scholar] [CrossRef]
- Hu, K.; Brambilla, L.; Sartori, P.; Moscheni, C.; Perrotta, C.; Zema, L.; Bertarelli, C.; Castiglioni, C. Development of Tailored Graphene Nanoparticles: Preparation, Sorting and Structure Assessment by Complementary Techniques. Molecules 2023, 28. [Google Scholar] [CrossRef] [PubMed]
- Liang, D.; Yan, L.; Huang, K.; Li, Y.; Ai, F.; Zhang, H.; Jiang, Z. Effect of Different Rotational Speeds on Graphene-Wrapped Sic Core-Shell Nanoparticles in Wet Milling Medium. Materials (Basel). 2021, 14, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Myekhlai, M.; Munkhbayar, B.; Lee, T.; Tanshen, M.R.; Chung, H.; Jeong, H. Experimental Investigation of the Mechanical Grinding Effect on Graphene Structure. RSC Adv. 2014, 4, 2495–2500. [Google Scholar] [CrossRef]
- Mahmoud, A.E.D.; Stolle, A.; Stelter, M. Sustainable Synthesis of High-Surface-Area Graphite Oxide via Dry Ball Milling. ACS Sustain. Chem. Eng. 2018, 6, 6358–6369. [Google Scholar] [CrossRef]
- Brandão, A.T.S.C.; Costa, R.; Silva, A.F.; Pereira, C.M. Sustainable Preparation of Nanoporous Carbons via Dry Ball Milling: Electrochemical Studies Using Nanocarbon Composite Electrodes and a Deep Eutectic Solvent as Electrolyte. Nanomaterials 2021, 11. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Cao, Y.; Zhang, J.; Zhang, W.; Xu, Y.; Guo, J.; Yang, W.; Liu, J. One-Step Preparation of Graphene Nanosheets via Ball Milling of Graphite and the Application in Lithium-Ion Batteries. J. Mater. Sci. 2016, 51, 3675–3683. [Google Scholar] [CrossRef]
- Dash, P.; Dash, T.; Rout, T.K.; Sahu, A.K.; Biswal, S.K.; Mishra, B.K. Preparation of Graphene Oxide by Dry Planetary Ball Milling Process from Natural Graphite. RSC Adv. 2016, 6, 12657–12668. [Google Scholar] [CrossRef]
- Jeon, I.Y.; Choi, H.J.; Jung, S.M.; Seo, J.M.; Kim, M.J.; Dai, L.; Baek, J.B. Large-Scale Production of Edge-Selectively Functionalized Graphene Nanoplatelets via Ball Milling and Their Use as Metal-Free Electrocatalysts for Oxygen Reduction Reaction. J. Am. Chem. Soc. 2013, 135, 1386–1393. [Google Scholar] [CrossRef] [PubMed]
- Yi, M.; Shen, Z.; Zhu, J. A Fluid Dynamics Route for Producing Graphene and Its Analogues. Chinese Sci. Bull. 2014, 59, 1794–1799. [Google Scholar] [CrossRef]
- Yi, M.; Shen, Z. Fluid Dynamics: An Emerging Route for the Scalable Production of Graphene in the Last Five Years. RSC Adv. 2016, 6, 72525–72536. [Google Scholar] [CrossRef]
- Pang, Y.X.; Yew, M.; Yan, Y.; Khine, P.; Filbert, A.; Manickam, S.; Foo, D.C.Y.; Sharmin, N.; Lester, E.; Wu, T.; et al. Application of Supercritical Fluid in the Synthesis of Graphene Materials: A Review. J. Nanoparticle Res. 2021, 23. [Google Scholar] [CrossRef]
- Morales Ibarra, R.; Goto, M.; García-Serna, J.; García Montes, S.M. Graphene Exfoliation with Supercritical Fluids. Carbon Lett. 2021, 31, 99–105. [Google Scholar] [CrossRef]
- Gao, H.; Hu, G. Graphene Production via Supercritical Fluids. RSC Adv. 2016, 6, 10132–10143. [Google Scholar] [CrossRef]
- Shang, T.; Feng, G.; Li, Q.; Zheng, Y. Production of Graphene Nanosheets by Supercritical CO2 Process Coupled with Micro-Jet Exfoliation. Fullerenes Nanotub. Carbon Nanostructures 2017, 25, 691–698. [Google Scholar] [CrossRef]
- Rangappa, D.; Sone, K.; Wang, M.; Gautam, U.K.; Golberg, D.; Itoh, H.; Ichihara, M.; Honma, I. Rapid and Direct Conversion of Graphite Crystals into High-Yielding, Good-Quality Graphene by Supercritical Fluid Exfoliation. Chem. - A Eur. J. 2010, 16, 6488–6494. [Google Scholar] [CrossRef] [PubMed]
- Hadi, A.; Karimi-Sabet, J.; Moosavian, S.M.A.; Ghorbanian, S. Optimization of Graphene Production by Exfoliation of Graphite in Supercritical Ethanol: A Response Surface Methodology Approach. J. Supercrit. Fluids 2016, 107, 92–105. [Google Scholar] [CrossRef]
- Ye, B.Y.; Wang, J.Y.; Geng, X.H.; An, C.W.; Ding, P.H. One-Step Synthesis of Graphene Nanosheets through Explosive Process. Inorg. Nano-Metal Chem. 2017, 47, 1216–1219. [Google Scholar] [CrossRef]
- Wright, J.P.; Sigdel, S.; Corkill, S.; Covarrubias, J.; LeBan, L.; Nepal, A.; Li, J.; Divigalpitiya, R.; Bossmann, S.H.; Sorensen, C.M. Synthesis of Turbostratic Nanoscale Graphene via Chamber Detonation of Oxygen/Acetylene Mixtures. Nano Sel. 2022, 3, 1054–1068. [Google Scholar] [CrossRef]
- Nepal, A.; Singh, G.P.; Flanders, B.N.; Sorensen, C.M. One-Step Synthesis of Graphene via Catalyst-Free Gas-Phase Hydrocarbon Detonation. Nanotechnology 2013, 24. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.H.; Zhuo, D.X.; Wu, L.X.; Ma, L.; Weng, Z.X.; Wang, R. A Facile and Efficient Method to Prepare Exfoliated and Reduced Graphene Nanosheets by Detonation. Adv. Mater. Res. 2014, 937, 260–266. [Google Scholar] [CrossRef]
- Dreyer, D.R.; Park, S.; Bielawski, C.W.; Ruoff, R.S. The Chemistry of Graphene Oxide. Chem. Soc. Rev. 2010, 39, 228–240. [Google Scholar] [CrossRef] [PubMed]
- Stankovich, S.; Dikin, D.A.; Piner, R.D.; Kohlhaas, K.A.; Kleinhammes, A.; Jia, Y.; Wu, Y.; Nguyen, S.B.T.; Ruoff, R.S. Synthesis of Graphene-Based Nanosheets via Chemical Reduction of Exfoliated Graphite Oxide. Carbon N. Y. 2007, 45, 1558–1565. [Google Scholar] [CrossRef]
- Eda, G.; Chhowalla, M. Chemically Derived Graphene Oxide: Towards Large-Area Thin-Film Electronics and Optoelectronics. Adv. Mater. 2010, 22, 2392–2415. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Murali, S.; Cai, W.; Li, X.; Suk, J.W.; Potts, J.R.; Ruoff, R.S. Graphene and Graphene Oxide: Synthesis, Properties, and Applications. Adv. Mater. 2010, 22, 3906–3924. [Google Scholar] [CrossRef] [PubMed]
- Pei, S.; Cheng, H.M. The Reduction of Graphene Oxide. Carbon N. Y. 2012, 50, 3210–3228. [Google Scholar] [CrossRef]
- Chua, C.K.; Pumera, M. Chemical Reduction of Graphene Oxide: A Synthetic Chemistry Viewpoint. Chem. Soc. Rev. 2014, 43, 291–312. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Jang, J.; Nagase, S. Hydrazine and Thermal Reduction of Graphene Oxide: Reaction Mechanisms, Product Structures, and Reaction Design. J. Phys. Chem. C 2010, 114, 832–842. [Google Scholar] [CrossRef]
- Chen, D.; Feng, H.; Li, J. Graphene Oxide: Preparation, Functionalization, and Electrochemical Applications. Chem. Rev. 2012, 112, 6027–6053. [Google Scholar] [CrossRef] [PubMed]
- Alam, S.N.; Sharma, N.; Kumar, L. Synthesis of Graphene Oxide (GO) by Modified Hummers Method and Its Thermal Reduction to Obtain Reduced Graphene Oxide (RGO)*. Graphene 2017, 06, 1–18. [Google Scholar] [CrossRef]
- Paton-Carrero, A.; Valverde, J.L.; Garcia-Alvarez, E.; Lavin-Lopez, M.P.; Romero, A. Influence of the Oxidizing Agent in the Synthesis of Graphite Oxide. J. Mater. Sci. 2020, 55, 2333–2342. [Google Scholar] [CrossRef]
- Das, P.; Ibrahim, S.; Chakraborty, K.; Ghosh, S.; Pal, T. Stepwise Reduction of Graphene Oxide and Studies on Defect-Controlled Physical Properties. Sci. Rep. 2024, 14, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Yao, X.; Li, H.; Liu, Z.; Ma, W.; Liang, X. Thermal Stability of Oxygen-Containing Functional Groups on Activated Carbon Surfaces in a Thermal Oxidative Environment. J. Chem. Eng. Japan 2014, 47, 21–27. [Google Scholar] [CrossRef]
- Lee, B.J.; Jeong, G.H. Thermal Oxidation of Synthesized Graphenes and Their Optical Property Characterization. J. Nanosci. Nanotechnol. 2011, 11, 6084–6088. [Google Scholar] [CrossRef] [PubMed]
- Bhullar, S.S.; Liu, W.W. A Review of the Effect of Different Electrolytes on the Synthesis of Graphene Sheets by Electrochemical Exfoliation. Int. J. Nanoelectron. Mater. 2024, 17, 279–283. [Google Scholar] [CrossRef]
- Pei, S.; Wei, Q.; Huang, K.; Cheng, H.M.; Ren, W. Green Synthesis of Graphene Oxide by Seconds Timescale Water Electrolytic Oxidation. Nat. Commun. 2018, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Lesiak, B.; Trykowski, G.; Tóth, J.; Biniak, S.; Kövér, L.; Rangam, N.; Stobinski, L.; Malolepszy, A. Chemical and Structural Properties of Reduced Graphene Oxide—Dependence on the Reducing Agent. J. Mater. Sci. 2021, 56, 3738–3754. [Google Scholar] [CrossRef]
- Cataldo, F.; Putz, M. V.; Ursini, O.; Angelini, G.; Garcia-Hernandez, D.A.; Manchado, A. A New Route to Graphene Starting from Heavily Ozonized Fullerenes: Part 1 - Thermal Reduction under Inert Atmosphere. Fullerenes Nanotub. Carbon Nanostructures 2016, 24, 52–61. [Google Scholar] [CrossRef]
- Sengunthar, P.; Patel, S.; Thankachen, N.; Joshi, U.S.; Pandya, R.J. Controlled Synthesis of Reduced Graphene Oxide Sheets on Large Scale Using Thermal Exfoliation. ECS Trans. 2022, 107, 19943–19948. [Google Scholar] [CrossRef]
- Feng, X.; Chen, W.; Yan, L. Electrochemical Reduction of Bulk Graphene Oxide Materials. RSC Adv. 2016, 6, 80106–80113. [Google Scholar] [CrossRef]
- Kholib, N.S.; Liu, W.W. Graphene Synthesis by Electrochemical Reduction of Graphene Oxide and Its Characterizations. Int. J. Nanoelectron. Mater. 2023, 16, 717–724. [Google Scholar] [CrossRef]
- Nair, S.S.; Saha, T.; Dey, P.; Bhadra, S. Thermal Oxidation of Graphite as the First Step for Graphene Preparation: Effect of Heating Temperature and Time. J. Mater. Sci. 2021, 56, 3675–3691. [Google Scholar] [CrossRef]
- Zhou, M.; Guo, L. ping; Lin, F. yun; Liu, H. xia Electrochemistry and Electrocatalysis of Polyoxometalate-Ordered Mesoporous Carbon Modified Electrode. Anal. Chim. Acta 2007, 587, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Zhang, L.; Jiao, X.; Qiu, Y.; Xu, W. The Electrochemical Performance of Reduced Graphene Oxide Prepared from Different Types of Natural Graphites. RSC Adv. 2021, 11, 4042–4052. [Google Scholar] [CrossRef] [PubMed]
- Mawatha, B.; Lanka, S. Spectroscopic Analysis of Mass-Scale Prepared GO and RGO from Vein Graphite through Compositional Improvement. Sri Lankan J. Phys. 2024, 25, 13–34. [Google Scholar] [CrossRef]
- Mhlongo, J.T.; Tlhaole, B.; Linganiso, L.Z.; Motaung, T.E.; Linganiso-Dziike, E.C. Microwave-Assisted Reduction of Graphene Oxide to Reduced Graphene Oxide. Processes 2025, 13, 1–15. [Google Scholar] [CrossRef]
- Hidayat, R.; Wahyuningsih, S.; Ramelan, A.H. Simple Synthesis of RGO (Reduced Graphene Oxide) by Thermal Reduction of GO (Graphene Oxide). IOP Conf. Ser. Mater. Sci. Eng. 2020, 858. [Google Scholar] [CrossRef]
- Thakur, A.; Kumar, S.; Pathania, P.; Pathak, D.; Rangra, V.S. SYNTHESIS of RGO-ZnO COMPOSITES for THERMAL, ELECTRICAL and ANTIBACTERIAL STUDIES. Surf. Rev. Lett. 2017, 24, 1–8. [Google Scholar] [CrossRef]
- Awoji, M.O.; Onoja, A.D.; Echi, M.I. Synthesis of Graphene Via Arc Discharge and Its Characterization: A Comparative Approach. East Eur. J. Phys. 2023, 2023, 252–257. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, B.; Ma, Y.; Huang, Y.; Li, N.; Zhang, F.; Chen, Y. Efficient and Large-Scale Synthesis of Few-Layered Graphene Using an Arc-Discharge Method and Conductivity Studies of the Resulting Films. Nano Res. 2010, 3, 661–669. [Google Scholar] [CrossRef]
- Li, N.; Wang, Z.; Zhao, K.; Shi, Z.; Gu, Z.; Xu, S. Large Scale Synthesis of N-Doped Multi-Layered Graphene Sheets by Simple Arc-Discharge Method. Carbon N. Y. 2010, 48, 255–259. [Google Scholar] [CrossRef]
- Wu, C.; Dong, G.; Guan, L. Production of Graphene Sheets by a Simple Helium Arc-Discharge. Phys. E Low-Dimensional Syst. Nanostructures 2010, 42, 1267–1271. [Google Scholar] [CrossRef]
- Kumar, R.; Singh, R.K.; Dubey, P.K.; Kumar, P.; Tiwari, R.S.; Oh, I.K. Pressure-Dependent Synthesis of High-Quality Few-Layer Graphene by Plasma-Enhanced Arc Discharge and Their Thermal Stability. J. Nanoparticle Res. 2013, 15. [Google Scholar] [CrossRef]
- Subrahmanyam, K.S.; Panchakarla, L.S.; Govindaraj, A.; Rao, C.N.R. Simple Method of Preparing Graphene Flakes by an Arc-Discharge Method. J. Phys. Chem. C 2009, 113, 4257–4259. [Google Scholar] [CrossRef]
- Wu, X.; Liu, Y.; Yang, H.; Shi, Z. Large-Scale Synthesis of High-Quality Graphene Sheets by an Improved Alternating Current Arc-Discharge Method. RSC Adv. 2016, 6, 93119–93124. [Google Scholar] [CrossRef]
- Antisari, M.V.; Gattia, D.M.; Brandão, L.; Marazzi, R.; Montone, A. Carbon Nanostructures Produced by an AC Arc Discharge. Mater. Sci. Forum 2010, 638–642, 1766–1771. [Google Scholar] [CrossRef]
- Gattia, D.M.; Vittori Antisari, M.; Marazzi, R. AC Arc Discharge Synthesis of Single-Walled Nanohorns and Highly Convoluted Graphene Sheets. Nanotechnology 2007, 18. [Google Scholar] [CrossRef]
- Kane, A.; Hinkov, I.; Brinza, O.; Hosni, M.; Barry, A.H.; Cherif, S.M.; Farhat, S. One-Step Synthesis of Graphene, Copper and Zinc Oxide Graphene Hybrids via Arc Discharge: Experiments and Modeling. Coatings 2020, 10, 1–24. [Google Scholar] [CrossRef]
- Wang, Z.; Li, N.; Shi, Z.; Gu, Z. Low-Cost and Large-Scale Synthesis of Graphene Nanosheets by Arc Discharge in Air. Nanotechnology 2010, 21. [Google Scholar] [CrossRef] [PubMed]
- Levchenko, I.; Cvelbar, U.; Keidar, M. Graphene Flakes in Arc Plasma: Conditions for the Fast Single-Layer Growth. Graphene 2016, 05, 81–89. [Google Scholar] [CrossRef]
- Volotskova, O.; Levchenko, I.; Shashurin, A.; Raitses, Y.; Ostrikov, K.; Keidar, M. Single-Step Synthesis and Magnetic Separation of Graphene and Carbon Nanotubes in Arc Discharge Plasmas. Nanoscale 2010, 2, 2281–2285. [Google Scholar] [CrossRef] [PubMed]
- Karmakar, S.; Kulkarni, N. V.; Nawale, A.B.; Lalla, N.P.; Mishra, R.; Sathe, V.G.; Bhoraskar, S. V.; Das, A.K. A Novel Approach towards Selective Bulk Synthesis of Few-Layer Graphenes in an Electric Arc. J. Phys. D. Appl. Phys. 2009, 42. [Google Scholar] [CrossRef]
- Pacheco, M.; Mendoza, D.; Valdivia-Barrientos, R.; Santana-Diaz, A.; Pacheco, J.; Alarcon, L.E.; Gutierrez, P.G.V.; Tu, X. Multilayer Graphene Growth Assisted by Sulfur Using the Arc Discharge Method at Ambient Conditions. IEEE Trans. Plasma Sci. 2018, 46, 2407–2412. [Google Scholar] [CrossRef]
- Kosynkin, D. V.; Higginbotham, A.L.; Sinitskii, A.; Lomeda, J.R.; Dimiev, A.; Price, B.K.; Tour, J.M. Longitudinal Unzipping of Carbon Nanotubes to Form Graphene Nanoribbons. Nature 2009, 458, 872–876. [Google Scholar] [CrossRef] [PubMed]
- Jiao, L.; Zhang, L.; Wang, X.; Diankov, G.; Dai, H. Narrow Graphene Nanoribbons from Carbon Nanotubes. Nature 2009, 458, 877–880. [Google Scholar] [CrossRef] [PubMed]
- Cano-marquez, A.G.; Rodríguez-macias, F.J.; Campos-delgado, J.; Espinosa-gonzalez, C.G.; Tristan-lopez, F.; Ramírez-gonzalez, D.; Cullen, D.A.; Smith, D.J.; Terrones, M.; Vega-cantu, Y.I. Ex-MWNTs: Graphene Sheets and Ribbons Produced by Lithium Intercalation and Exfoliation of Carbon Nanotubes. Nano Lett. 2009, 9, 1527–1533. [Google Scholar] [CrossRef] [PubMed]
- Dimiev, A.M.; Khannanov, A.; Vakhitov, I.; Kiiamov, A.; Shukhina, K.; Tour, J.M. Revisiting the Mechanism of Oxidative Unzipping of Multiwall Carbon Nanotubes to Graphene Nanoribbons. ACS Nano 2018, 12, 3985–3993. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Lim, J.; Cho, S.Y.; Kim, H.; Lee, C.; Lee, G.Y.; Sasikala, S.P.; Yun, T.; Choi, D.S.; Jeong, M.S.; et al. Intact Crystalline Semiconducting Graphene Nanoribbons from Unzipping Nitrogen-Doped Carbon Nanotubes. ACS Appl. Mater. Interfaces 2019, 11, 38006–38015. [Google Scholar] [CrossRef] [PubMed]
- Al-Tamimi, B.H.; Farid, S.B.H.; Chyad, F.A. Modified Unzipping Technique to Prepare Graphene Nano-Sheets. J. Phys. Conf. Ser. 2018, 1003. [Google Scholar] [CrossRef]
- Janowska, I.; Ersen, O.; Jacob, T.; Vennégues, P.; Begin, D.; Ledoux, M.J.; Pham-Huu, C. Catalytic Unzipping of Carbon Nanotubes to Few-Layer Graphene Sheets under Microwaves Irradiation. Appl. Catal. A Gen. 2009, 371, 22–30. [Google Scholar] [CrossRef]
- Zheng, Q.F.; Guo, Y.; Liang, Y.; Shen, Q. Graphene Nanoribbons from Electrostatic-Force-Controlled Electric Unzipping of Single- And Multi-Walled Carbon Nanotubes. ACS Appl. Nano Mater. 2020, 3, 4708–4716. [Google Scholar] [CrossRef]
- Hirsch, A. Unzipping Carbon Nanotubes: A Peeling Method for the Formation of Graphene Nanoribbons. Angew. Chemie - Int. Ed. 2009, 48, 6594–6596. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Hu, Y.; Huang, J.; Zhou, N.; Liu, Y.; Wei, L.; Chen, X.; Zhuang, N. One-Step Oxidation Preparation of Unfolded and Good Soluble Graphene Nanoribbons by Longitudinal Unzipping of Carbon Nanotubes. Nanotechnology 2018, 29, 0–14. [Google Scholar] [CrossRef] [PubMed]
- Jovanović, S.; Da Ross, T.; Ostric, A.; Tošić, D.; Prekodravac, J.; Marković, Z.; Todorović Marković, B. Raman Spectroscopy of Graphene Nanoribbons Synthesized by Longitudinal Unzipping of Multiwall Carbon Nanotubes. Phys. Scr. Top. Issues 2014, T162. [Google Scholar] [CrossRef]
- Ko, D.; Choi, J.; Yan, B.; Hwang, T.; Jin, X.; Kim, J.M.; Piao, Y. A Facile and Scalable Approach to Develop Electrochemical Unzipping of Multi-Walled Carbon Nanotubes to Graphene Nanoribbons. J. Mater. Chem. A 2020, 8, 22045–22053. [Google Scholar] [CrossRef]
- Xie, L.; Wang, H.; Jin, C.; Wang, X.; Jiao, L.; Suenaga, K.; Dai, H. Graphene Nanoribbons from Unzipped Carbon Nanotubes: Atomic Structures, Raman Spectroscopy, and Electrical Properties. J. Am. Chem. Soc. 2011, 133, 10394–10397. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Liu, M.; Gu, Y.; Guo, B.; Ma, H.X.; Wang, P.; Wang, X.; Zhang, R. Fast Chemical Exfoliation of Graphite to Few-Layer Graphene with High Quality and Large Size via a Two-Step Microwave-Assisted Process. Chem. Eng. J. 2020, 381. [Google Scholar] [CrossRef]
- Chacón-Torres, J.C.; Wirtz, L.; Pichler, T. Raman Spectroscopy of Graphite Intercalation Compounds: Charge Transfer, Strain, and Electron-Phonon Coupling in Graphene Layers. Phys. Status Solidi Basic Res. 2014, 251, 2337–2355. [Google Scholar] [CrossRef]
- MOOSA, A.A.; ABED, M.S. Graphene Preparation and Graphite Exfoliation. Turkish J. Chem. 2021, 45, 493–519. [Google Scholar] [CrossRef] [PubMed]
- Qamar, S.; Ramzan, N.; Aleem, W. Graphene Dispersion, Functionalization Techniques and Applications: A Review. Synth. Met. 2024, 307, 117697. [Google Scholar] [CrossRef]
- Du, W.; Lu, J.; Sun, P.; Zhu, Y.; Jiang, X. Organic Salt-Assisted Liquid-Phase Exfoliation of Graphite to Produce High-Quality Graphene. Chem. Phys. Lett. 2013, 568–569, 198–201. [Google Scholar] [CrossRef]
- Xu, Y.; Cao, H.; Xue, Y.; Li, B.; Cai, W. Liquid-Phase Exfoliation of Graphene: An Overview on Exfoliation Media, Techniques, and Challenges. Nanomaterials 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Bordes, E.; Morcos, B.; Bourgogne, D.; Andanson, J.M.; Bussière, P.O.; Santini, C.C.; Benayad, A.; Gomes, M.C.; Pádua, A.A.H. Dispersion and Stabilization of Exfoliated Graphene in Ionic Liquids. Front. Chem. 2019, 7. [Google Scholar] [CrossRef] [PubMed]
- Griffin, A.; Nisi, K.; Pepper, J.; Harvey, A.; Szydłowska, B.M.; Coleman, J.N.; Backes, C. Effect of Surfactant Choice and Concentration on the Dimensions and Yield of Liquid-Phase-Exfoliated Nanosheets. Chem. Mater. 2020, 32, 2852–2862. [Google Scholar] [CrossRef]
- Wang, S.; Yi, M.; Shen, Z. The Effect of Surfactants and Their Concentration on the Liquid Exfoliation of Graphene. RSC Adv. 2016, 6, 56705–56710. [Google Scholar] [CrossRef]
- Choi, E.Y.; Choi, W.S.; Lee, Y.B.; Noh, Y.Y. Production of Graphene by Exfoliation of Graphite in a Volatile Organic Solvent. Nanotechnology 2011, 22. [Google Scholar] [CrossRef] [PubMed]
- Neill, A.O.; Khan, U.; Nirmalraj, P.N.; Boland, J.; Coleman, J.N.; Lotya, M.; Hernandez, Y.; King, P.J.; Smith, R.J.; Nicolosi, V.; et al. Graphene Dispersion and Exfoliation in Low Boiling Point Solvents Graphene Dispersion and Exfoliation in Low Boiling Point Solvents. J. Phys. Chem. 2011, 115, 5422–5428. [Google Scholar] [CrossRef]
- Liu, W.W.; Aziz, A. Review on the Effects of Electrochemical Exfoliation Parameters on the Yield of Graphene Oxide. ACS Omega 2022, 7, 33719–33731. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Casiraghi, C.; Parvez, K. Electrochemical Exfoliation of 2D Materials beyond Graphene. Chem. Soc. Rev. 2024, 53, 3036–3064. [Google Scholar] [CrossRef] [PubMed]
- Achee, T.C.; Sun, W.; Hope, J.T.; Quitzau, S.G.; Sweeney, C.B.; Shah, S.A.; Habib, T.; Green, M.J. High-Yield Scalable Graphene Nanosheet Production from Compressed Graphite Using Electrochemical Exfoliation. Sci. Rep. 2018, 8, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Salverda, M.; Thiruppathi, A.R.; Pakravan, F.; Wood, P.C.; Chen, A. Electrochemical Exfoliation of Graphite to Graphene-Based Nanomaterials. Molecules 2022, 27. [Google Scholar] [CrossRef] [PubMed]
- Xia, Z.; Bellani, V.; Sun, J.; Palermo, V. Electrochemical Exfoliation of Graphite in H2SO4, Li2SO4and NaClO4solutions Monitored: In Situ by Raman Microscopy and Spectroscopy. Faraday Discuss. 2021, 227, 291–305. [Google Scholar] [CrossRef] [PubMed]
- Reina, A.; Jia, X.; Ho, J.; Nezich, D.; Son, H.; Bulovic, V.; Dresselhaus, M.S.; Kong, J. Large Area, Few-Layer Graphene Films on Arbitrary Substrates by Chemical Vapor Deposition. Am. Chem. Soc. 2008, 9, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Cai, W.; An, J.; Kim, S.; Nah, J.; Yang, D.; Piner, R.; Velamakanni, A.; Jung, I.; Tutuc, E.; et al. Large-Area Synthesis of High-Quality and Uniform Graphene Films on Copper Foils. Science (80-. ). 2009, 324, 1312–1314. [Google Scholar] [CrossRef] [PubMed]
- Bhaviripudi, S.; Jia, X.; Dresselhaus, M.S.; Kong, J. Role of Kinetic Factors in Chemical Vapor Deposition Synthesis of Uniform Large Area Graphene Using Copper Catalyst. Nano Lett. 2010, 10, 4128–4133. [Google Scholar] [CrossRef] [PubMed]
- Chae, S.J.; Güneş, F.; Kim, K.K.; Kim, E.S.; Han, G.H.; Kim, S.M.; Shin, H.; Yoon, S.M.; Choi, J.Y.; Park, M.H.; et al. Synthesis of Large-Area Graphene Layers on Poly-Nickel Substrate by Chemical Vapor Deposition: Wrinkle Formation. Adv. Mater. 2009, 21, 2328–2333. [Google Scholar] [CrossRef]
- Kostogrud, I.A.; Trusov, K. V.; Smovzh, D. V. Influence of Gas Mixture and Temperature on AP-CVD Synthesis of Graphene on Copper Foil. Adv. Mater. Interfaces 2016, 3, 1–6. [Google Scholar] [CrossRef]
- Memon, N.K.; Tse, S.D.; Chhowalla, M.; Kear, B.H. Role of Substrate, Temperature, and Hydrogen on the Flame Synthesis of Graphene Films. Proc. Combust. Inst. 2013, 34, 2163–2170. [Google Scholar] [CrossRef]
- Zhang, C.; Man, B.Y.; Jiang, S.Z.; Yang, C.; Liu, M.; Chen, C.S.; Xu, S.C.; Feng, D.J.; Bi, D.; Liu, F.Y.; et al. Facile Synthesis of Graphene on Single Mode Fiber via Chemical Vapor Deposition. Appl. Surf. Sci. 2014, 307, 327–332. [Google Scholar] [CrossRef]
- Saeed, M.; Alshammari, Y.; Majeed, S.A.; Al-Nasrallah, E. Chemical Vapour Deposition of Graphene—Synthesis, Characterisation, and Applications: A Review. Molecules 2020, 25. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Mehta, B.R. A Parametric Study on the Influence of Synthesis and Transfer Conditions on the Quality of Graphene. J. Nanosci. Nanotechnol. 2017, 17, 286–299. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Yuan, G.; Gao, L.; Yang, J.; Chhowalla, M.; Gharahcheshmeh, M.H.; Gleason, K.K.; Choi, Y.S.; Hong, B.H.; Liu, Z. Chemical Vapour Deposition. Primer 2021, 1. [Google Scholar] [CrossRef]
- Thodkar, K.; Plodinec, M.; Gramm, F.; Kunze, K. ISCOPEM2D V1.0: An In Situ Method to Characterize and Compare Chemical Vapor Deposition Graphene Films Using Quality Matrix Approaches. Phys. Status Solidi - Rapid Res. Lett. 2024, 18. [Google Scholar] [CrossRef]
- Donglah, N.A.B.H.; Adenan, N.B.M.; Sabet, M. Effects of Pressure Variations in the Quality of Graphene Production through Chemical Vapor Deposition by Regression. AIP Conf. Proc. 2023, 2643, 10–14. [Google Scholar] [CrossRef]
- Tursunkulov, O.; Allabergenov, B.; Abidov, A.; Kim, S.-Y.; Jeon, H.-W.; Jeong, S.-W.; Kim, S. Comparison Characteristic of Large Area Graphene Films Grown by Chemical Vapor Deposition with Nano-Graphite Structures. Int. J. Mater. Mech. Manuf. 2013, 324–327. [Google Scholar] [CrossRef]
- Lee, B.; Chu, W.; Li, W. Effects of Process Parameters on Graphene Growth via Low-Pressure Chemical Vapor Deposition. J. Micro Nano-Manufacturing 2020, 8, 1–7. [Google Scholar] [CrossRef]
- KAHYAOĞLU, A.; ÜNLÜ, Ö. Graphene Growth in Different Thickness by Chemical Vapor Deposition Method. Düzce Üniversitesi Bilim ve Teknol. Derg. 2023, 11, 787–798. [Google Scholar] [CrossRef]
- Arjmandi-Tash, H.; Lebedev, N.; van Deursen, P.M.G.; Aarts, J.; Schneider, G.F. Hybrid Cold and Hot-Wall Reaction Chamber for the Rapid Synthesis of Uniform Graphene. Carbon N. Y. 2017, 118, 438–442. [Google Scholar] [CrossRef]
- Jia, K.; Ci, H.; Zhang, J.; Sun, Z.; Ma, Z.; Zhu, Y.; Liu, S.; Liu, J.; Sun, L.; Liu, X.; et al. Superclean Growth of Graphene Using a Cold-Wall Chemical Vapor Deposition Approach. Angew. Chemie - Int. Ed. 2020, 59, 17214–17218. [Google Scholar] [CrossRef] [PubMed]
- Bosc, A.; Ladron-de-Guevara, A.; Pedros, J.; Martinez, J.; Fandan, R.; Calle, F. Parameter Space for Graphene Chemical Vapour Deposition in Cold-Wall Reactors under High Precursor Flux. Cryst. Growth Des. 2023, 23. [Google Scholar] [CrossRef]
- Das, S.; Drucker, J. Nucleation and Growth of Single Layer Graphene on Electrodeposited Cu by Cold Wall Chemical Vapor Deposition. Nanotechnology 2017, 28. [Google Scholar] [CrossRef] [PubMed]
- Chang, Q.H.; Huang, L.; Ji, L.C.; Wang, T.; Ling, B.; Yang, H.F. Few-Layer Graphene Direct Deposition on Ni and Cu Foil by Cold-Wall Chemical Vapor Deposition. Proc. - 2010 8th Int. Vac. Electron Sources Conf. Nanocarbon, IVESC 2010 NANOcarbon 2010 2010, 467–468. [CrossRef]
- Deng, B.; Liu, Z.; Peng, H. Toward Mass Production of CVD Graphene Films. Adv. Mater. 2019, 31, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Shen, C.-M.; Tian, Y.; Wang, G.-Q.; Lin, S.-X.; Zhang, Y.; Gu, C.-Z.; Li, J.-J.; Gao, H.-J. Influence of Reaction Parameters on Synthesis of High-Quality Single-Layer Graphene on Cu Using Chemical Vapor Deposition. Chinese Phys. B 2014, 23, 096803. [Google Scholar] [CrossRef]
- Anisur, M.R.; Raman, R.K.S.; Banerjee, P.C.; Al-Saadi, S.; Arya, A.K. Review of the Role of CVD Growth Parameters on Graphene Coating Characteristics and the Resulting Corrosion Resistance. Surf. Coatings Technol. 2024, 487, 130934. [Google Scholar] [CrossRef]
- Fauzi, F.B.; Ismail, E.; Ani, M.H.; Syed Abu Bakar, S.N.; Mohamed, M.A.; Majlis, B.Y.; Md Din, M.F.; Azam Mohd Abid, M.A. A Critical Review of the Effects of Fluid Dynamics on Graphene Growth in Atmospheric Pressure Chemical Vapor Deposition. J. Mater. Res. 2018, 33, 1088–1108. [Google Scholar] [CrossRef]
- Shinde, D.B.; Chaturvedi, P.; Vlassiouk, I. V.; Smirnov, S.N. Unique Role of Dimeric Carbon Precursors in Graphene Growth by Chemical Vapor Deposition. Carbon Trends 2021, 5, 100093. [Google Scholar] [CrossRef]
- Wang, J. Bin; Ren, Z.; Hou, Y.; Yan, X.L.; Liu, P.Z.; Zhang, H.; Zhang, H.X.; Guo, J.J. A Review of Graphene Synthesis at Low Temperatures by CVD Methods. Xinxing Tan Cailiao/New Carbon Mater. 2020, 35, 193–208. [Google Scholar] [CrossRef]
- Zafar, M.A.; Jacob, M. V. Plasma-Based Synthesis of Graphene and Applications: A Focused Review; Springer Nature Singapore, 2022; Vol. 6; ISBN 0123456789.
- Woehrl, N.; Ochedowski, O.; Gottlieb, S.; Shibasaki, K.; Schulz, S. Plasma-Enhanced Chemical Vapor Deposition of Graphene on Copper Substrates. AIP Adv. 2014, 4, 0–9. [Google Scholar] [CrossRef]
- Bekdüz, B.; Beckmann, Y.; Mischke, J.; Twellmann, J.; Mertin, W.; Bacher, G. Graphene Growth through a Recrystallization Process in Plasma Enhanced Chemical Vapor Deposition. Nanotechnology 2018, 29. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.M.; Pei, Y.H.; Wang, X.; Wang, H.; Meng, Q.N.; Tian, H.W.; Zheng, X.L.; Zheng, W.T.; Liu, Y.C. Synthesis of Graphene on a Polycrystalline Co Film by Radio-Frequency Plasma-Enhanced Chemical Vapour Deposition. J. Phys. D. Appl. Phys. 2010, 43. [Google Scholar] [CrossRef]
- Lee, S.; Park, W.K.; Yoon, Y.; Baek, B.; Yoo, J.S.; Kwon, S. Bin; Kim, D.H.; Hong, Y.J.; Kang, B.K.; Yoon, D.H.; et al. Quality Improvement of Fast-Synthesized Graphene Films by Rapid Thermal Chemical Vapor Deposition for Mass Production. Mater. Sci. Eng. B 2019, 242, 63–68. [Google Scholar] [CrossRef]
- Zhang, C.; Zhang, J.; Lin, K.; Huang, Y. Laser-Assisted Chemical Vapor Deposition Setup for Fast Synthesis of Graphene Patterns. Rev. Sci. Instrum. 2017, 88. [Google Scholar] [CrossRef] [PubMed]
- Riedl, C.; Coletti, C.; Starke, U. Structural and Electronic Properties of Epitaxial Graphene on SiC(0001): A Review of Growth, Characterization, Transfer Doping and Hydrogen Intercalation. J. Phys. D. Appl. Phys. 2010, 43. [Google Scholar] [CrossRef]
- Emtsev, K. V.; Bostwick, A.; Horn, K.; Jobst, J.; Kellogg, G.L.; Ley, L.; McChesney, J.L.; Ohta, T.; Reshanov, S.A.; Röhrl, J.; et al. Towards Wafer-Size Graphene Layers by Atmospheric Pressure Graphitization of Silicon Carbide. Nat. Mater. 2009, 8, 203–207. [Google Scholar] [CrossRef] [PubMed]
- Ouerghi, A.; Silly, M.G.; Marangolo, M.; Mathieu, C.; Eddrief, M.; Picher, M.; Sirotti, F.; El Moussaoui, S.; Belkhou, R. Large-Area and High-Quality Epitaxial Graphene on off-Axis Sic Wafers. ACS Nano 2012, 6, 6075–6082. [Google Scholar] [CrossRef] [PubMed]
- De Heer, W.A.; Berger, C.; Ruan, M.; Sprinkle, M.; Li, X.; Hu, Y.; Zhang, B.; Hankinson, J.; Conrad, E. Large Area and Structured Epitaxial Graphene Produced by Confinement Controlled Sublimation of Silicon Carbide. Proc. Natl. Acad. Sci. U. S. A. 2011, 108, 16900–16905. [Google Scholar] [CrossRef] [PubMed]
- Riedl, C.; Coletti, C.; Iwasaki, T.; Zakharov, A.A.; Starke, U. Quasi-Free-Standing Epitaxial Graphene on SiC Obtained by Hydrogen Intercalation. Phys. Rev. Lett. 2009, 103, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Tzalenchuk, A.; Lara-Avila, S.; Kalaboukhov, A.; Paolillo, S.; Syväjärvi, M.; Yakimova, R.; Kazakova, O.; Janssen, T.J.B.M.; Fal’Ko, V.; Kubatkin, S. Towards a Quantum Resistance Standard Based on Epitaxial Graphene. Nat. Nanotechnol. 2010, 5, 186–189. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Hwang, J.; Shields, V.B.; Tiwari, S.; Spencer, M.G.; Lee, J.W. SiC Surface Orientation and Si Loss Rate Effects on Epitaxial Graphene. Nanoscale Res. Lett. 2012, 7, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Zhang, Y.; Zhang, Y.; Lei, T.; Guo, H.; Wang, Y.; Tang, X.; Wang, H. Raman Analysis of Epitaxial Graphene on 6H-SiC (0001) Substrates under Low Pressure Environment. J. Semicond. 2011, 32. [Google Scholar] [CrossRef]
- Al-Temimy, A.; Riedl, C.; Starke, U. Growth and Characterization of Epitaxial Graphene on SiC Induced by Carbon Evaporation. Mater. Sci. Forum 2010, 645–648, 593–596. [Google Scholar] [CrossRef]
- Lei, T.M.; Deng, P.F.; Zhang, Y.M.; Guo, H. Epitaxial Graphene Growth on 6H-SiC (0001) Substrate by Confinement Controlled Sublimation of Silicon Carbide. Adv. Mater. Res. 2013, 709, 62–65. [Google Scholar] [CrossRef]
- Robinson, Z.R.; Jernigan, G.G.; Bussmann, K.M.; Nyakiti, L.O.; Garces, N.Y.; Nath, A.; Wheeler, V.D.; Myers-Ward, R.L.; Gaskill, D.K.; Eddy, C.R. Graphene Growth on SiC(000-1): Optimization of Surface Preparation and Growth Conditions. Carbon Nanotub. Graphene, Emerg. 2D Mater. Electron. Photonic Devices VIII 2015, 9552, 95520Y. [Google Scholar] [CrossRef]
- Vesapuisto, E.; Kim, W.; Novikov, S.; Lipsanen, H.; Kuivalainen, P. Growth Temperature Dependence of the Electrical and Structural Properties of Epitaxial Graphene on SiC(0001). Phys. Status Solidi Basic Res. 2011, 248, 1908–1914. [Google Scholar] [CrossRef]
- Palmer, J.; Kunc, J.; Hu, Y.; Hankinson, J.; Guo, Z.; Berger, C.; De Heer, W.A. Controlled Epitaxial Graphene Growth within Removable Amorphous Carbon Corrals. Appl. Phys. Lett. 2014, 105. [Google Scholar] [CrossRef]
- Göckeritz, R.; Schmidt, D.; Beleites, M.; Seifert, G.; Krischok, S.; Himmerlich, M.; Pezoldt, J. High Temperature Graphene Formation on Capped and Uncapped SiC. Mater. Sci. Forum 2011, 679–680, 785–788. [Google Scholar] [CrossRef]
- Mishra, N.; Boeckl, J.J.; Tadich, A.; Jones, R.T.; Pigram, P.J.; Edmonds, M.; Fuhrer, M.S.; Nichols, B.M.; Iacopi, F. Solid Source Growth of Graphene with Ni-Cu Catalysts: Towards High Quality in Situ Graphene on Silicon. J. Phys. D. Appl. Phys. 2017, 50. [Google Scholar] [CrossRef]
| Methods | FTIR | SEM | Raman | XRD | Ref. |
| Sonication during oxidation (Hummers method) | Decrease in O–H and C=O functional groups due to strong sonication | Flexible sheet structure with lower oxidation | Increased (many defects) | Intense (002) peak at 2θ = 26.5°, indicating dominant graphite phase | [26] |
| Sonication for 1–5 hours (with Tween 80) | – | Lateral size decreased from ~5 μm to 317 nm | Ratio increased gradually with longer sonication time | – | [27] |
| Sonication for 10/20 minutes (30/50 W) | O–H and COOH peak intensities decreased (sample S1 to S4) | Morphology becomes increasingly deformed (S1 to S4) | Increased from 0.84 to 0.95, indicating increased structural disorder | (002) peak shifted from 2θ = 11.34° to 11.14° | [28] |
| Sonication for 15–45 minutes (electrochemical method) | Appearance of C–O–H vibration, indicating the presence of hydroxyl group | – | – | (002) peak intensity weakened; crystallite size decreased | [29] |
| Sonication for 8.5 hours (ethanol: water = 20:80) | Peaks observed O–H, C=C, and C–H | Spherical morphology with particle size ~23–41 nm | ID/IG ratio approximately 0.65, suggesting a moderate level of defects | Broad (002) peak at 2θ ~25°; crystallite size ~20 nm | [30] |
| Wet Ball-Mill Method | |||||
| Sample | FTIR | Morphology (SEM/TEM) | Raman (ID/IG) | XRD | Ref. |
| Graphite + DMF | C=O stretching (~1700 cm⁻¹) | Thin sheets with folded edges; few-layer structure (0.8–1.8 nm) | Increased, indicating higher disorder due to milling | (002) peak broadened | [34] |
| Graphite + Water + KClO₄ | C–O stretching (~1060 cm⁻¹) | Small layered nanosheets, graphene oxide (GO) formed | Increased with longer milling time | (002) peak broadened | [31] |
| Graphite + Water | C=O functional group observed | Large aggregates (BOTTOM60), finer sheets (TOP60); few-layer structure | High value (TOP60), suggesting a small sheet size and more defects | - | [35] |
| Graphite + Ethanol: Water (7:3) | - | Graphene-encapsulated SiC; few-layer structure | Decreased with increasing speed, indicating improved quality | Graphite peak intensity decreased | [36] |
| Graphite + Water | - | More uniform particles; presence of individual sheets | Lowest value at 500 rpm (0.221), indicating high-quality graphene | (002) peak became sharper | [37] |
| Dry Ball-Mill Method | |||||
| Sample | FTIR | Morphology (SEM/TEM) | Raman (ID/IG) | XRD | Ref. |
| Graphite | - | Nanoparticles with irregular shapes | Increased, suggesting greater defect formation | Crystallinity decreased (weakened graphite peak) | [38] |
| Graphite | C=O, C–O functional groups | Shaft-like structure with reduced particle size | Increased, reflecting higher structural disorder | Graphite peak intensity decreased | [39] |
| Graphite | C=O, OH, COOH groups | Thin sheets with open structure; <10 layers | Increased, indicating defect generation during milling | Graphite peaks broadened | [40] |
| Graphite | OH, C=O groups | Rough surface morphology; reduced particle size | Increased (from 0.21 to 0.97), supporting oxidation process | 2θ peaks shifted and broadened | [41] |
| Graphite | - | Thin and layered flake morphology | - | 2θ peaks shifted and broadened | [32] |
| Parameters | VFD | PFD | MFD |
| Thickness | Ranges from < 1 nm to > 20 nm | Up to 79% ≤ 1.5 nm (after 8 hours at 15 MPa) | Average ~1.5 nm; up to 92% ≤ 1.5 nm (after 3 hours) |
| Number of Layers | 1 to > 20 layers | ≤ 5 layers: 29% (0.5 h), 63% (4 h), 79% (8 h) | Average < 5 layers; stable across various exfoliation durations |
| Lateral Size / Area | < 1 μm | Over 85% of flakes < 0.1 μm² (after 8 hours) | Average ~320 nm (AFM); ~0.5 μm (Raman in protein medium) |
| Thickness Distribution | Uneven; limited data available | Becomes thinner and more uniform over time | Remains stable around 1.5 nm; shifts toward thinner layers |
| Defect (Raman/XPS) | Minimal defects | Low defect levels, mainly at the edges | Very low defect levels; basal planes largely defect-free |
| Sample | AFM | Raman | XRD | Ref. |
| Graphite + SC-CO₂ | >10 layers | Weak 2D peak; high ID/IG ratio indicating limited exfoliation | Intense (002) peak with slight broadening, indicating minor delamination | [47] |
| Graphite + SC-CO₂ | Majority <3 layers (yield ~28%) | 88% <3 layers; sharp 2D peak confirms few-layer graphene | Clear graphene structure; no signs of oxidation | [48] |
| Graphite + SC ethanol | ~1.0–1.2 nm thickness; 6–10% monolayer content; stable | Low ID/IG ratio (~0.17); symmetric 2D peak at 2684 cm⁻¹ | (002) peak intensity decreased; increased interlayer spacing | [49] |
| Graphite + SC ethanol | Few layers (maximum yield ~18.5%) | Slight increase in ID/IG ratio; minor defects introduced | Decrease in (002) peak intensity; successful exfoliation | [50] |
| Methods | TEM | Raman (ID/IG) | XRD (2θ) | Ref. |
| C₂H₂ + O₂ gas detonation (ratio 0.4–0.8) | 2–3 layers; monolayers observed; lateral size increases with higher O₂ ratio | Decreases from ~1.33 to ~0.28, indicating reduced defects and improved crystallinity | (002) peak at 26.05°, close to graphite (26.6°), indicating preserved graphite structure | [53] |
| O₂/C₂H₂ detonation (O/C ratio 0.25–0.75) | 8–30 layers; turbostratic structure; lateral size 20–200 nm | Decreases when O/C > 0.5, suggesting fewer defects and a more ordered structure | (002) peak shifts from 25.33° to 25.74°, lower than graphite, indicating increased interlayer spacing | [52] |
| Solid explosive: CaCO₃ + Mg + RDX | <10 layers; transparent and crumpled sheets | ~0.26, indicating very few defects and high-quality graphene | (002) peak at 26.04°, close to graphite, suggesting good crystallinity | [51] |
| GO to ER-GO (thermal reduction at 100°C) | Thin, transparent sheets | No numerical value reported; D and G bands are present, indicating moderate defect density | GO: 7.9° (d ≈ 1.09 nm); ER-GO: approximately 26.3°, indicating partial restoration of graphite-like structure | [54] |
| Oxidation | Reduction | FTIR | Raman (ID/IG) | XRD | Ref. |
| Thermal | Thermal | High intensity of –OH, C=O, and C–O functional groups | Decreases with increasing temperature and time | (002) peak at 26.5°, interlayer spacing d ≈ 3.36 Å | [75] |
| Chemical (Hummers) | Chemical (NaBH₄) | High –OH and C=O intensity; epoxy group is reduced | 0.98 | Interlayer spacing d = 0.388 nm; average number of layers ≈ 1.4; crystallite size ≈ 22 nm | [70] |
| Electrochemical | Chemical (hydrazine) | Presence of –OH, C–O–C, C–C, and C=O groups | 0.849 | 2θ = 26.52°; grain size ≈ 23 nm | [76] |
| Chemical (Hummers) | Electrochemical | Decrease in –OH, C=O, and C–O functional groups | 1.24 | Interlayer spacing d = 0.3554 nm | [77] |
| Chemical (Hummers) | Chemical (Ascorbic acid) | Decrease in C=O, C–OH, and C–O–C; partial restoration of sp² structure | Decreases from 0.805 to 0.788 with increasing temperature | GO peak at ~11.9°; rGO peaks between 24.8° and 25.2°; d-spacing ≈ 3.55 Å | [78] |
| Chemical (Hummers) | Chemical (hydrazine) | C=O and C–O functional groups are reduced | 2.23 | GO: ~10.9° (001); rGO: ~26.4° (002); interlayer spacing decreases | [79] |
| Chemical (Hummers) | Thermal | C=O and C–OH groups are reduced | - | GO: ~10.5°; rGO: 24.7° to 26.2°; d-spacing for GO ≈ 0.84 nm; for rGO ≈ 0.34–0.36 nm | [80] |
| Chemical (Hummers) | Chemical (Zn metal) | Decrease in C–O–C, C–OH, and C=O groups; oxygen-containing groups are weakened | 1.01 | GO (002) peak at ~26°; additional ZnO peak with wurtzite structure observed | [81] |
| Types of Gas | Result | Ref. |
| Ar | ~12% graphene sheets comprising 1–10 layers; interlayer spacing of 0.34–0.39 nm | [85] |
| Ar | High-quality, very pure 4-layer graphene | [86] |
| H2 | 2–4-layer graphene, free of nanotube contaminants | [87] |
| H2-N2 | Up to 5 layers of graphene, low defect density, suitable for mass production | [88] |
| Aspects | Air conditioning | DC | Ref. |
| Structure | Nanohorns, carbon onions, 1–5 layers of graphene | Carbon nanotubes, 2–4 layers of graphene | [89],[88],[86],[87] |
| Purity | Very pure; minimal carbon contamination | Less pure; mixed with non-graphitic carbon | [89], [86] |
| Process & Control | Flexible, frequency-controlled process | Less flexible; dependent on gas, metal catalyst & pressure | [89],[90],[91] |
| Graphene Quality | Low defects, optimal with N₂/H₂ mixture | Minimal defects, optimal with argon gas | [88],[86] |
| Scalability | Large-scale, suitable for industry | Suitable for small–to medium-scale and specialized applications | [88],[91] |
| Efficiency & Results | High efficiency when optimizing gas composition & frequency | Generally lower efficiency than the air-conditioning method | [88],[90] |
| Pressure | Number of Layers | Purity | Ref. |
| Low | Formation of nanohorns and nanospheres, no coated graphene layers | Low purity, a hybrid of various carbon nanostructures | [92] |
| Moderate | Approximately 4-layer graphene, thermally and structurally stable | High purity, well-ordered structure without toxic intercalates | [86] |
| High | Graphene with 2–10 layers, pronounced condensation observed | High purity, uniform, defect-free graphene structure | [92] |
| Temperature | Number of Layers | Purity | Ref. |
| Low | Single-layer graphene grown at low energy | High purity, low-carbon atomic mobility, minimal defects | [93] |
| Moderate | 2–4 layer graphene with balanced growth energy | High purity, stable plasma, minimal fouling | [86], [87] |
| High | > 4-layer graphene; rapid growth due to high energy | Lower purity, increased mobility lead to more defects and contamination | [93], [94] |
| Duration | Number of Layers | Purity | Ref. |
| Short | 2–4 layer graphene | High purity; slightly distorted structure | [87], [95] |
| Moderate | 4–6 layer graphene | High purity; some layer non-uniformity and minor structural flaws | [82], [94] |
| Long | Up to ~20 layers graphene | Decreased purity; more defects, mitigable with buffer gas | [83] |
| Chamber Type | Number of Layers | Purity | Ref. |
| Closed chamber | Approximately 4 layers of graphene | High purity, minimal defects | [82],[86] |
| Semi-open chamber | Moderate, depending on parameters | Moderate purity, possible contamination | [82] |
| Open chamber | Multilayer graphene (many layers) | Low purity, high defect density | [96] |
| Method | Number of Layers | Purity | Ref. |
| Oxidative | Single to multi-layer graphene | Multiple defects; presence of oxygen-containing functional groups | [100],[106],[107] |
| Catalytic | 4 – 8 layers graphene | Slight defects, traces of residual metal catalyst | [103] |
| Electrochemical | Single to multi-layer graphene | High purity, minimal defects | [108] |
| Sonochemistry | Predominantly bilayer graphene, with some monolayer | Smooth edges, low interference | [109] |
| Ionic Liquids | SEG (mg/mL) | Number of Layers |
| [C₄C₁im][Ntf₂] | ~1.8 | ≤5 |
| [Pyrr₄,₁][Ntf₂] | ~1.8 | ≤5 |
| [N₄,₁,₁,₁][Ntf₂] | ~1.8 | ≤5 |
| [C₁₀C₁im][Ntf₂] | <1.8 | ≤5 |
| [BnzmC₁im][Ntf₂] | <1.8 | ≤5 |
| [C₄C₁im][C(CN)₃] | <0.5 | >30 |
| [C₂C₁im][N(CN)₂] | <0.5 | ≤5 |
| [C₄C₁im][C₁SO₄] | <0.5 | ≤5 |
| [C₂C₁im][OTF] | <0.5 | <5 |
| Surfactant | CG Max (mg/mL) | Optimal Csur (mg/mL) | CMC (mg/mL) |
| SDOC | ~0.10 | ~1.0 | 5.0 |
| SDBS | ~0.11 | ~0.7 | 0.7 |
| SDS | ~0.09 | ~2.0 | 2.3 |
| HTAB | ~0.12 | ~0.3 | 0.33 |
| Tween 80 | ~0.08 | ~0.015 | 0.0157 |
| Triton X-100 | ~0.29 | ~1.0 | 0.343 |
| Solvent | Boiling Point (°C) | Concentration (mg/mL) | Result |
| Acetone | 56 | ~0.08 | Low concentration, suitable for low-boiling dispersion. |
| Chloroform | 61 | ~0.5 | Stable (≥75 % remains suspended after 100 h), produces medium-sized flakes. |
| Isopropanol | 82 | ~0.5 | Highly stable (>90% fixed suspended >200 hours); produces good quality flakes |
| Cyclohexa-none | 156 | ~1.0 | Effectively exfoliates graphene; high boiling point makes solvent removal challenging. |
| NMP | 204 | ~1.0 | Effectively exfoliates graphene; high boiling point makes solvent removal challenging. |
| DMF | 153 | ~1.0 | Effectively exfoliates graphene; high boiling point makes solvent removal challenging. |
| CVD Temperature | Layers | Defects | Substrate Interactions | Ref. |
| Low | Slow film growth, non-uniform coverage | High defects, insufficient energy | Weak interactions, delamination potential. | [132] |
| High | Fast carbon diffusion, uniform and continuous graphene film | Reduced defects, more perfect structure | Strong interaction, adhesion and stability are better. | [133], [134] |
| Ultra-High | Rapid growth, increased film thickness | Further reduced defects, though new defects may emerge | Potential substrate etching and adverse interactions | [135],[136] |
| CVD Pressure | Coating & Uniformity | Defects | Substrate Interactions | Ref. |
| APCVD | Large area, fairly uniform, limited control | High defect, decreased purity | Strong substrate interaction, difficult to transfer | [138],[133] |
| LPCVD | Uniform, high-quality monolayer | Low defects, high purity | Weak interaction, easy to transfer | [139] |
| Ultra-Vacuum | Highly uniform, high precision | Very low defects, optimal purity | Minimal interaction, ideal for transfer | [140] |
| Aspects | Cold Wall CVD | Hot Wall CVD | Ref. |
| Number of Layers | Uniform and thin layer formation | Thicker layers, less uniform | [142],[143] |
| Defects | Low defects, high purity | Higher defect density, reduced purity | [142],[144] |
| Substrate Influence | Better process control, high-quality graphene | Greater substrate influence; increased defect formation | [145],[133] |
| CVD Method | Coating & Uniformity | Defects | Ref. |
| Continuous | Uniform, suitable for large areas | Low defect density, high material purity | [146],[147] |
| Diconected | Less uniform | Higher defect density, lower purity | [148] |
| Pulsed | Controlled and tunable deposition | Low defect density, high material purity | [135],[136] |
| CVD System | Gas Flow & Dynamics | Defects & Purity | Scalability | Ref. |
| Open | Continuous flow (methane, H₂), stable reaction | Slight defects, high purity | Suitable for large-scale, even gas distribution | [138] |
| Closed | Static gas, layer growth control | High crystallinity & purity, sensitive to reaction conditions | Less suitable for large-scale use without process optimization | [150] |
| Method | Defects & Purity | Layer | Efficiency | Ref. |
| PECVD | Low defects, high purity | SLG–FLG (dependent on parameters) | Fast, low temperature, suitable for large-scale | [152],[153], [154],[155] |
| TCVD | Higher defect density; RT-CVD offers improvement | Generally monolayer | Slow, high temperature, RT-CVD is more efficient | [156] |
| Laser-CVD | Low defects with precise control | Multi-layer (dependent on laser parameters) | Fast, ideal for specific patterns | [157] |
| Parameters | Si-face | C-Face |
| Number of layers | 1–2 layers (monolayer) | Up to ~30 layers (multilayer) |
| Raman Spectrum | ID/IG < 0.02 (sharp G and 2D peaks) | ID/IG < 0.05, broader peaks with creases |
| Electron mobility | Relatively low | Relatively high |
| Temperature | Number of Layers | Electron Mobility | Description |
| Low | 0 – 0.6 layers | Very low (up to 81 cm²/Vs) | Graphene is not fully formed; significant exposure of SiC surface remains |
| Optimal | ~1.2 – 1.4 layers | Highest (~370 cm²/Vs) | Nearly monolayer graphene, maximum mobility, uniform surface morphology |
| High | >1.6 layers | Decreasing (up to 77 cm²/Vs) | Multilayer graphene, presence of grain boundaries and "giraffe stripe" patterns |
| Pressure | Raman | Surface Morphology | Graphene Quality |
| Low Pressure (∼10⁻⁷ mbar) | Weak G and 2D bands, dominant D band | Rough surface with many defects | Low-quality, non-uniform graphene |
| Inert Atmosphere (∼10⁻³ mbar) | Strong G and 2D bands, D band nearly absent | Smooth surface with uniform morphology | High-quality, thin, and uniform graphene layers |
| Method | Number of Layers | Raman | Surface Morphology | Graphene Quality |
| No Catalyst | 6–7 layers | G and 2D wideband, ID/IG > 0.4 | Rough surface, many defects, non-uniform | Low-quality, multilayer graphene with small crystallite |
| With Ni–Cu Catalyst | Monolayer | G and 2D sharp band, ID/IG ~0.24, 2D > G | Smooth surface, uniform morphology | High quality, uniform monolayer graphene with large crystallites (35–60 nm) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




