Submitted:
11 July 2025
Posted:
14 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
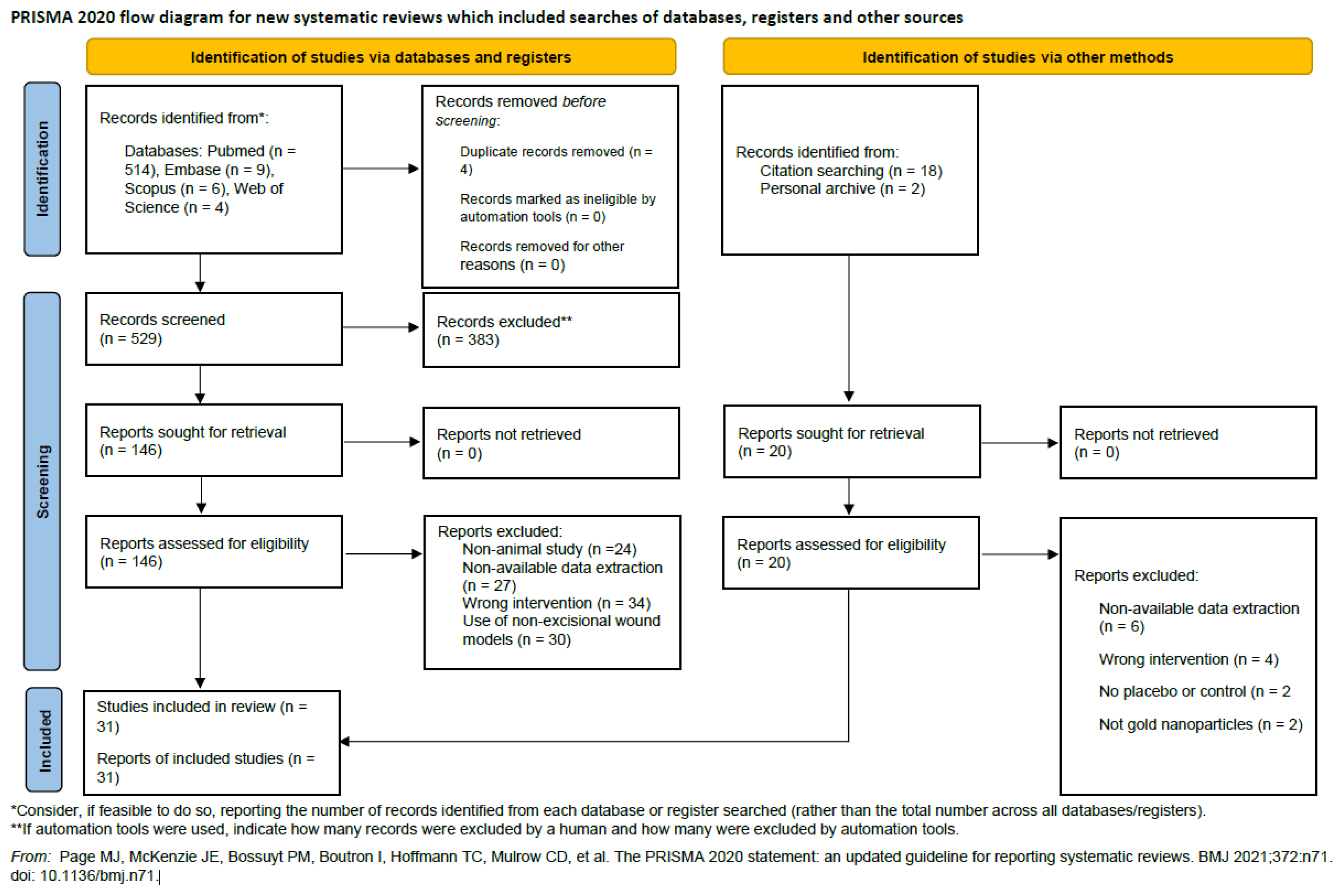
3.1. Study selection
3.2. Study Characteristics
3.4. Dealing with Missing Data
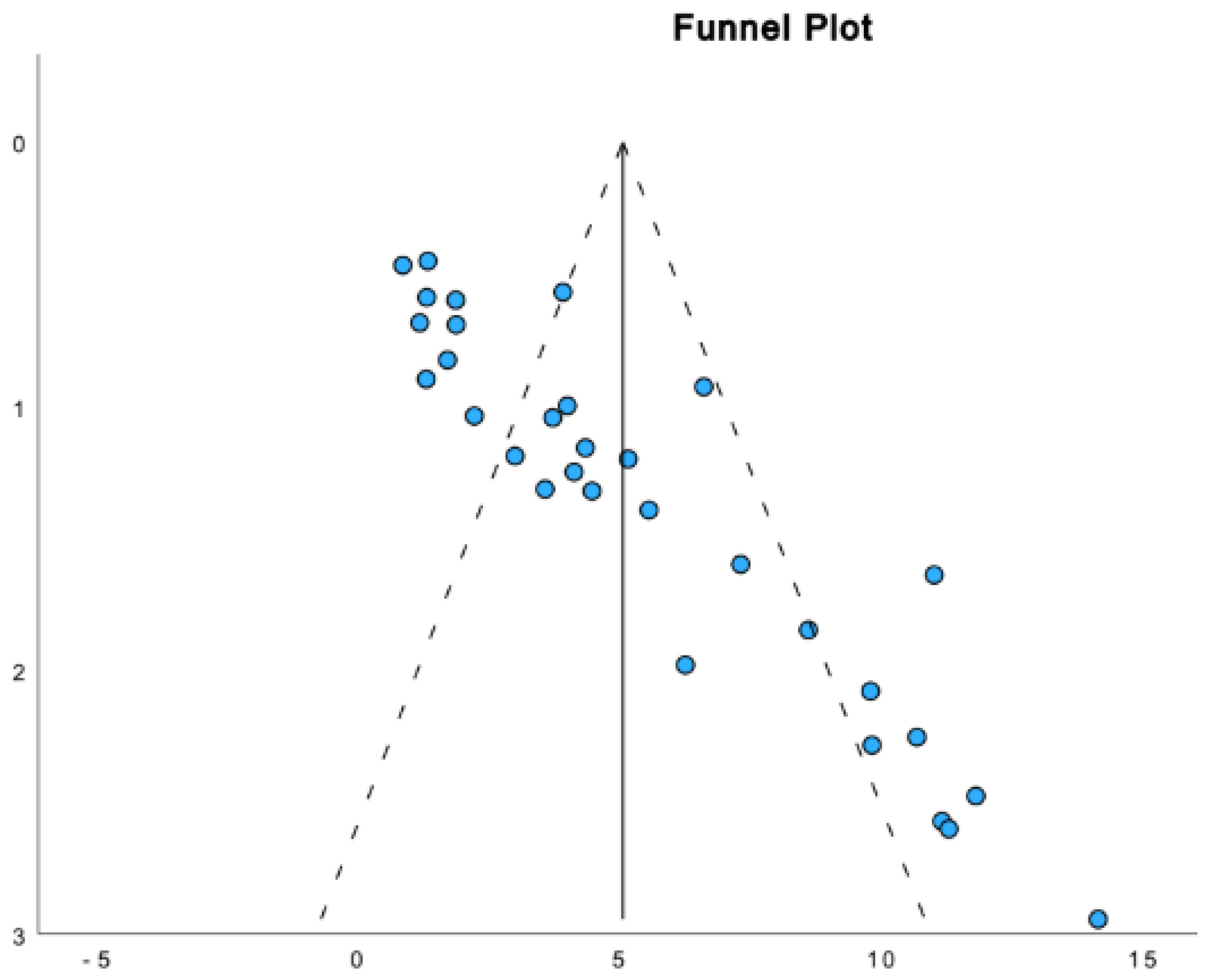
3.5. Statistical Analysis and Pooling
3.6. Meta-Analysis Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
Appendix A
Appendix A.1. Search Strategy
| Source | Thesaurus headings/free text and truncation | Results | Date of search |
| Pubmed | "gold nanoparticle*"[Text Word] "gold nanoparticles"[Text Word] "gold nanorod*"[Text Word] "gold nanoshell*"[Text Word] "AuNPs"[Text Word] "GNPs"[Text Word] "gold nanostructure"[Text Word] "gold nanomaterials"[Text Word] |
39,136 | 14-05-2025 |
| "wound*"[Text Word] "wound healing"[Text Word] "wound repair"[Text Word] "wound treatment"[Text Word] "tissue regeneration"[Text Word] "tissue repair"[Text Word] "skin regeneration"[Text Word] "skin repair"[Text Word] "Wound Healing/drug effects"[MeSH] "Re-Epithelialization/drug effects"[MeSH] |
514,331 | ||
| "randomized controlled trial"[Text Word] "randomised controlled trial"[Text Word] "RCT"[Text Word] "randomized clinical trial"[Text Word] |
723,707 | ||
| (("gold nanoparticle*" OR "gold nanoparticles" OR "gold nanorod*" OR "gold nanoshell*" OR "AuNPs" OR "GNPs" OR "gold nanostructure" OR "gold nanomaterials") AND (wound* OR "wound healing" OR "wound repair" OR "wound treatment" OR "tissue regeneration" OR "tissue repair" OR "skin regeneration" OR "skin repair" OR "Wound Healing/drug effects" OR "Re-Epithelialization/drug effects" OR "randomized controlled trial" OR "randomised controlled trial" OR "RCT" OR "randomized clinical trial")) | 519 | ||
| Embase | (gold nanoparticle* OR gold nanorod* OR gold nanoshell* OR "AuNPs" OR "GNPs") | 15,897 | 14-05-2025 |
| (randomized controlled trial* OR randomised controlled trial* OR random allocation OR random* OR "RCT") | 2,431,698 | ||
| (wound healing OR wound repair OR wound* OR tissue regeneration OR tissue repair OR skin regeneration OR skin repair) | 614,077 | ||
| (gold nanoparticle* OR gold nanorod* OR gold nanoshell* OR "AuNPs" OR "GNPs") AND (randomized controlled trial* OR randomised controlled trial* OR random allocation OR random* OR "RCT") AND (wound healing OR wound repair OR wound* OR tissue regeneration OR tissue repair OR skin regeneration OR skin repair) | 9 | ||
| Cochrane | (("gold" NEXT nanoparticle*) OR ("gold" NEXT nanorod*) OR ("gold" NEXT nanoshell*) OR "AuNPs" OR "GNPs") | 35 | 14-05-2025 |
| (randomized controlled trial OR randomised controlled trial OR randomized OR randomised OR RCT) | 8942 | ||
| (wound NEXT healing OR wound NEXT repair OR wound* OR tissue NEXT regeneration OR tissue NEXT repair OR skin NEXT regeneration OR skin NEXT repair) | 494 | ||
| (("gold" NEXT nanoparticle*) OR ("gold" NEXT nanorod*) OR ("gold" NEXT nanoshell*) OR "AuNPs" OR "GNPs") AND (randomized controlled trial OR randomised controlled trial OR randomized OR randomised OR RCT) AND (wound NEXT healing OR wound NEXT repair OR wound* OR tissue NEXT regeneration OR tissue NEXT repair OR skin NEXT regeneration OR skin NEXT repair) |
|||
| Scopus | (TITLE-ABS-KEY("gold nanoparticle*" OR "gold nanorod*" OR "gold nanoshell*" OR "AuNPs" OR "GNPs")) | 101,410 | 14-05-2025 |
| (TITLE-ABS-KEY("randomized controlled trial" OR "randomised controlled trial" OR "randomized" OR "randomised" OR "RCT")) | 1,465,658 | ||
| (TITLE-ABS-KEY("wound healing" OR "wound repair" OR "wound*" OR "tissue regeneration" OR "tissue repair" OR "skin regeneration" OR "skin repair")) | 750,089 | ||
| (TITLE-ABS-KEY("gold nanoparticle*" OR "gold nanorod*" OR "gold nanoshell*" OR "AuNPs" OR "GNPs")) AND (TITLE-ABS-KEY("randomized controlled trial" OR "randomised controlled trial" OR "randomized" OR "randomised" OR "RCT")) AND (TITLE-ABS-KEY("wound healing" OR "wound repair" OR "wound*" OR "tissue regeneration" OR "tissue repair" OR "skin regeneration" OR "skin repair")) |
|||
| Web of Science | TS=("gold nanoparticle*" OR "gold nanorod*" OR "gold nanoshell*" OR "AuNPs" OR "GNPs") |
127,319 | 14-05-2025 |
| (TS=("randomized controlled trial" OR "randomised controlled trial" OR "randomized" OR "randomised" OR "RCT")) | 1,153,119 | ||
| TS=("wound healing" OR "wound repair" OR "wound*" OR "tissue regeneration" OR "tissue repair" OR "skin regeneration" OR "skin repair") |
341,754 | ||
| (TS=("gold nanoparticle*" OR "gold nanorod*" OR "gold nanoshell*" OR "AuNPs" OR "GNPs")) AND (TS=("randomized controlled trial" OR "randomised controlled trial" OR "randomized" OR "randomised" OR "RCT")) AND (TS=("wound healing" OR "wound repair" OR "wound*" OR "tissue regeneration" OR "tissue repair" OR "skin regeneration" OR "skin repair")) |
5 |
References
- Raziyeva, K.; Kim, Y.; Zharkinbekov, Z.; Kassymbek, K.; Jimi, S.; Saparov, A. Immunology of Acute and Chronic Wound Healing. Biomolecules 2021, 11, 700. [CrossRef]
- Choudhary, V.; Choudhary, M.; Bollag, W.B. Exploring Skin Wound Healing Models and the Impact of Natural Lipids on the Healing Process. International Journal of Molecular Sciences 2024, 25, 3790. [CrossRef]
- Landén, N.X.; Li, D.; Ståhle, M. Transition from inflammation to proliferation: a critical step during wound healing. Cell Mol Life Sci 2016, 73, 3861. [CrossRef]
- Atkin, L. Chronic wounds: The challenges of appropriate management. Br J Community Nurs 2019, 24, S26. [CrossRef]
- Chan, B.C.L. Hypericin and Pheophorbide a Mediated Photodynamic Therapy Fighting MRSA Wound Infections: A Translational Study from In Vitro to In Vivo. Pharmaceutics 2021, 13, 1399. [CrossRef]
- Bui, U.T. Assessment, management and prevention of chronic wounds in the Australian context: a scoping review. Wound Practice and Research 2023, 31, 120. [CrossRef]
- Sen, C.K. Human Wound and Its Burden: Updated 2020 Compendium of Estimates. Adv Wound Care (New Rochelle) 2021, 10, 281. [CrossRef]
- Frykberg, R.G.; Banks, J. Challenges in the Treatment of Chronic Wounds. Adv Wound Care (New Rochelle) 2015, 4, 560. [CrossRef]
- Lau, P.S. Influence of gold nanoparticles on wound healing treatment in rat model: Photobiomodulation therapy. Lasers Surg Med 2017, 49, 380. [CrossRef]
- Korani, S. Evaluation of Antimicrobial and Wound Healing Effects of Gold Nanoparticles Containing Abelmoschus esculentus (L.) Aqueous Extract. Bioinorg Chem Appl 2021, 2021, 7019130. [CrossRef]
- Verma, R.; Gupta, P.P.; Satapathy, T.; Roy, A. A review of wound healing activity on different wound models. Journal of Applied Pharmaceutical Research 2019, 7, 1. [CrossRef]
- Nukaly, H.Y.; Ansari, S.A. An Insight Into the Physicochemical Properties of Gold Nanoparticles in Relation to Their Clinical and Diagnostic Applications. Cureus 2023, 15. [CrossRef]
- Bessa, L.J.; Fazii, P.; Di Giulio, M.; Cellini, L. Bacterial isolates from infected wounds and their antibiotic susceptibility pattern: some remarks about wound infection. Int Wound J 2015, 12, 47. [CrossRef]
- Martinengo, L. et al. Prevalence of chronic wounds in the general population: systematic review and meta-analysis of observational studies. Ann Epidemiol 2019, 29, 8. [CrossRef]
- Gould, L. et al. Chronic Wound Repair and Healing in Older Adults: Current Status and Future Research. Wound Repair Regen 2015, 23, 1. [CrossRef]
- Gethin, G.; Van Netten, J.J.; Probst, S.; Care, W. The impact of patient health and lifestyle factors on wound healing, part 2. Journal of Wound Management 2022.
- Mustoe, T.A.; O’Shaughnessy, K.; Kloeters, O. Chronic wound pathogenesis and current treatment strategies: A unifying hypothesis. Plast Reconstr Surg 2006, 117. [CrossRef]
- Batool, Z. et al. Hydrogel assisted synthesis of gold nanoparticles with enhanced microbicidal and in vivo wound healing potential. Sci Rep 2022, 12. [CrossRef]
- Apelqvist, J. Diagnostics and treatment of the diabetic foot. Endocrine 2012, 41, 384. [CrossRef]
- Lipsky, B.A. et al. Guidelines on the diagnosis and treatment of foot infection in persons with diabetes (IWGDF 2019 update). Diabetes Metab Res Rev 2020, 36. [CrossRef]
- Uçkay, I.; Gariani, K.; Pataky, Z.; Lipsky, B.A. Diabetic foot infections: state-of-the-art. Diabetes Obes Metab 2014, 16, 305. [CrossRef]
- Mariadoss, A.V.A.; Sivakumar, A.S.; Lee, C.H.; Kim, S.J. Diabetes mellitus and diabetic foot ulcer: Etiology, biochemical and molecular based treatment strategies via gene and nanotherapy. Biomedicine & Pharmacotherapy 2022, 151, 113134. [CrossRef]
- Martínez, S.P.H. et al. A novel gold calreticulin nanocomposite based on chitosan for wound healing in a diabetic mice model. Nanomaterials 2019, 9. [CrossRef]
- Edsberg, L.E.; Black, J.M.; Goldberg, M.; McNichol, L.; Moore, L.; Sieggreen, M. Revised National Pressure Ulcer Advisory Panel Pressure Injury Staging System. J Wound Ostomy Continence Nurs 2016, 43, 585. [CrossRef]
- Coleman, S. et al. Patient risk factors for pressure ulcer development: Systematic review. Int J Nurs Stud 2013, 50, 974. [CrossRef]
- Li, Z.; Lin, F.; Thalib, L.; Chaboyer, W. Global prevalence and incidence of pressure injuries in hospitalised adult patients: A systematic review and meta-analysis. Int J Nurs Stud 2020, 105, 103546. [CrossRef]
- Borojeny, L.; Albatineh, A.; Dehkordi, A.; Gheshlagh, R. The incidence of pressure ulcers and its associations in different wards of the hospital: A systematic review and meta-analysis. Int J Prev Med 2020, 11, 171. [CrossRef]
- Bai, Z.; Wang, H.; Sun, H.; Cui, L. Effect of hyperbaric oxygen therapy on the patients with venous leg ulcer: A systematic review and meta-analysis. Asian J Surg 2023, 46, 4131. [CrossRef]
- Nelson, E.A.; Adderley, U. Venous leg ulcers. BMJ Clin Evid 2016. [PMCID: PMC4714578].
- Neumann, H.A.M. et al. Evidence-based (S3) guidelines for diagnostics and treatment of venous leg ulcers. J Eur Acad Dermatol Venereol 2016, 30, 1843. [CrossRef]
- Probst, S.; et al. Prevalence and incidence of venous leg ulcers—A systematic review and meta-analysis. Int Wound J 2023, 20, 3906–3921. [CrossRef]
- Rittié, L. Cellular mechanisms of skin repair in humans and other mammals. J Cell Commun Signal 2016, 10, 103–120. [CrossRef]
- Dabiri, G.; Damstetter, E.; Phillips, T. Choosing a Wound Dressing Based on Common Wound Characteristics. Adv Wound Care (New Rochelle) 2016, 5, 32–41. [CrossRef]
- Tan, S.H.; Ngo, Z.H.; Sci, D.B.; Leavesley, D.; Liang, K. Recent Advances in the Design of Three-Dimensional and Bioprinted Scaffolds for Full-Thickness Wound Healing. Tissue Eng Part B Rev 2022, 28, 160–181. [CrossRef]
- Cherng, J.H.; et al. Hemostasis and Anti-Inflammatory Abilities of AuNPs-Coated Chitosan Dressing for Burn Wounds. J Pers Med 2022, 12, 1089. [CrossRef]
- Zhang, W.; et al. Hydrogel-based dressings designed to facilitate wound healing. Mater Adv 2024, 5, 1364–1394. [CrossRef]
- Chen, W.; et al. A novel wound dressing based on a gold nanoparticle self-assembled hydrogel to promote wound healing. Mater Adv 2023, 4, 2918–2925. [CrossRef]
- Dong, H.; et al. Ultrasmall gold nanoparticles/carboxymethyl chitosan composite hydrogel: Tough, restorable, biocompatible antimicrobial dressing for wound healing. Appl Mater Today 2024, 38, 102206. [CrossRef]
- Korani, S.; et al. Evaluation of Antimicrobial and Wound Healing Effects of Gold Nanoparticles Containing Abelmoschus esculentus (L.) Aqueous Extract. Bioinorg Chem Appl 2021, 2021, 7019130. [CrossRef]
- Chen, W.; et al. A novel wound dressing based on a gold nanoparticle self-assembled hydrogel to promote wound healing. Mater Adv 2023, 4, 2918–2925. [CrossRef]
- Danscher, G.; Rasmussen, S. nanoGold and µGold inhibit autoimmune inflammation: a review. Histochem. Cell Biol. 2023, 159, 225–232. [CrossRef]
- Rasmussen, S.; Frederickson, C.; Danscher, G. Inhibition of Local Inflammation by Implanted Gold: A Narrative Review of the History and Use of Gold. J. Rheumatol. 2023, 50, 704–705. [CrossRef]
- Rasmussen, S.; et al. Intra-articular injection of gold micro-particles with hyaluronic acid for painful knee osteoarthritis. BMC Musculoskelet. Disord. 2024, 25, 1–9. [CrossRef]
- Rasmussen, S.; Skjoldemose, E.; Jørgensen, N.K. Intraarticular gold microparticles using hyaluronic acid as the carrier for hip osteoarthritis. A 2-year follow-up pilot study. Sci. Rep. 2024, 14, 1–7. [CrossRef]
- Fu, C.; Jiang, Y.; Yang, X.; Wang, Y.; Ji, W.; Jia, G. Mussel-inspired gold nanoparticle and PLGA/L-lysine-g-graphene oxide composite scaffolds for bone defect repair. Int. J. Nanomedicine 2021, 16, 6693–6718. [CrossRef]
- Mostafa, A.A.; El-Sayed, M.M.H.; Emam, A.N.; Abd-Rabou, A.A.; Dawood, R.M.; Oudadesse, H. Bioactive glass doped with noble metal nanoparticles for bone regeneration: in vitro kinetics and proliferative impact on human bone cell line. RSC Adv. 2021, 11, 25628–25638. [CrossRef]
- Wang, T.; et al. Multifunctional gold clusterzymes with distinct glucose depletion and macrophage reprogramming capability towards regulating the regeneration cascade. Chem. Eng. J. 2024, 482, 149068. [CrossRef]
- Wang, Y.; Zhang, M.; Yan, Z.; Ji, S.; Xiao, S.; Gao, J. Metal nanoparticle hybrid hydrogels: the state-of-the-art of combining hard and soft materials to promote wound healing. Theranostics 2024, 14, 1534. [CrossRef]
- Zhou, S.; Xie, M.; Su, J.; Cai, B.; Li, J.; Zhang, K. New insights into balancing wound healing and scarless skin repair. J. Tissue Eng. 2023, 14, Article ID. [CrossRef]
- Li, Y.Y.; Ji, S.F.; Fu, X.B.; Jiang, Y.F.; Sun, X.Y. Biomaterial-based mechanical regulation facilitates scarless wound healing with functional skin appendage regeneration. Mil. Med. Res. 2024, 11, 1–24. [CrossRef]
- Y. Liu, J. Tan, A. Thomas, D. Ou-Yang, and V. R. Muzykantov. The shape of things to come: importance of design in nanotechnology for drug delivery. Ther Deliv 2012, 3, 181. [CrossRef]
- L. Zhang et al. Tumor Chemo-Radiotherapy with Rod-Shaped and Spherical Gold Nano Probes: Shape and Active Targeting Both Matter. Theranostics 2019, 9, 1893. [CrossRef]
- M. R. Kumalasari, R. Alfanaar, and A. S. Andreani. Gold nanoparticles (AuNPs): A versatile material for biosensor application. Talanta Open 2024, 9, 100327. [CrossRef]
- S. Siddique and J. C. L. Chow. Gold Nanoparticles for Drug Delivery and Cancer Therapy. Appl. Sci. 2020, 10, 3824. [CrossRef]
- W. Yang et al. Shape effects of gold nanoparticles in photothermal cancer therapy. Mater. Today Sustain. 2021, 13, 100078. [CrossRef]
- T. Muthukumar, Sudhakumari, B. Sambandam, A. Aravinthan, T. P. Sastry, and J. H. Kim. Green synthesis of gold nanoparticles and their enhanced synergistic antitumor activity using HepG2 and MCF7 cells and its antibacterial effects. Process Biochem. 2016, 51, 384–391. [CrossRef]
- P. Baei, S. Jalili-Firoozinezhad, S. Rajabi-Zeleti, M. Tafazzoli-Shadpour, H. Baharvand, and N. Aghdami. Electrically conductive gold nanoparticle-chitosan thermosensitive hydrogels for cardiac tissue engineering. Mater. Sci. Eng. C 2016, 63, 131–141. [CrossRef]
- K. F. Bruggeman, R. J. Williams, and D. R. Nisbet. Dynamic and Responsive Growth Factor Delivery from Electrospun and Hydrogel Tissue Engineering Materials. Adv. Healthc. Mater. 2018, 7, 1700836. [CrossRef]
- Z. Wang, W. Hu, W. Wang, Y. Xiao, Y. Chen, and X. Wang. Antibacterial Electrospun Nanofibrous Materials for Wound Healing. Adv. Fiber Mater. 2022, 5, 107–129. [CrossRef]
- P. Pan et al. Recent Advances in Multifunctional Microneedle Patches for Wound Healing and Health Monitoring. Adv. Nanobiomed Res. 2023, 3, 2200126. [CrossRef]
- Priya, S.; Tomar, Y.; Desai, V.M.; Singhvi, G. Enhanced skin drug delivery using dissolving microneedles: a potential approach for the management of skin disorders. Expert Opin Drug Deliv 2023, 20, 721–738. [CrossRef]
- Guillot, A.J.; Martínez-Navarrete, M.; Zinchuk-Mironova, V.; Melero, A. Microneedle-assisted transdermal delivery of nanoparticles: Recent insights and prospects. Wiley Interdiscip Rev Nanomed Nanobiotechnol 2023, 15, e1884. [CrossRef]
- Paula, A.; Nozaki, M.; Helena, M.; Lima, M.; Ângela, ·; Moraes, M. Sprayable Bioactive Dressings for Skin Wounds: Recent Developments and Future Prospects. Biomed Mater Devices 2022, 1, 569–586. [CrossRef]
- Patel, V.N.; et al. Comprehensive developmental investigation on simvastatin enriched bioactive film forming spray using the quality by design paradigm: a prospective strategy for improved wound healing. J Drug Target 2024. [CrossRef]
- Page, M.J.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. J Clin Epidemiol 2021, 134, 178–189. [CrossRef]
- Jiang, Y.; Xu, X.; Xiao, L.; Wang, L.; Qiang, S. The Role of microRNA in the Inflammatory Response of Wound Healing. Front Immunol 2022, 13, 852419. [CrossRef]
- Hannoodee, S.; Nasuruddin, D.N. Acute Inflammatory Response. Nature 2024, 206, 20. [CrossRef]
- Ryan, R.; Hill, S.; Prictor, M.; McKenzie, J. Study Quality Guide; Cochrane Consumers and Communication Review Group: Melbourne, Australia, May 2013. Available online: https://cccrg.cochrane.org/sites/cccrg.cochrane.org/files/uploads/StudyQualityGuide_May%202013.pdf.
- Higgins, J.P.T.; et al. Cochrane Handbook for Systematic Reviews of Interventions | Cochrane Training. Cochrane. Available online: https://training.cochrane.org/handbook (accessed on 7 December 2024).
- Macleod, M.R.; O’Collins, T.; Howells, D.W.; Donnan, G.A. Pooling of animal experimental data reveals influence of study design and publication bias. Stroke 2004, 35, 1203–1208. [CrossRef]
- Naraginti, S.; Kumari, P.L.; Das, R.K.; Sivakumar, A.; Patil, S.H.; Andhalkar, V.V. Amelioration of excision wounds by topical application of green synthesized, formulated silver and gold nanoparticles in albino Wistar rats. Mater. Sci. Eng. C 2016, 62, 293–300. [CrossRef]
- Lau, P.S.; et al. Influence of gold nanoparticles on wound healing treatment in rat model: Photobiomodulation therapy. Lasers Surg. Med. 2017, 49, 380–386. [CrossRef]
- Comune, M.; et al. Antimicrobial peptide-gold nanoscale therapeutic formulation with high skin regenerative potential. J. Control. Release 2017, 262, 58–71. [CrossRef]
- Pan, A.; et al. Topical Application of Keratinocyte Growth Factor Conjugated Gold Nanoparticles Accelerate Wound Healing. Nanomedicine 2018, 14, 1619–1628. [CrossRef]
- Wang, L.; et al. Mercaptophenylboronic Acid-Activated Gold Nanoparticles as Nanoantibiotics against Multidrug-Resistant Bacteria. ACS Appl. Mater. Interfaces 2020, 12, 51148–51159. [CrossRef]
- Hu, W.C.; Younis, M.R.; Zhou, Y.; Wang, C.; Xia, X.H. In Situ Fabrication of Ultrasmall Gold Nanoparticles/2D MOFs Hybrid as Nanozyme for Antibacterial Therapy. Small 2020, 16, 2000553. [CrossRef]
- Al-Musawi, S.; et al. Antibacterial activity of honey/chitosan nanofibers loaded with capsaicin and gold nanoparticles for wound dressing. Molecules 2020, 25, 4770. [CrossRef]
- Nor Azlan, A.Y.H.; Katas, H.; Mohamad Zin, N.; Fauzi, M.B. Dual action gels containing DsiRNA loaded gold nanoparticles: Augmenting diabetic wound healing by promoting angiogenesis and inhibiting infection. Eur. J. Pharm. Biopharm. 2021, 169, 78–90. [CrossRef]
- Chen, W.; et al. A novel wound dressing based on a gold nanoparticle self-assembled hydrogel to promote wound healing. Mater. Adv. 2023, 4, 2918–2925. [CrossRef]
- Wang, L.; Zheng, W.; Hou, Q.; Zhong, L.; Li, Q.; Jiang, X. Breathable and Stretchable Dressings for Accelerating Healing of Infected Wounds. Adv. Healthc. Mater. 2022, 11, 2201053. [CrossRef]
- Du, N.; et al. Upcycling of Expanded Polystyrene Waste into Multifunctional Antibacterial Platforms for Microbial Control. ACS Appl. Mater. Interfaces 2025. [CrossRef]
- Luo, J.; et al. Piezoelectric dual-network tough hydrogel with on-demand thermal contraction and sonopiezoelectric effect for promoting infected-joint-skin-wound healing via FAK and AKT signaling pathways. Natl. Sci. Rev. 2025, 12. [CrossRef]
- Choodari Gharehpapagh, A.; Farahpour, M.R.; Jafarirad, S. The biological synthesis of gold/perlite nanocomposite using Urtica dioica extract and its chitosan-capped derivative for healing wounds infected with methicillin-resistant Staphylococcus aureus. Int. J. Biol. Macromol. 2021, 183, 447–456. [CrossRef]
- Sun, Z.; et al. Albumin Broadens the Antibacterial Capabilities of Nonantibiotic Small Molecule-Capped Gold Nanoparticles. ACS Appl. Mater. Interfaces 2019, 11, 45381–45389. [CrossRef]
- Peng, J.; et al. Construction of multifunctional hydrogel containing pH-responsive gold nanozyme for bacteria-infected wound healing. Int. J. Biol. Macromol. 2024, 283. [CrossRef]
- Zhou, Y.; et al. Flexible PDMS-SERS platform for culture-free diagnosis of bacterial infections in clinical wound care. Talanta 2025, 293. [CrossRef]
- Li, S.; et al. Improved stability of KGF by conjugation with gold nanoparticles for diabetic wound therapy. Nanomedicine 2019, 14, 2909–2923. [CrossRef]
- Yao, M.Y.; et al. 白细胞介素4修饰的金纳米酶对糖尿病小鼠全层皮肤缺损的作用. 中华烧伤与创面修复杂志 2023, 39, 15–24. [CrossRef]
- Chao, F.; et al. Sprayable Hydrogel for pH-Responsive Nanozyme-Derived Bacteria-Infected Wound Healing. ACS Appl. Mater. Interfaces 2025. [CrossRef]
- Leu, J.G.; et al. The effects of gold nanoparticles in wound healing with antioxidant epigallocatechin gallate and α-lipoic acid. Nanomedicine 2012, 8, 767–775. [CrossRef]
- Jiang, N.; Liu, X.; Sui, B.; Wang, J.; Liu, X.; Zhang, Z. Using Hybrid MnO₂-Au Nanoflowers to Accelerate ROS Scavenging and Wound Healing in Diabetes. Pharmaceutics 2024, 16, 1244. [CrossRef]
- Xu, X.; et al. Controlled-temperature photothermal and oxidative bacteria killing and acceleration of wound healing by polydopamine-assisted Au-hydroxyapatite nanorods. Acta Biomater. 2018, 77, 352–364. [CrossRef]
- Li, W.; et al. Development of an Antiswelling Hydrogel System Incorporating M2-Exosomes and Photothermal Effect for Diabetic Wound Healing. ACS Nano 2023, 17, 22106–22120. [CrossRef]
- Gerile, S.; Wu, X.; Kang, J.; Qi, Y.; Dong, A. Thiol-terminated N-halamine ligands to photothermal gold nanorods for synergistically combating antibiotic-resistant bacteria. Soft Matter 2024. [CrossRef]
- Raghuwanshi, N.; et al. Synergistic effects of Woodfordia fruticosa gold nanoparticles in preventing microbial adhesion and accelerating wound healing in Wistar albino rats in vivo. Mater. Sci. Eng. C 2017, 80, 252–262. [CrossRef]
- Cai, R.; et al. Sericin-Assisted Green Synthesis of Gold Nanoparticles as Broad-Spectrum Antimicrobial and Biofilm-Disrupting Agents for Therapy of Bacterial Infection. Int. J. Nanomedicine 2025, 20, 3559–3574. [CrossRef]
- Biswal, A.; et al. Nano CaCO₃ mediated in vitro and in vivo wound healing characteristics of chitosan films without added drugs. Int. J. Biol. Macromol. 2025, 307. [CrossRef]
- Ma, P.; et al. Injectable Light-Responsive Hydrogel Dressing Promotes Diabetic Wound Healing by Enhancing Wound Angiogenesis and Inhibiting Inflammation. Polymers (Basel) 2025, 17, 607. [CrossRef]
- Salama, A.; et al. Curcumin-loaded gold nanoparticles with enhanced antibacterial efficacy and wound healing properties in diabetic rats. Int. J. Pharm. 2024, 666, 124761. [CrossRef]
- Yang, F.; Bai, X.; Dai, X.; Li, Y. The Biological Processes During Wound Healing. Regen. Med. 2021, 16, 373–390. [CrossRef]
- Li, S.; et al. Improved stability of KGF by conjugation with gold nanoparticles for diabetic wound therapy. Nanomedicine 2019, 14, 2909–2923. [CrossRef]
- Jiang, M.; Nie, R.; Kang, J.; Li, P.; Dong, A. Mild Phototherapy Strategies for Preventing Pathogen Infection and Enhancing Cell Proliferation in Diabetic Wound. Adv. Healthc. Mater. 2025. [CrossRef]
- Hammami, I.; Alabdallah, N.M.; Al jomaa, A.; Kamoun, M. Gold nanoparticles: Synthesis properties and applications. J. King Saud Univ. Sci. 2021, 33, 101560. [CrossRef]
- Rodríguez-León, E.; et al. Synthesis of Gold Nanoparticles Using Mimosa tenuiflora Extract, Assessments of Cytotoxicity, Cellular Uptake, and Catalysis. Nanoscale Res. Lett. 2019, 14, 1–16. [CrossRef]
- Mahmoud, N.N.; et al. Investigating Inflammatory Markers in Wound Healing: Understanding Implications and Identifying Artifacts. ACS Pharmacol. Transl. Sci. 2024, 7, 18–27. [CrossRef]
- Zhao, L.; et al. Topical drug delivery strategies for enhancing drug effectiveness by skin barriers, drug delivery systems and individualized dosing. Front. Pharmacol. 2024, 14, 1333986. [CrossRef]
- Yang, W.; et al. Shape effects of gold nanoparticles in photothermal cancer therapy. Mater. Today Sustain. 2021, 13, 100078. [CrossRef]
- Diller, R.B.; Tabor, A.J. The Role of the Extracellular Matrix (ECM) in Wound Healing: A Review. Biomimetics 2022, 7, 87. [CrossRef]
- Inbathamizh, L.; Varthan, M.K.H.; Kumar, R.S.R.; Rohinth, M.; Tawfeeq Ahmed, Z.H. Nanotechnology: Ethical Impacts, Health Issues, and Safety Issues. In Modern Nanotechnology: Volume 2: Green Synthesis, Sustainable Energy and Impacts; Springer: Cham, Switzerland, 2023; pp. 455–477. [CrossRef]
- Jakic, K.; et al. Long-Term Accumulation, Biological Effects and Toxicity of BSA-Coated Gold Nanoparticles in the Mouse Liver, Spleen, and Kidneys. Int. J. Nanomedicine 2024, 19, 4103. [CrossRef]
- Li, Q.; Li, X.; Zhao, C. Strategies to Obtain Encapsulation and Controlled Release of Small Hydrophilic Molecules. Front. Bioeng. Biotechnol. 2020, 8, 437. [CrossRef]
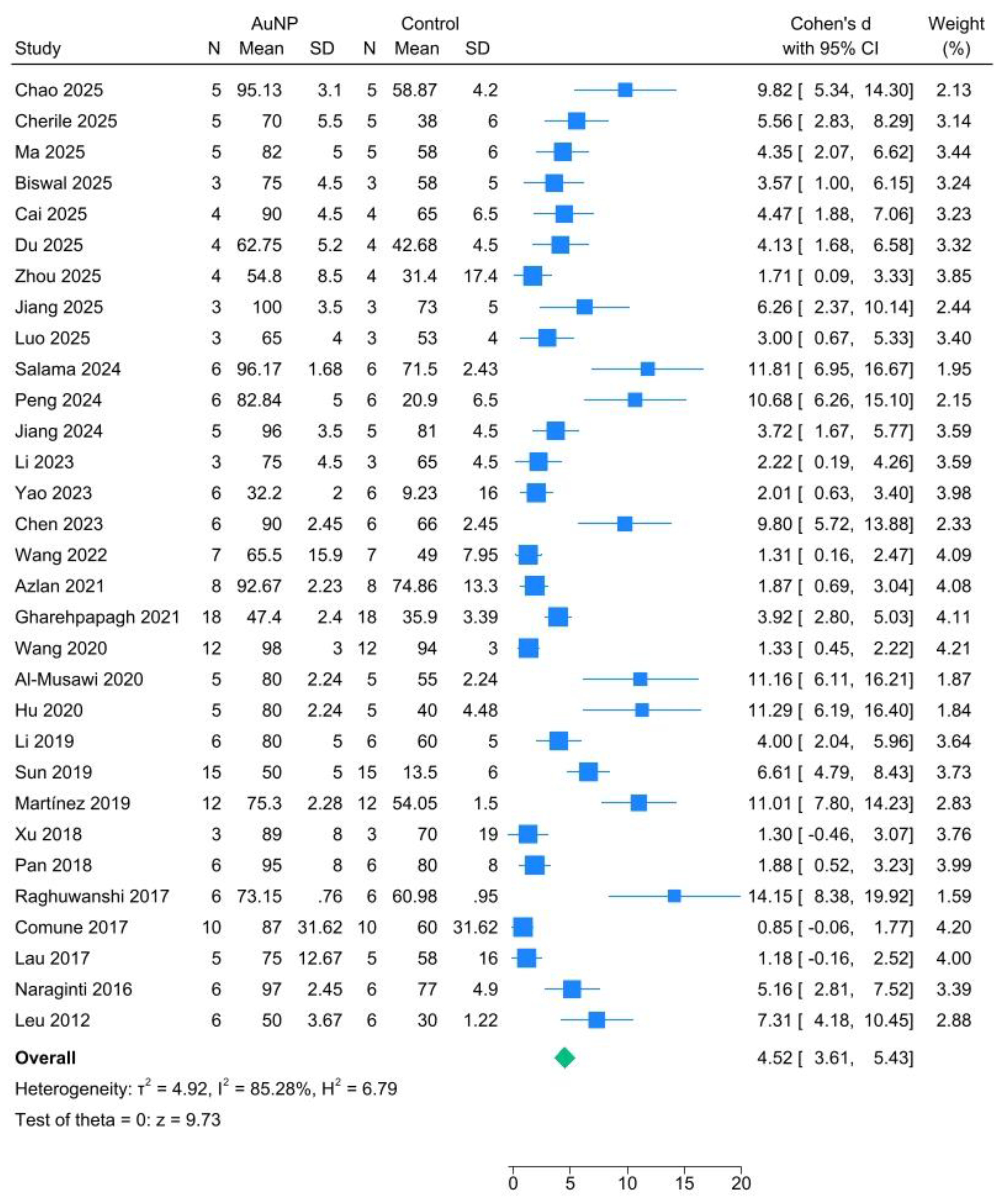
| Study | Intervention group, n | Control group, n | Wound type | Animal model | Day of peak effect | Study quality |
| 1. Chao et al., 2025 | 5 | 5 | Full thickness | Mice | Day 7 | 7/14 |
| 2. Gerile et al., 2025 | 5 | 5 | Full thickness | Mice | Day 7 | 7/14 |
| 3. Ma et al., 2025 | 5 | 5 | Full thickness | Mice | Day 7 | 6/14 |
| 4. Biswal et al., 2025 | 3 | 3 | Full thickness | Rats | Day 7 | 6/14 |
| 5. Cai et al., 2025 | 4 | 4 | Full thickness | Rats | Day 8 | 8/14 |
| 6. Du et al., 2025 | 4 | 4 | Full thickness | Mice | Day 9 | 7/14 |
| 7. Zhou et al., 2025 | 4 | 4 | Full thickness | Mice | Day 3 | 5/14 |
| 8. Jiang et al., 2025 | 3 | 3 | Full thickness | Mice | Day 13 | 7/14 |
| 9. Luo et al., 2025 | 3 | 3 | Full thickness | Mice | Day 12 | 7/14 |
| 10.Salama et al., 2024 | 6 | 6 | Full thickness | Rats | Day 9 | 7/14 |
| 11. Peng et al., 2024 | 6 | 6 | Full thickness | Rats | Day 12 | 7/14 |
| 12. Jiang et al., 2024 | 5 | 5 | Full thickness | Mice | Day 12 | 10/14 |
| 13. Li et al., 2023 | 3 | 3 | Full thickness | Mice | Day 14 | 10/14 |
| 14. Yao et al., 2023 | 6 | 6 | Full thickness | Mice | Day 15 |
7/14 |
| 15. Chen et al., 2023 | 6 | 6 | Full thickness | Rats | Day 13 |
8/14 |
| 16.Wang et al., 2022 | 7 | 7 | Full thickness | Rats | Day 7 |
10/14 |
| 17.Azlan et al., 2021 | 8 | 8 | Full thickness | Rats | Day 3 |
8/14 |
| 18.Gharehpapagh et al., 2021 | 18 | 18 | Full thickness | Mice | Day 7 |
9/14 |
| 19.Wang et al., 2020 | 12 | 12 | Full thickness | Rats | Day 10 |
10/14 |
| 20.Al-Musawi et al., 2020 | 5 | 5 | Full thickness | Rabbits | Day 7 |
7/14 |
| 21. Hu et al., 2020 | 5 | 5 | Full thickness | Mice | Day 4 |
7/14 |
| 22. Li et al., 2019 | 6 | 6 | Full thickness | Rats | Day 3 |
9/14 |
| 23. Sun et al., 2019 | 15 | 15 | Full-thickness | Mice | Day 7 | 7/14 |
| 24. Martínez et al., 2019 | 12 | 12 | Full thickness | Mice | Day 10 |
10/14 |
| 25. Xu et al., 2018 | 3 | 3 | Full thickness | Rats | Day 10 |
10/14 |
| 26. Pan et al., 2018 | 6 | 6 | Full thickness | Rats | Day 10 |
10/14 |
| 27. Raghuwanshi et al., 2017 | 6 | 6 | Full thickness | Rats | Day 9 |
10/14 |
| 28. Comune et al., 2017 | 10 | 10 | Full thickness | Mice | Day 7 | 10/14 |
| 29. Lau et al., 2017 | 5 | 5 | Full thickness | Rats | Day 7 | 9/14 |
| 30. Naraginti et al., 2016 | 6 | 6 | Full thickness | Rats | Day 9 | 10/14 |
| 31. Leu et al., 2012 | 6 | 6 | Full thickness | Mice | Day 7 | 10/14 |
| Id | Randomization | Allocation | Blinding | Assessment | Data | Reporting | Other |
| 1. Chao 2025 | Low | Unclear | High | High | Low | Low | Low |
| 2. Cherile 2025 | Low | Unclear | High | High | Low | Low | Low |
| 3. Ma 2025 | Low | Unclear | High | High | Low | Low | Low |
| 4. Biswal 2025 | Low | Unclear | High | High | Low | Low | Low |
| 5. Cai 2025 | Low | Unclear | High | High | Low | Low | Low |
| 6. Du 2025 | Low | Unclear | High | High | Low | Low | Low |
| 7. Zhou 2025 | Low | Unclear | High | High | Low | Low | Low |
| 8. Jiang 2025 | Low | Unclear | High | High | Low | Low | Low |
| 9. Luo 2025 | Low | Unclear | High | High | Low | Low | Low |
| 10. Salama 2024 | Low | Unclear | High | High | Low | Low | Low |
| 11. Peng 2024 | Low | Unclear | High | High | Low | Low | Low |
| 12. Jiang 2024 | Low | Unclear | High | High | Low | Low | Low |
| 13. Li 2024 | Low | Unclear | High | High | Low | Low | Low |
| 14. Yao 2023 | Low | Unclear | High | High | Low | Low | Low |
| 15. Chen 2023 | Low | Unclear | High | High | Low | Low | Low |
| 16. Wang 2022 | Low | Unclear | High | High | Low | Low | Low |
| 17. Azlan 2021 | Low | Unclear | High | High | Low | Low | Low |
| 18.Gharehpapagh 2021 | Low | Unclear | High | High | Low | Low | Low |
| 19. Wang 2020 | Low | Unclear | High | High | Low | Low | Low |
| 20.Al-Musawi 2020 | Low | Unclear | High | High | Low | Low | Low |
| 21. Hu 2020 | Low | Unclear | High | High | Low | Low | Low |
| 22. Li 2019 | Low | Unclear | High | High | Low | Low | Low |
| 23. Sun 2019 | Low | Unclear | High | High | Low | Low | Low |
| 24.Martínez 2019 | Low | Unclear | High | High | Low | Low | Low |
| 25. Xu 2018 | Low | Unclear | High | High | Low | Low | Low |
| 26. Pan 2018 | Low | Unclear | High | High | Low | Low | Low |
| 27.Raghuwanshi 2017 | Low | Unclear | High | High | Low | Low | Low |
| 28.Comune 2017 | Low | Unclear | High | High | Low | Low | Low |
| 29. Lau 2017 | Low | Unclear | High | High | Low | Low | Low |
| 30.Naraginti 2016 | Low | Unclear | High | High | Low | Low | Low |
| 31. Leu 2012 | Low | Unclear | High | High | Low | Low | Low |
| Publication | Year | (1) | (2) | (3) | (4) | (5) | (6) | (7) | (8) | (9) | (10) | (11) | (12) | (13) | (14) | Score |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Chao | 2025 | X | X | X | X | X | X | X | 7 | |||||||
| Cherile | 2025 | X | X | X | X | X | X | X | 7 | |||||||
| Ma | 2025 | X | X | X | X | X | X | 6 | ||||||||
| Biswal | 2025 | X | X | X | X | X | X | 6 | ||||||||
| Cai | 2025 | X | X | X | X | X | X | X | X | 8 | ||||||
| Du | 2025 | X | X | X | X | X | X | X | 7 | |||||||
| Zhou | 2025 | X | X | X | X | X | 5 | |||||||||
| Jiang | 2025 | X | X | X | X | X | X | X | 7 | |||||||
| Luo | 2025 | X | X | X | X | X | X | X | 7 | |||||||
| Salama | 2024 | X | X | X | X | X | X | X | 7 | |||||||
| Peng | 2024 | X | X | X | X | X | X | X | 7 | |||||||
| Jiang | 2024 | X | X | X | X | X | X | X | X | X | X | 10 | ||||
| Li | 2023 | X | X | X | X | X | X | X | X | X | X | 10 | ||||
| Yao | 2023 | X | X | X | X | X | X | X | 7 | |||||||
| Chen | 2023 | X | X | X | X | X | X | X | X | 8 | ||||||
| Wang | 2022 | X | X | X | X | X | X | X | X | X | X | 10 | ||||
| Azlan | 2021 | X | X | X | X | X | X | X | X | 8 | ||||||
| Gharehpapagh | 2021 | X | X | X | X | X | X | X | X | X | 9 | |||||
| Wang | 2020 | X | X | X | X | X | X | X | X | X | X | 10 | ||||
| Al-Musawi | 2020 | X | X | X | X | X | X | X | 7 | |||||||
| Hu | 2020 | X | X | X | X | X | X | X | 7 | |||||||
| Li | 2019 | X | X | X | X | X | X | X | X | X | 9 | |||||
| Sun | 2019 | X | X | X | X | X | X | X | 7 | |||||||
| Martínez | 2019 | X | X | X | X | X | X | X | X | X | X | 10 | ||||
| Xu | 2018 | X | X | X | X | X | X | X | X | X | X | 10 | ||||
| Pan | 2018 | X | X | X | X | X | X | X | X | X | X | 10 | ||||
| Raghuwanshi | 2017 | X | X | X | X | X | X | X | X | X | X | 10 | ||||
| Comune | 2017 | X | X | X | X | X | X | X | X | X | X | 10 | ||||
| Lau | 2017 | X | X | X | X | X | X | X | X | X | 9 | |||||
| Naraginti | 2016 | X | X | X | X | X | X | X | X | X | X | 10 | ||||
| Leu | 2012 | X | X | X | X | X | X | X | X | X | X | 10 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




