Submitted:
02 July 2025
Posted:
03 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Epigenetic Alterations as Danger Signals: Conceptual Framework
3. Molecular Mechanisms of Epigenetic Alteration Recognition by PRRs
3.1. Sensing Unmethylated DNA Patterns
3.2. Extracellular Chromatin and Histones as DAMPs
3.3. Epitranscriptomic Modifications and RNA Sensing
4. Evidence from Cancer Models and Disease Contexts
4.1. Viral Mimicry in Tumors: Endogenous Retroviruses and Interferon Activation
4.2. DNA Damage, Micronuclei and cGAS-STING in Cancer
4.3. Immunogenic Cell Death and Epigenetic Modulation
5. Can Epigenetic Readers Function as Pattern Recognition Receptors?
6. Conclusion and Perspectives
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Janeway, C.A., Jr., Approaching the asymptote? Evolution and revolution in immunology. Cold Spring Harb Symp Quant Biol, 1989. 54 Pt 1: p. 1-13.
- Janeway, C.A., Jr. and R. Medzhitov, Innate immune recognition. Annu Rev Immunol, 2002. 20: p. 197-216.
- Medzhitov, R. and C. Janeway, Jr., Innate immune recognition: mechanisms and pathways. Immunol Rev, 2000. 173: p. 89-97.
- Matzinger, P. Tolerance, danger, and the extended family. Annu Rev Immunol 1994, 12, 991–1045. [Google Scholar] [CrossRef]
- Matzinger, P. The danger model: a renewed sense of self. Science 2002, 296, 301–305. [Google Scholar] [CrossRef] [PubMed]
- Seong, S.Y.; Matzinger, P. Hydrophobicity: an ancient damage-associated molecular pattern that initiates innate immune responses. Nat Rev Immunol 2004, 4, 469–478. [Google Scholar] [CrossRef]
- Ma, M.; Jiang, W.; Zhou, R. DAMPs and DAMP-sensing receptors in inflammation and diseases. Immunity 2024, 57, 752–771. [Google Scholar] [CrossRef] [PubMed]
- Kouzarides, T. Chromatin modifications and their function. Cell 2007, 128, 693–705. [Google Scholar] [CrossRef] [PubMed]
- Mattei, A.L.; Bailly, N.; Meissner, A. DNA methylation: a historical perspective. Trends Genet 2022, 38, 676–707. [Google Scholar] [CrossRef]
- Li, X.; Ye, Y.; Peng, K.; Zeng, Z.; Chen, L.; Zeng, Y. Histones: The critical players in innate immunity. Front Immunol 2022, 13, 1030610. [Google Scholar] [CrossRef]
- Silk, E.; Zhao, H.; Weng, H.; Ma, D. The role of extracellular histone in organ injury. Cell Death Dis 2017, 8, e2812. [Google Scholar] [CrossRef]
- Yang, T.; Peng, J.; Zhang, Z.; Chen, Y.; Liu, Z.; Jiang, L.; Jin, L.; Han, M.; Su, B.; Li, Y. Emerging therapeutic strategies targeting extracellular histones for critical and inflammatory diseases: an updated narrative review. Front Immunol 2024, 15, 1438984. [Google Scholar] [CrossRef]
- Latz, E.; Schoenemeyer, A.; Visintin, A.; Fitzgerald, K.A.; Monks, B.G.; Knetter, C.F.; Lien, E.; Nilsen, N.J.; Espevik, T.; Golenbock, D.T. TLR9 signals after translocating from the ER to CpG DNA in the lysosome. Nat Immunol 2004, 5, 190–198. [Google Scholar] [CrossRef]
- Hemmi, H.; Takeuchi, O.; Kawai, T.; Kaisho, T.; Sato, S.; Sanjo, H.; Matsumoto, M.; Hoshino, K.; Wagner, H.; Takeda, K.; et al. A Toll-like receptor recognizes bacterial DNA. Nature 2000, 408, 740–745. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.Z.; Liu, Z.; Liu, J.; Ren, J.X.; Sun, T.S. Mitochondrial DNA induces inflammation and increases TLR9/NF-kappaB expression in lung tissue. Int J Mol Med 2014, 33, 817–824. [Google Scholar] [CrossRef] [PubMed]
- West, A.P.; Shadel, G.S. Mitochondrial DNA in innate immune responses and inflammatory pathology. Nat Rev Immunol 2017, 17, 363–375. [Google Scholar] [CrossRef] [PubMed]
- Beltran-Garcia, J.; Osca-Verdegal, R.; Perez-Cremades, D.; Novella, S.; Hermenegildo, C.; Pallardo, F.V.; Garcia-Gimenez, J.L. Extracellular Histones Activate Endothelial NLRP3 Inflammasome and are Associated with a Severe Sepsis Phenotype. J Inflamm Res 2022, 15, 4217–4238. [Google Scholar] [CrossRef]
- Chen, G.Y.; Nunez, G. Sterile inflammation: sensing and reacting to damage. Nat Rev Immunol 2010, 10, 826–837. [Google Scholar] [CrossRef]
- Xu, J.; Zhang, X.; Monestier, M.; Esmon, N.L.; Esmon, C.T. Extracellular histones are mediators of death through TLR2 and TLR4 in mouse fatal liver injury. J Immunol 2011, 187, 2626–2631. [Google Scholar] [CrossRef]
- Chen, R.; Ishak, C.A.; De Carvalho, D.D. Endogenous Retroelements and the Viral Mimicry Response in Cancer Therapy and Cellular Homeostasis. Cancer Discov 2021, 11, 2707–2725. [Google Scholar] [CrossRef]
- Esteller, M.; Dawson, M.A.; Kadoch, C.; Rassool, F.V.; Jones, P.A.; Baylin, S.B. The Epigenetic Hallmarks of Cancer. Cancer Discov 2024, 14, 1783–1809. [Google Scholar] [CrossRef]
- Nishiyama, A.; Nakanishi, M. Navigating the DNA methylation landscape of cancer. Trends Genet 2021, 37, 1012–1027. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, M.; Wang, Y. The roles of histone modifications in tumorigenesis and associated inhibitors in cancer therapy. J Natl Cancer Cent 2022, 2, 277–290. [Google Scholar] [CrossRef]
- Locatelli, M.; Faure-Dupuy, S. Virus hijacking of host epigenetic machinery to impair immune response. J Virol 2023, 97, e0065823. [Google Scholar] [CrossRef]
- Roh, J.S.; Sohn, D.H. Damage-Associated Molecular Patterns in Inflammatory Diseases. Immune Netw 2018, 18, e27. [Google Scholar] [CrossRef]
- Miller, S.I.; Ernst, R.K.; Bader, M.W. LPS, TLR4 and infectious disease diversity. Nat Rev Microbiol 2005, 3, 36–46. [Google Scholar] [CrossRef]
- Smith, K.D.; Andersen-Nissen, E.; Hayashi, F.; Strobe, K.; Bergman, M.A.; Barrett, S.L.; Cookson, B.T.; Aderem, A. Toll-like receptor 5 recognizes a conserved site on flagellin required for protofilament formation and bacterial motility. Nat Immunol 2003, 4, 1247–1253. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Zhang, H.; Wang, J.; Chen, J.; Guo, Z.; Liu, Y.; Hua, H. Exploiting RIG-I-like receptor pathway for cancer immunotherapy. J Hematol Oncol 2023, 16, 8. [Google Scholar] [CrossRef] [PubMed]
- Zhong, H.; Li, X.; Zhou, S.; Jiang, P.; Liu, X.; Ouyang, M.; Nie, Y.; Chen, X.; Zhang, L.; Liu, Y.; et al. Interplay between RAGE and TLR4 Regulates HMGB1-Induced Inflammation by Promoting Cell Surface Expression of RAGE and TLR4. J Immunol 2020, 205, 767–775. [Google Scholar] [CrossRef]
- Ronning, K.E.; Dechelle-Marquet, P.A.; Che, Y.; Guillonneau, X.; Sennlaub, F.; Delarasse, C. The P2X7 Receptor, a Multifaceted Receptor in Alzheimer's Disease. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef] [PubMed]
- Vande Walle, L.; Lamkanfi, M. Drugging the NLRP3 inflammasome: from signalling mechanisms to therapeutic targets. Nat Rev Drug Discov 2024, 23, 43–66. [Google Scholar] [CrossRef]
- Yasuda, K.; Richez, C.; Uccellini, M.B.; Richards, R.J.; Bonegio, R.G.; Akira, S.; Monestier, M.; Corley, R.B.; Viglianti, G.A.; Marshak-Rothstein, A.; et al. Requirement for DNA CpG content in TLR9-dependent dendritic cell activation induced by DNA-containing immune complexes. J Immunol 2009, 183, 3109–3117. [Google Scholar] [CrossRef]
- Amadio, R.; Piperno, G.M.; Benvenuti, F. Self-DNA Sensing by cGAS-STING and TLR9 in Autoimmunity: Is the Cytoskeleton in Control? Front Immunol 2021, 12, 657344. [Google Scholar] [CrossRef]
- Liu, R.; Zhao, E.; Yu, H.; Yuan, C.; Abbas, M.N.; Cui, H. Methylation across the central dogma in health and diseases: new therapeutic strategies. Signal Transduct Target Ther 2023, 8, 310. [Google Scholar] [CrossRef] [PubMed]
- Perez, R.F.; Tejedor, J.R.; Bayon, G.F.; Fernandez, A.F.; Fraga, M.F. Distinct chromatin signatures of DNA hypomethylation in aging and cancer. Aging Cell 2018, 17, e12744. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Ganz, P.A.; Sehl, M.E. DNA Methylation, Aging, and Cancer Risk: A Mini-Review. Front Bioinform 2022, 2, 847629. [Google Scholar] [CrossRef]
- Wu, J.; Sun, L.; Chen, X.; Du, F.; Shi, H.; Chen, C.; Chen, Z.J. Cyclic GMP-AMP is an endogenous second messenger in innate immune signaling by cytosolic DNA. Science 2013, 339, 826–830. [Google Scholar] [CrossRef]
- Sun, L.; Wu, J.; Du, F.; Chen, X.; Chen, Z.J. Cyclic GMP-AMP synthase is a cytosolic DNA sensor that activates the type I interferon pathway. Science 2013, 339, 786–791. [Google Scholar] [CrossRef] [PubMed]
- Beck, M.A.; Fischer, H.; Grabner, L.M.; Groffics, T.; Winter, M.; Tangermann, S.; Meischel, T.; Zaussinger-Haas, B.; Wagner, P.; Fischer, C.; et al. DNA hypomethylation leads to cGAS-induced autoinflammation in the epidermis. EMBO J 2021, 40, e108234. [Google Scholar] [CrossRef]
- Lu, Y.; Zhao, M.; Chen, L.; Wang, Y.; Liu, T.; Liu, H. cGAS: action in the nucleus. Front Immunol 2024, 15, 1380517. [Google Scholar] [CrossRef]
- Bai, J.; Liu, F. Nuclear cGAS: sequestration and beyond. Protein Cell 2022, 13, 90–101. [Google Scholar] [CrossRef]
- Wang, H.; Kim, S.J.; Lei, Y.; Wang, S.; Wang, H.; Huang, H.; Zhang, H.; Tsung, A. Neutrophil extracellular traps in homeostasis and disease. Signal Transduct Target Ther 2024, 9, 235. [Google Scholar] [CrossRef]
- Thiam, H.R.; Wong, S.L.; Wagner, D.D.; Waterman, C.M. Cellular Mechanisms of NETosis. Annu Rev Cell Dev Biol 2020, 36, 191–218. [Google Scholar] [CrossRef]
- Allam, R.; Darisipudi, M.N.; Tschopp, J.; Anders, H.J. Histones trigger sterile inflammation by activating the NLRP3 inflammasome. Eur J Immunol 2013, 43, 3336–3342. [Google Scholar] [CrossRef]
- Huang, H.; Chen, H.W.; Evankovich, J.; Yan, W.; Rosborough, B.R.; Nace, G.W.; Ding, Q.; Loughran, P.; Beer-Stolz, D.; Billiar, T.R.; et al. Histones activate the NLRP3 inflammasome in Kupffer cells during sterile inflammatory liver injury. J Immunol 2013, 191, 2665–2679. [Google Scholar] [CrossRef]
- Sun, W.; Lv, S.; Li, H.; Cui, W.; Wang, L. Enhancing the Anticancer Efficacy of Immunotherapy through Combination with Histone Modification Inhibitors. Genes (Basel) 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Lee, K.; Kim, K.; Yi, S.J. The role of histone modifications: from neurodevelopment to neurodiseases. Signal Transduct Target Ther 2022, 7, 217. [Google Scholar] [CrossRef]
- Leshner, M.; Wang, S.; Lewis, C.; Zheng, H.; Chen, X.A.; Santy, L.; Wang, Y. PAD4 mediated histone hypercitrullination induces heterochromatin decondensation and chromatin unfolding to form neutrophil extracellular trap-like structures. Front Immunol 2012, 3, 307. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.L.; Wagner, D.D. Peptidylarginine deiminase 4: a nuclear button triggering neutrophil extracellular traps in inflammatory diseases and aging. FASEB J 2018, 32, fj201800691R. [Google Scholar] [CrossRef] [PubMed]
- Peng, W.; Wu, S.; Wang, W. Correlation of serum citrullinated histone H3 levels with disease activity in patients with rheumatoid arthritis. Clin Exp Rheumatol 2023, 41, 1792–1800. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, S. Cracking the epitranscriptome. RNA 2016, 22, 169–174. [Google Scholar] [CrossRef]
- Yoon, K.J. Voyages to map unexplored parts of the epitranscriptomic world. Exp Mol Med 2022, 54, 1599–1600. [Google Scholar] [CrossRef]
- Roundtree, I.A.; Evans, M.E.; Pan, T.; He, C. Dynamic RNA Modifications in Gene Expression Regulation. Cell 2017, 169, 1187–1200. [Google Scholar] [CrossRef]
- Kim, G.W.; Imam, H.; Khan, M.; Siddiqui, A. N(6)-Methyladenosine modification of hepatitis B and C viral RNAs attenuates host innate immunity via RIG-I signaling. J Biol Chem 2020, 295, 13123–13133. [Google Scholar] [CrossRef]
- Lu, M.; Zhang, Z.; Xue, M.; Zhao, B.S.; Harder, O.; Li, A.; Liang, X.; Gao, T.Z.; Xu, Y.; Zhou, J.; et al. N(6)-methyladenosine modification enables viral RNA to escape recognition by RNA sensor RIG-I. Nat Microbiol 2020, 5, 584–598. [Google Scholar] [CrossRef]
- Zhao, B.; Wang, W.; Zhao, Y.; Qiao, H.; Gao, Z.; Chuai, X. Regulation of Antiviral Immune Response by N (6)-Methyladenosine of mRNA. Front Microbiol 2021, 12, 789605. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Hui, H.; Bray, B.; Gonzalez, G.M.; Zeller, M.; Anderson, K.G.; Knight, R.; Smith, D.; Wang, Y.; Carlin, A.F.; et al. METTL3 regulates viral m6A RNA modification and host cell innate immune responses during SARS-CoV-2 infection. Cell Rep 2021, 35, 109091. [Google Scholar] [CrossRef]
- Kariko, K.; Buckstein, M.; Ni, H.; Weissman, D. Suppression of RNA recognition by Toll-like receptors: the impact of nucleoside modification and the evolutionary origin of RNA. Immunity 2005, 23, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Jansz, N.; Faulkner, G.J. Endogenous retroviruses in the origins and treatment of cancer. Genome Biol 2021, 22, 147. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Qiu, P.; Ai, J.; Liu, B.; Han, G.Z.; Zhu, F.; Zhang, W.; Cui, J. Endogenous retrovirus activation: potential for immunology and clinical applications. Natl Sci Rev 2024, 11, nwae034. [Google Scholar] [CrossRef] [PubMed]
- Dias Junior, A.G.; Sampaio, N.G.; Rehwinkel, J. A Balancing Act: MDA5 in Antiviral Immunity and Autoinflammation. Trends Microbiol 2019, 27, 75–85. [Google Scholar] [CrossRef]
- Rehwinkel, J.; Mehdipour, P. ADAR1: from basic mechanisms to inhibitors. Trends Cell Biol 2025, 35, 59–73. [Google Scholar] [CrossRef]
- Wang, R.; Dong, X.; Zhang, X.; Liao, J.; Cui, W.; Li, W. Exploring viral mimicry combined with epigenetics and tumor immunity: new perspectives in cancer therapy. Int J Biol Sci 2025, 21, 958–973. [Google Scholar] [CrossRef]
- Liu, K.; Li, Y.; Shen, M.; Xu, W.; Wu, S.; Yang, X.; Zhang, B.; Lin, N. Epigenetic Regulation of Stromal and Immune Cells and Therapeutic Targets in the Tumor Microenvironment. Biomolecules 2025, 15. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Zhou, Z.; Yang, S.; Zhang, S.; Shao, B. Epigenetic regulation and therapeutic targets in the tumor microenvironment. Mol Biomed 2023, 4, 17. [Google Scholar] [CrossRef]
- Chiappinelli, K.B.; Strissel, P.L.; Desrichard, A.; Li, H.; Henke, C.; Akman, B.; Hein, A.; Rote, N.S.; Cope, L.M.; Snyder, A.; et al. Inhibiting DNA Methylation Causes an Interferon Response in Cancer via dsRNA Including Endogenous Retroviruses. Cell 2015, 162, 974–986. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Zhang, B.; Li, B.; Wu, H.; Jiang, M. Cold and hot tumors: from molecular mechanisms to targeted therapy. Signal Transduct Target Ther 2024, 9, 274. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Cui, G.; Liu, H.; Han, Y.; Cai, C.; Feng, Z.; Shen, H.; Zeng, S. Converting "cold" to "hot": epigenetics strategies to improve immune therapy effect by regulating tumor-associated immune suppressive cells. Cancer Commun (Lond) 2024, 44, 601–636. [Google Scholar] [CrossRef]
- Chen, M.; Linstra, R.; van Vugt, M. Genomic instability, inflammatory signaling and response to cancer immunotherapy. Biochim Biophys Acta Rev Cancer 2022, 1877, 188661. [Google Scholar] [CrossRef]
- Hosea, R.; Hillary, S.; Naqvi, S.; Wu, S.; Kasim, V. The two sides of chromosomal instability: drivers and brakes in cancer. Signal Transduct Target Ther 2024, 9, 75. [Google Scholar] [CrossRef]
- Kwon, J.; Bakhoum, S.F. The Cytosolic DNA-Sensing cGAS-STING Pathway in Cancer. Cancer Discov 2020, 10, 26–39. [Google Scholar] [CrossRef]
- Tripathi, R.; Modur, V.; Senovilla, L.; Kroemer, G.; Komurov, K. Suppression of tumor antigen presentation during aneuploid tumor evolution contributes to immune evasion. Oncoimmunology 2019, 8, 1657374. [Google Scholar] [CrossRef]
- MacDonald, K.M.; Benguerfi, S.; Harding, S.M. Alerting the immune system to DNA damage: micronuclei as mediators. Essays Biochem 2020, 64, 753–764. [Google Scholar] [CrossRef]
- Bharti, V.; Kumar, A.; Wang, Y.; Roychowdhury, N.; de Lima Bellan, D.; Kassaye, B.B.; Watkins, R.; Capece, M.; Chung, C.G.; Hilinski, G.; et al. TTK inhibitor OSU13 promotes immunotherapy responses by activating tumor STING. JCI Insight 2024, 9. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.Y.; Chiu, D.K.; Yuen, V.W.; Law, C.T.; Wong, B.P.; Thu, K.L.; Cescon, D.W.; Soria-Bretones, I.; Cheu, J.W.; Lee, D.; et al. CFI-402257, a TTK inhibitor, effectively suppresses hepatocellular carcinoma. Proc Natl Acad Sci U S A 2022, 119, e2119514119. [Google Scholar] [CrossRef]
- Hu, Y.; Manasrah, B.K.; McGregor, S.M.; Lera, R.F.; Norman, R.X.; Tucker, J.B.; Scribano, C.M.; Yan, R.E.; Humayun, M.; Wisinski, K.B.; et al. Paclitaxel Induces Micronucleation and Activates Pro-Inflammatory cGAS-STING Signaling in Triple-Negative Breast Cancer. Mol Cancer Ther 2021, 20, 2553–2567. [Google Scholar] [CrossRef] [PubMed]
- Yue, B.; Gao, W.; Lovell, J.F.; Jin, H.; Huang, J. The cGAS-STING pathway in cancer immunity: dual roles, therapeutic strategies, and clinical challenges. Essays Biochem 2025, 69. [Google Scholar] [CrossRef]
- Naik, A.; Lattab, B.; Qasem, H.; Decock, J. Cancer testis antigens: Emerging therapeutic targets leveraging genomic instability in cancer. Mol Ther Oncol 2024, 32, 200768. [Google Scholar] [CrossRef]
- Ahmed, A.; Tait, S.W.G. Targeting immunogenic cell death in cancer. Mol Oncol 2020, 14, 2994–3006. [Google Scholar] [CrossRef] [PubMed]
- Ge, Y.; Huang, M.; Yao, Y.M. The Effect and Regulatory Mechanism of High Mobility Group Box-1 Protein on Immune Cells in Inflammatory Diseases. Cells 2021, 10. [Google Scholar] [CrossRef]
- Rivas-Yanez, E.; Barrera-Avalos, C.; Parra-Tello, B.; Briceno, P.; Rosemblatt, M.V.; Saavedra-Almarza, J.; Rosemblatt, M.; Acuna-Castillo, C.; Bono, M.R.; Sauma, D. P2X7 Receptor at the Crossroads of T Cell Fate. Int J Mol Sci 2020, 21. [Google Scholar] [CrossRef] [PubMed]
- Dhatchinamoorthy, K.; Colbert, J.D.; Rock, K.L. Cancer Immune Evasion Through Loss of MHC Class I Antigen Presentation. Front Immunol 2021, 12, 636568. [Google Scholar] [CrossRef]
- Wang, Y.; Bedford, M.T. Effectors and effects of arginine methylation. Biochem Soc Trans 2023, 51, 725–734. [Google Scholar] [CrossRef]
- Xu, Y.; Zhang, W.; Shen, F.; Yang, X.; Liu, H.; Dai, S.; Sun, X.; Huang, J.; Guo, Q. YTH Domain Proteins: A Family of m(6)A Readers in Cancer Progression. Front Oncol 2021, 11, 629560. [Google Scholar] [CrossRef] [PubMed]
- Boulias, K.; Greer, E.L. Means, mechanisms and consequences of adenine methylation in DNA. Nat Rev Genet 2022, 23, 411–428. [Google Scholar] [CrossRef] [PubMed]
- Zheng, K.; Lyu, Z.; Chen, J.; Chen, G. 5-Hydroxymethylcytosine: Far Beyond the Intermediate of DNA Demethylation. Int J Mol Sci 2024, 25. [Google Scholar] [CrossRef] [PubMed]
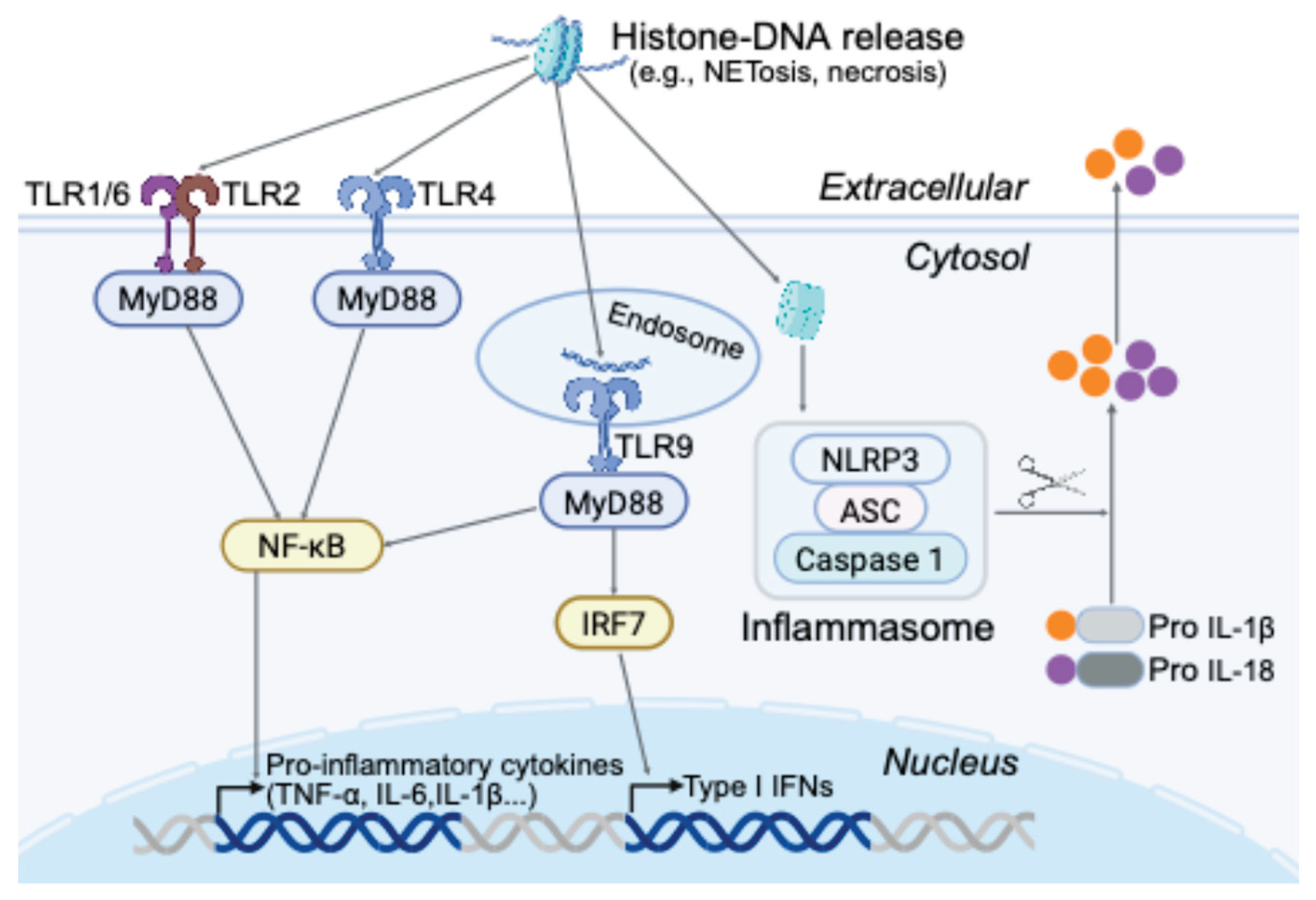
| Category of Signal | Source | Recognized by PRRs (Examples) |
|---|---|---|
| Pathogen-Associated Molecular Patterns (PAMPs) |
Microbial molecules |
TLR4 recognizes LPS on bacteria[24]. TLR5 recognizes flagellin[25]. RIG-I/MDA5 sense viral RNA[26]. TLR9 recognizes microbial DNA[12]. |
| Damage/Danger- Associated Molecular Patterns (DAMPs) |
Host molecules released due to damage/danger |
TLR4 and RAGE bind HMGB1[27]. P2X7 receptor senses ATP[28]. NLRP3 inflammasome is activated by diverse DAMP-induced stress[29]. |
| Epigenetic alteration– Associated Molecular Patterns (EAMPs) |
Epigenetic modifications that produce abnormal or pathogen-mimicking patterns without altering the DNA sequence. |
TLR9 can respond to host DNA containing unmethylated CpG motifs (normally suppressed by methylation)[30]. extracellular DNA–Histone complexes engage TLR2/4[17]. cGAS detects any cytosolic DNA (self or viral) leading to STING activation[31]. |
| Type | Modification | Targets | Role in immune recognition/surveillance |
|---|---|---|---|
| DNA | 5-Methylcytosine (5mC) |
Cytosine (CpG sites) |
Silences immune and viral genes. |
| 5-Hydroxymethylcytosine (5hmC) |
Oxidized 5mC | May influence immune tolerance. Not directly recognized by PRRs. |
|
| 6-Methyladenine (6mA) |
Adenine | Bacterial 6mA may act as PAMPs. | |
| RNA | N6-Methyladenosine (m6A) |
Adenosine | Mask viral RNA from RIG-I/MDA5 detection. |
| N1-Methyladenosine (m1A) |
Adenosine | Emerging role in regulating translation and stress reponses. | |
| 5-Methylcytosine (5mC) |
Cytosine | Influences RNA stability and immune recognition (emerging). | |
| 2′-O-Methylation (Nm) |
Ribose 2′-OH group |
Discriminates self from non-self RNA; lack of 2′-O-Me on viral RNA caps activates RIG-I, MDA5, IFITs. | |
| Pseudouridine (Ψ) |
Uridine | Ψ-containing RNA is less immunogenic; used in mRNA vaccines to evade immune detection. |
|
| RNA Editing (A-to-I) |
Adenosine | Prevents detection of self-dsRNA by MDA5/RIG-I. | |
| RNA Editing (C-to-U) |
Cytidine | Hypermutation of viral RNA; serves as antiviral restriction factor. | |
| m7G Cap (5′ cap) |
Guanosine | Shields self-RNA from RIG-I and IFITs. Absent or improper capping triggers immune detection. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




