Submitted:
26 June 2025
Posted:
27 June 2025
You are already at the latest version
Abstract
Keywords:
The Thalamocortical System, Gatekeeper of Experience
Layer 6b, A Conductor of Higher-Order Thalamocortical Activity
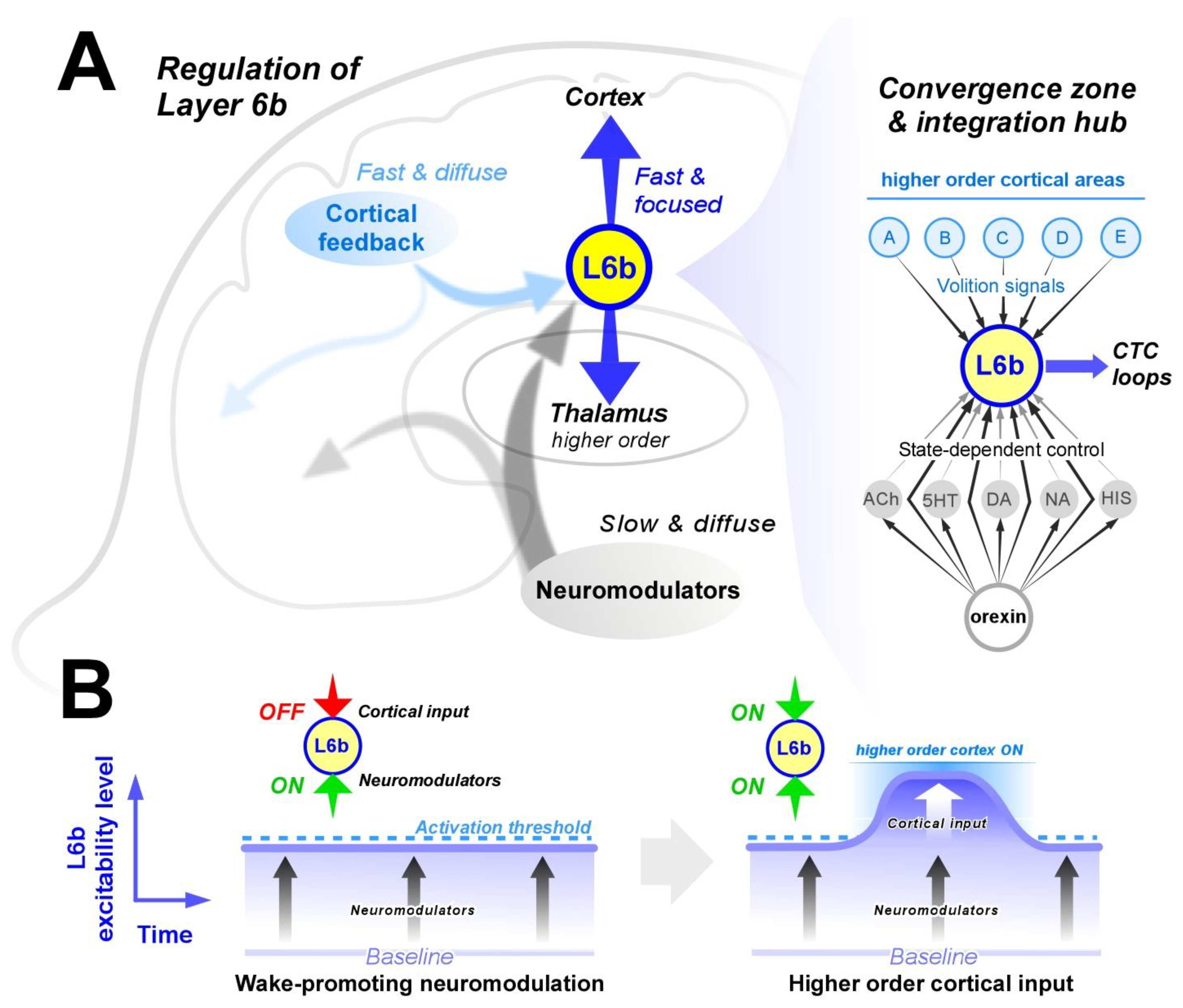
L6b Is a Convergence Zone of Arousal-Promoting Neuromodulation
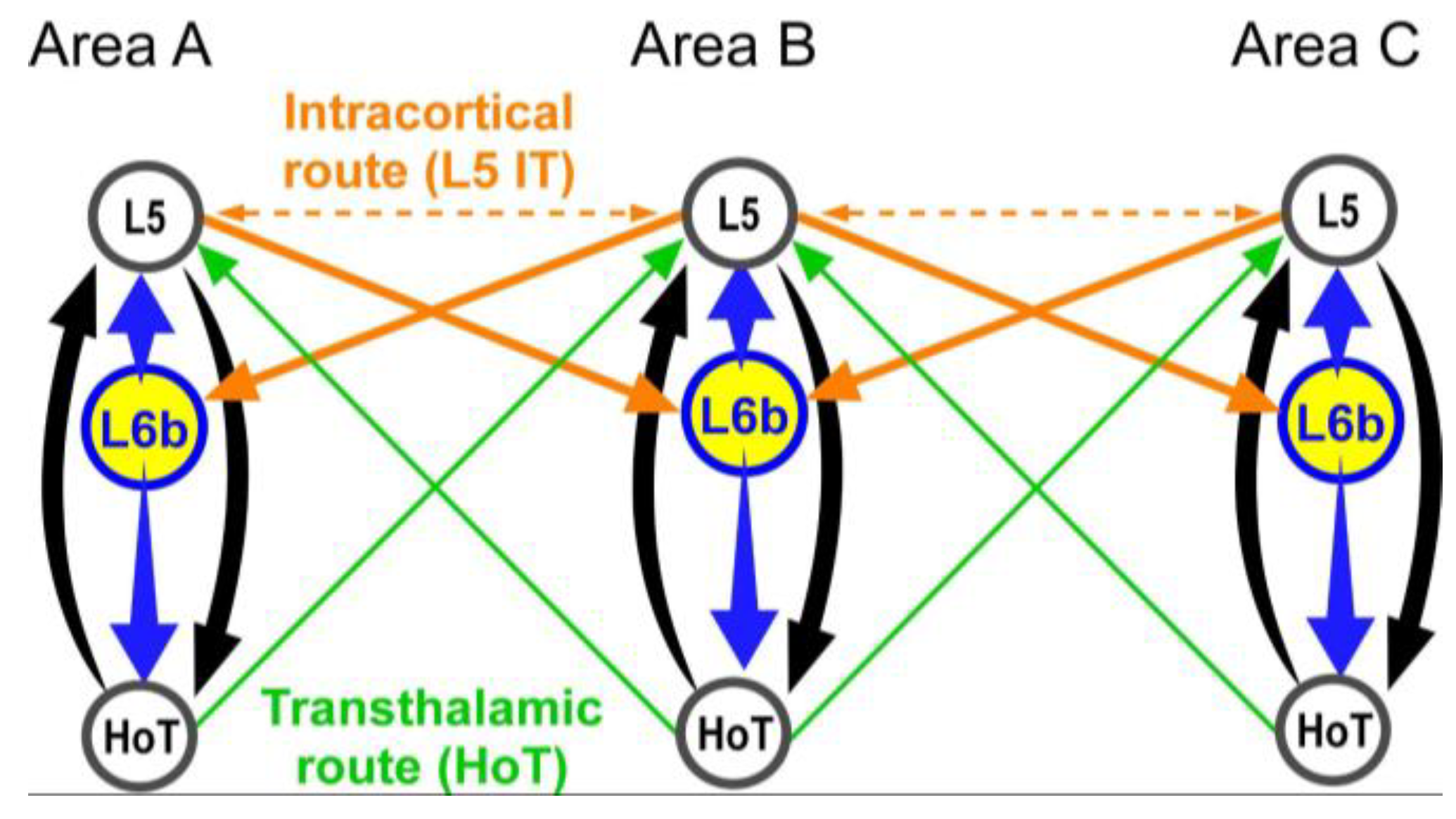
L6b Delivers Fast and Focused Control of Higher-Order Thalamocortical Loops
Layer 6b, An Engine OF Attention
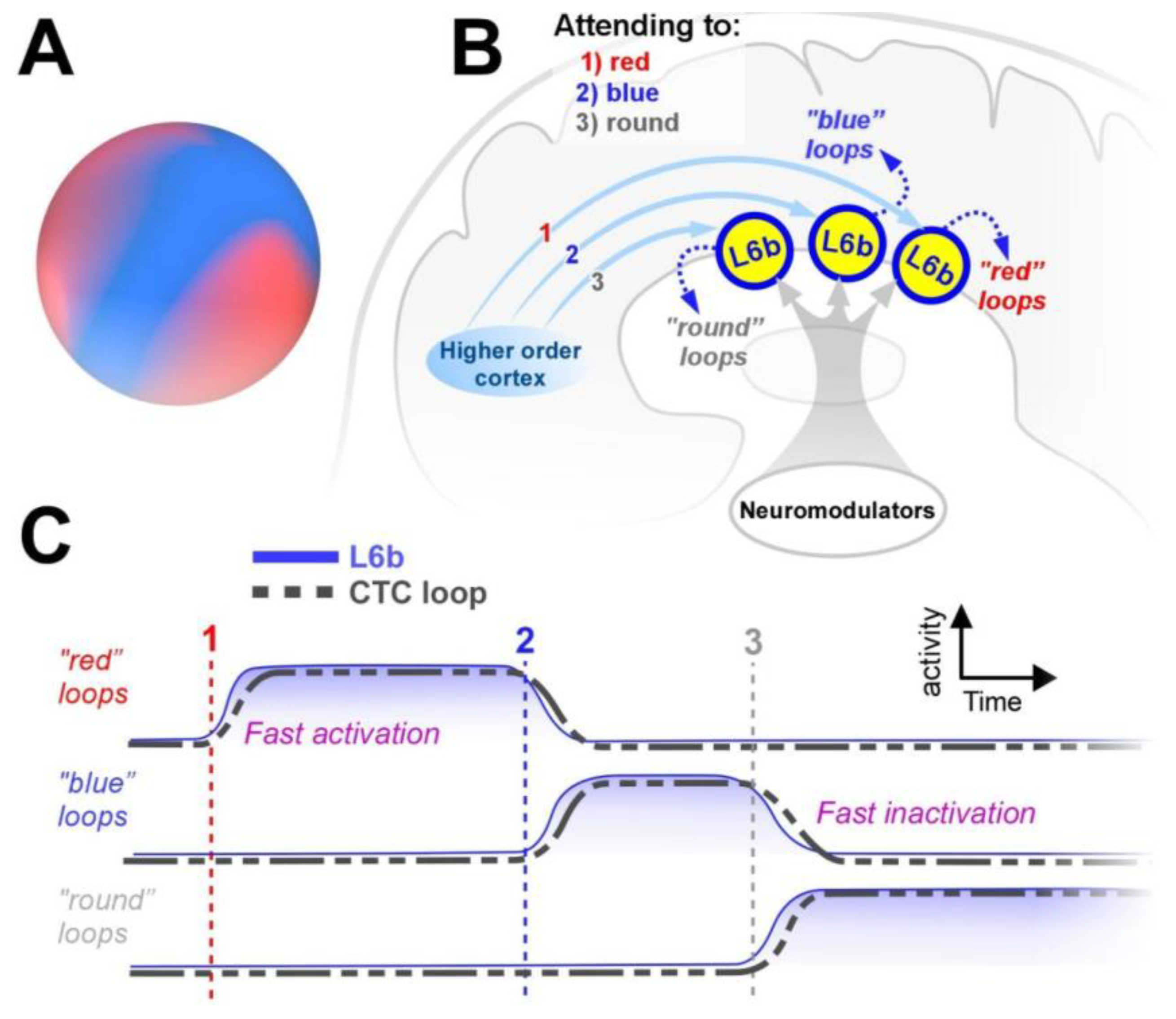
Focused Attention, L6b’s Focused Projections Specifically Target CTC Loops
Sustained Attention, L6b’s Persistent Synapses Maintain Activity in CTC Loops
Attentional Flexibility, L6b’s Fast Synapses Enable Precise Temporal Control of CTC Loops
L6b and the Superior Colliculus
Layer 6b and Attention-Associated Cognitive Functions
Perceptual Binding, L6b’s Powerful long range Influence May Help Coordinate CTC Loops
When Layer 6b Malfunctions
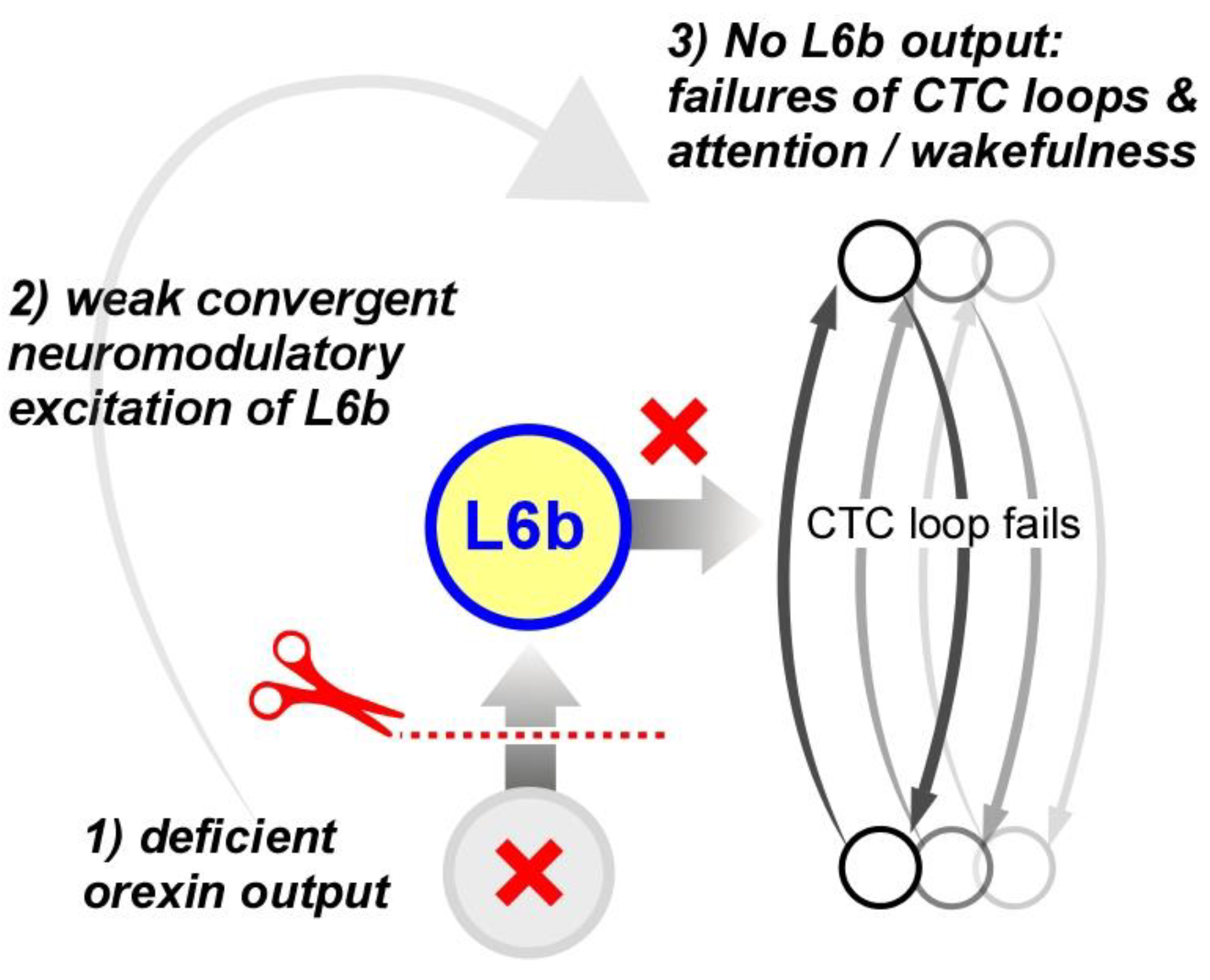
Narcolepsy
Schizophrenia
Conclusions
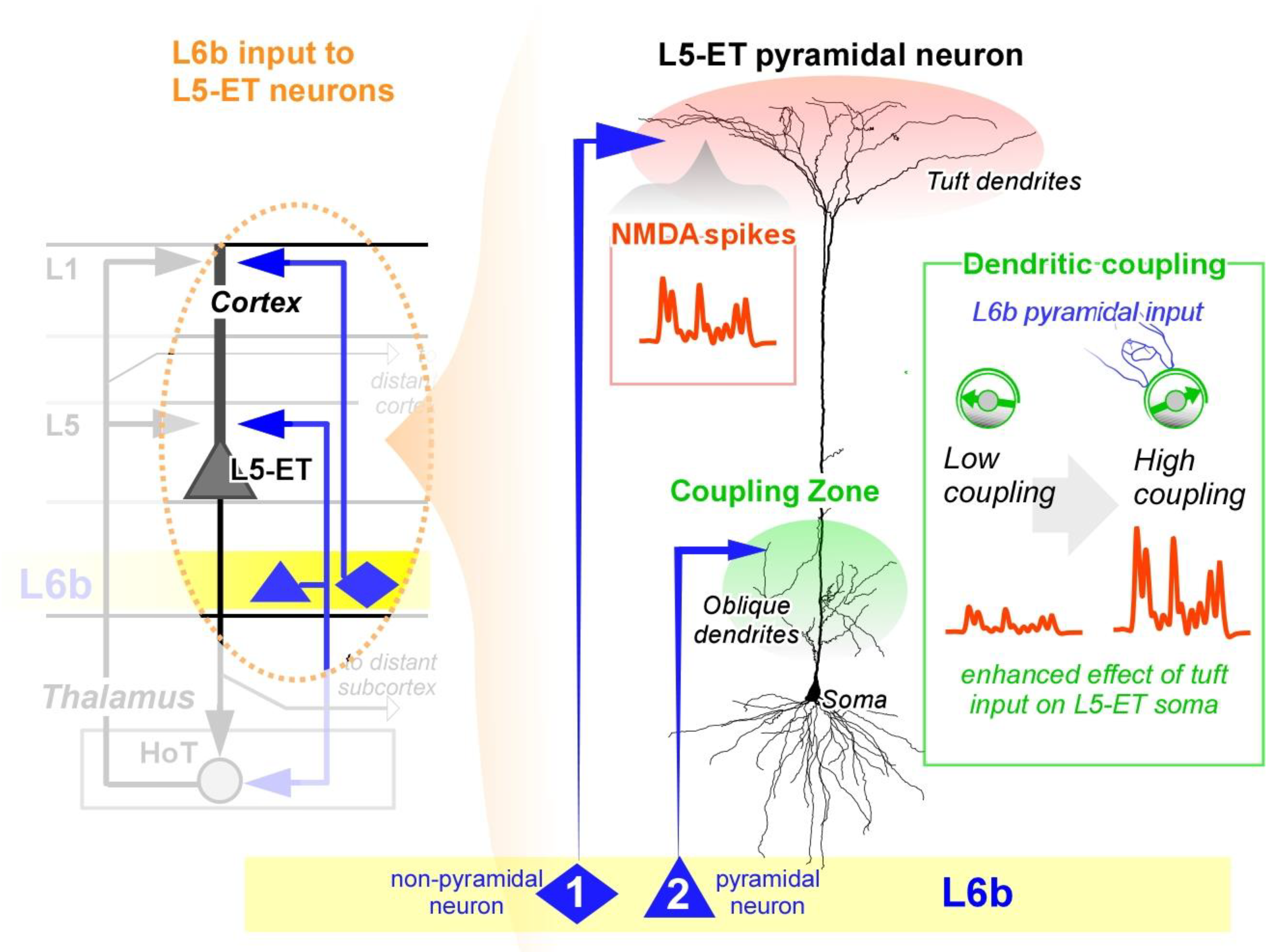
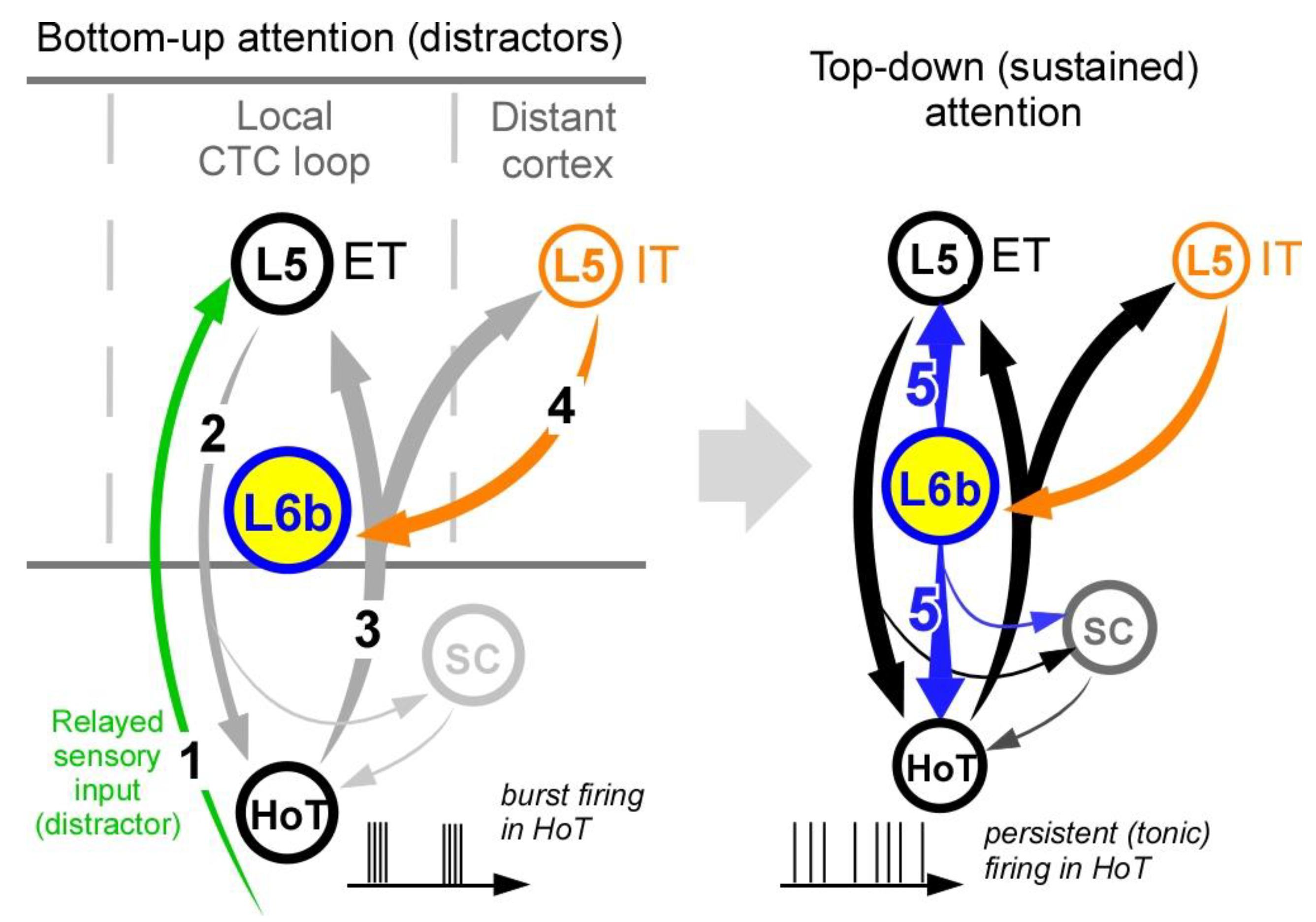
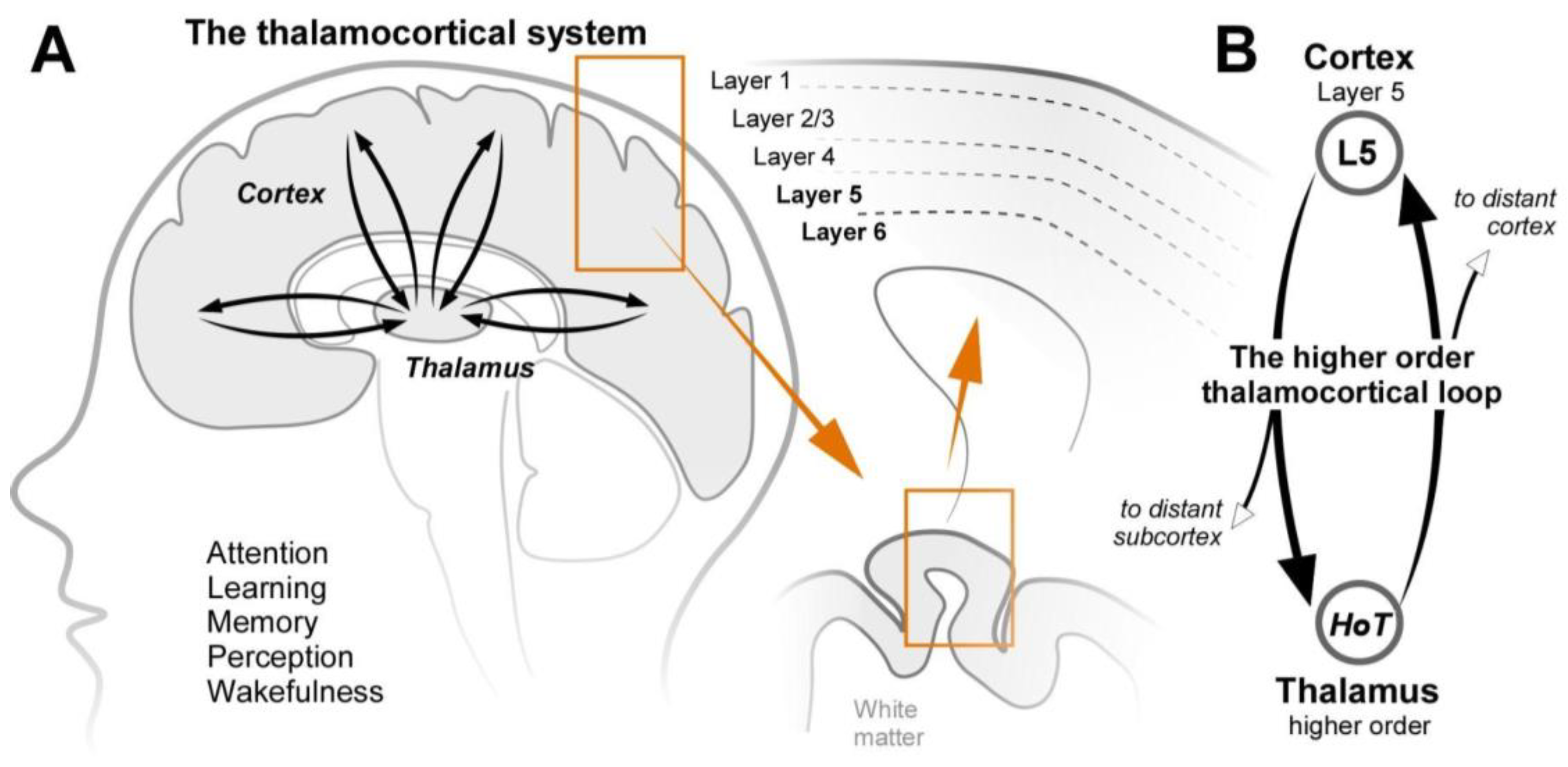
- (1)
- The thalamocortical system plays an important role in learning. (La Terra et al., 2022; Perry et al., 2023). Interestingly, L6b has highly plastic synapses that undergo long-term activity-dependent enhancement. (Zolnik et al., 2024a), thought to be the synaptic signature of learning. Since L6b drives thalamocortical loops associated with perception, could L6b also contribute to perceptual learning, such as learning to distinguish similar sounds in a foreign language through practice)?
- (2)
- The thalamocortical system is linked to memory recall. (Slotnick et al., 2002; Staudigl et al., 2012). When we remember an event. (episodic memory), our brain is thought to re-engages the same or similar cortical-thalamic circuits of the original experience. (Waldhauser et al., 2016; La Terra et al., 2022). Some L6b neurons project to the apical tuft dendrites in layer 1, considered a “memory layer”. (Shin et al., 2021) and also the hippocampus. (Ben-Simon et al., 2022), suggesting an important role for these neurons in memory. Could L6b play a role in supporting episodic memory by reactivating the appropriate thalamocortical loops?
- (3)
- Predictive coding theory suggests that our brain experiences the world by generating predictions generated from higher-order cortex that are fed to lower level circuits. Layer 5 pyramidal neurons are thought to play a critical role in this process. (Bastos et al., 2012; Shipp, 2016). L6b is primarily driven by higher-order cortical feedback, and in turn drives L5 pyramidal neurons, raising the question, Does L6b play an essential role in predictive coding?
- (4)
- During early brain development, L6b. (then called the subplate) plays a unique role by wiring up thalamocortical connections. (Kanold and Luhmann, 2010; Hoerder-Suabedissen and Molnár, 2015). At this time, subplate/L6b neurons receive direct sensory thalamic input, which is lost later in development - input that could help associate sensory input to top down. (attention) signals. Given the idea that L6b drives attention, could this early wiring function of L6b be necessary to prepare thalamocortical circuits for future. (functional) input from L6b that directs attention to the correct sensory signals? In this view, L6b/subplate wires up thalamocortical connections in relation to top-down input that can later properly drive attention.
- (5)
- L6b excitatory neurons drive local interneurons that provide inhibitory feedback. (Zolnik et al., 2020, 2024a). Such excitatory-inhibitory loops are thought to generate oscillations. In the case of L6b, could these excitatory-inhibitory loops set the oscillation frequency of L6b and CTC loops?
- (6)
- Attention and conscious experience are tightly intertwined, with some philosophers considering the two identical. If L6b is critical for promoting attention, is L6b also critical for consciousness itself?
References
- Adamantidis, A.R.; Zhang, F.; Aravanis, A.M.; Deisseroth, K.; de Lecea, L. Neural substrates of awakening probed with optogenetic control of hypocretin neurons. Nature 2007, 450, 420–424. [Google Scholar] [CrossRef] [PubMed]
- Alkire, M.T.; Hudetz, A.G.; Tononi, G. Consciousness and Anesthesia. Science 2008, 322, 876–880. [Google Scholar] [CrossRef] [PubMed]
- Amen, D.G.; Henderson, T.A.; Newberg, A. SPECT Functional Neuroimaging Distinguishes Adult Attention Deficit Hyperactivity Disorder From Healthy Controls in Big Data Imaging Cohorts. Front Psychiatry 2021, 12, 725788. [Google Scholar] [CrossRef]
- Anderson, S.A.; Volk, D.W.; Lewis, D.A. () Increased density of microtubule associated protein 2-immunoreactive neurons in the prefrontal white matter of schizophrenic subjects. Schizophr Res. 1996, 19, 111–119. [Google Scholar] [CrossRef]
- Aru, J.; Suzuki, M.; Larkum, M.E. Cellular Mechanisms of Conscious Processing. Trends Cogn Sci. 2020, S1364661320301753. [Google Scholar] [CrossRef]
- Aru, J.; Suzuki, M.; Rutiku, R.; Larkum, M.E.; Bachmann, T. Coupling the State and Contents of Consciousness. Front 2019, 13, 43. [Google Scholar] [CrossRef]
- Bachmann, T.; Suzuki, M.; Aru, J. Dendritic integration theory, A thalamo-cortical theory of state and content of consciousness. Philos, 31 October 8946. [Google Scholar]
- Bastos, A.M.; Usrey, W.M.; Adams, R.A.; Mangun, G.R.; Fries, P.; Friston, K.J. Canonical microcircuits for predictive coding. Neuron 2012, 76, 695–711. [Google Scholar] [CrossRef]
- Bayer, L.; Serafin, M.; Eggermann, E.; Saint-Mleux, B.; Machard, D.; Jones, B.E.; Mühlethaler, M. Exclusive Postsynaptic Action of Hypocretin-Orexin on Sublayer 6b Cortical Neurons. J 2004, 24, 6760–6764. [Google Scholar] [CrossRef]
- Ben-Simon, Y.; Kaefer, K.; Velicky, P.; Csicsvari, J.; Danzl, J.G.; Jonas, P. A direct excitatory projection from entorhinal layer 6b neurons to the hippocampus contributes to spatial coding and memory. Nat 2022, 13, 4826. [Google Scholar] [CrossRef]
- Boroujeni, K.B.; Helfrich, R.F.; Fiebelkorn, I.C.; Bentley, N.; Lin, J.J.; Knight, R.T.; Kastner, S. (2025) Fast Attentional Information Routing via High-Frequency Bursts in the Human Brain., 2024.09.11.612548 Available at, https, //www.biorxiv.org/content/10.1101/2024.09.11.612548v2 [Accessed , 2025]. 24 April.
- Bruguier, H.; Suarez, R.; Manger, P.; Hoerder-Suabedissen, A.; Shelton, A.M.; Oliver, D.K.; Packer, A.M.; Ferran, J.L.; García-Moreno, F.; Puelles, L.; Molnár, Z. In search of common developmental and evolutionary origin of the claustrum and subplate. J 2020, 528, 2956–2977. [Google Scholar] [CrossRef]
- Brunk, M.G.K.; Deane, K.E.; Kisse, M.; Deliano, M.; Vieweg, S.; Ohl, F.W.; Lippert, M.T.; Happel, M.F.K. Optogenetic stimulation of the VTA modulates a frequency-specific gain of thalamocortical inputs in infragranular layers of the auditory cortex. Sci 2019, 9, 20385. [Google Scholar] [CrossRef] [PubMed]
- Bureau, I.; Paul Fvon, S.; Svoboda, K. Interdigitated Paralemniscal and Lemniscal Pathways in the Mouse Barrel Cortex. PLOS 2006, 4, e382. [Google Scholar] [CrossRef] [PubMed]
- Cano, C.A.; Harel, B.T.; Scammell, T.E. Impaired cognition in narcolepsy, clinical and neurobiological perspectives. Sleep 2024, 47, zsae150. [Google Scholar] [CrossRef] [PubMed]
- Clancy, B.; Cauller, L.J. (1999) Widespread projections from subgriseal neurons. (layer VII) to layer I in adult rat cortex. J Comp Neurol 407, 275–286.
- Constantinidis, C.; Funahashi, S.; Lee, D.; Murray, J.D.; Qi, X.-L.; Wang, M.; Arnsten, A.F.T. Persistent Spiking Activity Underlies Working Memory. J 2018, 38, 7020–7028. [Google Scholar] [CrossRef]
- Corbetta, M.; Shulman, G.L. Control of goal-directed and stimulus-driven attention in the brain. Nat Rev Neurosci 2002, 3, 201–215. [Google Scholar] [CrossRef]
- Crabtree, G.W.; Sun, Z.; Kvajo, M.; Broek, J.A.C.; Fénelon, K.; McKellar, H.; Xiao, L.; Xu, B.; Bahn, S.; O’Donnell, J.M.; Gogos, J.A. (2017) Alteration of Neuronal Excitability and Short-Term Synaptic Plasticity in the Prefrontal Cortex of a Mouse Model of Mental Illness. J Neurosci Off J Soc Neurosci 37, 4158–4180.
- Dehaene, S.; Charles, L.; King, J.-R.; Marti, S. Toward a computational theory of conscious processing. Curr Opin Neurobiol 2014, 25, 76–84. [Google Scholar] [CrossRef]
- Desimone, R.; Duncan, J. Neural mechanisms of selective visual attention. Annu Rev Neurosci 1995, 18, 193–222. [Google Scholar] [CrossRef]
- Ernst, M.; Zametkin, A.J.; Matochik, J.A.; Jons, P.H.; Cohen, R.M. DOPA Decarboxylase Activity in Attention Deficit Hyperactivity Disorder Adults. A [Fluorine-18]Fluorodopa Positron Emission Tomographic Study. J Neurosci 1998, 18, 5901–5907. [Google Scholar] [CrossRef]
- Fang, Z.; Dang, Y.; Ping, A.; Wang, C.; Zhao, Q.; Zhao, H.; Li, X.; Zhang, M. Human high-order thalamic nuclei gate conscious perception through the thalamofrontal loop. Science 2025, 388, eadr3675. [Google Scholar] [CrossRef]
- Feldman, J. The neural binding problem(s). Cogn 2013, 7, 1–11. [Google Scholar] [CrossRef]
- Feldmeyer, D. (2023a) Structure and function of neocortical layer 6b. Front Cell Neurosci 17, 1257803.
- Feldmeyer, D. (2023b) Structure and function of neocortical layer 6b. Front Cell Neurosci 17, 1257803.
- Fénelon, K.; Xu, B.; Lai, C.S.; Mukai, J.; Markx, S.; Stark, K.L.; Hsu, P.-K.; Gan, W.-B.; Fischbach, G.D.; MacDermott, A.B.; Karayiorgou, M.; Gogos, J.A. The Pattern of Cortical Dysfunction in a Mouse Model of a Schizophrenia-Related Microdeletion. J 2013, 33, 14825–14839. [Google Scholar] [CrossRef] [PubMed]
- Findley, L.J.; Suratt, P.M.; Dinges, D.F. Time-on-task decrements in “steer clear” performance of patients with sleep apnea and narcolepsy. Sleep 1999, 22, 804–809. [Google Scholar] [CrossRef] [PubMed]
- Fogerson, P.M.; Huguenard, J.R. Tapping the Brakes, Cellular and Synaptic Mechanisms that Regulate Thalamic Oscillations. Neuron 2016, 92, 687–704. [Google Scholar] [CrossRef] [PubMed]
- Fries, P. Neuronal Gamma-Band Synchronization as a Fundamental Process in Cortical Computation. Annu Rev Neurosci 2009, 32, 209–224. [Google Scholar] [CrossRef]
- Fronczek, R.; Middelkoop, H.A.M.; van Dijk, J.G.; Lammers, G.J. Focusing on vigilance instead of sleepiness in the assessment of narcolepsy, high sensitivity of the Sustained Attention to Response Task. (SART). Sleep 2006, 29, 187–191. [Google Scholar]
- Gent, T.C.; Bandarabadi, M.; Herrera, C.G.; Adamantidis, A.R. Thalamic dual control of sleep and wakefulness. Nat 2018, 21, 974–984. [Google Scholar] [CrossRef]
- Goldman-Rakic, P.S. Cellular basis of working memory. Neuron 1995, 14, 477–485. [Google Scholar] [CrossRef]
- Granato, A.; Phillips, W.A.; Schulz, J.M.; Suzuki, M.; Larkum, M.E. Dysfunctions of cellular context-sensitivity in neurodevelopmental learning disabilities. Neurosci Biobehav Rev 2024, 161, 105688. [Google Scholar] [CrossRef]
- Haas, H.L.; Sergeeva, O.A.; Selbach, O. Histamine in the Nervous System. Physiol 2008, 88, 1183–1241. [Google Scholar] [CrossRef]
- Halassa, M.M.; Kastner, S. Thalamic functions in distributed cognitive control. Nat 2017, 20, 1669–1679. [Google Scholar] [CrossRef]
- Halassa, M.M.; Sherman, S.M. Thalamocortical Circuit Motifs, A General Framework. Neuron 2019, 103, 762–770. [Google Scholar] [CrossRef] [PubMed]
- Hatanaka, Y.; Namikawa, T.; Yamauchi, K.; Kawaguchi, Y. Cortical Divergent Projections in Mice Originate from Two Sequentially Generated, Distinct Populations of Excitatory Cortical Neurons with Different Initial Axonal Outgrowth Characteristics. Cereb 2016, 26, 2257–2270. [Google Scholar] [CrossRef] [PubMed]
- Hoerder-Suabedissen, A.; Hayashi, S.; Upton, L.; Nolan, Z.; Casas-Torremocha, D.; Grant, E.; Viswanathan, S.; Kanold, P.O.; Clasca, F.; Kim, Y.; Molnár, Z. Subset of Cortical Layer 6b Neurons Selectively Innervates Higher Order Thalamic Nuclei in Mice. Cereb 2018, 28, 1882–1897. [Google Scholar] [CrossRef] [PubMed]
- Hoerder-Suabedissen, A.; Molnár, Z. Development, evolution and pathology of neocortical subplate neurons. Nat Rev Neurosci 2015, 16, 133–146. [Google Scholar] [CrossRef]
- Hoerder-Suabedissen, A.; Oeschger, F.M.; Krishnan, M.L.; Belgard, T.G.; Wang, W.Z.; Lee, S.; Webber, C.; Petretto, E.; Edwards, A.D.; Molnár, Z. Expression profiling of mouse subplate reveals a dynamic gene network and disease association with autism and schizophrenia. Proc Natl Acad Sci 2013, 110, 3555–3560. [Google Scholar] [CrossRef]
- Jackman, S.L.; Regehr, W.G. The Mechanisms and Functions of Synaptic Facilitation. Neuron 2017, 94, 447–464. [Google Scholar] [CrossRef]
- Javitt, D.C.; Zukin, S.R. Recent advances in the phencyclidine model of schizophrenia. Am J Psychiatry 1991, 148, 1301–1308. [Google Scholar]
- Jennings, K.J.; de Lecea, L. (2019) Chapter 1 - Hypocretins. (Orexins), Twenty Years of Dissecting Arousal Circuits. In, The Orexin/Hypocretin System. (Burk JA, Fadel JR, eds), pp 1–29. Academic Press. Available at, https, //www.sciencedirect.com/science/article/pii/B9780128137512000012 [Accessed , 2024]. 5 November.
- Jiao Zet, a.l. (2025) Projectome-based characterization of hypothalamic peptidergic neurons in male mice. Nat Neurosci, 1–16.
- Kanold, P.O.; Luhmann, H.J. The Subplate and Early Cortical Circuits. Annu Rev Neurosci 2010, 33, 23–48. [Google Scholar] [CrossRef]
- Kim, E.J.; Juavinett, A.L.; Kyubwa, E.M.; Jacobs, M.W.; Callaway, E.M. Three Types of Cortical Layer 5 Neurons That Differ in Brain-wide Connectivity and Function. Neuron 2015, 88, 1253–1267. [Google Scholar] [CrossRef]
- Kim, J.; Lee, G.-H.; Sung, S.M.; Jung, D.S.; Pak, K. Prevalence of attention deficit hyperactivity disorder symptoms in narcolepsy, a systematic review. Sleep 2020, 65, 84–88. [Google Scholar] [CrossRef]
- Kim, M.; Lee, T.H.; Park, H.; Moon, S.-Y.; Lho, S.K.; Kwon, J.S. Thalamocortical dysrhythmia in patients with schizophrenia spectrum disorder and individuals at clinical high risk for psychosis. Neuropsychopharmacology 2022, 47, 673–680. [Google Scholar] [CrossRef] [PubMed]
- Kinney, H.C.; Korein, J.; Panigrahy, A.; Dikkes, P.; Goode, R. Neuropathological Findings in the Brain of Karen Ann Quinlan – The Role of the Thalamus in the Persistent Vegetative State. N Engl J Med 1994, 330, 1469–1475. [Google Scholar] [CrossRef]
- Klatzmann, U.; Froudist-Walsh, S.; Bliss, D.P.; Theodoni, P.; Mejías, J.; Niu, M.; Rapan, L.; Palomero-Gallagher, N.; Sergent, C.; Dehaene, S.; Wang, X.-J. A dynamic bifurcation mechanism explains cortex-wide neural correlates of conscious access. Cell, 8 April 2211. [Google Scholar]
- Koehler, S.; Lauer, P.; Schreppel, T.; Jacob, C.; Heine, M.; Boreatti-Hümmer, A.; Fallgatter, A.J.; Herrmann, M.J. (2009) Increased EEG power density in alpha and theta bands in adult ADHD patients. J Neural Transm Vienna Austria 1996, 116, 97–104. [Google Scholar] [CrossRef]
- Krauzlis, R.J.; Lovejoy, L.P.; Zénon, A. Superior Colliculus and Visual Spatial Attention. Annu Rev Neurosci 2013, 36, 165–182. [Google Scholar] [CrossRef]
- Kronemer SIet, a.l. Human visual consciousness involves large scale cortical and subcortical networks independent of task report and eye movement activity. Nat Commun 2022, 13, 7342. [Google Scholar] [CrossRef]
- Kubo, K. Increased densities of white matter neurons as a cross-disease feature of neuropsychiatric disorders. Psychiatry Clin Neurosci 2020, 74, 166–175. [Google Scholar] [CrossRef]
- La Terra, D.; Bjerre, A.-S.; Rosier, M.; Masuda, R.; Ryan, T.J.; Palmer, L.M. The role of higher-order thalamus during learning and correct performance in goal-directed behavior Abdus-Saboor I, Gold JI, eds. eLife 2022, 11, e77177. [Google Scholar] [CrossRef]
- Larkum, M. A cellular mechanism for cortical associations, an organizing principle for the cerebral cortex. Trends Neurosci 2013, 36, 141–151. [Google Scholar] [CrossRef]
- Larkum, M.E.; Zhu, J.J.; Sakmann, B. Dendritic mechanisms underlying the coupling of the dendritic with the axonal action potential initiation zone of adult rat layer 5 pyramidal neurons. J Physiol 2001, 533, 447–466. [Google Scholar] [CrossRef]
- Laureys, S. Tracking the recovery of consciousness from coma. J Clin Invest 2006, 116, 1823–1825. [Google Scholar] [CrossRef]
- Law, R.G.; Pugliese, S.; Shin, H.; Sliva, D.D.; Lee, S.; Neymotin, S.; Moore, C.; Jones, S.R. Thalamocortical Mechanisms Regulating the Relationship between Transient Beta Events and Human Tactile Perception. Cereb Cortex 2022, 32, 668–688. [Google Scholar] [CrossRef] [PubMed]
- Ledderose, J.M.T.; Zolnik, T.A.; Toumazou, M.; Trimbuch, T.; Rosenmund, C.; Eickholt, B.J.; Jaeger, D.; Larkum, M.E.; Sachdev, R.N.S. Layer 1 of somatosensory cortex, an important site for input to a tiny cortical compartment. Cereb Cortex 2023, 33, 11354–11372. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Sroubek, A.; Kelly, M.S.; Lesser, I.; Sussman, E.; He, Y.; Branch, C.; Foxe, J.J. Atypical pulvinar-cortical pathways during sustained attention performance in children with attention-deficit/hyperactivity disorder. J Am Acad Child Adolesc Psychiatry 2012, 51, 1197–1207.e4. [Google Scholar] [CrossRef] [PubMed]
- Llinás, R. Consciousness and the thalamocortical loop. Int Congr Ser 2003, 1250, 409–416. [Google Scholar] [CrossRef]
- Llinas, R.R. (2002) I of the Vortex, From Neurons to Self. MIT Press.
- Llinás, R.R.; Ribary, U.; Jeanmonod, D.; Kronberg, E.; Mitra, P.P. Thalamocortical dysrhythmia, A neurological and neuropsychiatric syndrome characterized by magnetoencephalography. Proc Natl Acad Sci 1999, 96, 15222–15227. [Google Scholar] [CrossRef]
- Mah, W. Aberrant Thalamocortical Synchrony Associated with Behavioral Manifestations in Git1-/- Mice. Exp Neurobiol 2015, 24, 126–132. [Google Scholar] [CrossRef]
- Mahoney, C.E.; Cogswell, A.; Koralnik, I.J.; Scammell, T.E. The neurobiological basis of narcolepsy. Nat Rev Neurosci 2019, 20, 83–93. [Google Scholar] [CrossRef]
- Marx, M.; Feldmeyer, D. Morphology and Physiology of Excitatory Neurons in Layer 6b of the Somatosensory Rat Barrel Cortex. Cereb Cortex 2013, 23, 2803–2817. [Google Scholar] [CrossRef]
- Marx, M.; Qi, G.; Hanganu-Opatz, I.L.; Kilb, W.; Luhmann, H.J.; Feldmeyer, D. Neocortical Layer 6B as a Remnant of the Subplate - A Morphological Comparison. Cereb Cortex 2017, 27, 1011–1026. [Google Scholar] [CrossRef]
- Masse, N.Y.; Yang, G.R.; Song, H.F.; Wang, X.-J.; Freedman, D.J. Circuit mechanisms for the maintenance and manipulation of information in working memory. Nat Neurosci 2019, 22, 1159–1167. [Google Scholar] [CrossRef]
- McCormick, D.A.; Nestvogel, D.B.; He, B.J. Neuromodulation of Brain State and Behavior. Annu Rev Neurosci 2020, 43, 391–415. [Google Scholar] [CrossRef] [PubMed]
- Meijer, E.J.; Mueller, M.; Krone, L.B.; Yamagata, T.; Hoerder-Suabedissen, A.; Wilcox, S.; Alfonsa, H.; Chakrabarty, A.; Guidi, L.; Oliver, P.L.; Vyazovskiy, V.V.; Molnár, Z. (2025) Cortical layer 6b mediates state-dependent changes in brain activity and effects of orexin on waking and sleep., 2024.10.26.620399 Available at, https, //www.biorxiv.org/content/10.1101/2024.10.26.620399v2 [Accessed , 2025]. 24 April.
- Merker, B.H. (2013) The efference cascade, consciousness, and its self, naturalizing the first person pivot of action control. Front Psychol 4 Available at, https, //www.frontiersin.orghttps, //www.frontiersin.org/journals/psychology/articles/10.3389/fpsyg.2013.00501/full [Accessed , 2025]. 25 April.
- Miao, S.; Han, J.; Gu, Y.; Wang, X.; Song, W.; Li, D.; Liu, Z.; Yang, J.; Li, X. (2017) Reduced Prefrontal Cortex Activation in Children with Attention-Deficit/Hyperactivity Disorder during Go/No-Go Task, A Functional Near-Infrared Spectroscopy Study. Front Neurosci 11 Available at, https, //www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2017.00367/full [Accessed , 2025]. 8 April.
- Moberg, S.; Takahashi, N. (2022) Neocortical layer 5 subclasses, From cellular properties to roles in behavior. Front Synaptic Neurosci 14 Available at, https, //www.frontiersin.org/journals/synaptic-neuroscience/articles/10.3389/fnsyn.2022.1006773/full [Accessed , 2024]. 31 October.
- Moghaddam, B.; Javitt, D. From Revolution to Evolution, The Glutamate Hypothesis of Schizophrenia and its Implication for Treatment. Neuropsychopharmacology 2012, 37, 4–15. [Google Scholar] [CrossRef] [PubMed]
- Mohn, A.R.; Gainetdinov, R.R.; Caron, M.G.; Koller, B.H. Mice with reduced NMDA receptor expression display behaviors related to schizophrenia. Cell 1999, 98, 427–436. [Google Scholar] [CrossRef] [PubMed]
- Molnár, Z. Cortical layer with no known function. Eur J Neurosci 2018, 49, 957. [Google Scholar] [CrossRef]
- Mongillo, G.; Barak, O.; Tsodyks, M. Synaptic Theory of Working Memory. Science 2008, 319, 1543–1546. [Google Scholar] [CrossRef]
- Noirhomme, Q.; Soddu, A.; Lehembre, R.; Vanhaudenhuyse, A.; Boveroux, P.; Boly, M.; Laureys, S. Brain Connectivity in Pathological and Pharmacological Coma. Front Syst Neurosci, 31 October 3389. [Google Scholar]
- Ohno, S.; Kuramoto, E.; Furuta, T.; Hioki, H.; Tanaka, Y.R.; Fujiyama, F.; Sonomura, T.; Uemura, M.; Sugiyama, K.; Kaneko, T. A Morphological Analysis of Thalamocortical Axon Fibers of Rat Posterior Thalamic Nuclei, A Single Neuron Tracing Study with Viral Vectors. Cereb Cortex 2012, 22, 2840–2857. [Google Scholar] [CrossRef]
- Pereira, M.; Faivre, N.; Bernasconi, F.; Brandmeir, N.; Suffridge, J.; Tran, K.; Wang, S.; Finomore, V.; Konrad, P.; Rezai, A.; Blanke, O. Subcortical correlates of consciousness with human single neuron recordings. eLife, 16 March 9527. [Google Scholar]
- Perry, B.A.L.; Mendez, J.C.; Mitchell, A.S. Cortico-thalamocortical interactions for learning, memory and decision-making. J Physiol 2023, 601, 25–35. [Google Scholar] [CrossRef]
- Phillips, W.A. (2023) The Cooperative Neuron, Cellular Foundations of Mental Life, 1st ed. Oxford University PressOxford. Available at, https, //academic.oup.com/book/46060 [Accessed , 2023]. 16 November.
- Phillips, W.A.; Bachmann, T.; Spratling, M.W.; Muckli, L.; Petro, L.S.; Zolnik, T. Cellular psychology, relating cognition to context-sensitive pyramidal cells. Trends Cogn Sci 2025, 29, 28–40. [Google Scholar] [CrossRef]
- Portas, C.M.; Rees, G.; Howseman, A.M.; Josephs, O.; Turner, R.; Frith, C.D. A Specific Role for the Thalamus in Mediating the Interaction of Attention and Arousal in Humans. J Neurosci 1998, 18, 8979–8989. [Google Scholar] [CrossRef]
- Posada-Quintero, H.F.; Reljin, N.; Bolkhovsky, J.B.; Orjuela-Cañón, A.D.; Chon, K.H. Brain Activity Correlates With Cognitive Performance Deterioration During Sleep Deprivation. Front Neurosci 2019, 13, 1001. [Google Scholar] [CrossRef]
- Qi, G.; Yang, D.; Messore, F.; Bast, A.; Yáñez, F.; Oberlaender, M.; Feldmeyer, D. FOXP2-immunoreactive corticothalamic neurons in neocortical layers 6a and 6b are tightly regulated by neuromodulatory systems. iScience, 1 April 2589. [Google Scholar]
- Rao, N.; Northoff, G.; Tagore, A.; Rusjan, P.; Kenk, M.; Wilson, A.; Houle, S.; Strafella, A.; Remington, G.; Mizrahi, R. Impaired Prefrontal Cortical Dopamine Release in Schizophrenia During a Cognitive Task, A [11C]FLB 457 Positron Emission Tomography Study. Schizophr Bull 2019, 45, 670–679. [Google Scholar] [CrossRef] [PubMed]
- Reichova, I.; Sherman, S.M. Somatosensory Corticothalamic Projections, Distinguishing Drivers From Modulators. J Neurophysiol 2004, 92, 2185–2197. [Google Scholar] [CrossRef] [PubMed]
- Rockland, K.S. Collateral branching of long-distance cortical projections in monkey. J Comp Neurol 2013, 521, 4112–4123. [Google Scholar] [CrossRef] [PubMed]
- Rockland, K.S.; Drash, G.W. Collateralized divergent feedback connections that target multiple cortical areas. J Comp Neurol 1996, 373, 529–548. [Google Scholar] [CrossRef]
- Rodriguez, E.; George, N.; Lachaux, J.P.; Martinerie, J.; Renault, B.; Varela, F.J. Perception’s shadow, long-distance synchronization of human brain activity. Nature 1999, 397, 430–433. [Google Scholar] [CrossRef]
- Roskies, A.L. The Binding Problem. Neuron 1999, 24, 7–9. [Google Scholar] [CrossRef]
- Ross, B.; Fujioka, T. 40-Hz oscillations underlying perceptual binding in young and older adults. Psychophysiology 2016, 53, 974–990. [Google Scholar] [CrossRef]
- Saalmann, Y.B.; Kastner, S. Cognitive and Perceptual Functions of the Visual Thalamus. Neuron 2011, 71, 209–223. [Google Scholar] [CrossRef]
- Saalmann, Y.B.; Pinsk, M.A.; Wang, L.; Li, X.; Kastner, S. The Pulvinar Regulates Information Transmission Between Cortical Areas Based on Attention Demands. Science 2012, 337, 753–756. [Google Scholar] [CrossRef]
- Sakurai, T.; Saito, Y.C. ; Yanagisawa M(2021) Interaction between Orexin Neurons Monoaminergic Systems In Frontiers of Neurology Neuroscience (Steiner, M.A.; Yanagisawa, M.; Clozel, M.; eds); pp 11–21,,, S. Karger AG. Available at, https, //karger.com/books/book/, 5 November 5563. [Google Scholar]
- Sergent, C.; Corazzol, M.; Labouret, G.; Stockart, F.; Wexler, M.; King, J.-R.; Meyniel, F.; Pressnitzer, D. Bifurcation in brain dynamics reveals a signature of conscious processing independent of report. Nat Commun 2021, 12, 1149. [Google Scholar] [CrossRef]
- Sermet, B.S.; Truschow, P.; Feyerabend, M.; Mayrhofer, J.M.; Oram, T.B.; Yizhar, O.; Staiger, J.F.; Petersen, C.C. (2019) Pathway-, layer- and cell-type-specific thalamic input to mouse barrel cortex Nelson SB, Marder E, Nelson SB, Feldman DE, eds. eLife 8, e52665.
- Shepherd, G.M.G.; Yamawaki, N. Untangling the cortico-thalamo-cortical loop, cellular pieces of a knotty circuit puzzle. Nat Rev Neurosci 2021, 22, 389–406. [Google Scholar] [CrossRef] [PubMed]
- Sherman, S.M. Thalamus plays a central role in ongoing cortical functioning. Nat Neurosci 2016, 19, 533–541. [Google Scholar] [CrossRef] [PubMed]
- Sherman, S.M.; Usrey, W.M. Transthalamic Pathways for Cortical Function. J Neurosci 2024, 44, e0909242024. [Google Scholar] [CrossRef]
- Shin, J.N.; Doron, G.; Larkum, M.E. (2021) Neocortical Layer 1 - The Memory Layer? Science 374, 538–539.
- Shine, J. (2020) The thalamus integrates the macrosystems of the brain to facilitate complex, adaptive brain network dynamics. Prog Neurobiol, 31 October 9740. [Google Scholar]
- Shine, J.M.; Lewis, L.D.; Garrett, D.D.; Hwang, K. The impact of the human thalamus on brain-wide information processing. Nat Rev Neurosci 2023, 24, 416–430. [Google Scholar] [CrossRef]
- Shipp, S. (2016) Neural Elements for Predictive Coding. Front Psychol 7 Available at, https, //www.frontiersin.orghttps, //www.frontiersin.org/journals/psychology/articles/10.3389/fpsyg.2016.01792/full [Accessed , 2025]. 25 April.
- Slifstein, M.; van de Giessen, E.; Van Snellenberg, J.; Thompson, J.L.; Narendran, R.; Gil, R.; Hackett, E.; Girgis, R.; Ojeil, N.; Moore, H.; D’Souza, D.; Malison, R.T.; Huang, Y.; Lim, K.; Nabulsi, N.; Carson, R.E.; Lieberman, J.A.; Abi-Dargham, A. Deficits in Prefrontal Cortical and Extrastriatal Dopamine Release in Schizophrenia, A Positron Emission Tomographic Functional Magnetic Resonance Imaging Study. JAMA Psychiatry 2015, 72, 316–324. [Google Scholar] [CrossRef]
- Slotnick, S.D.; Moo, L.R.; Kraut, M.A.; Lesser, R.P.; Hart, J. (2002) Interactions between thalamic and cortical rhythms during semantic memory recall in human. Proc Natl Acad Sci 99, 6440–6443.
- Staudigl, T.; Zaehle, T.; Voges, J.; Hanslmayr, S.; Esslinger, C.; Hinrichs, H.; Schmitt, F.C.; Heinze, H.-J.; Richardson-Klavehn, A. Memory signals from the thalamus, Early thalamocortical phase synchronization entrains gamma oscillations during long-term memory retrieval. Neuropsychologia 2012, 50, 3519–3527. [Google Scholar] [CrossRef]
- Steriade, M. Corticothalamic resonance, states of vigilance and mentation. Neuroscience 2000, 101, 243–276. [Google Scholar] [CrossRef]
- Steriade, M.; McCormick, D.A.; Sejnowski, T.J. Thalamocortical Oscillations in the Sleeping and Aroused Brain. Science 1993, 262, 679–685. [Google Scholar] [CrossRef]
- Stokes, M.G. ‘Activity-silent’ working memory in prefrontal cortex, a dynamic coding framework. Trends Cogn Sci 2015, 19, 394–405. [Google Scholar] [CrossRef]
- Suzuki, M.; Larkum, M.E. General anesthesia decouples cortical pyramidal neurons. Cell 2020, 180, 666–676.e13. [Google Scholar] [CrossRef]
- Takahashi, N.; Ebner, C.; Sigl-Glöckner, J.; Moberg, S.; Nierwetberg, S.; Larkum, M.E. Active dendritic currents gate descending cortical outputs in perception. Nat Neurosci 2020, 23, 1277–1285. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, N.; Oertner, T.G.; Hegemann, P.; Larkum, M.E. Active cortical dendrites modulate perception. Science 2016, 354, 1587–1590. [Google Scholar] [CrossRef] [PubMed]
- Tolner, E.A.; Sheikh, A.; Yukin, A.Y.; Kaila, K.; Kanold, P.O. Subplate Neurons Promote Spindle Bursts and Thalamocortical Patterning in the Neonatal Rat Somatosensory Cortex. J Neurosci 2012, 32, 692–702. [Google Scholar] [CrossRef]
- Treisman, A. The binding problem. Curr Opin Neurobiol 1996, 6, 171–178. [Google Scholar] [CrossRef]
- Treisman, A.M.; Gelade, G. A feature-integration theory of attention. Cognit Psychol 1980, 12, 97–136. [Google Scholar] [CrossRef]
- Tsai, S.-H.; Tsao, C.-Y.; Lee, L.-J. (2020) Altered White Matter and Layer VIb Neurons in Heterozygous Disc1 Mutant, a Mouse Model of Schizophrenia. Front Neuroanat 14 Available at, https, //www.frontiersin.org/journals/neuroanatomy/articles/10.3389/fnana.2020.605029/full [Accessed , 2025]. 31 March.
- Vanneste, S.; Song, J.-J.; De Ridder, D. Thalamocortical dysrhythmia detected by machine learning. Nat Commun 2018, 9, 1103. [Google Scholar] [CrossRef]
- Venkatesan, S.; Chen, T.; Liu, Y.; Turner, E.E.; Tripathy, S.J.; Lambe, E.K. (2023) Chrna5 and lynx prototoxins identify acetylcholine super-responder subplate neurons. iScience 26 Available at, https, //www.cell.com/iscience/abstract/S2589-0042(23)00069-X [Accessed , 2025]. 2 April.
- Waldhauser, G.T.; Braun, V.; Hanslmayr, S. Episodic Memory Retrieval Functionally Relies on Very Rapid Reactivation of Sensory Information. J Neurosci 2016, 36, 251–260. [Google Scholar] [CrossRef]
- Wenger Combremont, A.-L.; Bayer, L. ; Dupré,, A.; Mühlethaler, M.; Serafin, M. (2016) Slow Bursting Neurons of Mouse Cortical Layer 6b Are Depolarized by Hypocretin/Orexin and Major Transmitters of Arousal. Front Neurol 7 Available at, https, //www.frontiersin.org/journals/neurology/articles/10.3389/fneur.2016.00088/full [Accessed , 2024]. 5 November.
- Whitmire, C.J.; Liew, Y.J.; Stanley, G.B. Thalamic state influences timing precision in the thalamocortical circuit. J Neurophysiol 2021, 125, 1833–1850. [Google Scholar] [CrossRef]
- Whyte, C.J.; Redinbaugh, M.J.; Shine, J.M.; Saalmann, Y.B. (2024) Thalamic contributions to the state and contents of consciousness. Neuron 112, 1611–1625.
- Wimmer, R.D.; Schmitt, L.I.; Davidson, T.J.; Nakajima, M.; Deisseroth, K.; Halassa, M.M. Thalamic control of sensory selection in divided attention. Nature 2015, 526, 705–709. [Google Scholar] [CrossRef]
- Zametkin, A.J.; Nordahl, T.E.; Gross, M.; King, A.C.; Semple, W.E.; Rumsey, J.; Hamburger, S.; Cohen, R.M. (1990) Cerebral Glucose Metabolism in Adults with Hyperactivity of Childhood Onset. N Engl J Med 323, 1361–1366.
- Zhang, S.; Xu, M.; Kamigaki, T.; Hoang Do, J.P.; Chang, W.-C.; Jenvay, S.; Miyamichi, K.; Luo, L.; Dan, Y. Long-range and local circuits for top-down modulation of visual cortex processing. Science 2014, 345, 660–665. [Google Scholar] [CrossRef]
- Zhou, J.; Liu, X.; Song, W.; Yang, Y.; Zhao, Z.; Ling, F.; Hudetz, A.; Li, S.-J. Specific and nonspecific thalamocortical functional connectivity in normal and vegetative states. Conscious Cogn 2011, 20, 257–268. [Google Scholar] [CrossRef] [PubMed]
- Zolnik, T.A.; Bronec, A.; Ross, A.; Staab, M.; Sachdev, R.N.S.; Molnár, Z.; Eickholt, B.J.; Larkum, M.E. (2024a) Layer 6b controls brain state via apical dendrites and the higher-order thalamocortical system. Neuron 112, 805-820.e4.
- Zolnik, T.A.; Ledderose, J.; Toumazou, M.; Trimbuch, T.; Oram, T.; Rosenmund, C.; Eickholt, B.J.; Sachdev, R.N.S.; Larkum, M.E. Layer 6b Is Driven by Intracortical Long-Range Projection Neurons. Cell Rep 2020, 30, 3492–3505.e5. [Google Scholar] [CrossRef] [PubMed]
- Zolnik TA, Meenakshisundaram A, Mueller M, Onken J, Sauvigny T, Thomale U-W, Schneider UC.
- Sachdev, R.N.; Fidzinski, P.; Holtkamp, M.; Schmitz, D.; Kaindl, A.M.; Geiger, J.; Molnar, Z.; Eickholt, B.J.; Larkum, M.E. (2024b) It’s all in your head, layer 6b and the orexin-activated neurons of the human cortex. 382.13, Soc Neurosci Poster Present.
Disclaimer/Publisher’s Note, The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



