Submitted:
03 June 2025
Posted:
04 June 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. K/BxN Mice
2.2. Reagents
2.3. Arthritis Scoring and Treatment
2.4. Organs Examined
2.5. Statistical Analysis
3. Results
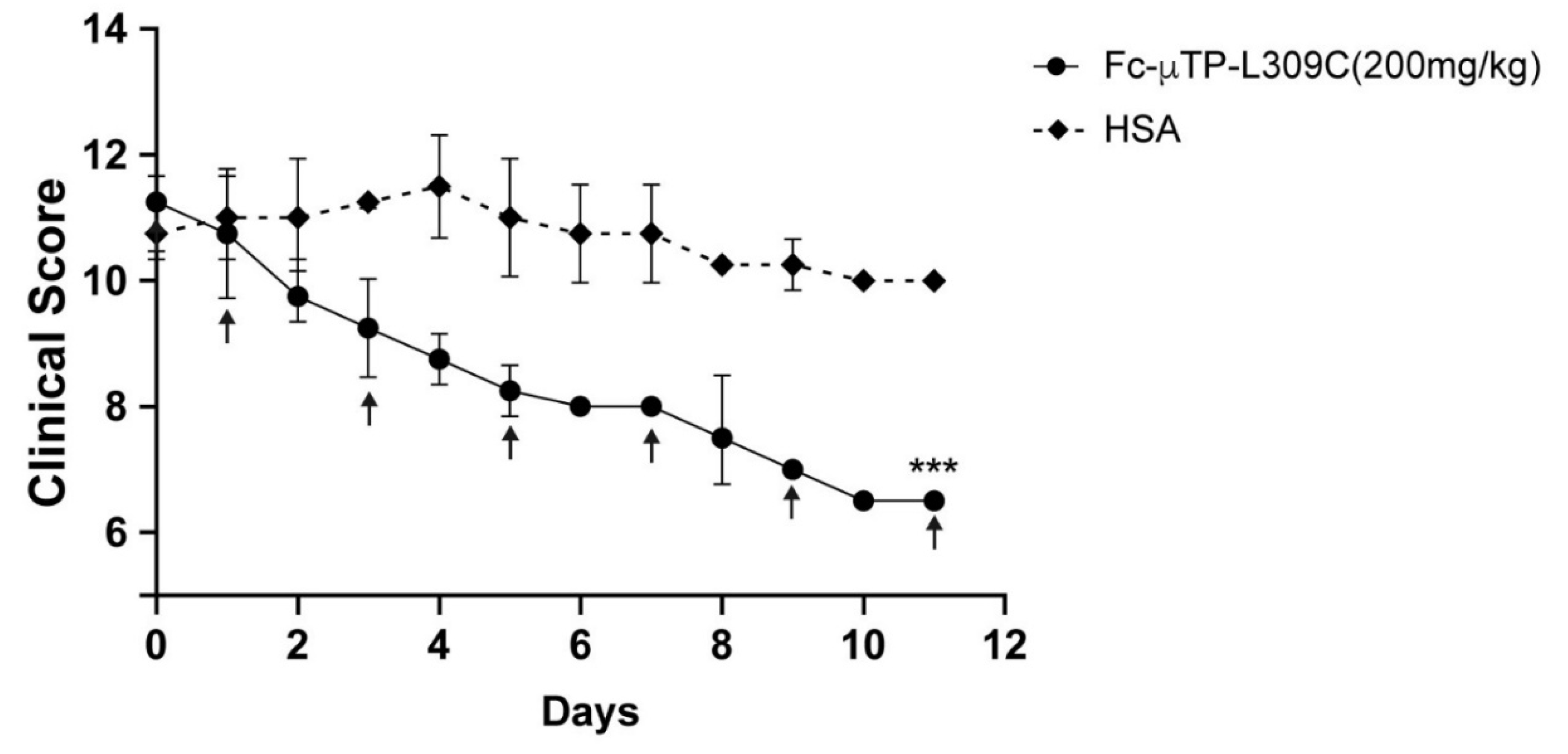
3.1. rFc-µTP-L309C Ameliorates Endogenous Arthritis in K/BxN Mice
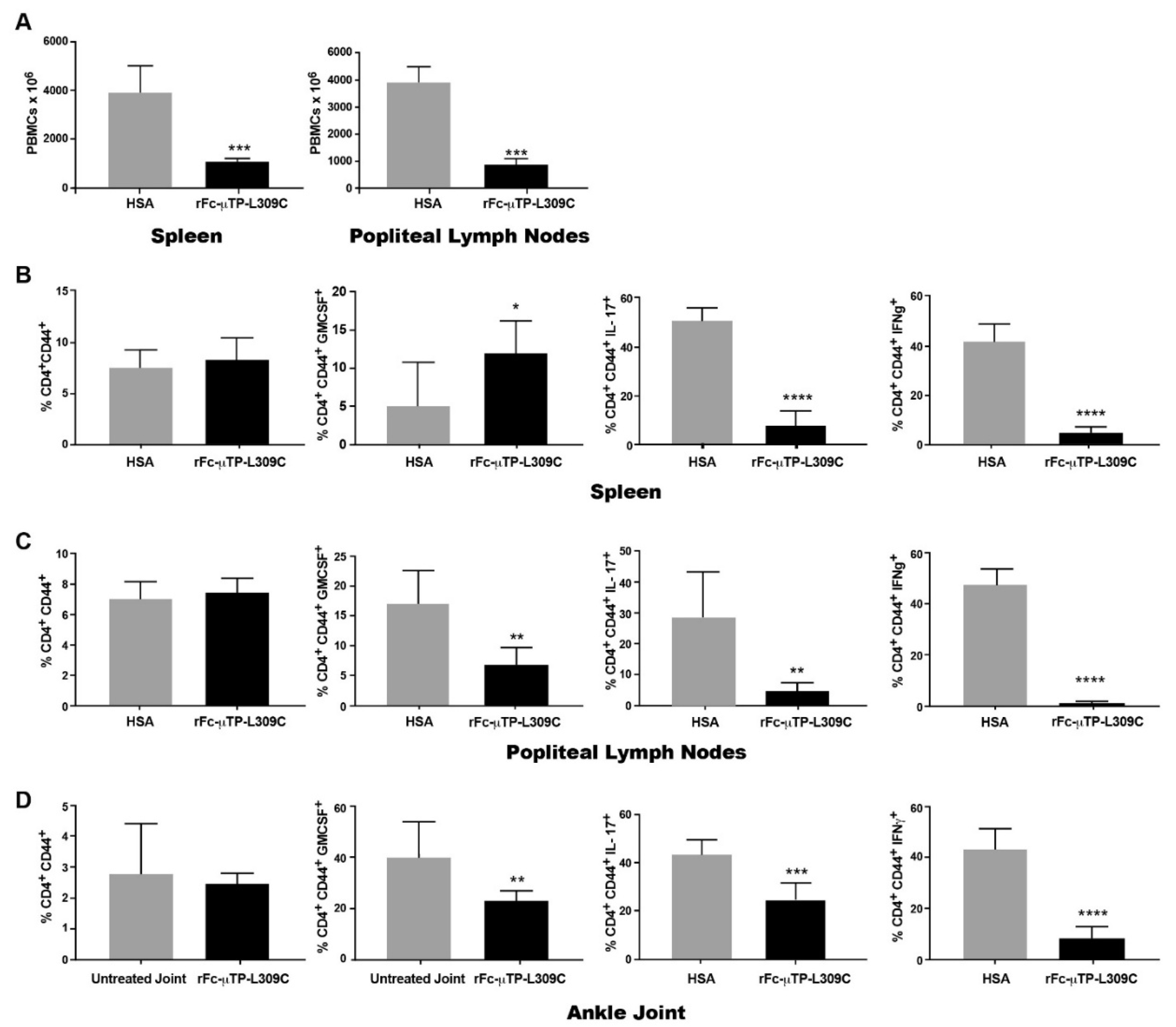
3.2. Th1 and Th17 T-Cells Are Decreased in the Spleen, Lymph Nodes and Joints of K/BxN Mice After rFc-µTP-L309C Treatment
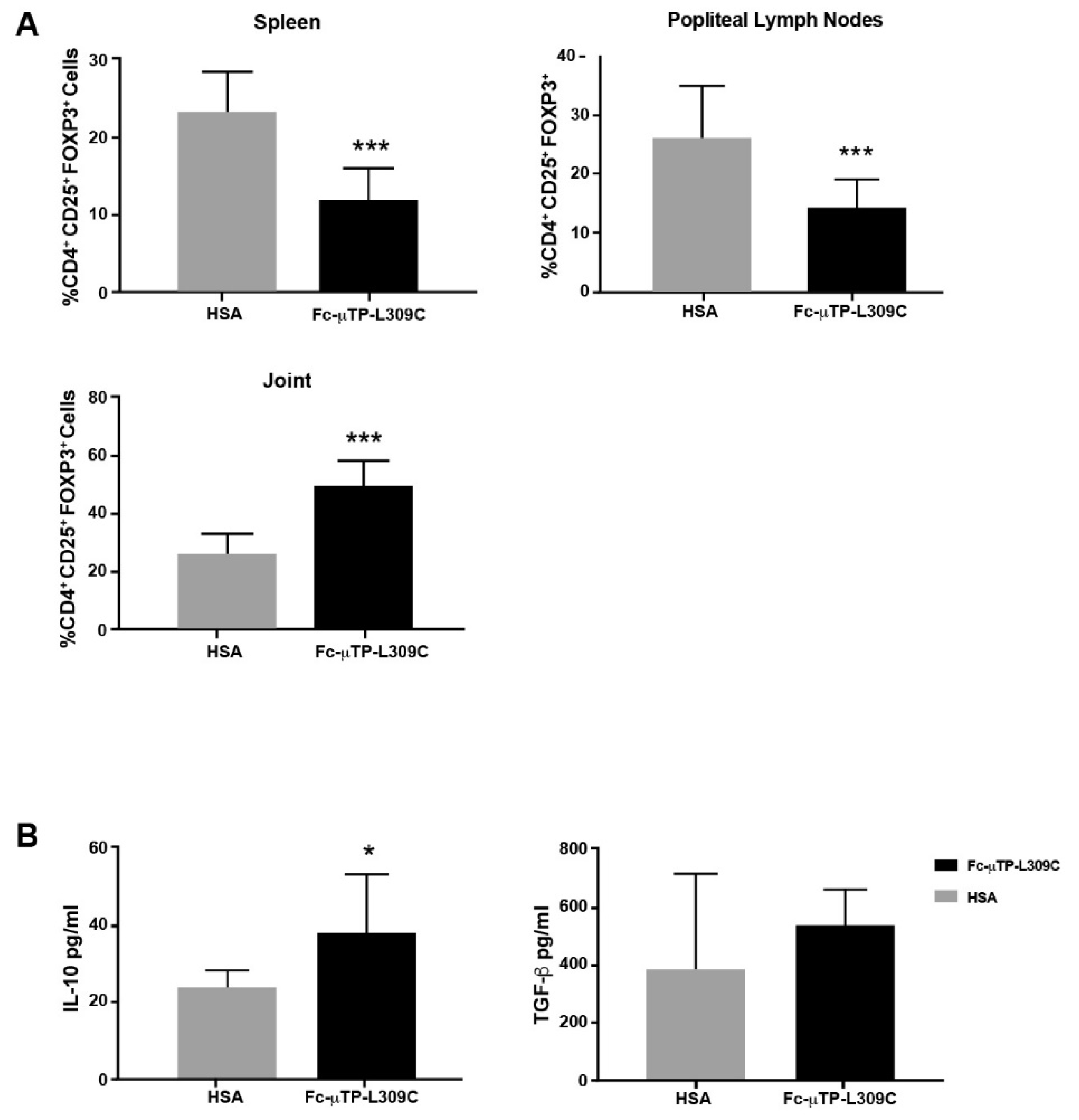
3.3. Tregs and IL-10 Are Increased in the Spleen, Lymph Nodes
4. Discussion
Funding
Author contributions
Institutional Review Board Statement
Data availability
Acknowledgements
References
- Anthony, R.M.; Nimmerjahn, F.; Ashline, D.J.; Reinhold, V.N.; Paulson, J.C.; Ravetch, JV. Recapitulation of IVIG anti-inflammatory activity with a recombinant IgG Fc. Science 2008, 320, 373–376. [Google Scholar] [CrossRef] [PubMed]
- Anthony, R.M.; Wermeling, F.; Karlsson, M.C.; Ravetch, J.V. Identification of a receptor required for the anti-inflammatory activity of IVIG. Proc. Nat. Acad. Sci. USA 2008, 105, 19571–19578. [Google Scholar] [CrossRef] [PubMed]
- Anthony, R.M.; Kobayashi, T.; Wermeling, F.; Ravetch, J.V. (2011). Intravenous gammaglobulin suppresses inflammation through a novel TH2 pathway. Nature 2011, 475, 110–113. [Google Scholar] [CrossRef] [PubMed]
- Campbell, I.K.; Miescher, S.; Branch, D.R.; Mott, P.J.; Lazarus, A.H.; Han, D.; Maraskovsky, E.; Zuercher, A.W.; Neschadim, A.; Leontyev, D.; McKenzie, B.S. Therapeutic effect of IVIG on inflammatory arthritis in mice is dependent on the Fc portion and independent of sialylation or basophils. J. Immunol. 2014, 192, 5031–5038. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, Y.; Nimmerjahn, F.; Ravetch, J. V. Anti-inflammatory activity of immunoglobulin G resulting from Fc sialylation. Science 2006, 313, 670–673. [Google Scholar] [CrossRef] [PubMed]
- Lewis, B.J.B.; Ville, J.; Blacquiere, M.; Cen., S.; Spirig, R.; Zuercher, A.W.; Fasermann, F.; Branch, D.R. Using the K/BxN mouse model of endogenous, chronic, rheumatoid arthritis for the evaluation of potential immunoglobulin-based therapeutic agents, including IVIg and Fc-μTP-L309C, a recombinant IgG1 Fc hexamer. BMC Immunol. 2019, 20, 44. [Google Scholar] [CrossRef] [PubMed]
- Almizraq, R.J.; Frias Boligan, K.; Lewis, B.J.B.; Cen, S.; Whetstone, H.; Spirig, R.; Kasermann, F.; Campbell, I.K.; von Gunten, S.; Branch, D.R. Modulation of neutrophil function by recombinant human IgG1 Fc hexamer in the endogenous K/BxN mouse model of rheumatoid arthritis. Pharmacol. 2023, 108, 176–187. [Google Scholar] [CrossRef] [PubMed]
- Campbell, I.K.; Leong, D.; Edwards, K.M.; Rayzman, V.; Ng, M.; Goldberg, G.L.; et al. Therapeutic targeting of the G-CSF receptor reduces neutrophil trafficking and joint inflammation in antibody-mediated inflammatory arthritis. J. Immunol. 2016, 197, 4392–4402. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.T.; Jacobs, J.; Mathis, D.; Benoist, C. Where FoxP3-edpendent regulatory T cells impinge on the development of inflammatory arthritis. Arthritis & Rheum. 2007, 56, 509–520. [Google Scholar]
- Fujio, K.; Okamura, T.; Sumitomo, S.; Yamamoto, K. Regulatory T cell-mediated control of autoantibody-induced inflammation. Front. Immunol. 2012, 3, 28. [Google Scholar] [CrossRef] [PubMed]
- Maloy, K.J.; Salaun, L.; Cahill, R.; Dougan, G.; Saunders, N.J.; Powrie, F. CD4+ CD25+ TR cells suppress innate immune pathology through cytokine-dependent mechanisms. J. Exp. Med. 2003, 197, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Moore, K.W.; de Waal Malefyt, R.; Coffman, R.L.; O’Garra, A. Interleukin-10 and the interleukin-10 receptor. Ann. Rev. Immunol. 2001, 19, 683–765. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



