Submitted:
30 May 2025
Posted:
30 May 2025
You are already at the latest version
Abstract
Keywords:
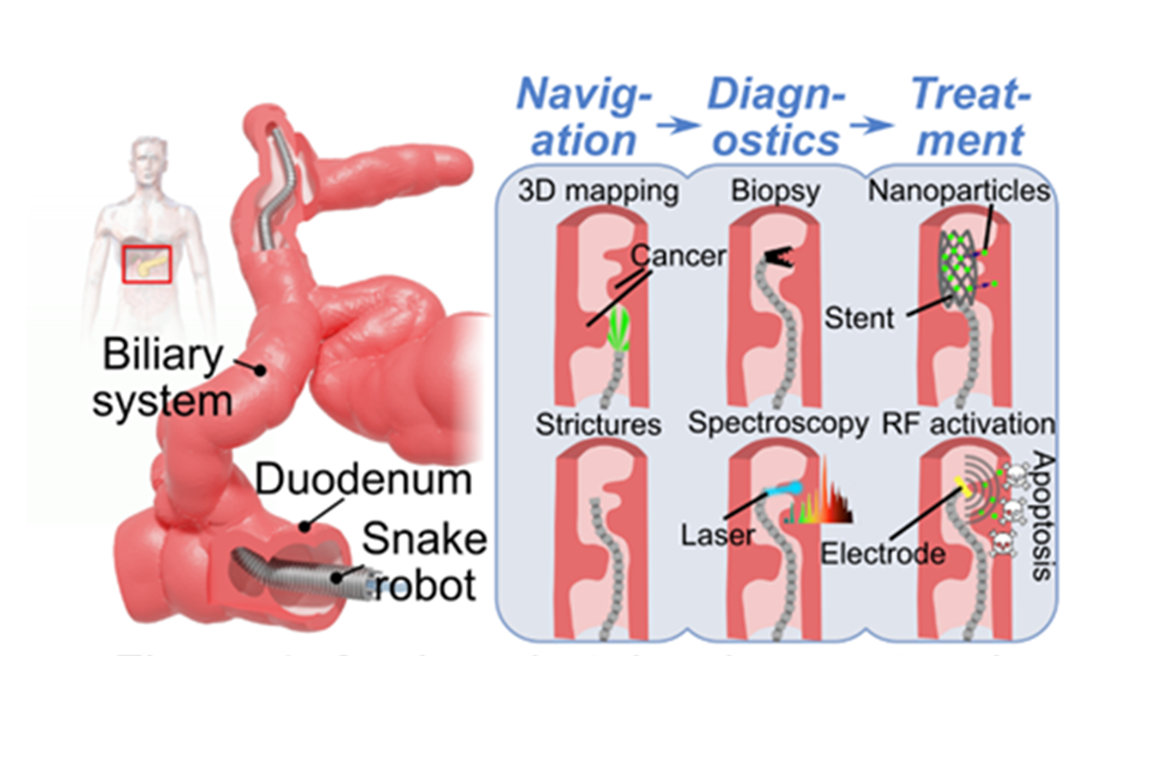
1. Introduction
2. Diagnostic Techniques and Pathway
3. Surgical Treatment Options
- Extrahepatic tumours may be treated through the Whipple procedure which removes the pancreatic head, parts of the duodenum and bile duct, and the gallbladder.
- Intrahepatic tumours may be treated by surgical liver resection.
- Hilar tumours (at the Y-junction/bifurcation where the left and right hepatic ducts merge) require complex procedures that involve major hepatic resection.
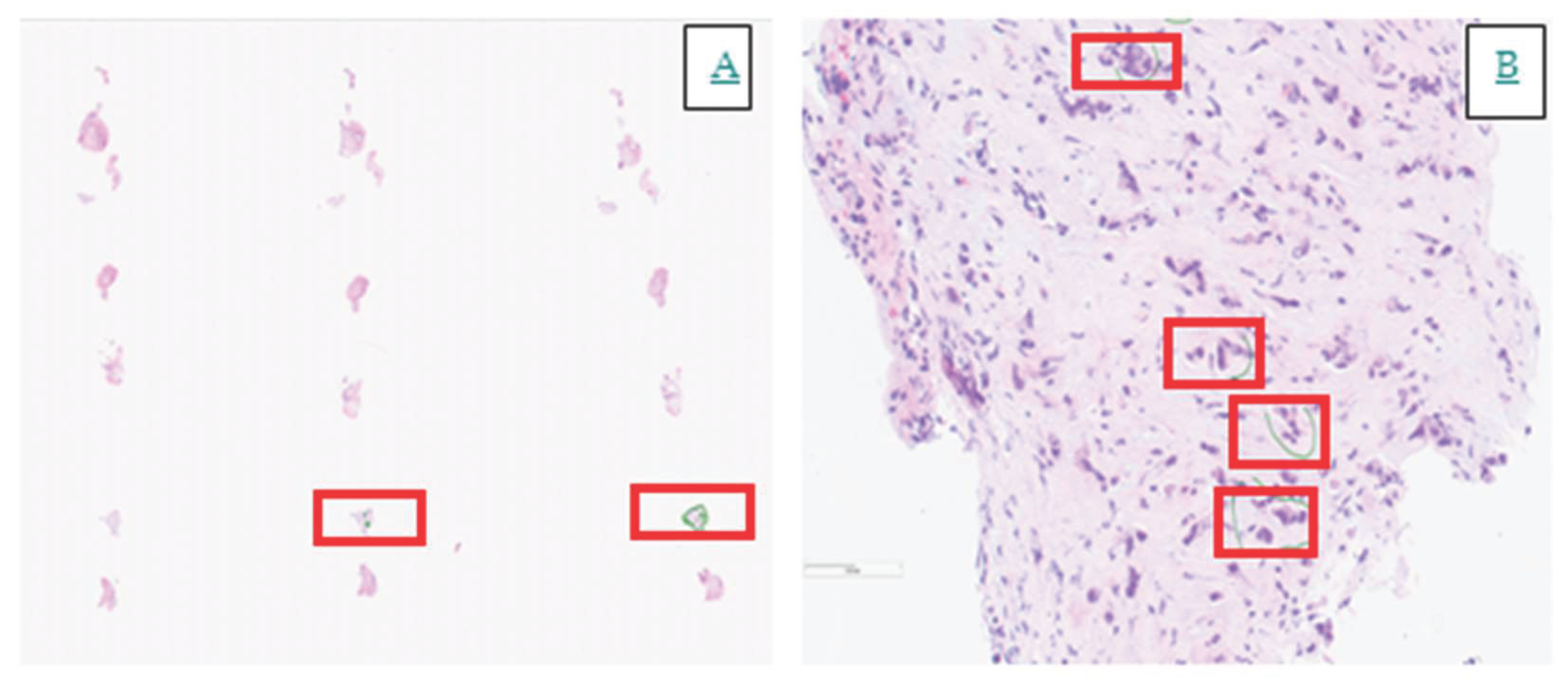
4. Histopathology
5. Oncology
6. Next Generation Technologies
7. Improving Stenting
8. Ethically Obtaining Samples for Molecular Mapping and Research
9. Discussion: Engineering Life for Advancing Care
10. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Saluja SS, Sharma R, Pal S, Sahni P, Chattopadhyay TK. Differentiation between benign and malignant hilar obstructions using laboratory and radiological investigations: a prospective study. HPB (Oxford). 2007, 9, 373–382. [Google Scholar] [CrossRef] [PubMed]
- Rosch T, Meining A, Fruhmorgen S, Zillinger C, Schusdziarra V, Hellerhoff K, et al. A prospective comparison of the diagnostic accuracy of ERCP, MRCP, CT, and EUS in biliary strictures. Gastrointest Endosc. 2002, 55, 870–876. [Google Scholar] [CrossRef] [PubMed]
- Vardar BU, Dupuis CS, Goldstein AJ, Vardar Z, Kim YH. Ultrasonographic evaluation of patients with abnormal liver function tests in the emergency department. Ultrasonography. 2022, 41, 243–262. [Google Scholar] [CrossRef]
- Hori Y, Chari ST, Tsuji Y, Takahashi N, Inoue D, Hart PA, et al. Diagnosing Biliary Strictures: Distinguishing IgG4-Related Sclerosing Cholangitis From Cholangiocarcinoma and Primary Sclerosing Cholangitis. Mayo Clin Proc Innov Qual Outcomes. 2021, 5, 535–541. [Google Scholar] [CrossRef]
- Stassen PMC, Goodchild G, de Jonge PJF, Erler NS, Anderloni A, Cennamo V, et al. Diagnostic accuracy and interobserver agreement of digital single-operator cholangioscopy for indeterminate biliary strictures. Gastrointest Endosc. 2021, 94, 1059–1068. [Google Scholar] [CrossRef] [PubMed]
- Blechacz B, Komuta M, Roskams T, Gores GJ. Clinical diagnosis and staging of cholangiocarcinoma. Nat Rev Gastroenterol Hepatol. 2011, 8, 512–522. [Google Scholar] [CrossRef]
- Valle JW, Borbath I, Khan SA, Huguet F, Gruenberger T, Arnold D, et al. Biliary cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2016, 27 (suppl 5), v28–v37. [Google Scholar] [CrossRef]
- Rushbrook SM, Kendall TJ, Zen Y, Albazaz R, Manoharan P, Pereira SP, et al. British Society of Gastroenterology guidelines for the diagnosis and management of cholangiocarcinoma. Gut. 2023, 73, 16–46. [Google Scholar]
- Skorepa P, Ford KL, Alsuwaylihi A, O'Connor D, Prado CM, Gomez D, et al. The impact of prehabilitation on outcomes in frail and high-risk patients undergoing major abdominal surgery: A systematic review and meta-analysis. Clin Nutr. 2024, 43, 629–648. [Google Scholar] [CrossRef]
- McClements J, Valle JW, Blackburn L, Brooks A, Prachalias A, Dasari BVM, et al. Variation in treatment of intrahepatic cholangiocarcinoma: a nationwide multicentre study. Br J Surg. 2023, 110, 1673–1676. [Google Scholar] [CrossRef]
- Hewitt DB, Brown ZJ, Pawlik TM. Current Perspectives on the Surgical Management of Perihilar Cholangiocarcinoma. Cancers 2022, 14. [Google Scholar] [CrossRef] [PubMed]
- Franken LC, Schreuder AM, Roos E, van Dieren S, Busch OR, Besselink MG, et al. Morbidity and mortality after major liver resection in patients with perihilar cholangiocarcinoma: A systematic review and meta-analysis. Surgery. 2019, 165, 918–928. [Google Scholar] [CrossRef] [PubMed]
- Mueller M, Breuer E, Mizuno T, Bartsch F, Ratti F, Benzing C, et al. Perihilar Cholangiocarcinoma - Novel Benchmark Values for Surgical and Oncological Outcomes From 24 Expert Centers. Ann Surg. 2021, 274, 780–788. [Google Scholar] [CrossRef] [PubMed]
- Zhang XF, Beal EW, Chakedis J, Chen Q, Lv Y, Ethun CG, et al. Defining Early Recurrence of Hilar Cholangiocarcinoma After Curative-intent Surgery: A Multi-institutional Study from the US Extrahepatic Biliary Malignancy Consortium. World J Surg. 2018, 42, 2919–2929. [Google Scholar] [CrossRef]
- Groot Koerkamp B, Wiggers JK, Allen PJ, Besselink MG, Blumgart LH, Busch OR, et al. Recurrence Rate and Pattern of Perihilar Cholangiocarcinoma after Curative Intent Resection. J Am Coll Surg. 2015, 221, 1041–1049. [Google Scholar] [CrossRef]
- Labib PL, Russell TB, Denson JL, Puckett MA, Ausania F, Pando E, et al. Patterns, timing and predictors of recurrence following pancreaticoduodenectomy for distal cholangiocarcinoma: An international multicentre retrospective cohort study. Eur J Surg Oncol. 2024, 50, 108353. [Google Scholar] [CrossRef]
- Roos E, Soer EC, Klompmaker S, Meijer LL, Besselink MG, Giovannetti E, et al. Crossing borders: A systematic review with quantitative analysis of genetic mutations of carcinomas of the biliary tract. Crit Rev Oncol Hematol. 2019, 140, 8–16. [Google Scholar] [CrossRef]
- Borger DR, Tanabe KK, Fan KC, Lopez HU, Fantin VR, Straley KS, et al. Frequent mutation of isocitrate dehydrogenase (IDH)1 and IDH2 in cholangiocarcinoma identified through broad-based tumor genotyping. Oncologist. 2012, 17, 72–79. [Google Scholar] [CrossRef]
- Arai Y, Totoki Y, Hosoda F, Shirota T, Hama N, Nakamura H, et al. Fibroblast growth factor receptor 2 tyrosine kinase fusions define a unique molecular subtype of cholangiocarcinoma. Hepatology. 2014, 59, 1427–1434. [Google Scholar] [CrossRef]
- Graham RP, Barr Fritcher EG, Pestova E, Schulz J, Sitailo LA, Vasmatzis G, et al. Fibroblast growth factor receptor 2 translocations in intrahepatic cholangiocarcinoma. Hum Pathol. 2014, 45, 1630–1638. [Google Scholar] [CrossRef]
- Abou-Alfa GK, Macarulla T, Javle MM, Kelley RK, Lubner SJ, Adeva J, et al. Ivosidenib in IDH1-mutant, chemotherapy-refractory cholangiocarcinoma (ClarIDHy): a multicentre, randomised, double-blind, placebo-controlled, phase 3 study. Lancet Oncol. 2020, 21, 796–807. [Google Scholar] [CrossRef] [PubMed]
- Lowery MA, Burris HA, 3rd, Janku F, Shroff RT, Cleary JM, Azad NS, et al. Safety and activity of ivosidenib in patients with IDH1-mutant advanced cholangiocarcinoma: a phase 1 study. Lancet Gastroenterol Hepatol. 2019, 4, 711–720. [Google Scholar] [CrossRef] [PubMed]
- Goyal L, Meric-Bernstam F, Hollebecque A, Valle JW, Morizane C, Karasic TB, et al. Futibatinib for FGFR2-Rearranged Intrahepatic Cholangiocarcinoma. N Engl J Med. 2023, 388, 228–239. [Google Scholar] [CrossRef] [PubMed]
- Maio M, Ascierto PA, Manzyuk L, Motola-Kuba D, Penel N, Cassier PA, et al. Pembrolizumab in microsatellite instability high or mismatch repair deficient cancers: updated analysis from the phase II KEYNOTE-158 study. Ann Oncol. 2022, 33, 929–938. [Google Scholar] [CrossRef]
- Primrose JN, Fox RP, Palmer DH, Malik HZ, Prasad R, Mirza D, et al. Capecitabine compared with observation in resected biliary tract cancer (BILCAP): a randomised, controlled, multicentre, phase 3 study. Lancet Oncol. 2019, 20, 663–673. [Google Scholar] [CrossRef]
- Kelley RK, Ueno M, Yoo C, Finn RS, Furuse J, Ren Z, et al. Pembrolizumab in combination with gemcitabine and cisplatin compared with gemcitabine and cisplatin alone for patients with advanced biliary tract cancer (KEYNOTE-966): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2023, 401, 1853–1865. [Google Scholar] [CrossRef]
- Abou-Alfa GK, Sahai V, Hollebecque A, Vaccaro G, Melisi D, Al-Rajabi R, et al. Pemigatinib for previously treated, locally advanced or metastatic cholangiocarcinoma: a multicentre, open-label, phase 2 study. Lancet Oncol. 2020, 21, 671–684. [Google Scholar] [CrossRef]
- Lamarca A, Palmer DH, Wasan HS, Ross PJ, Ma YT, Arora A, et al. Second-line FOLFOX chemotherapy versus active symptom control for advanced biliary tract cancer (ABC-06): a phase 3, open-label, randomised, controlled trial. Lancet Oncol. 2021, 22, 690–701. [Google Scholar] [CrossRef]
- Marabelle A, Le DT, Ascierto PA, Di Giacomo AM, De Jesus-Acosta A, Delord JP, et al. Efficacy of Pembrolizumab in Patients With Noncolorectal High Microsatellite Instability/Mismatch Repair-Deficient Cancer: Results From the Phase II KEYNOTE-158 Study. J Clin Oncol. 2020, 38, 1–10. [Google Scholar] [CrossRef]
- Subbiah V, Kreitman RJ, Wainberg ZA, Gazzah A, Lassen U, Stein A, et al. Dabrafenib plus trametinib in BRAFV600E-mutated rare cancers: the phase 2 ROAR trial. Nat Med. 2023, 29, 1103–1112. [Google Scholar] [CrossRef]
- Wong CY, Seshadri P, Parks GT. Automatic Borescope Damage Assessments for Gas Turbine Blades via Deep Learning. AIAA Scitech 2021 Forum. AIAA SciTech Forum: American Institute of Aeronautics and Astronautics; 2021.
- Troncoso DA, Robles-Linares JA, Russo M, Elbanna MA, Wild S, Dong X, et al. A Continuum Robot for Remote Applications: From Industrial to Medical Surgery With Slender Continuum Robots. IEEE Robotics & Automation Magazine. 2023, 30, 94–105. [Google Scholar]
- Stiburek M, Ondrackova P, Tuckova T, Turtaev S, Siler M, Pikalek T, et al. 110 mum thin endo-microscope for deep-brain in vivo observations of neuronal connectivity, activity and blood flow dynamics. Nat Commun. 2023, 14, 1897. [Google Scholar] [CrossRef] [PubMed]
- Wen Z, Dong Z, Deng Q, Pang C, Kaminski CF, Xu X, et al. Single multimode fibre for in vivo light-field-encoded endoscopic imaging. Nature Photonics. 2023, 17, 679–687. [Google Scholar] [CrossRef]
- George SDG, James J, Maria PA, Travis S, Calum W, Catherine RMF, et al. Quantitative phase and polarization imaging through an optical fiber applied to detection of early esophageal tumorigenesis. Journal of Biomedical Optics. 2019, 24, 126004. [Google Scholar]
- Jane C, George SDG. Ultra-miniature dual-wavelength spatial frequency domain imaging for micro-endoscopy. Journal of Biomedical Optics. 2024, 29, 026002. [Google Scholar]
- Sweer JA, Chen MT, Salimian KJ, Battafarano RJ, Durr NJ. Wide-field optical property mapping and structured light imaging of the esophagus with spatial frequency domain imaging. Journal of Biophotonics. 2019, 12, e201900005. [Google Scholar]
- Angelo JP, van de Giessen M, Gioux S. Real-time endoscopic optical properties imaging. Biomed Opt Express. 2017, 8, 5113–5126. [Google Scholar] [CrossRef]
- Awe AM, Rendell VR, Lubner MG, Winslow ER. Texture Analysis: An Emerging Clinical Tool for Pancreatic Lesions. Pancreas. 2020, 49. [Google Scholar]
- Shipp DW, Sinjab F, Notingher I. Raman spectroscopy: techniques and applications in the life sciences. Adv Opt Photon. 2017, 9, 315–428. [Google Scholar] [CrossRef]
- Kong K, Rowlands CJ, Varma S, Perkins W, Leach IH, Koloydenko AA, et al. Diagnosis of tumors during tissue-conserving surgery with integrated autofluorescence and Raman scattering microscopy. Proceedings of the National Academy of Sciences. 2013, 110, 15189–15194. [Google Scholar] [CrossRef]
- Boitor R, de Wolf C, Weesie F, Shipp DA-O, Varma S, Veitch D, et al. Clinical integration of fast Raman spectroscopy for Mohs micrographic surgery of basal cell carcinoma. Biomed Opt Express 2021, 12, 2015–2026. [Google Scholar] [CrossRef] [PubMed]
- Boitor R, Varma S, Sharma A, Elsheikh S, Kulkarni K, Eldib K, et al. Ex vivo assessment of basal cell carcinoma surgical margins in Mohs surgery by autofluorescence-Raman spectroscopy: A pilot study. JEADV Clinical Practice. 2024, 3, 498–507. [Google Scholar] [CrossRef]
- Corden C, Boitor R, Dusanjh PK, Harwood A, Mukherjee A, Gomez D, et al. Autofluorescence-Raman Spectroscopy for Ex Vivo Mapping Colorectal Liver Metastases and Liver Tissue. (1095-8673 (Electronic)).
- Barkur S, Boitor RA, Mihai R, Gopal NSR, Leeney S, Koloydenko AA, et al. Intraoperative spectroscopic evaluation of sentinel lymph nodes in breast cancer surgery. Breast Cancer Research and Treatment. 2024, 207, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Mangiavillano B, Pagano N, Baron TH, Luigiano C. Outcome of stenting in biliary and pancreatic benign and malignant diseases: A comprehensive review. World J Gastroenterol. 2015, 21, 9038–9054. [Google Scholar] [CrossRef]
- Song G, Zhao HQ, Liu Q, Fan Z. A review on biodegradable biliary stents: materials and future trends. Bioact Mater. 2022, 17, 488–495. [Google Scholar]
- Lam R, Muniraj T. Fully covered metal biliary stents: A review of the literature. World J Gastroenterol. 2021, 27, 6357–6373. [Google Scholar] [CrossRef]
- Yang K, Sun W, Cui L, Zou Y, Wen C, Zeng R. Advances in functional coatings on biliary stents. Regen Biomater. 2024, 11, rbae001. [Google Scholar]
- Suk KT, Kim JW, Kim HS, Baik SK, Oh SJ, Lee SJ, et al. Human application of a metallic stent covered with a paclitaxel-incorporated membrane for malignant biliary obstruction: multicenter pilot study. Gastrointest Endosc. 2007, 66, 798–803. [Google Scholar] [CrossRef]
- Chung MJ, Kim H, Kim KS, Park S, Chung JB, Park SW. Safety evaluation of self-expanding metallic biliary stents eluting gemcitabine in a porcine model. J Gastroenterol Hepatol. 2012, 27, 261–267. [Google Scholar] [CrossRef]
- Kim DH, Jeong YI, Chung CW, Kim CH, Kwak TW, Lee HM, et al. Preclinical evaluation of sorafenib-eluting stent for suppression of human cholangiocarcinoma cells. Int J Nanomedicine. 2013, 8, 1697–1711. [Google Scholar]
- Kwak TW, Lee HL, Song YH, Kim C, Kim J, Seo SJ, et al. Vorinostat-eluting poly(DL-lactide-co-glycolide) nanofiber-coated stent for inhibition of cholangiocarcinoma cells. Int J Nanomedicine. 2017, 12, 7669–7680. [Google Scholar] [CrossRef] [PubMed]
- Wang HW, Li XJ, Li SJ, Lu JR, He DF. Biliary stent combined with iodine-125 seed strand implantation in malignant obstructive jaundice. World J Clin Cases. 2021, 9, 801–811. [Google Scholar] [CrossRef] [PubMed]
- Yang F, Ren Z, Chai Q, Cui G, Jiang L, Chen H, et al. A novel biliary stent coated with silver nanoparticles prolongs the unobstructed period and survival via anti-bacterial activity. Sci Rep. 2016, 6, 21714. [Google Scholar] [CrossRef] [PubMed]
- Yamabe A, Irisawa A, Kunogi Y, Kashima K, Nagashima K, Minaguchi T, et al. Development of biliary stent applying the antibacterial activity of silver: A literature review. Biomed Mater Eng. 2021, 32, 63–71. [Google Scholar]
- Park JS, Yim KH, Jeong S, Lee DH, Kim DG. A Novel High-Visibility Radiopaque Tantalum Marker for Biliary Self-Expandable Metal Stents. Gut Liver. 2019, 13, 366–372. [Google Scholar] [CrossRef]
- Kobayashi, M. Development of a biliary multi-hole self-expandable metallic stent for bile tract diseases: A case report. World J Clin Cases. 2019, 7, 1323–1328. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



