Submitted:
22 May 2025
Posted:
22 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials, Electrodes, Li-Ion Battery Cell Elaboration and Fabrications
2.2. Methods
2.2.1. X-ray Diffraction (XRD)
2.2.2. Specific Surface Area Determination by the Brunauer-Emmett-Teller (BET) Method
2.2.3. Scanning Electron Microscopy and Energy Dispersive X-ray Spectroscopy
2.2.4. Conductivity Measurement of the Activated Carbons
2.2.5. Electrochemical Characterisation

3. Results and Discussion
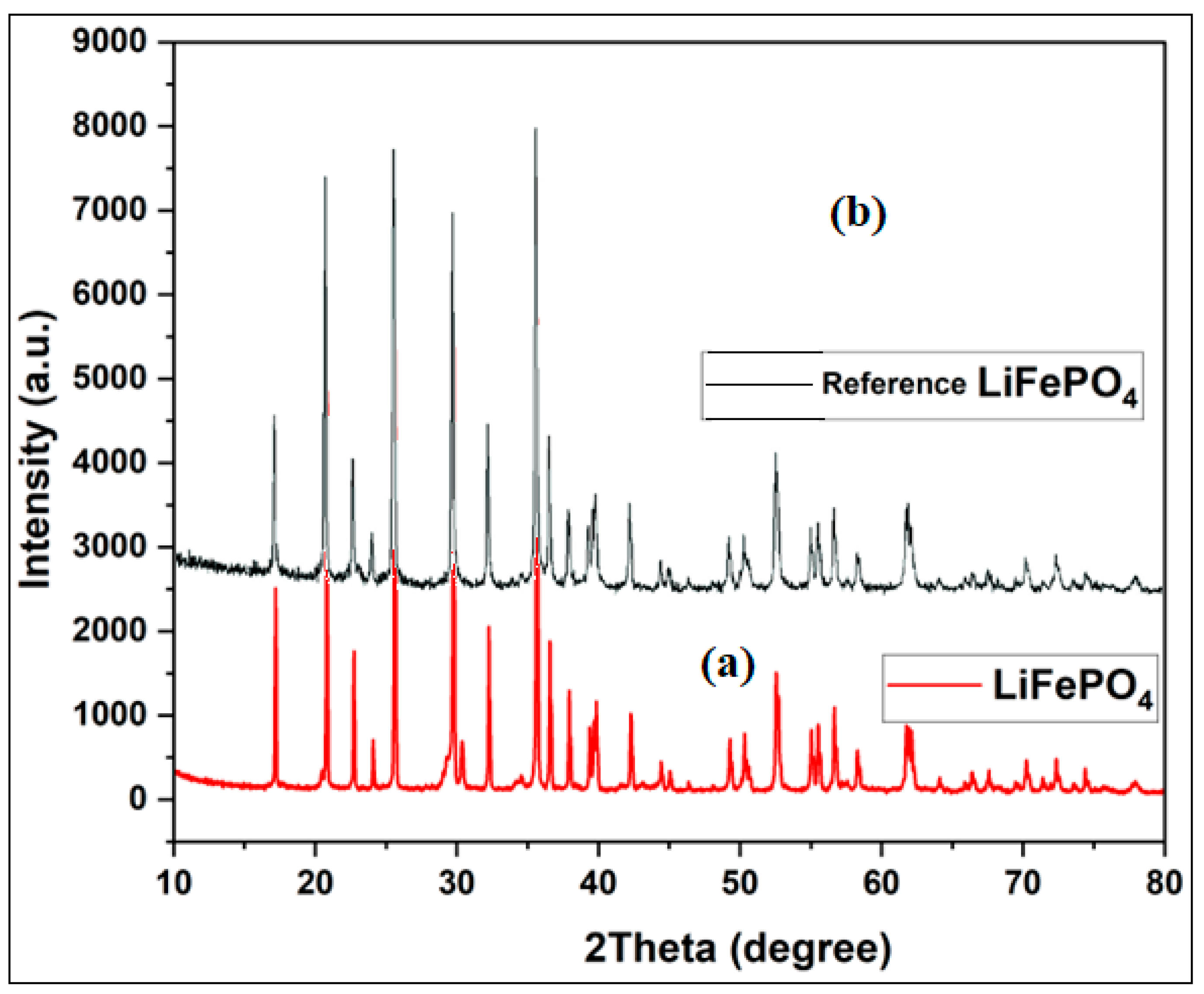
3.1. Morphological and Structural Characterization of Pure LiFePO4
3.2. Chemical and Physical Characterisation of Activated Carbons from Millet Cob and Water Hyacinth
3.2.1. Pore Size, Pore Distribution and BET Surface Determination of Activated Carbons from Millet Cob and Water Hyacinth
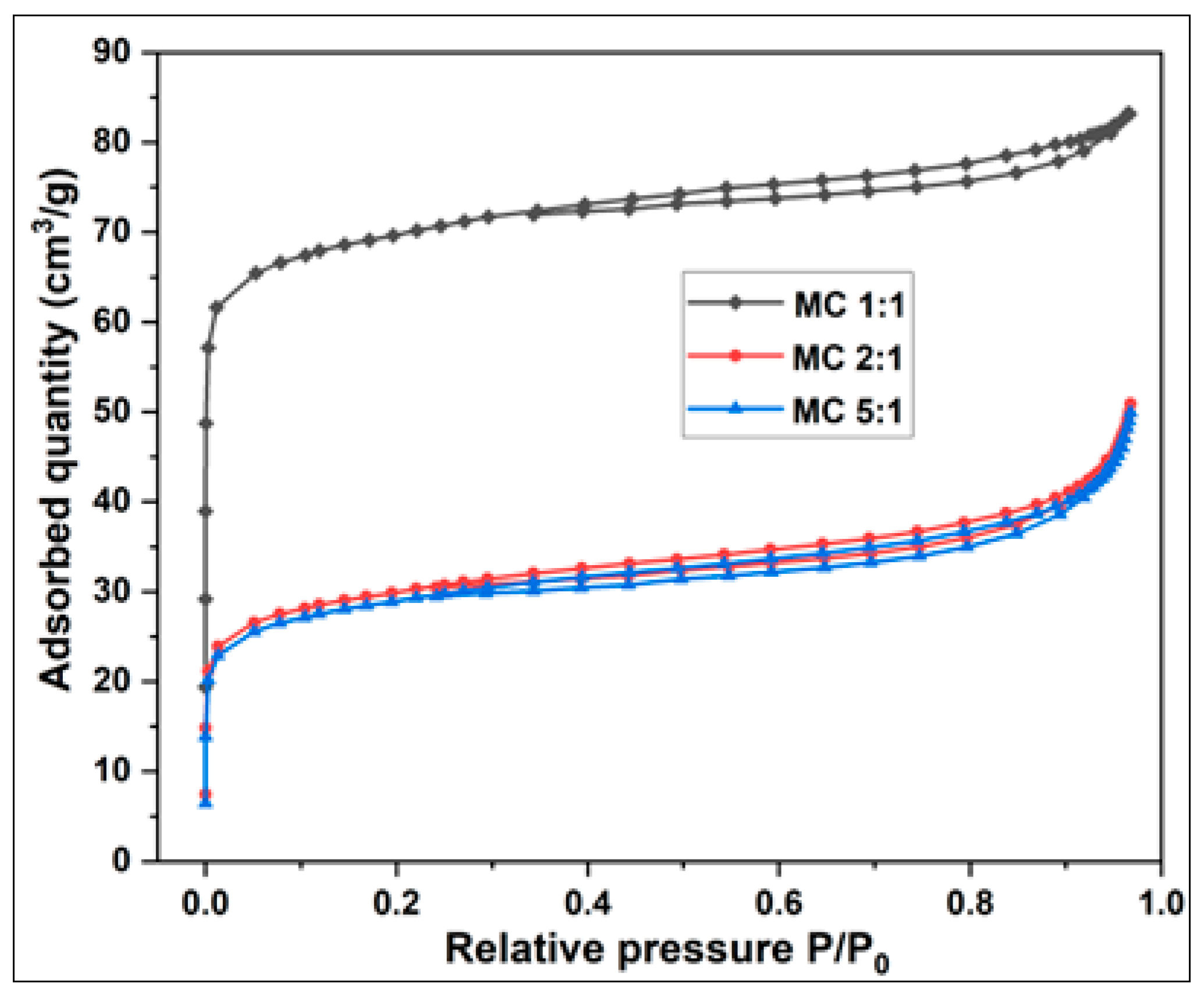
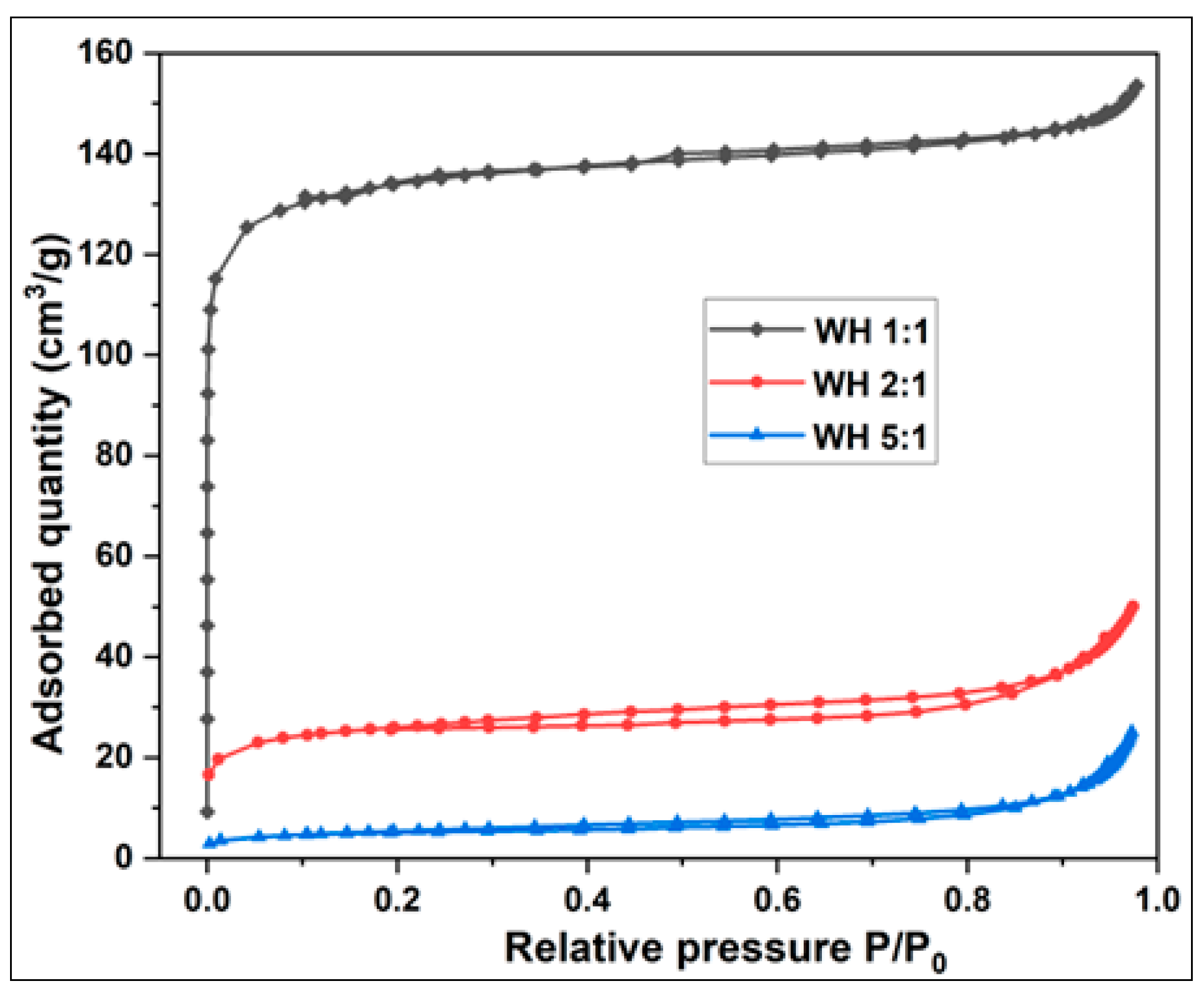
3.2.1.1. Adsorption and Desorption Isotherms of Activated Carbon from Millet Cob and Water Hyacinth
3.2.1.2. Pore Size Distribution
3.2.1.2.1. Water Hyacinth (WH) Sample
3.2.1.2.2. Millet Cob (MC) sample
3.2.1.3. BET Surface Area of Activated Carbons
3.2.2. SEM Characterisation of Activated Carbons from Millet Cob and Water Hyacinth
3.2.3. Elemental Composition of Porous Carbons from MC and WH Using EDS
3.2.4. X-ray Diffraction Studies of Porous Carbons from Millet Cob and Water Hyacinth
3.2.5. Electric Conductivity of Activated Carbons from Millet Cob and Water Hyacinth
3.3. Electrochemical Characterisation
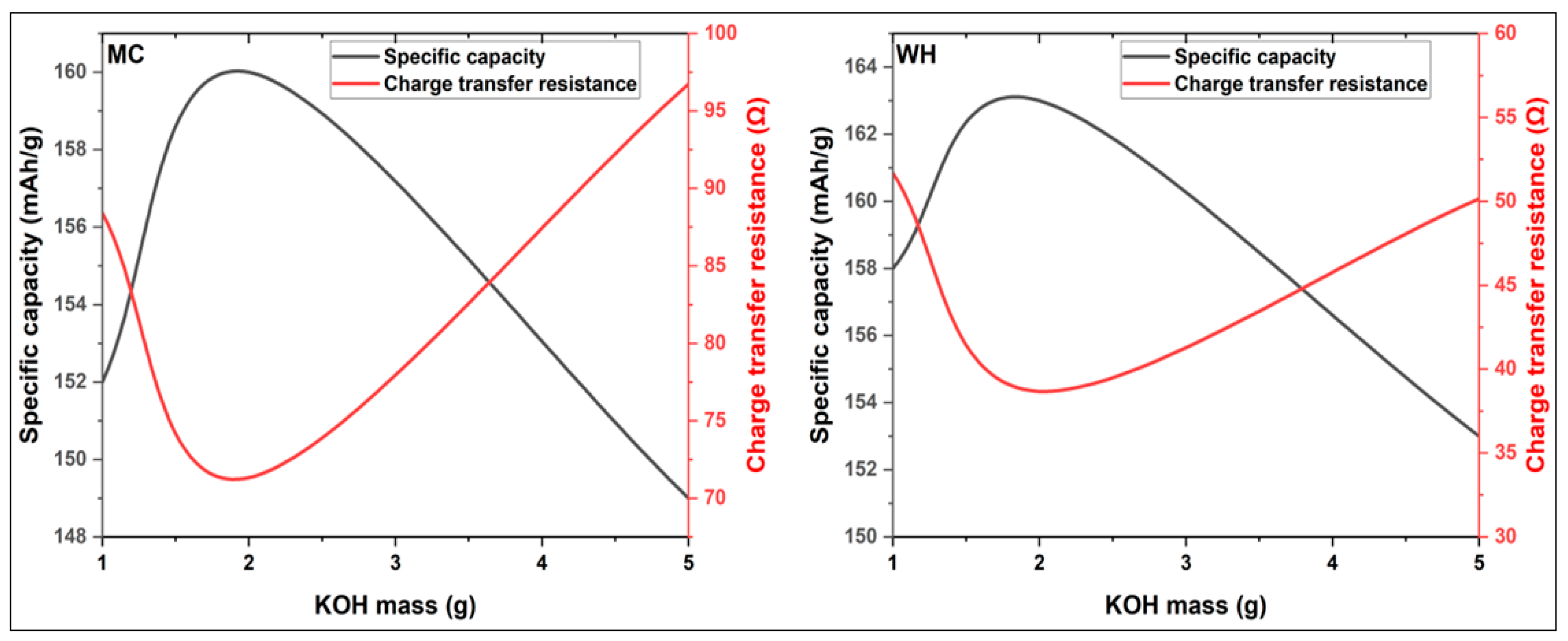
3.3.1. Specific Capacity of Synthesized Activated Carbon
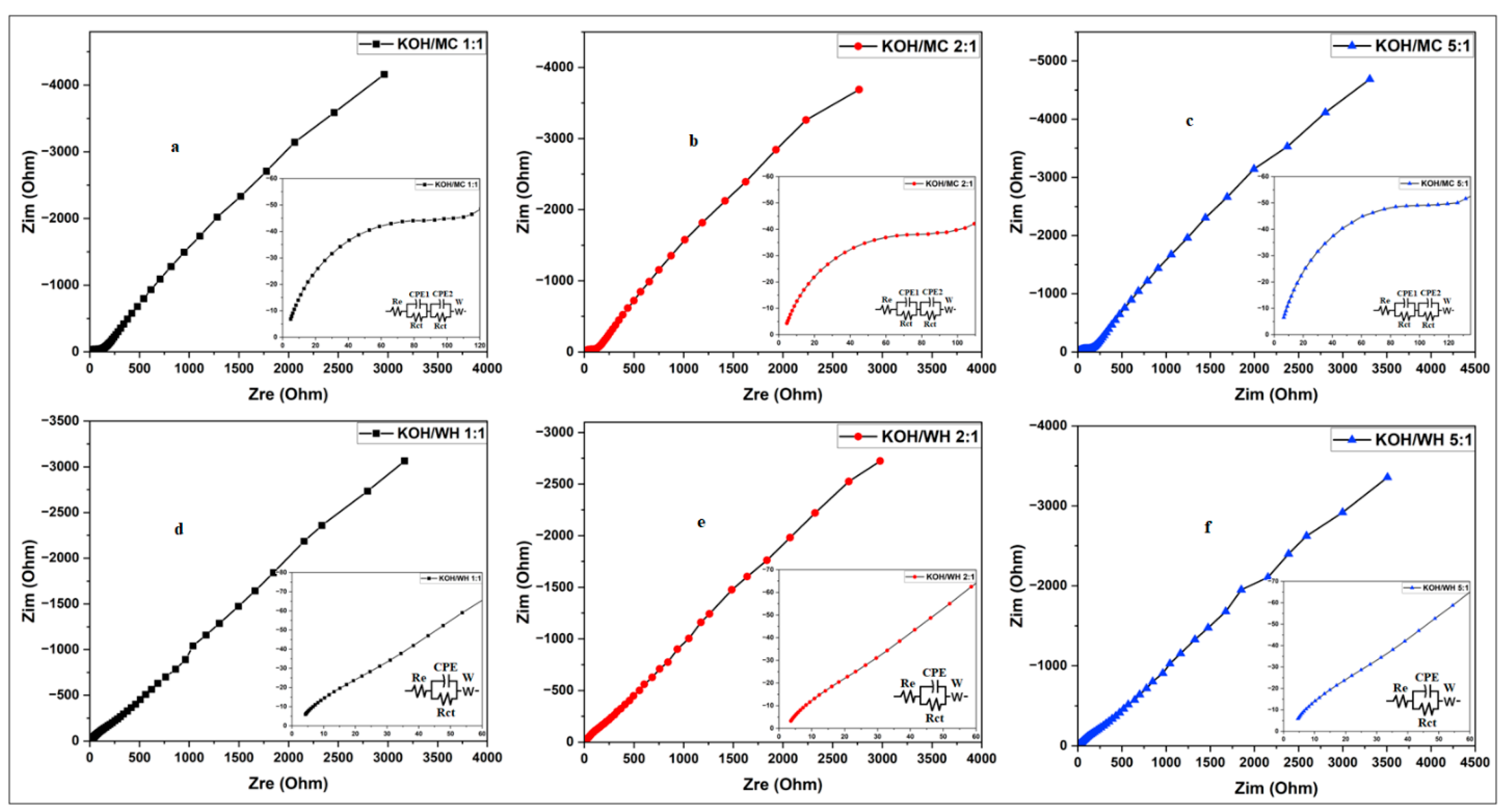
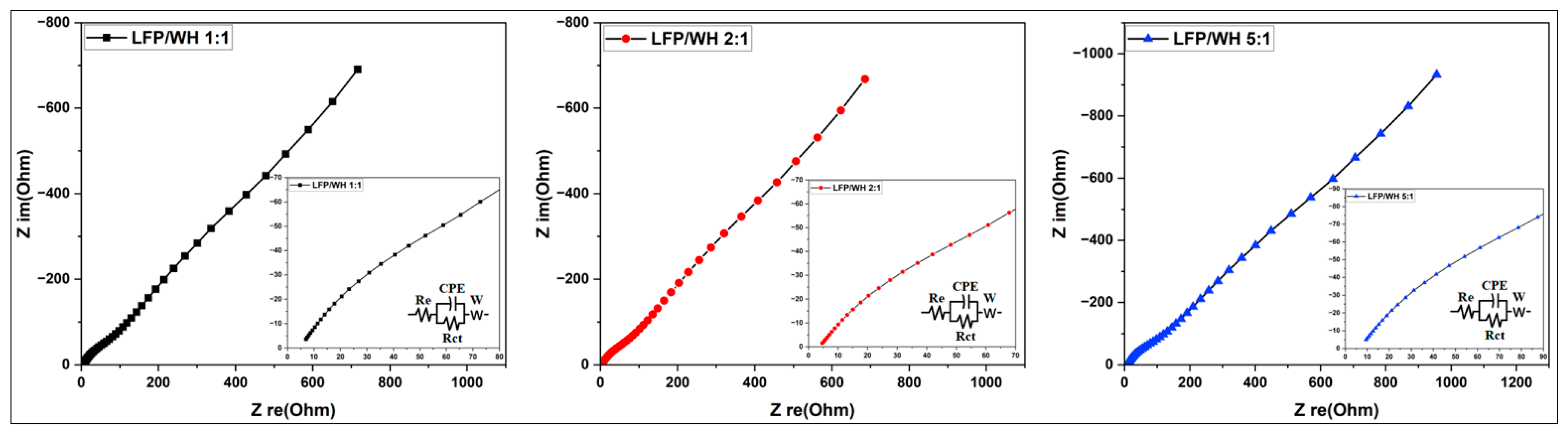
3.3.2. Electrochemical Impedance Spectroscopy Study of Synthesized Activated Carbon
3.3.3. Analysis of the Electrochemical Performance of the LiFePO4/C Cathode
3.3.3.1. Study of Coulombic Efficiency
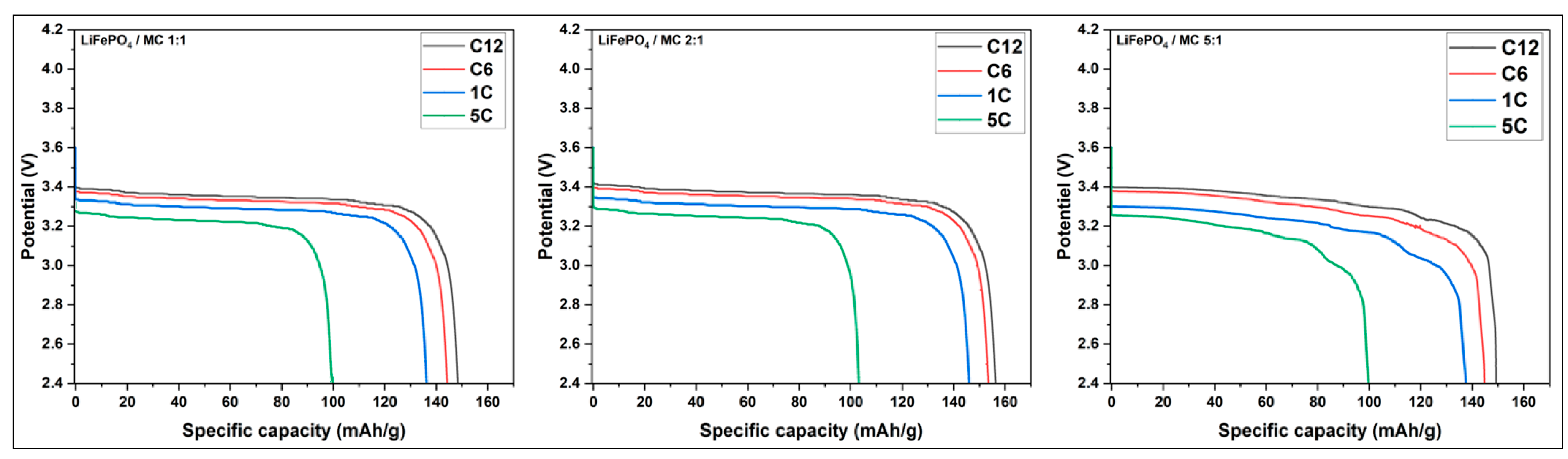
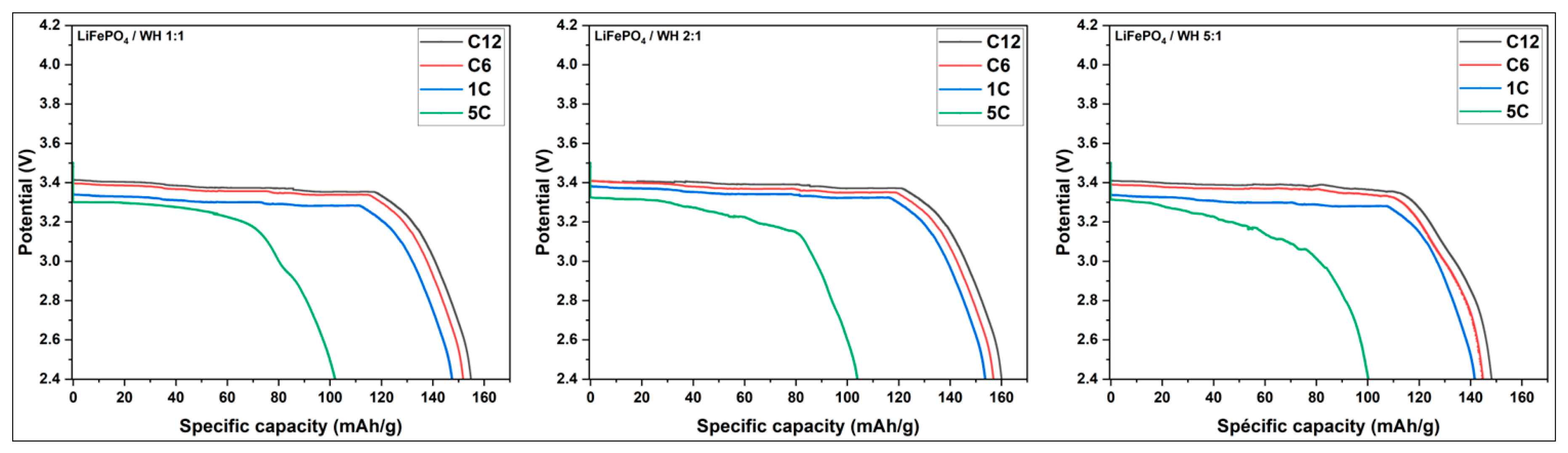
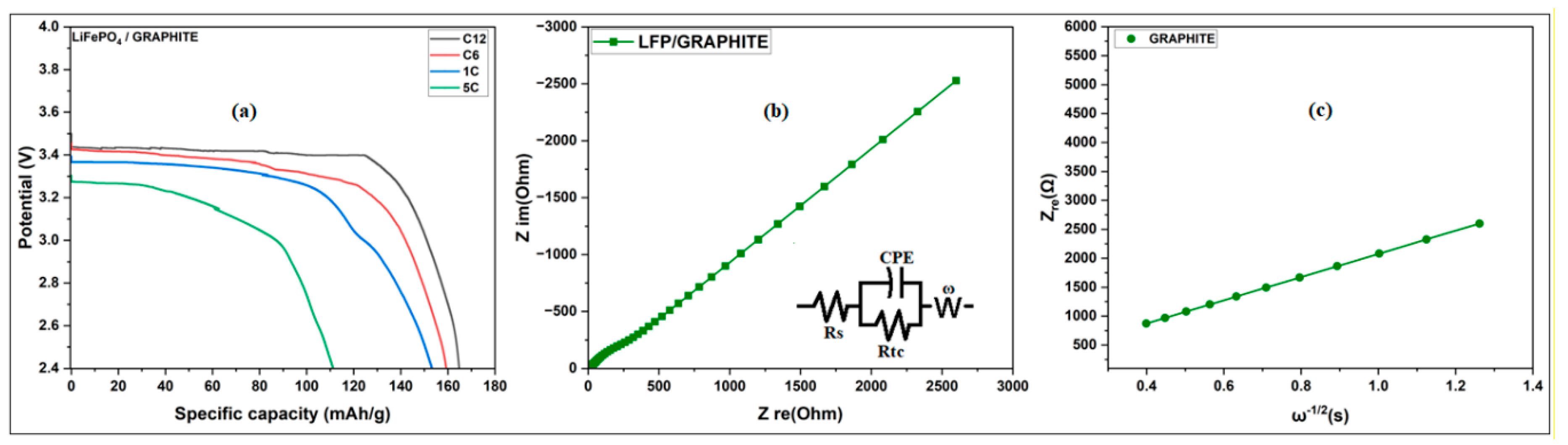
3.3.3.2. Study of the Discharge of LiFePO4/C at Different Current Rates
| Current rate | SC (mAh/g) WE 1:1 | SC (mAh/g) WE 2:1 | SC (mAh/g) WE 5:1 | SC(mAh/g) |
| C12 | 158 | 163 | 153 | 167 |
| C6 | 155 | 160 | 149 | 163 |
| 1C | 151 | 157 | 145 | 161 |
| 5C | 108 | 110 | 106 | 120 |
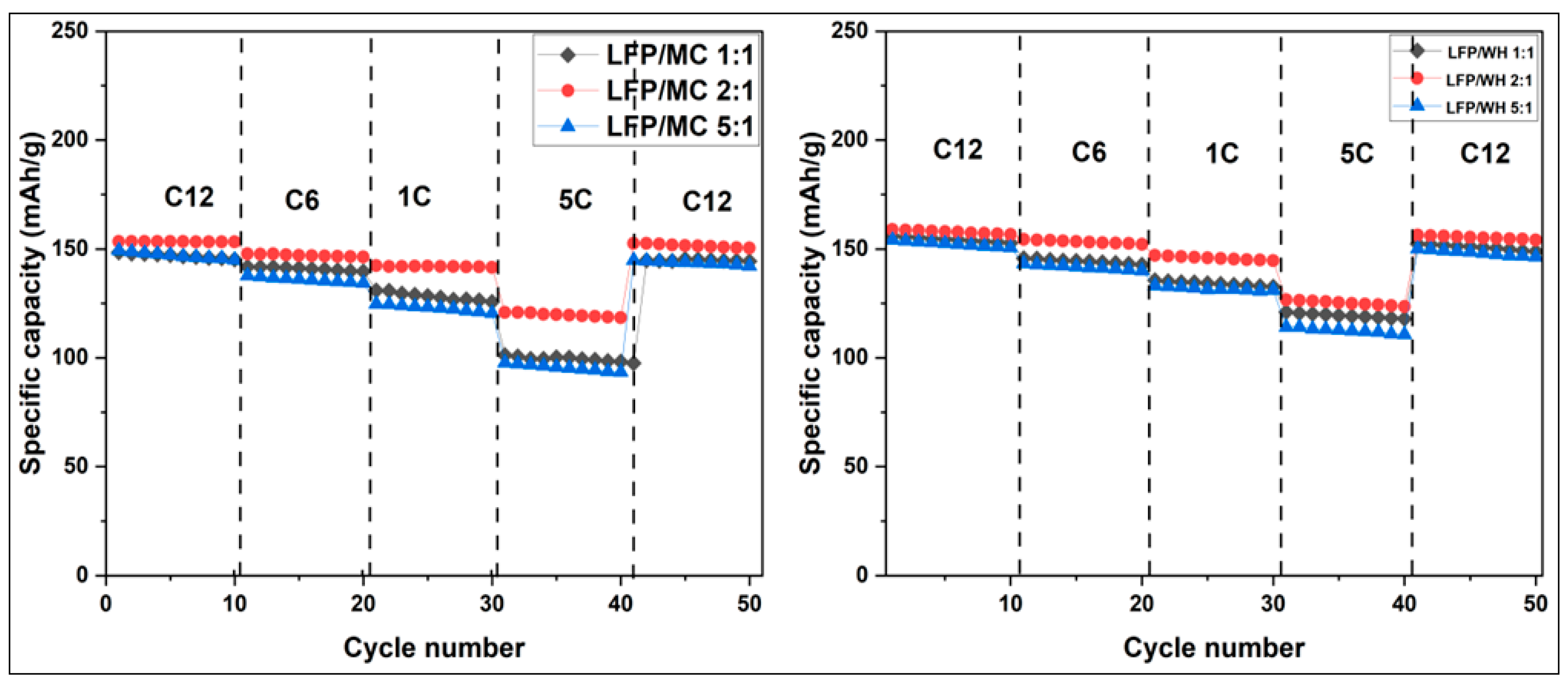
3.3.3.3. Cycling Performance of LiFePO4/C Sample
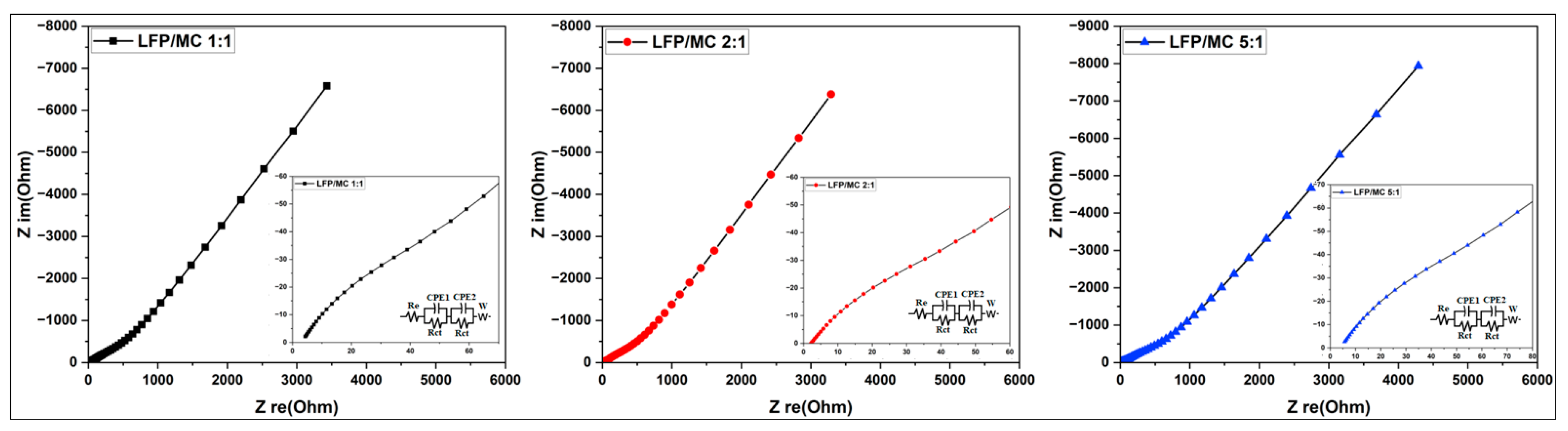
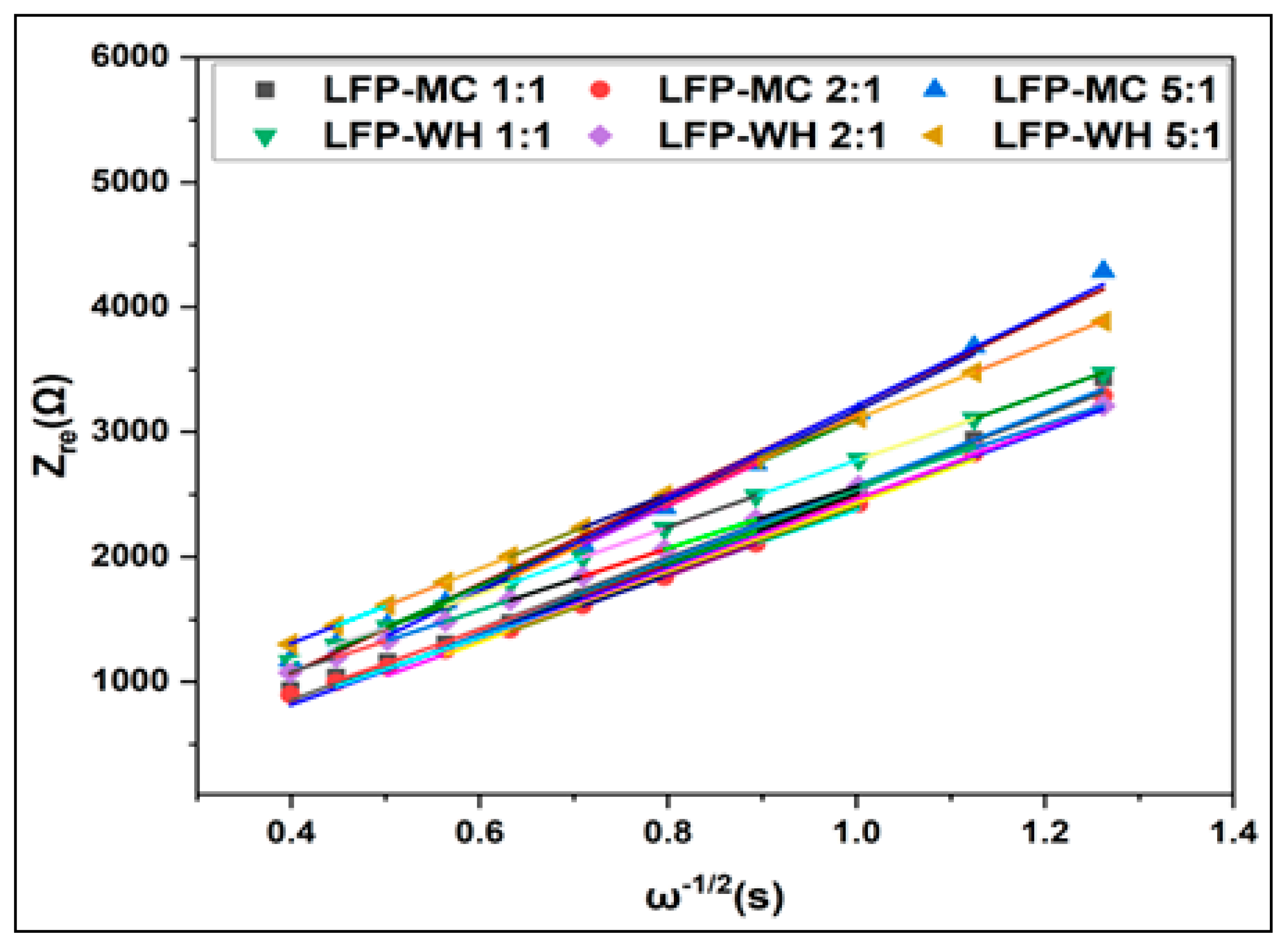
3.3.3.4. Electrochemical Impedance Spectroscopy (EIS) Analysis
| Samples | Re (Ω) | Rct (Ω) | DLi (cm²/s) |
| LFP/MC 1:1 | 4.39 | 99.91 | 1.69x10-13 |
| LFP/ MC 2:1 | 2.74 | 95.87 | 1.84x10-13 |
| LFP/ MC 5:1 | 5.72 | 124.88 | 1.07x10-13 |
| LFP/WH 1:1 | 2.70 | 98.6 | 1.94x10-13 |
| LFP/ WH 2:1 | 2.49 | 91.12 | 2.28x10-13 |
| LFP/ WH 5:1 | 3.02 | 110.38 | 1.53x10-13 |
| LFP/graphite | 2.34 | 73.81 | 3.55x10-13 |
4. Comments on Main Results
5. Conclusions
Author Contributions
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BET | Brunauer, Emmett, and Teller |
| BJH | Barrett, Joyner, and Halenda |
| C | Carbon, Current Rate |
| SC | Specific Capacitances |
| D | Diameter |
| DMAC | Dimethylacetamide |
| XRD | X-ray Diffraction |
| EIS | Electrochemical Impedance Spectroscopy |
| MC | Millet Cob |
| EDS | Energy Dispersive Spectroscopy |
| HCl | Hydrogen Chloride |
| HK | Horváth-Kawazoe |
| H₃PO4 | Phosphoric Acid |
| WH | Water Hyacinth |
| KOH | Potassium Hydroxide |
| LiFePO4 or LFP | Lithium Iron Phosphate |
| Li+ | Lithium Ion |
| LiPF6 | Lithium Hexafluorophosphate |
| CM | Carbonaceous Materials |
| SEM | Scanning Electron Microscopy |
| N2 | Nitrogen |
| NLDFT | Non-Local Density Functional Theory |
| PVDF | Polyvinylidene Fluoride |
| Rct | Charge Transfer Resistance |
| Re | Electrolyte Resistance |
References
- K. Padhi, K. S. Nanjundaswamy, and J. B. Goodenough, “Phospho-olivines as positive-electrode materials for rechargeable lithium batteries,” Journal of the electrochemical society, vol. 144, no. 4, p. 1188, 1997.
- S. Lee, I. Jang, H. a. Lim, V. Aravindan, H. Kim, and Y. Lee, “Preparation and electrochemical characterization of LiFePO4 nanoparticles with high rate capability by a sol–gel method,” Journal of Alloys and Compounds, vol. 491, no. 1-2, pp. 668-672, 2010. [CrossRef]
- Z. Li, D. Zhang, and F. Yang, “Developments of lithium-ion batteries and challenges of LiFePO4 as one promising cathode material,” Journal of materials science, vol. 44, no. 10, pp. 2435-2443, 2009. [CrossRef]
- D.-H. Kim and J. Kim, “Synthesis of LiFePO4 nanoparticles in polyol medium and their electrochemical properties,” Electrochemical and Solid-State Letters, vol. 9, no. 9, p. A439, 2006. [CrossRef]
- N. J. Yun, H.-W. Ha, K. H. Jeong, H.-Y. Park, and K. Kim, “Synthesis and electrochemical properties of olivine-type LiFePO4/C composite cathode material prepared from a poly (vinyl alcohol)-containing precursor,” Journal of Power Sources, vol. 160, no. 2, pp. 1361-1368, 2006.
- A. Ritchie and W. Howard, “Recent developments and likely advances in lithium-ion batteries,” Journal of Power Sources, vol. 162, no. 2, pp. 809-812, 2006. [CrossRef]
- T. Nakamura, Y. Miwa, M. Tabuchi, and Y. Yamada, “Structural and surface modifications of LiFePO4 olivine particles and their electrochemical properties,” Journal of the Electrochemical Society, vol. 153, no. 6, p. A1108, 2006. [CrossRef]
- Y. Guan et al., “LiFePO4/activated carbon/graphene composite with capacitive-battery characteristics for superior high-rate lithium-ion storage,” Electrochimica Acta, vol. 294, pp. 148-155, 2019. [CrossRef]
- H. Zhang, D. Liu, X. Qian, C. Zhao, and Y. Xu, “A novel nano structured LiFePO4/C composite as cathode for Li-ion batteries,” Journal of Power Sources, vol. 249, pp. 431-434, 2014. [CrossRef]
- H. Wang et al., “In-situ self-polymerization restriction to form core-shell LiFePO4/C nanocomposite with ultrafast rate capability for high-power Li-ion batteries,” Nano Energy, vol. 39, pp. 346-354, 2017. [CrossRef]
- J. Tu, K. Wu, H. Tang, H. Zhou, and S. Jiao, “Mg–Ti co-doping behavior of porous LiFePO 4 microspheres for high-rate lithium-ion batteries,” Journal of Materials Chemistry A, vol. 5, no. 32, pp. 17021-17028, 2017.
- M. Talebi-Esfandarani, “Synthesis, Characterization and Modification of LifeP04 by Doping with Platinum and Palladium for Lithium-Ion Batteries,” École Polytechnique de Montréal, 2013.
- Wang, W. Al Abdulla, D. Wang, and X. Zhao, “A three-dimensional porous LiFePO 4 cathode material modified with a nitrogen-doped graphene aerogel for high-power lithium ion batteries,” Energy & Environmental Science, vol. 8, no. 3, pp. 869-875, 2015. [CrossRef]
- Z. Jinli et al., “High-performance lithium iron phosphate with phosphorus-doped carbon layers for lithium ion batteries,” Journal of Materials Chemistry A, vol. 3, no. 5, pp. 2043-2049, 2015. [CrossRef]
- Y. Zhou et al., “Nitrogen-doped graphene guided formation of monodisperse microspheres of LiFePO 4 nanoplates as the positive electrode material of lithium-ion batteries,” Journal of Materials Chemistry A, vol. 4, no. 31, pp. 12065-12072, 2016. [CrossRef]
- Y. Wang, Y. Wang, E. Hosono, K. Wang, and H. Zhou, “The design of a LiFePO4/carbon nanocomposite with a core–shell structure and its synthesis by an in situ polymerization restriction method,” Angewandte Chemie International Edition, vol. 47, no. 39, pp. 7461-7465, 2008.
- H. Huang, S.-C. Yin, and L. s. Nazar, “Approaching theoretical capacity of LiFePO4 at room temperature at high rates,” Electrochemical and solid-state letters, vol. 4, no. 10, p. A170, 2001. [CrossRef]
- R. Shahid and S. Murugavel, “Synthesis and characterization of olivine phosphate cathode material with different particle sizes for rechargeable lithium-ion batteries,” Materials Chemistry and Physics, vol. 140, no. 2-3, pp. 659-664, 2013. [CrossRef]
- S.-Y. Chung, J. T. Bloking, and Y.-M. Chiang, “Electronically conductive phospho-olivines as lithium storage electrodes,” Nature materials, vol. 1, no. 2, pp. 123-128, 2002.
- M. Talebi-Esfandarani and O. Savadogo, “Effects of palladium doping on the structure and electrochemical properties of LiFePO4/C prepared using the sol-gel method,” J New Mater Electrochem Syst, vol. 17, no. 2, pp. 91-97, 2014.
- M. Talebi-Esfandarani and O. Savadogo, “Enhancement of electrochemical properties of platinum doped LiFePO4/C cathode material synthesized using hydrothermal method,” Solid State Ionics, vol. 261, pp. 81-86, 2014. [CrossRef]
- M. Talebi-Esfandarani and O. Savadogo, “Synthesis and characterization of Pt-doped LiFePO 4/C composites using the sol–gel method as the cathode material in lithium-ion batteries,” Journal of Applied Electrochemistry, vol. 44, pp. 555-562, 2014.
- N. Ravet, A. Abouimrane, and M. Armand, “On the electronic conductivity of phospho-olivines as lithium storage electrodes,” Nature Materials, vol. 2, no. 11, pp. 702-702, 2003. [CrossRef]
- M. Shi, R. Li, and Y. Liu, “In situ preparation of LiFePO4/C with unique copolymer carbon resource for superior performance lithium-ion batteries,” Journal of Alloys and Compounds, vol. 854, p. 157162, 2021.
- N. Ravet, S. Besner, M. Simoneau, A. Vallée, M. Armand, and J.-F. Magnan, “Electrode materials with high surface conductivity,” ed: Google Patents, 2005.
- Z. Chen and J. Dahn, “Reducing carbon in LiFePO4/C composite electrodes to maximize specific energy, volumetric energy, and tap density,” Journal of the Electrochemical Society, vol. 149, no. 9, p. A1184, 2002.
- R. Dominko et al., “Impact of the carbon coating thickness on the electrochemical performance of LiFePO4/C composites,” Journal of the Electrochemical Society, vol. 152, no. 3, p. A607, 2005.
- M. M. Doeff, J. D. Wilcox, R. Kostecki, and G. Lau, “Optimization of carbon coatings on LiFePO4,” Journal of power sources, vol. 163, no. 1, pp. 180-184, 2006. [CrossRef]
- M. M. Doeff, J. D. Wilcox, R. Yu, A. Aumentado, M. Marcinek, and R. Kostecki, “Impact of carbon structure and morphology on the electrochemical performance of LiFePO 4/C composites,” Journal of Solid State Electrochemistry, vol. 12, no. 7, pp. 995-1001, 2008.
- M. Talebi-Esfandarani and O. Savadogo, “Improvement of electrochemical and electrical properties of LiFePO 4 coated with citric acid,” Rare Metals, vol. 35, no. 4, pp. 303-308, 2016. [CrossRef]
- X. Zhi, G. Liang, L. Wang, X. Ou, L. Gao, and X. Jie, “Optimization of carbon coatings on LiFePO4: Carbonization temperature and carbon content,” Journal of Alloys and Compounds, vol. 503, no. 2, pp. 370-374, 2010. [CrossRef]
- E. Sohouli, K. Adib, B. Maddah, and M. Najafi, “Manganese dioxide/cobalt tungstate/ nitrogen-doped carbon nano-onions nanocomposite as new supercapacitor electrode,” Ceramics International, vol. 48, no. 1, pp. 295-303, 2022/01/01/ 2022. [CrossRef]
- E. Sohouli, K. Adib, B. Maddah, and M. Najafi, “Preparation of a supercapacitor electrode based on carbon nano-onions/manganese dioxide/iron oxide nanocomposite,” Journal of Energy Storage, vol. 52, p. 104987, 2022/08/25/ 2022. [CrossRef]
- M. A. Albo Hay Allah and H. A. Alshamsi, “Green synthesis of ZnO NPs using Pontederia crassipes leaf extract: characterization, their adsorption behavior and anti-cancer property,” Biomass Conversion and Biorefinery, vol. 14, no. 9, pp. 10487-10500, 2024/05/01 2024. [CrossRef]
- G. Wilson, S. Zilinskaite, S. Unka, R. Boston, and N. Reeves-McLaren, “Establishing operando diffraction capability through the study of Li-ion (de) intercalation in LiFePO4,” Energy Reports, vol. 6, pp. 174-179, 2020.
- J. Rouquerol, F. Rouquerol, P. Llewellyn, G. Maurin, and K. Sing, Adsorption by powders and porous solids: principles, methodology and applications. Academic press, 2013.
- E. P. Barrett, L. G. Joyner, and P. P. Halenda, “The determination of pore volume and area distributions in porous substances. I. Computations from nitrogen isotherms,” Journal of the American Chemical society, vol. 73, no. 1, pp. 373-380, 1951.
- G. Horváth and K. Kawazoe, “Method for the calculation of effective pore size distribution in molecular sieve carbon,” Journal of Chemical Engineering of Japan, vol. 16, no. 6, pp. 470-475, 1983. [CrossRef]
- K. S. Sing, “Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity (Recommendations 1984),” Pure and applied chemistry, vol. 57, no. 4, pp. 603-619, 1985.
- S. Yu, J. Bo, L. Fengli, and L. Jiegang, “Structure and fractal characteristic of micro-and meso-pores in low, middle-rank tectonic deformed coals by CO2 and N2 adsorption,” Microporous and Mesoporous Materials, vol. 253, pp. 191-202, 2017.
- W. Han et al., “Experimental analysis of the pore structure and fractal characteristics of different metamorphic coal based on mercury intrusion-nitrogen adsorption porosimetry,” Powder Technology, vol. 362, pp. 386-398, 2020. [CrossRef]
- J. Tang, L. Feng, Y. Li, J. Liu, and X. Liu, “Fractal and pore structure analysis of Shengli lignite during drying process,” Powder Technology, vol. 303, pp. 251-259, 2016. [CrossRef]
- A. Saito and H. Foley, “Curvature and parametric sensitivity in models for adsorption in micropores,” AIChE journal, vol. 37, no. 3, pp. 429-436, 1991. [CrossRef]
- P. I. Ravikovitch, G. L. Haller, and A. V. Neimark, “Density functional theory model for calculating pore size distributions: pore structure of nanoporous catalysts,” Advances in colloid and interface science, vol. 76, pp. 203-226, 1998. [CrossRef]
- R. S. Mikhail, S. Brunauer, and E. Bodor, “Investigations of a complete pore structure analysis: I. Analysis of micropores,” Journal of Colloid and Interface Science, vol. 26, no. 1, pp. 45-53, 1968.
- F. Rouquerol, J. Rouquerol, and K. Sing, “Assessment of mesoporosity,” Adsorption by powders and porous solids, pp. 191-217, 1999.
- N. A. Rahma, A. Kurniasari, Y. D. S. Pambudi, H. M. Bintang, A. Zulfia, and C. Hudaya, “Characteristics of Corncob-Originated Activated Carbon Using Two Different Chemical Agent,” in IOP Conference Series: Materials Science and Engineering, 2019, vol. 622, no. 1: IOP Publishing, p. 012030.
- S. Wang and G. Lu*, “A comprehensive study on carbon dioxide reforming of methane over Ni/γ-Al2O3 catalysts,” Industrial & engineering chemistry research, vol. 38, no. 7, pp. 2615-2625, 1999.
- S. R. Kamath and A. Proctor, “Silica gel from rice hull ash: preparation and characterization,” Cereal Chemistry, vol. 75, no. 4, pp. 484-487, 1998. [CrossRef]
- A. Omri and M. Benzina, “Characterization of activated carbon prepared from a new raw lignocellulosic material: Ziziphus spina-christi seeds,” Journal de la Société Chimique de Tunisie, vol. 14, pp. 175-183, 2012.
- S. Ahmed, M. Parvaz, R. Johari, and M. Rafat, “Studies on activated carbon derived from neem (azadirachta indica) bio-waste, and its application as supercapacitor electrode,” Materials Research Express, vol. 5, no. 4, p. 045601, 2018.
- Y. Chen et al., “Application studies of activated carbon derived from rice husks produced by chemical-thermal process—A review,” Advances in colloid and interface science, vol. 163, no. 1, pp. 39-52, 2011. [CrossRef]
- De and N. Karak, “A green and facile approach for the synthesis of water soluble fluorescent carbon dots from banana juice,” Rsc Advances, vol. 3, no. 22, pp. 8286-8290, 2013. [CrossRef]
- T. Qiu, J.-G. Yang, X.-J. Bai, and Y.-L. Wang, “The preparation of synthetic graphite materials with hierarchical pores from lignite by one-step impregnation and their characterization as dye absorbents,” RSC advances, vol. 9, no. 22, pp. 12737-12746, 2019. [CrossRef]
- A. Barroso-Bogeat, M. Alexandre-Franco, C. Fernández-González, J. Sánchez-González, and V. Gómez-Serrano, “Electrical conductivity of metal (hydr) oxide–activated carbon composites under compression. A comparison study,” Materials Chemistry and Physics, vol. 152, pp. 113-122, 2015.
- Celzard, J. Marêché, F. Payot, and G. Furdin, “Electrical conductivity of carbonaceous powders,” Carbon, vol. 40, no. 15, pp. 2801-2815, 2002. [CrossRef]
- T. Adinaveen, J. J. Vijaya, and L. J. Kennedy, “Comparative study of electrical conductivity on activated carbons prepared from various cellulose materials,” Arabian Journal for Science and Engineering, vol. 41, pp. 55-65, 2016. [CrossRef]
- S. Ryu, S. Kim, N. Gallego, and D. Edie, “Physical properties of silver-containing pitch-based activated carbon fibers,” Carbon, vol. 37, no. 10, pp. 1619-1625, 1999. [CrossRef]
- Z. Hashisho, M. J. Rood, S. Barot, and J. Bernhard, “Role of functional groups on the microwave attenuation and electric resistivity of activated carbon fiber cloth,” Carbon, vol. 47, no. 7, pp. 1814-1823, 2009. [CrossRef]
- Y. Huang, “Electrical and thermal properties of activated carbon fibers,” in Activated carbon fiber and textiles: Elsevier, 2017, pp. 181-192.
- J. R. Dahn, T. Zheng, Y. Liu, and J. Xue, “Mechanisms for lithium insertion in carbonaceous materials,” Science, vol. 270, no. 5236, pp. 590-593, 1995. [CrossRef]
- K. Ciosek Högström et al., “The influence of PMS-additive on the electrode/electrolyte interfaces in LiFePO4/graphite Li-ion batteries,” The Journal of Physical Chemistry C, vol. 117, no. 45, pp. 23476-23486, 2013.
- H. Ekström and G. Lindbergh, “A model for predicting capacity fade due to SEI formation in a commercial graphite/LiFePO4 cell,” Journal of The Electrochemical Society, vol. 162, no. 6, p. A1003, 2015. [CrossRef]
- F. Yang, X. Song, G. Dong, and K.-L. Tsui, “A coulombic efficiency-based model for prognostics and health estimation of lithium-ion batteries,” Energy, vol. 171, pp. 1173-1182, 2019. [CrossRef]
- J. B. Goodenough and K.-S. Park, “The Li-ion rechargeable battery: a perspective,” Journal of the American Chemical Society, vol. 135, no. 4, pp. 1167-1176, 2013. [CrossRef]
- J. B. Goodenough and Y. Kim, “Challenges for rechargeable Li batteries,” Chemistry of materials, vol. 22, no. 3, pp. 587-603, 2010.
- S. S. Prabaharan and M. S. Michael, Nanotechnology in Advanced Electrochemical Power Sources. CRC Press, 2014.
- L. Deleebeeck and S. Veltzé, “Electrochemical impedance spectroscopy study of commercial Li-ion phosphate batteries: a metrology perspective,” International Journal of Energy Research, vol. 44, no. 9, pp. 7158-7182, 2020. [CrossRef]
- Y. Cui, X. Zhao, and R. Guo, “Improved electrochemical performance of La0. 7Sr0. 3MnO3 and carbon co-coated LiFePO4 synthesized by freeze-drying process,” Electrochimica Acta, vol. 55, no. 3, pp. 922-926, 2010. [CrossRef]

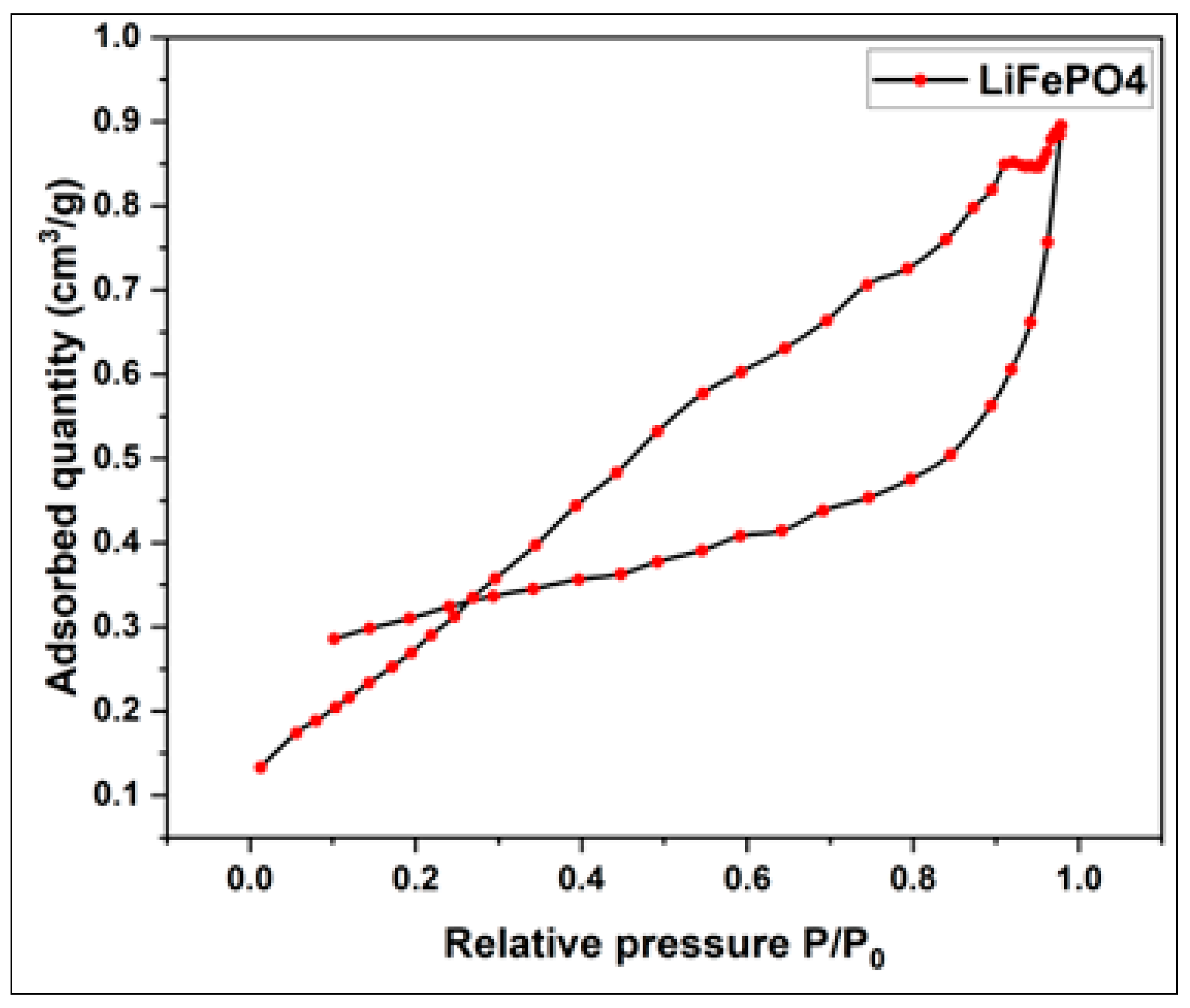
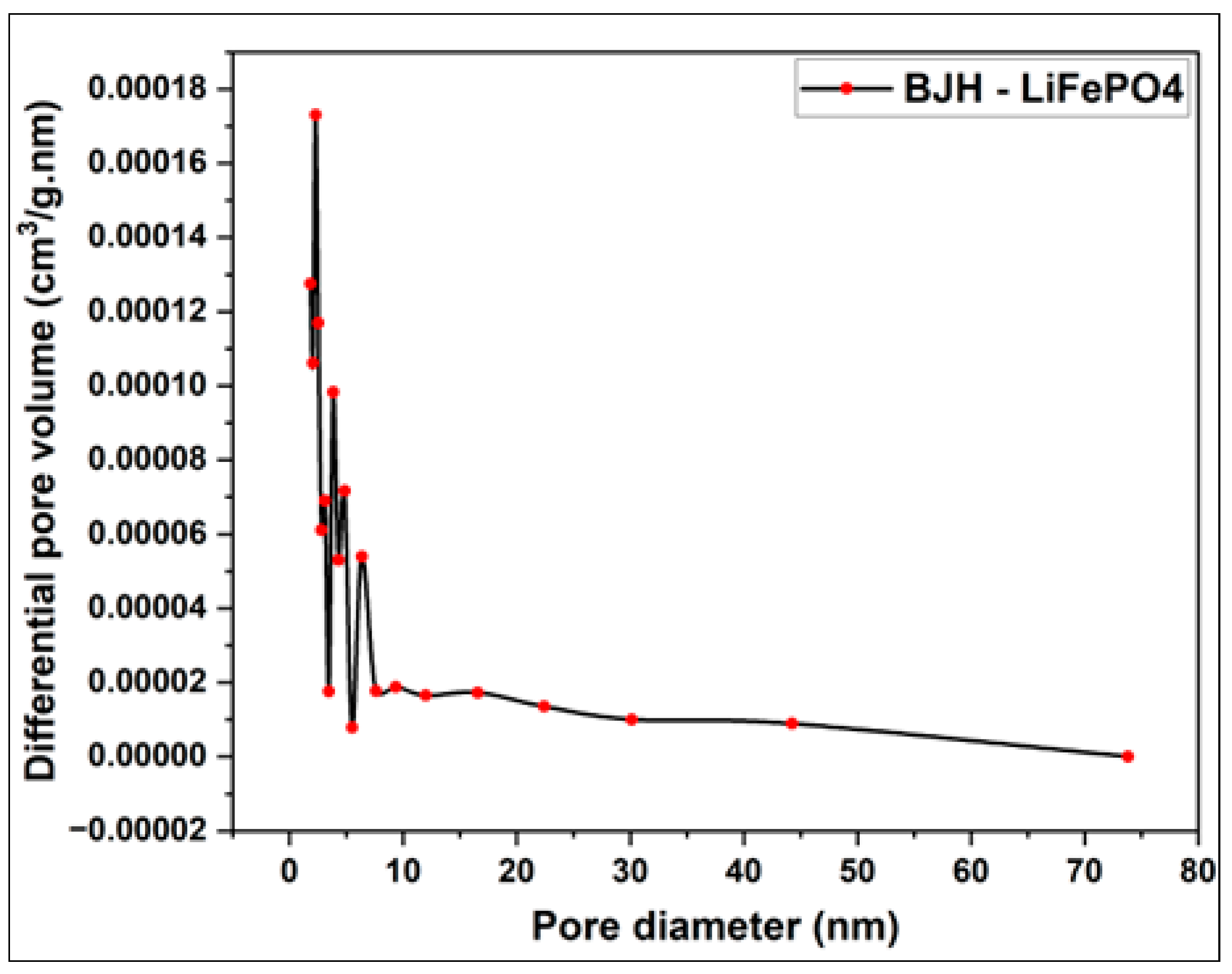
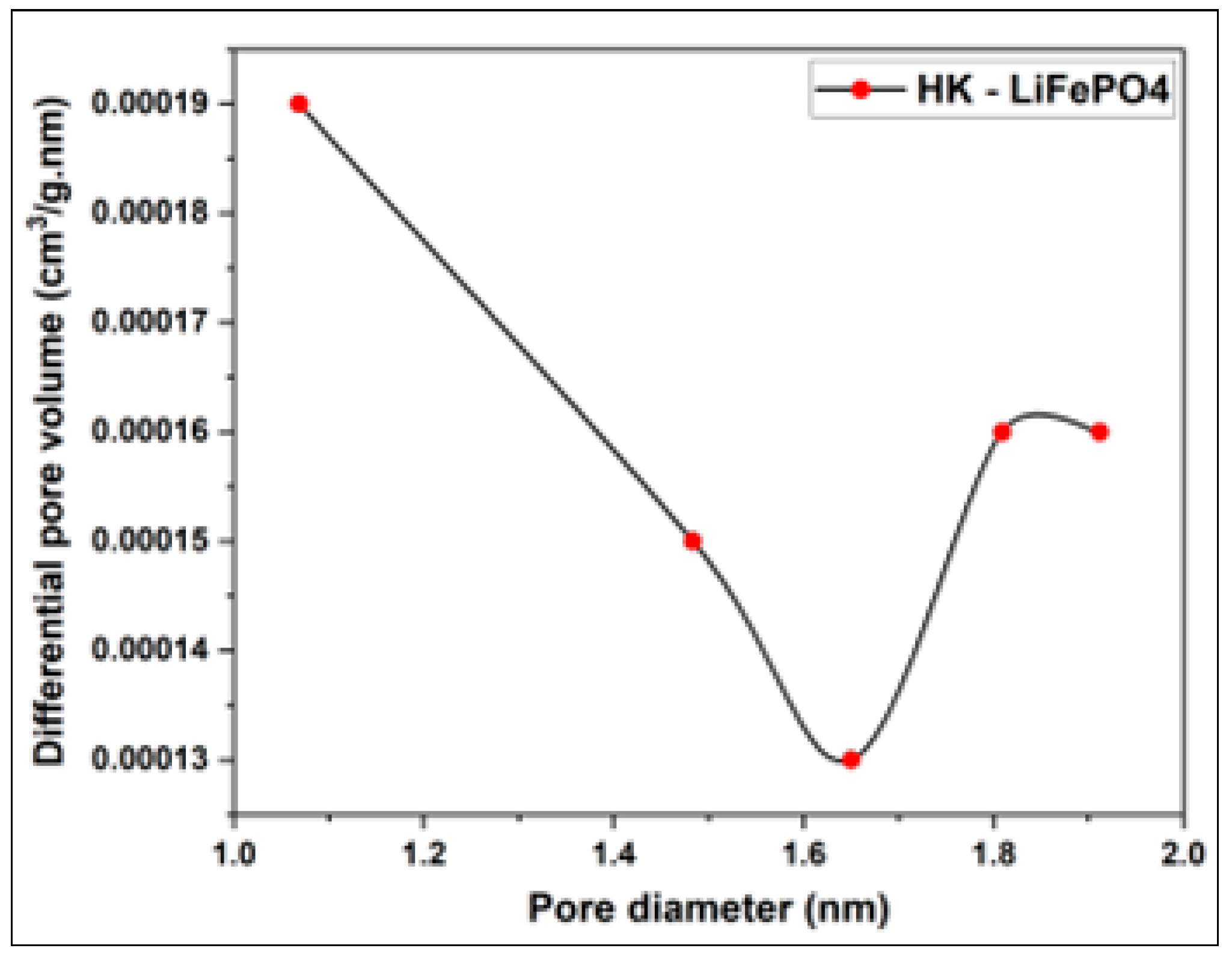
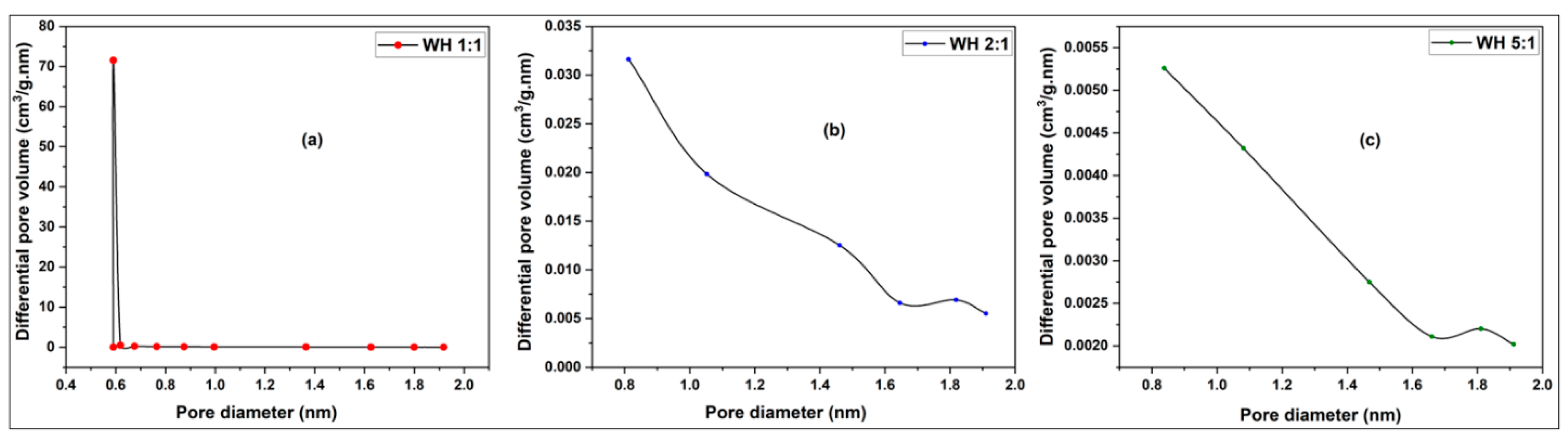
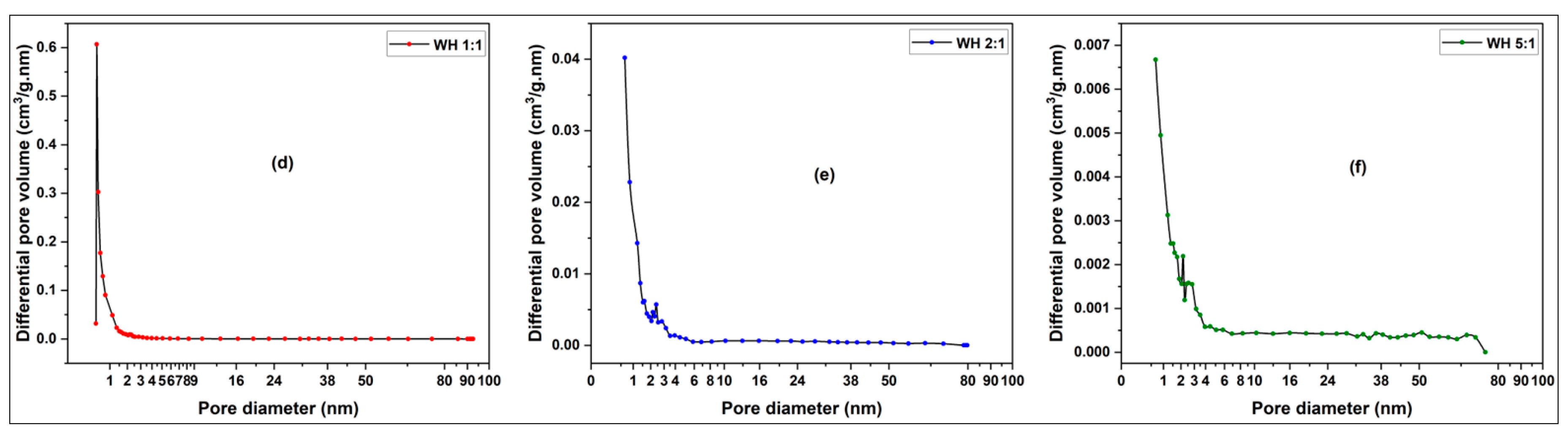
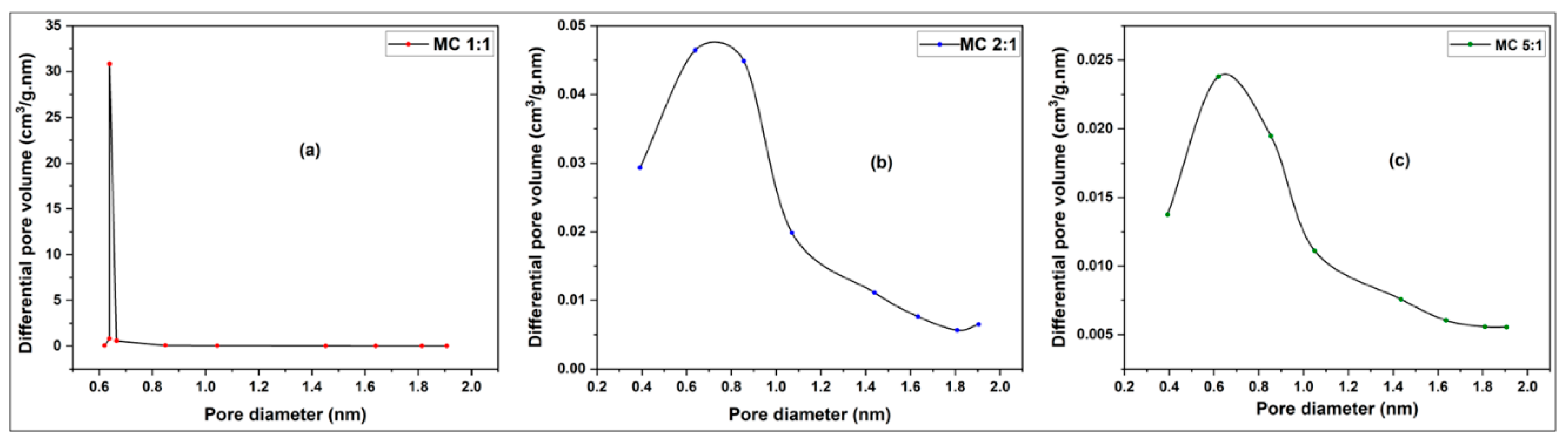
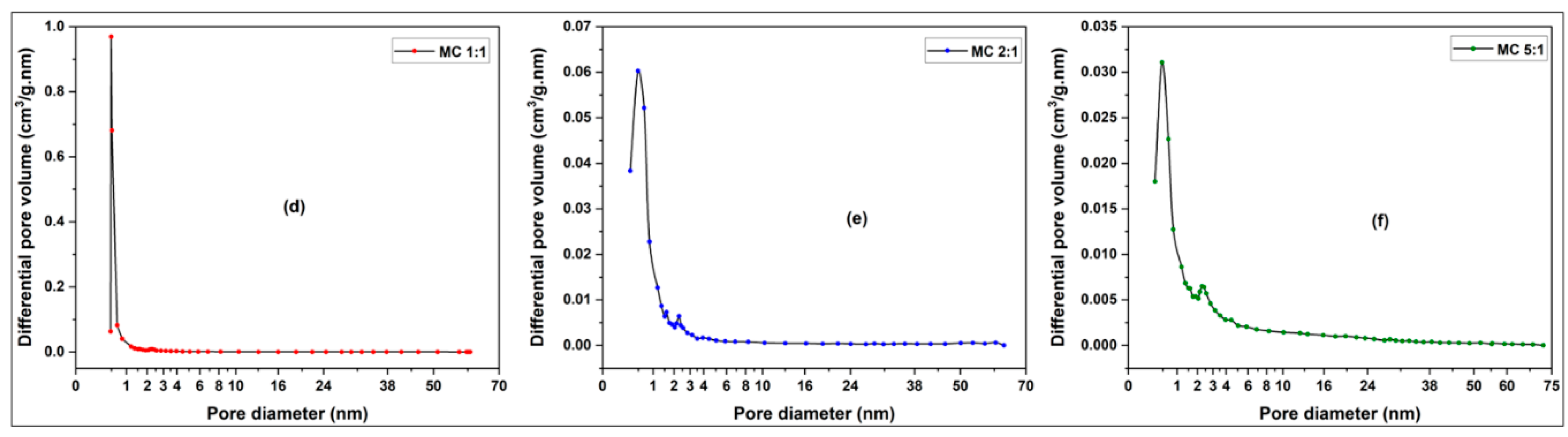

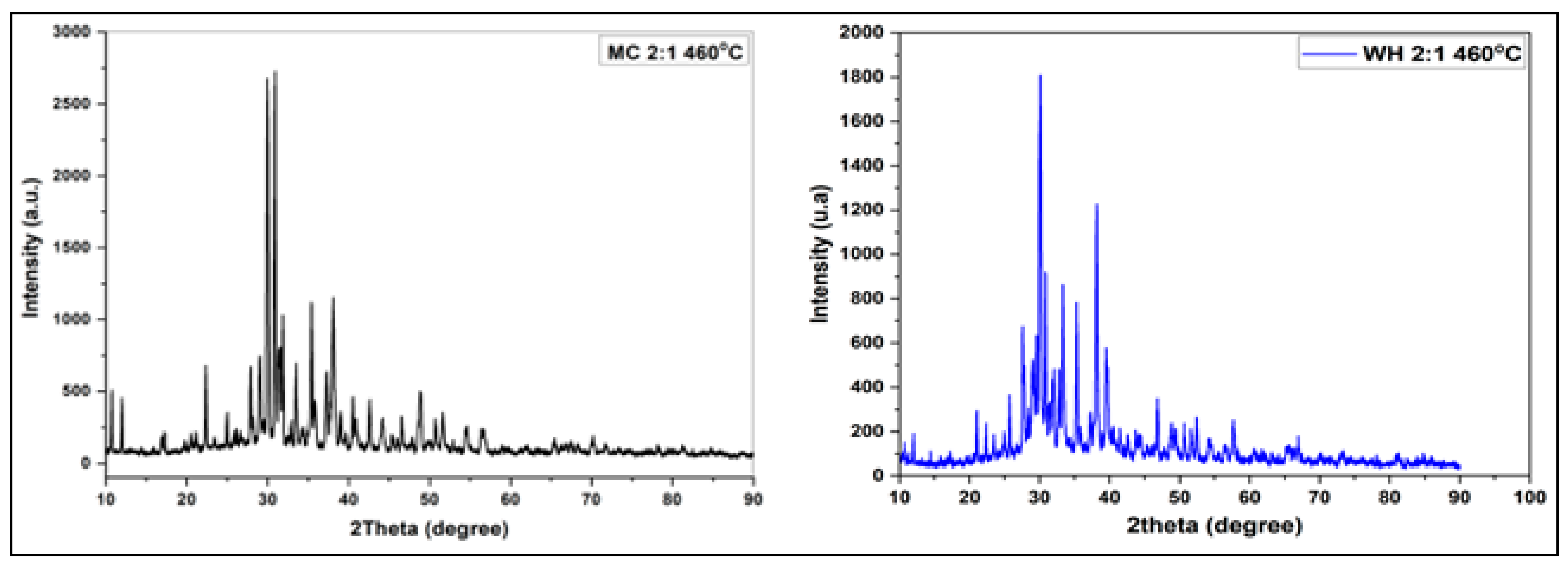
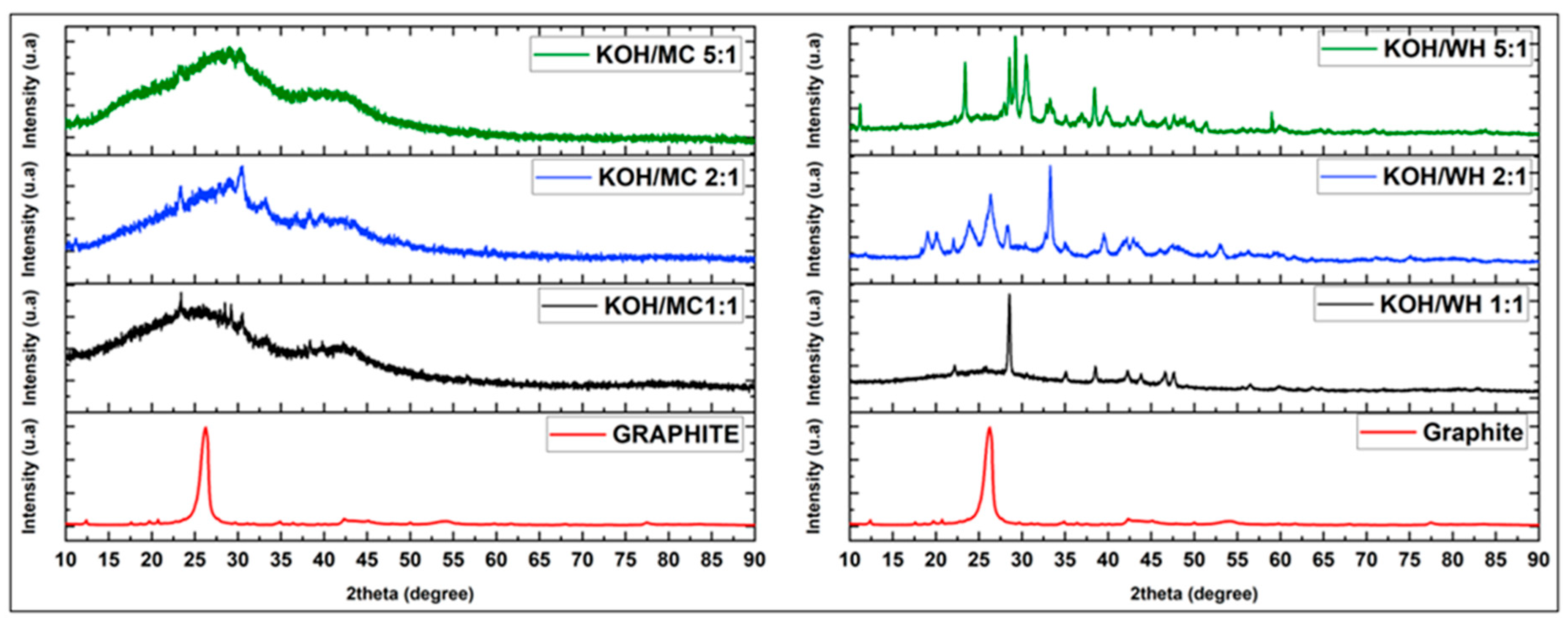
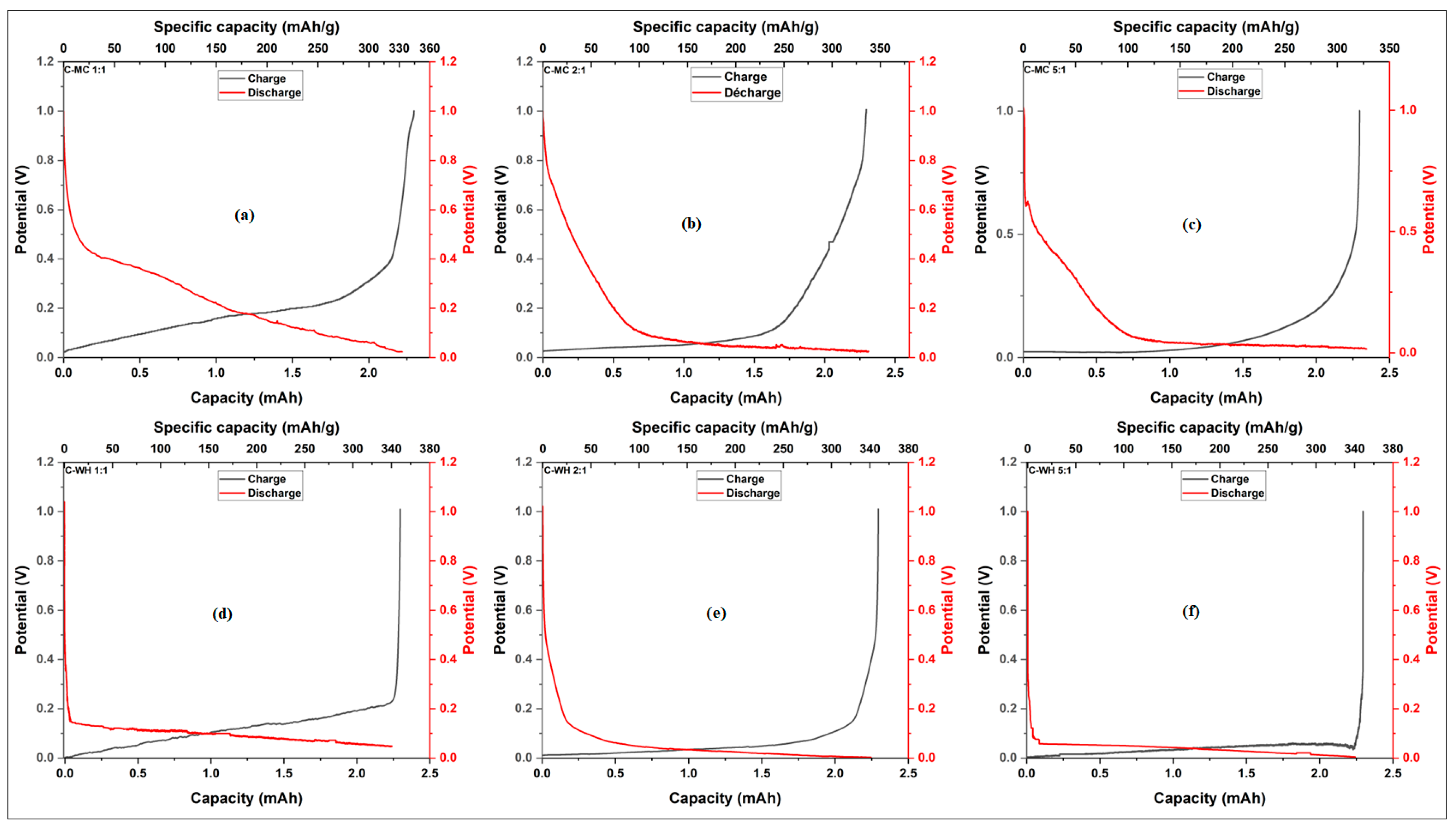
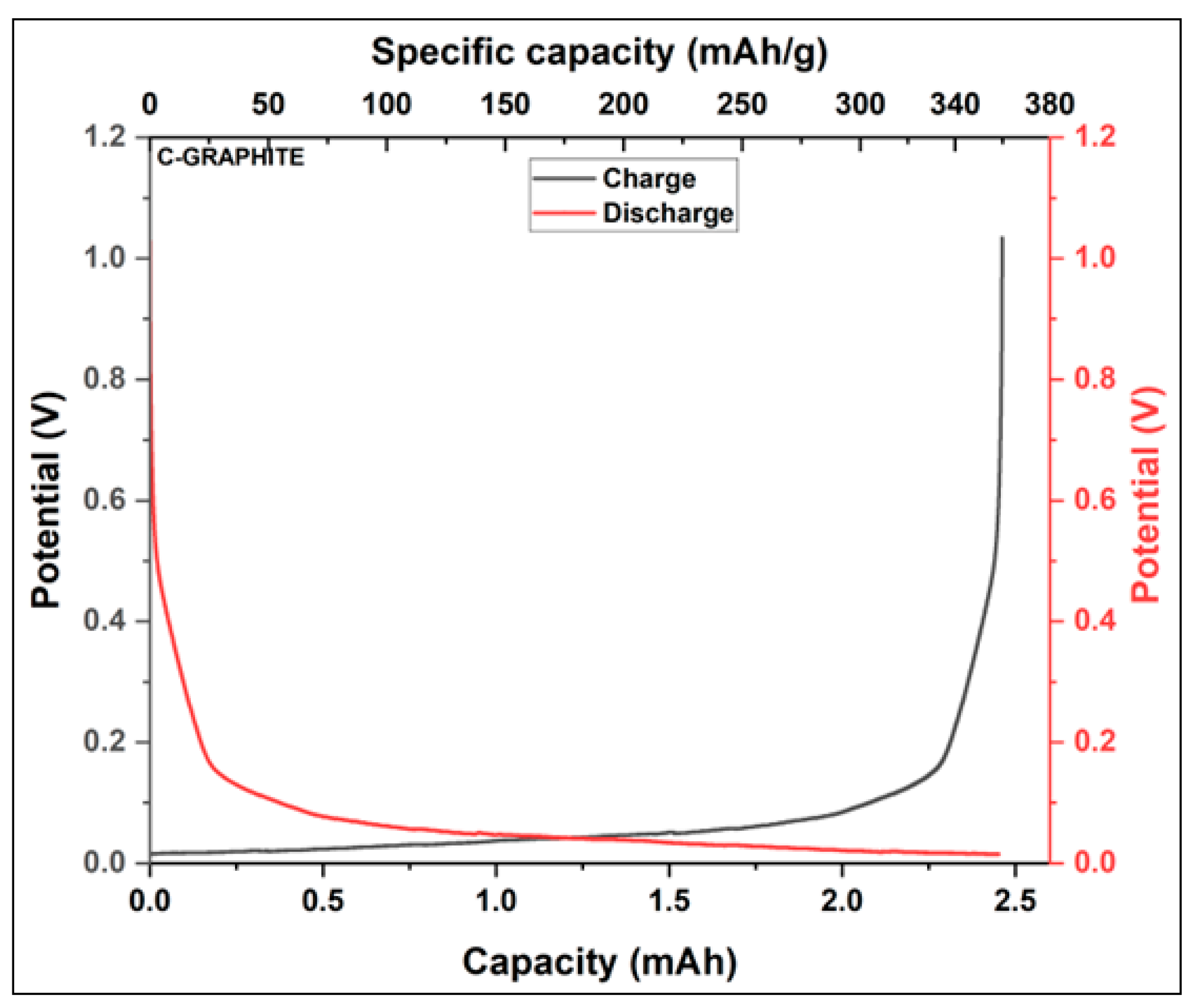
| Sample | BET surface area (m2/g) | Total pore volume (cm3/g) | Vmicropore (cm3/g) | Vmesopore (cm3/g) | Average pore diameter (nm) |
| LiFePO4 | 1.2012 | 0.0014 | 0.0005 | 0.0009 | 4.6620 |
| Sample | BET Surface area (m2/g) | Total pore volume (cm3/g) | Vmicropore (cm3/g) | V mesopore (cm3/g) | Average pore diameter (nm) |
| WH 1:1 | 413.03 | 0.2376 | 0.2031 | 0.0343 | 2.3001 |
| WH 2:1 | 83.17 | 0.0774 | 0.0391 | 0.0382 | 3.7181 |
| WH 5:1 | 18.53 | 0.0389 | 0.0075 | 0.0129 | 8.3790 |
| MC 1:1 | 216.34 | 0.1286 | 0.1043 | 0.0243 | 2.3778 |
| MC 2:1 | 95.75 | 0.0787 | 0.0469 | 0.0311 | 3.2878 |
| MC 5:1 | 57.79 | 0.0763 | 0.0280 | 0.0480 | 5.2819 |
| Samples | KOH/CM | C (%) | O (%) | K (%) | Na (%) | Cl (%) | Al (%) | Si (%) | Mg (%) | P (%) |
| MC 460oC | 2 : 1 | 30 | 33 | 35 | - | - | 0.2 | 1.7 | 0.1 | - |
| WH 460oC | 2 : 1 | 26 | 41 | 30 | 0.3 | 1.4 | 0.1 | 0.4 | 0.3 | 0.1 |
| Samples | KOH/C | Chemical elements (% mass) | |||||||||||
| C | O | K | Na | Cl | Al | Si | Fe | Ca | Mg | S | P | ||
| Millet cob | 1 : 1 | 85.5 | 12 | 2 | - | 0.3 | - | 0.2 | - | - | |||
| 2 : 1 | 71.5 | 19 | 6 | 0.1 | 0.8 | 0.8 | 1.1 | 0.1 | 0.1 | 0.1 | 0.4 | - | |
| 5 : 1 | 71 | 20 | 8.3 | - | 0.1 | 0.2 | 0.3 | - | - | - | 0.1 | - | |
| Water hyacinth | 1 : 1 | 80 | 13 | 2 | - | 2 | 0.2 | 0.5 | 0.4 | 0.8 | 0.1 | 0.9 | 0.1 |
| 2 : 1 | 70 | 18 | 8 | 0.1 | 1.8 | 0.2 | 0.7 | 0.1 | 0.6 | 0.2 | 0.2 | 0.1 | |
| 5 : 1 | 70 | 18 | 8 | 0.1 | 2.4 | 0.1 | 0.4 | 0.2 | 0.2 | 0.1 | 0.5 | - | |
| Sample | Mixing ration (KOH/CM) | Electrical conductivity (S.cm-1) | Total pore volume (cm3/g) | Average pore diameter (nm) | |
| Millet cob | 1 : 1 | 6,74 10-3 | 0.2376 | 2.3001 | |
| 2 : 1 | 8,42 10-3 | 0.0774 | 3.7181 | ||
| 5 : 1 | 4,99 10-3 | 0.0389 | 8.3790 | ||
| Water hyacinth | 1 : 1 | 8,92 10-3 | 0.1286 | 2.3778 | |
| 2 : 1 | 1,47 10-2 | 0.0787 | 3.2878 | ||
| 5 : 1 | 6,40 10-3 | 0.0763 | 5.2819 |
| Mixing ratio (KOH/CM) | Mixing ratio (KOH/CM) | Mixing ratio (KOH/CM) | ||
| Sample | 1.1 | 2:1 | 5:1 | |
| Millet cob | 333 | 335 | 330 | |
| Water hyacinth | 336 | 339 | 332 |
| Samples | Coulombic efficiency (%) +/- 1% | ||
| Cycle 1 | Cycle 2 | Cycle 3 | |
| LFP/MC 1:1 | 95.02 | 100 | 99.79 |
| LFP/MC 2:1 | 95.86 | 100 | 99.82 |
| LFP/MC 5:1 | 95.48 | 100 | 99.73 |
| LFP/WH 1:1 | 96.05 | 100 | 99.88 |
| LFP/WH 2:1 | 96.36 | 100 | 99.96 |
| LFP/WH 5:1 | 95.88 | 100 | 99.84 |
| Current rate | SC (mAh/g) MC 1:1 | SC (mAh/g) MC 2:1 | SC (mAh/g) MC 5:1 |
| C12 | 152 | 160 | 149 |
| C6 | 148 | 157 | 145 |
| 1C | 140 | 150 | 138 |
| 5C | 102 | 106 | 100 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




