Submitted:
08 May 2025
Posted:
12 May 2025
You are already at the latest version
Abstract
Keywords:
“Il buono, il brutto, il cattivo“(Movie by Sergio Leone)
1. Introduction
2. . Nociception and Pain
2.1. Causes and Classification of Pain
2.1.1. Genesis and Site of Origin
2.1.1.1. Cutaneous Pain
2.1.1.2. Visceral Pain
2.1.1.3. Neuropathic Pain
2.1.1.3.1. Case Report: Acute Neuropathic Pain Following Herpes Zoster Infection
2.1.1.4. Inflammatory Pain
2.1.1.4.1. Case Report: Acute Inflammatory Pain in Septic Arthritis of the Knee
2.2. Time Course
3. From PNS to CNS
3.1. Nociceptors
3.1.1. Types of Nociceptors
3.1.2. Nociceptive Afferents
3.1.3. Nociceptors, Immune Cells and Cytokines
3.1.3.1. Case Report: Acute Burn Injury with Nociceptor Sensitization
3.1.4. To-Do-List
4. Spinal Cord
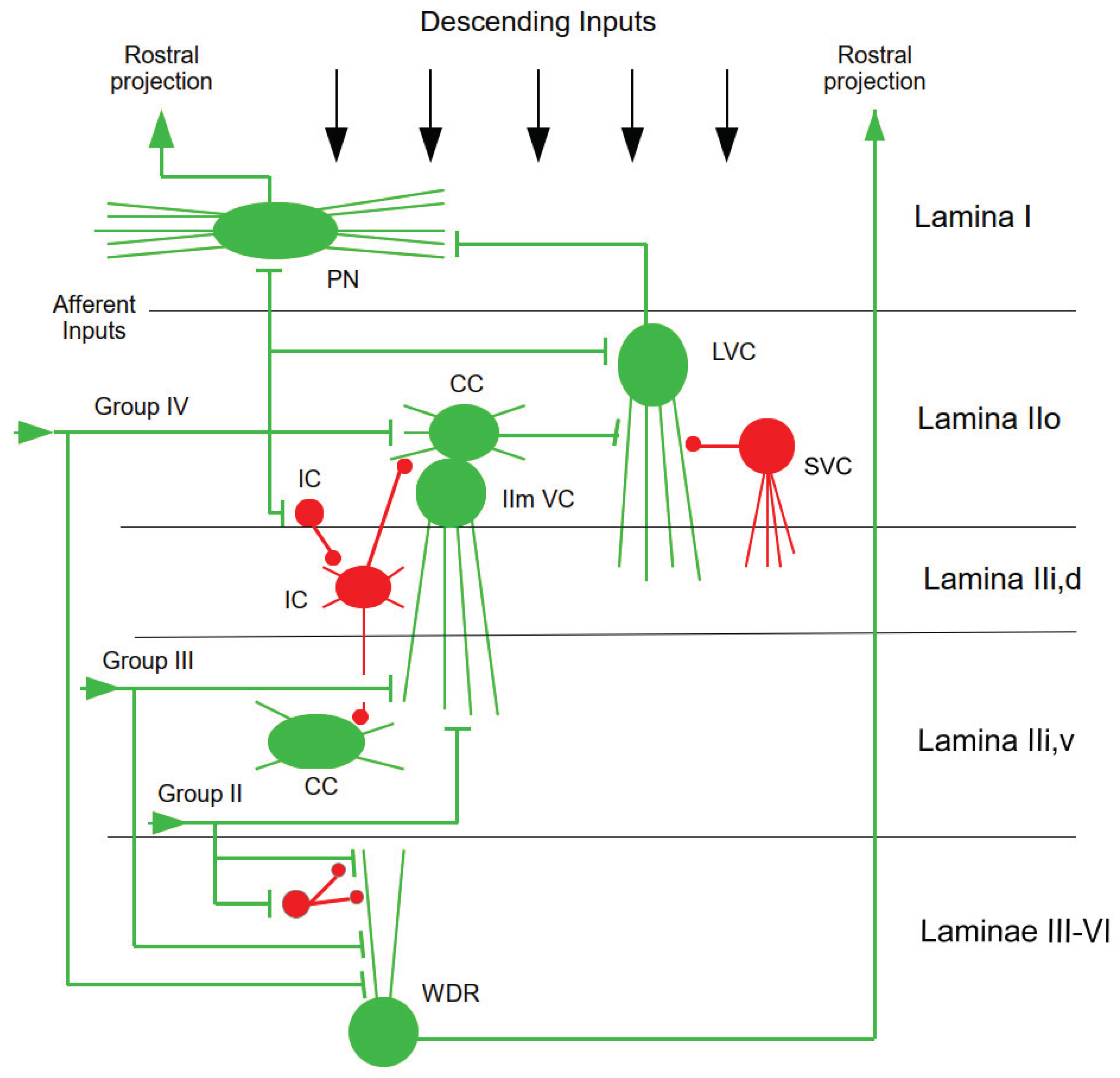
4.1. Dorsal Horn (DH)
4.2. Spinal Signal Transmission
4.2.1. Synaptic Effects
4.2.2. Presynaptic Inhibition (PSI)
4.3. Case Report: Acute Myofascial Pain Following Eccentric Exercise
5. Towards Supraspinal Structures
5.1. Spinal Projection Neurons with Multiple Supraspinal Targets
5.2. Itch Pathways
5.3. Nucleus Tractus Solitarii (NTS)
5.4. Parabrachial Nucleus (PBN)
5.5. Cerebellum
5.6. The PAG-Triad Connection
5.6.1. Peri-Aqueductal Gray (PAG)
5.6.2. Rostral Ventro-Medial Medulla (RVM)
5.6.3. Caudal Ventro-Lateral Medulla (CVLM)
5.6.4. Dorsal Reticular Nucleus (DReN)
5.6.5. Case Report: Acute Thoracoabdominal Trauma with Severe Pain and Autonomic Dysregulation
5.7. Hypothalamus (HYP)
5.8. Midbrain Dopamine (DA) Neurons
5.8.1. General Inputs
5.8.2. Nocicpeptive Inputs
5.8.3. Outputs
5.9. Locus Coeruleus (LC) and Other Cell Groups
5.9.1. Locus Coeruleus (LC)
5.9.2. A and C Cell Groups
5.10. Raphé Nuclei
5.11. Basal Ganglia (BG)
5.11.1. Striatum
5.11.2. Nucleus Accumbens (NAc)
5.11.3. Subthalamic Nucleus (STN)
5.12. Amygdala (AMY)
5.13. Habenula (Hb)
5.14. Thalamus (THAL)
5.15. Cerebral Cortex
5.15.1. Functional Differentiation of the Cerebral Cortex
5.15.2. Somatosensory Cortex
5.15.3. Prefrontal Cortex (PFC)
5.15.4. Cingulate Cortex (CC)
5.15.4.1. Anterior Cingulate Cortex (ACC)
5.15.4.2. Mid-Cingulate Cortex (MCC)
5.15.4.3. Posterior Cingulate Cortex (PCC)
5.15.5. Insular Cortex (IC)
5.15.6. Hippocampus (HIPP)
6. Motor Reactions to Noxious Stimuli
6.1. Vocalizations
6.2. Facial Expressions
6.3. Withdrawal Reactions
6.4. Nociceptive Reflex Modulation
6.5. Pain and Sleep
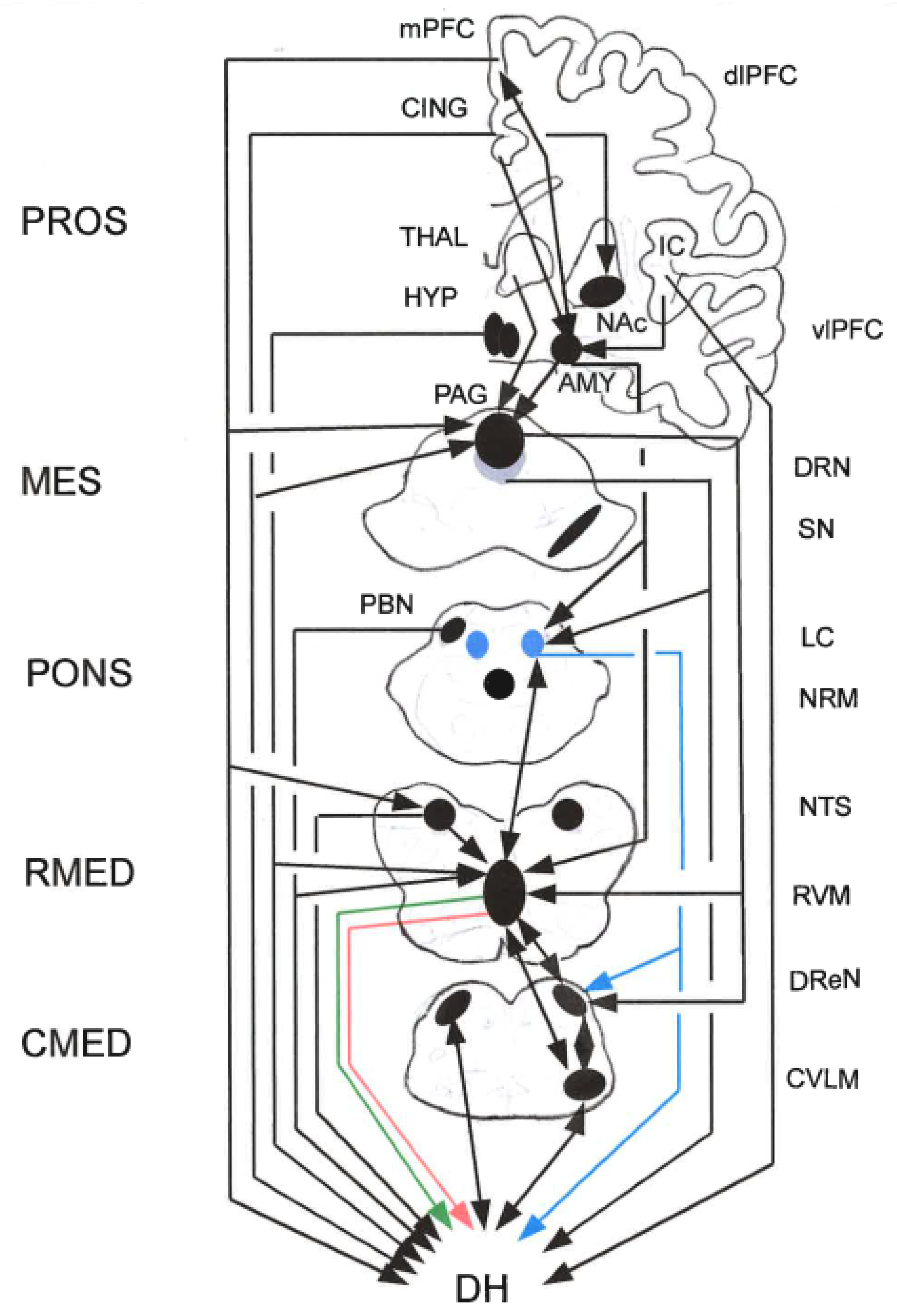
7. Descending Pain Control
7.1. Cerebral Cortex
7.1.1. Somatosensory Cortices
7.1.2. Primary Motor Cortex (M1)
7.1.3. Prefrontal Cortex (PFC)
7.1.3.1. Medial Prefrontal Cortex (mPFC)
7.1.3.2. Ventro-Medial Prefrontal Cortex (vmPFC)
7.1.3.3. Dorso-Lateral Prefrontal Cortex (dlPFC)
7.1.3.4. Ventro-Lateral Prefrontal Cortex (vlPFC)
7.1.3.5. OFC-THAL-PAG Connection
7.1.4. Cingulate Cortex (CC)
7.1.5. Insular Cortex (IC)
7.2. Basal Ganglia (BG)
7.2.1. Striatum
7.2.2. Nucleus Accumbens (NAc)
7.2.3. Subthalamic Nucleus (STN)
7.3. Amygdala (AMY)
7.4. Thalamus (THAL)
7.5. Habenula (Hb)
7.6. Hypothalamus (HYP)
7.6.1. Neurons Releasing Corticotropin-Releasing Hormone (CRH)
7.6.2. Neurons Releasing Oxytocin (OXT) and Vasopressin (AVP)
7.6.2.1. Neurons Releasing Oxytocin (OXT)
7.6.2.2. Neurons Releasing Vasopressin (AVP)
7.6.3. Neurons Releasing Orexin (ORX)
7.6.4. Neurons Releasing Hypothalamic Dopamine (DA) Neurons
7.7. Midbrain Neurons Releasing Dopamine (DA)
7.8. Brainstem Noradrenergic (NA) Cell Groups
7.8.1. Locus Coeruleus (LC)
7.8.2. Noradrenergic (NA) A Cell Groups
7.9. Peri-Aqueductal Gray (PAG)
7.10. Nuclei in the Reticular Formation (RF)
7.10.1. Rostral Ventro-Medial Medulla (RVM)
7.10.2. Caudal Ventro-Lateral Medulla (CVLM)
7.10.3. Dorsal Reticular Nucleus (DReN)
7.10.4. Dorsal Raphé Nucleus (DRN)
7.10.5. Sub-Nucleus Reticularis Dorsalis (SRD)
7.10.6. Acupuncture Against Visceral Pain
7.11. Cerebellum
7.12. Nucleus Tractus Solitarii (NTS)
7.13. Spinal Dorsal Horn (DH)
8. Clinical Syndromes
9. Concluding Remarks
Funding
Acknowledgments
Conflict of Interest
Ethics Approval and Consent to Participate
Abbreviations
References
- Adamaszek M, D´Agata F, Ferrucci R, Habas C, Keulen S, Kirkby KC, Leggio M, Mariёn P, Molinari M, Moulton E, Orsi L, Van Overwalle F, Papadelis C, Priori A, Sacchetti L, Schutter DJ, Styliadis C, Verhoeven J (2017) Consensus Paper: Cerebellum and emotion. Cerebellum 16(2):552-576.
- Agostinelli LJ, Mix MR, Hefti MM, Scammell TE, Bassuk AG (2021) Input-output connections of LJA5 prodynorphin neurons. J Comp Neurol 529(3):635-654.
- Akhter F, Haque T, Sato F, Kato T, Ohara H, Fujio T, Tsutsumi K, Uchino K, Sessler BJ, Yoshida A (2014) Projections from the dorsal peduncular cortex to the trigeminal subnucleus caudalis (medullary dorsal horn) and other lower brainstem areas in rats. Neuroscience 266:23-37. [CrossRef]
- Allen HN, Bobnar HJ, Kolber BJ (2021) Left and right hemispheric lateralization of the amygdala in pain. Prog Neurobiol 196:101891.
- Almeida TF, Roizenblatt S, Tufik S (2004) Afferent pain pathways: a neuroanatomical review. Brain Res 1000:40-56.
- Amorim D, Viisanen H, Wei H, Almeida A, Pertovaara A, Pinto-Ribeiro F (2015) Galanin-mediated behavioural hyperalgesia from the dorsomedial nucleus of the hypothalamus involves two independent descending pronociceptive pathways. PLoS One 10(11):e0142919.
- Antunes GF, Pinheiro Campos AC, Varin de Assis D, Gouveia FV, Dias de Jesus Seno M, Pagano RL, Ruiz Martinez RC (2022) Habenula activation patterns in a preclinical model of neuropathic pain accompanied by depressive-like behaviour. PloS One 17(7):e0271295.
- Apkarian AV, Bushnell MC, Treede R-D, Zubieta J-K (2005) Human brain mechanisms of pain perception and regulation in health and disease. Eur J Pain 9(4):463-484.
- Arendt-Nielsen L, Graven-Nielsen T (2009) Muscle pain, including fibromyalgia. In: Binder MD, Hirokawa N, Windhorst U (eds) Encyclopedia of neuroscience. Springer-Verlag, Berlin Heidelberg, pp 2505-2508.
- Arnsten AFT (2009) Stress signalling pathways that impair prefrontal cortex structure and function. Nat Rev Neurosci 10(6):410-422.
- Arnsten AFT, Joyce MKP, Roberts AC (2023) The aversive lens: Stress effects on the prefrontal-cingulate cortical pathways that regulate emotion. Neurosci Biobehav Rev 145:105000. [CrossRef]
- Aston-Jones G, Bloom FE (1981) Activity of norepinephrine-containing locus coeruleus neurons in behaving rats anticipates fluctuations in the sleep-waking cycle. J Neurosci 1(8):876-886. [CrossRef]
- Aston-Jones G, Waterhouse B (2016) Locus coeruleus: from global projection system to adaptive regulation of behavior. Brain Res 1645:75-78.
- Bajic D, Commons KG (2010) Visualizing acute pain-morphine interaction in descending monoamine nuclei with Fos. Brain Res 1306:29-38.
- Baker PM, Mathis V, Lecourtier L, Simmons SC, Nugent FS, Hill S, Mizumori SJY (2022) Lateral habenula beyond avoidance: Roles in stress, memory, and decision-making with implications for psychiatric disorders. Front Syst Neurosci 16:826475.
- Baker PM, Mizumori SJY (2017) Control of behavioral flexibility by the lateral habenula. Pharmacol Biochem Behav 162:62-68.
- Bali A, Singh N, Singh Jaggi A (2014) Neuropeptides as therapeutical targets to combat stress-associated behavioral and neuroendocrinoloical effects. CNS&Neurological Disorders – Drug Targets 13:347-368.
- Baliki MN, Apkarian AV (2015) Nociception, pain, negative moods, and behavior selection. Neuron 87(3):474-491.
- Bandler R, Shipley MT (1994) Columnar organization in the midbrain periaqueductal gray: modules for emotional expression? Trends Neurosci 7(9):379-389.
- Banks PJM, Peper CE, Marinus J, Beek PJ, van Hilten JJ (2013) Motor consequences of experimentally induced limb pain: a systematic review. Eur J Pain 17(2):145-157.
- Bannister K, Dickenson AH (2016) What do monoamines do in pain modulation? Curr Opin Support Palliat Care 10:143-148.
- Barceló AC, Filippini B, Pazo JH (2012) The striatum and pain modulation. Cell Mol Neurobiol 32(1):1-12.
- Bari BA, Chokshi V, Schmidt K (2019) Locus coeruleus-norepinephrine: basic functions and insights into Parkinson’s disease. Neural Regen Res 15(6):1006–1013.
- Bartfai T (2001) Telling the brain about pain. Nature 410:425-427.
- Basbaum AI, Bautista DM, Scherrer G, Julius D (2009) Cellular and molecular mechanisms of pain. Cell 139:267-284.
- Baumann O, Borra RJ, Bower JM, Cullen KE, Habas C, Ivry RB, Leggio M, Mattingley JB, Molinari M, Moulton EA, Paulin MG, Pavlova MA, Schmahmann JD, Sokolov AA (2015) Consensus paper: the role of the cerebellum in perceptual processes. Cerebellum 14(2):197-220.
- Becerra L, Navratilova E, Porreca F, Borsook D (2013) Analogous responses in the nucleus accumbens and cingulate cortex to pain onset (aversion) and offset (relief) in rats and humans. J Neurophysiol 110:1221-1226. [CrossRef]
- Belforte JE, Pazo JH (2005) Striatal inhibition of nociceptive responses evoked in trigeminal sensory neurons by tooth pulp stimulation. J Neurophysiol 93(3):1730-1741. [CrossRef]
- Benarroch EE (2006) Pain-autonomic interactions. Neurol Sci 27 Suppl 2:S130-123.
- Benarroch EE (2020) Physiology and pathophysiology of the autonomic nervous system. Continuum 26(1):12-24.
- Benke D (2022) GABAB receptors and pain. Curr Top Behav Neurosci 52:213-239.
- Berger JV, Knaepen L, Janssen SPM, Jaken RJP, Marcus MAE, Joosten EAJ, Deumens R (2011) Cellular and molecular insights into neuropathy-induced pain hypersensitivity for mechanism-based treatment approaches. Brain Res Rev 67:282-310. [CrossRef]
- Berridge CW, Waterhouse BD (2003) The locus coeruleus-NA system: modulation of behavioral state and state-dependent cognitive processes. Brain Res Brain Res Rev 42(1):33-84.
- Bielefeldt K (2009) Visceral pain. In: Binder MD, Hirokawa N, Windhorst U (eds) Encyclopedia of neuroscience. Springer-Verlag, Berlin Heidelberg, pp 4262-4273.
- Binshtok AM, Wang H, Zimmermann K, Amaya F, Vardeh D, Shi L, Brenner GJ, Ji RR, Bean BP, Woolf CJ, Samad TA (2008) Nociceptors are interleukin-1beta sensors. J Neurosci 28:14062-14073.
- Björklund M, Radovanovic S, Ljubisavljevic M, Windhorst U, Johansson H (2004) Muscle stretch-induced modulation of noxiously activated dorsal horn neurons of feline spinal cord. Neurosci Res 2004 Feb;48(2):175-184. [CrossRef]
- Boadas-Vaello P, Homs J, Reina F, Carrera A, Verdú E (2017) Neuroplasticity of supraspinal structures associated with pathological pain. Anat Rec 300:1481-1501.
- Boccella S, Marabese I, Guida F, Luongo L, Maione S, Palazzo E (2020) The modulation of pain by metabotropic glutamate receptors 7 and 8 in the dorsal striatum. Curr Neuropharmacol 18(1):34-50.
- Bokiniec P, Zampieri N, Lewin GR, Poulet JFA (2018) The neural circuits of thermal perception. Curr Opin Neurobiol 52:98-106.
- Borsook D (2012) Neurological diseases and pain. Brain 135:320-344.
- Boscan P, Pickering AE, Paton JFR (2002) Nucleus of the solitary tract: an integrating station for nociceptive and cardiorespiratory afferents. Exp Physiol 87(2):259-266.
- Bostan AC, Strick PL (2018) The basal ganglia and the cerebellum nodes in an integrated network. Nat Rev Neurosci 19:338-350.
- Bouchet CA, Ingram SL (2020) Cannabinoids in the descending pain modulatory circuit: Role in inflammation. Pharmacol Ther 209:107495.
- Bouhassira D, Bing Z, Le Bars D (1990) Studies of the brain structures involved in diffuse noxious inhibitory controls: the mesencephalon. J Neurophysiol 64(6):1712-1723.
- Boulos L-J, Darcq E, Kieffer BL (2017) Translating the habenula -- from rodents to humans. Biol Psychiatry 81(4):296-305.
- Bowker RM, Reddy VK, Fung SJ, Chan JY, Barnes CD (1987) Serotonergic and non-serotonergic raphe neurons projecting to the feline lumbar and cervical spinal cord: a quantitative horseradish peroxidase-immunocytochemical study. Neurosci Lett 1987 75(1):31-37. [CrossRef]
- Braz J, Solorzano C, Wang X, Basbaum AI (2014) Transmitting pain and itch messages: a contemporary view of the spinal cord circuits that generate gate control. Neuron 82(3):522-536. [CrossRef]
- Brockway DF, Crowley NA (2020) Turning the tides on neuropsychiatric diseases: The role of peptides in the prefrontal cortex. Front Behav Neurosci 14:588400.
- Brodal A (1981) Neurological anatomy. In relation to clinical medicine, 3rd edn. Oxford University Press, New York.
- Brodal P (2017) A neurobiologist´s attempt to understand persistent pain. Scand J Pain 15:140-147.
- Browne TJ, Hughes DI, Dayas CV, Callister RJ, Graham BA (2020) Projection neuron axon collaterals in the dorsal horn: placing a new player in spinal cord pain proccessing. Front Physiol 11:560802. [CrossRef]
- Bruinstroop E, Cano G, Vanderhorst VGJM, Cavalcante JC, Wirth J, Sena-Esteves M, Saper CB (2012) Spinal projections of the A5, A6 (locus coeruleus), and A7 noradrenergic cell groups in rats. J Comp Neurol 520(9):1985-2001.
- Bushnell MC, Čeko M, Low LA (2013) Cognitive and emotional control of pain and its disruption in chronic pain. Nat Rev Neurosci 14:502-511.
- Butler RK, Finn DP (2009) Stress-induced analgesia. Prog Neurobiol 88:184-202.
- Cao L, DeLeo JA (2009) Immune system and pain. In: Binder MD, Hirokawa N, Windhorst U (Eds) Encyclopedia of neuroscience. Springer-Verlag, Berlin Heidelberg, pp 1919-1922.
- Caputi FF, Rullo L, Stamatakos S, Candeletti S, Romualdi P (2019) Modulation of the negative affective dimension of pain: focus on selected neuropeptidergic system contributions. Int J Mol Sci 20, 4010; [CrossRef]
- Carlton SM (2014) Nociceptive primary afferents: they have a mind of their own. J Physiol (Lond) 592:3403-3411.
- Cathenaut L, Schlichter R, Hugel S (2023) Short-term plasticity in the spinal nociceptive system. Pain 164(11):2411-2424.
- Cevikbas F, Lerner EA (2020) Physiology and pathophysiology of itch. Physiol Rev 100(3):945-982.
- Charles K-A, Molpeceres Sierra E, Bouali-Benazzouz R, Tibar H, Oudaha K, Naudet F, Duveau A, Fossat P, Benazzouz A (2025) Interplay between subthalamic nucleus and spinal cord controls parkinsonian nociceptive disorders. Brain 148(1):313-330. [CrossRef]
- Chen LM (2018) Cortical representation of pain and touch: evidence from combined functional neuroimaging and electrophysiology in non-human primates. Neurosci Bull Neurosci Bull 34(1):165-177. [CrossRef]
- Chen Q, Roeder Z, Li M-H, Zhang Y, Ingram SL, Heinricher MM (2017) Optogenetic evidence for a direct circuit linking nociceptive transmission through the parabrachial complex with pain-modulating neurons of the rostral ventromedial medulla (RVM). eNeuro 4(3):ENEURO.0202-17.2017. [CrossRef]
- Chen T, Koga K, Descalzi G, Qiu S, Wang J, Zhang L-S, Zhang Z-J, He X-B, Qin X, Xu F-Q, Hu J, Wei F, Huganir RL, Li Y-Q, Zhuo M (2014) Postsynaptic potentiation of corticospinal projecting neurons in the anterior cingulate cortex after nerve injury. Mol Pain 10:33. [CrossRef]
- Chen X, Tang S-J (2024) Neural circuitry polarization in the spinal dorsal horn (SDH): A novel form of dysregulated circuitry plasticity during pain pathogenesis. Cells 13(5):398.
- Chen Y, Song Y, Wang H, Zhang Y, Hu X, Wang K, Lu Y, Zhang Z, Li S, Li A, Bao L, Xu F, Li C, Zhang X (2022) Distinct neural networks derived from galanin-containing nociceptors and neurotensin-expressing pruriceptors. Proc Natl Acad Sci USA 119(33):e2118501119. [CrossRef]
- Chen Q-Y, Zhuo M (2023) Glutamate acts as a key neurotransmitter for itch in the mammalian spinal cord. Mol Pain 19:17448069231152101.
- Chiang C, Aston-Jones G (1993) Response of locus coeruleus neurons to foot shock stimulation is mediated by neurons in the rostral ventral medulla. Neuroscience 53(3):705-715.
- Chiang MC, Bowen A, Schier LA, Tupone D, Uddin O, Heinricher MM (2019) Parabrachial complex: A hub for pain and aversion. J Neurosci 39(42):8225–8230.
- Chiang MC, Nguyen EK, Canto-Bustos M, Papale AE, Oswald A-MM, Ross SE (2020) Divergent neural pathways emanating from the lateral parabrachial nucleus mediate distinct components of the pain response. Neuron 106(6):927-939.e5. [CrossRef]
- Chudler EH, Dong WK (1995) The role of the basal ganglia in nociception and pain. Pain 60(1):3-38.
- Chudler EH, Sugiyama K, Dong WK (1993) Nociceptive responses in the neostriatum and globus pallidus of the anesthetized rat. J Neurophysiol 69(6):1890-1903.
- Cid-Jofré V, Moreno M, Reyes-Parada M, Renard GM (2021) Role of oxytocin and vasopressin in neuropsychiatric disorders: therapeutic potential of agonists and antagonists. Int J Mol Sci 22(21):12077. [CrossRef]
- Clarke RW, Harris J (2004) The organization of motor responses to noxious stimuli. Brain Res Rev 46:163-172.
- Cobos A, Lima D, Almeida A, Tavares I (2003) Brain afferents to the lateral caudal ventrolateral medulla: a retrograde and anterograde tracing study in the rat. Neuroscience 120(2):485-498. [CrossRef]
- Coghill RC (2020) The distributed nociceptive system: A framework for understanding pain. Trends Neurosci 43(10):780-794.
- Coghill RC, McHaffie JG, Yen Y-F (2003) Neural correlates of interindividual differences in the subjective experience of pain. Proc Natl Acad Sci USA 100:8538-8542.
- Coizet V, Dommett EJ, Klop EM, Redgrave P, Overton PG (2010) The parabrachial nucleus is a critical link in the transmission of short latency nociceptive information to midbrain dopaminergic neurons. Neuroscience 2168(1):263-272. [CrossRef]
- Colloca L, Ludman T, Bouhassira D, Baron R, Dickenson AH, Yarnitsky D, Freeman R, Truini A, Attal N, Finnerup NB, Eccleston C, Kalso E, Bennett DL, Dworkin RH, Raja SN (2017) Neuropathic pain. Nat Rev Dis Primers 3:17002.
- Comitato A, Bardoni R (2021) Presynaptic inhibition of pain and touch in the spinal cord: From Receptors to Circuits. Int J Mol Sci 22(1):414.
- Cordero-Erausquin M, Inquimbert P, Schlichter R, Hugel S (2016) Neuronal networks and nociceptive processing in the dorsal horn of the spinal cord. Neuroscience 338:230-247.
- Cortes-Altamirano JL, Olmos-Hernández A, Jaime HB, Carrillo-Mora P, Bandala C, Reyes-Long S, Alfaro-Rodriguez A (2018) Review: 5-HT1, 5-HT2, 5-HT3 and 5-HT7 receptors and their role in the modulation of pain response in the central nervous system. Curr Neuropharmacol 16(2):210–221. [CrossRef]
- Condés-Lara M, Martínez-Lorenzana G, Espinosa de Los Monteros-Zúñiga A, López-Córdoba G, Córdova-Quiroga A, Flores-Bojórquez SA, González-Hernández A (2024) Hypothalamic paraventricular stimulation inhibits nociceptive wide dynamic range trigeminocervical complex cells via oxytocinergic transmission. .J Neurosci 44(17):e1501232024.
- Costigan M, Scholz J, Woolf CJ (2009) Neuropathic pain: a maladaptive response of the nervous system to damage. Annu Rev Neurosci 32:1-32.
- Craig AD (2002) How do we feel? Interoception: the sense of the physiological condition of the body. Nat Rev Neurosci 3:655-666.
- Craig AD (2003) Pain mechanisms: labeled lines versus convergence in central processing. Annu Rev Neurosci 26:1-30.
- Cui C-X, Liu H-Y, Yue N, Du Y-R, Che L-M, Yu J-S (2023) Research progress on the mechanism of chronic neuropathic pain. IBRO Neurosci Rep 14:80-85.
- Curatolo M (2023) Is central sensitization relevant in acute low back pain? Eur J Pain 27(4):457-458.
- Dai D, Li W, Chen A, Gao X-F, Xiong L (2022) Lateral habenula and its potential roles in pain and related behaviors. ACS Chem Neurosci 13(8):1108-1118.
- D’Amico F, Kelleher E, D’Andria Ursoleo J, Yavorovskiy AG, Turi S, Zaffaroni S, Agosta VT, Ajello S, Landoni G (2025) The effect of Neurokinin-1 receptor antagonists on postoperative pain: A meta-analysis of randomized controlled trials. J Clin Anesth 102:111772.
- Da Silva JT, Seminowicz DA (2019) Neuroimaging of pain in animal models: a review of recent literature. Pain Rep 4(4): e732.
- Davis OC, Dickie AC, Mustapa MB, Boyle KA, Browne TJ, Gradwell MA, Smith KM, Polgár E, Bell AM, Kókai É, Watanabe M, Wildner H, Zeilhofer HU, Ginty DD, Callister RJ, Graham BA, Todd AJ, Hughes DI (2023) Calretinin-expressing islet cells are a source of pre- and post-synaptic inhibition of non-peptidergic nociceptor input to the mouse spinal cord. Sci Rep 13:11561. [CrossRef]
- De Felice M, Ossipov MH (2016) Cortical and subcortical modulation of pain. Pain Manag 6(2):111-120.
- Delbono O, Wang Z-M, Messi ML (2022) Brainstem noradrenergic neurons: Identifying a hub at the intersection of cognition, motility, and skeletal muscle regulation. Acta Physiol (Oxf) 236(3):e13887.
- DeLong MR, Wichmann T (2007) Circuits and circuit disorders of the basal ganglia. Arch Neurol 64:20-24.
- De Preter CC, Heinricher MM (2024) The `ìn´s´ and `out´s´ of descending pain modulation from the rostral ventromedial medulla. Trends Neurosci 47:447-460.
- Dibaj P, Safavi-Abbasi E, Asadollahi E (2024) In vivo spectrally unmixed multi-photon imaging of longitudinal axon-glia changes in injured spinal white matter. Neurosci Lett 841:137959. [CrossRef]
- Dibaj P, Windhorst U (2024a) Motor-control notions in health and disease (what controls motor control). Preprints. [CrossRef]
- Dibaj P, Windhorst U (2024b) Muscle fatigue in health and disease. Preprints. [CrossRef]
- Dib-Hajj SD, Waxman SG (2019) Sodium channels in human pain disorders: Genetics and pharmacogenomics. Annu Rev Neurosci 42:87-106.
- Djouhri L, Lawson SN (2004) A-fiber nociceptive primary afferent neurons: a review of incidence and properties in relation to other afferent A-fiber neurons in mammals. Brain Res Rev 46:131-145. [CrossRef]
- D´Mello R, Dickenson AH (2008) Spinal cord mechanisms of pain. Br J Anaesth 101:8-16.
- Doan L, Manders T, Wang J (2015) Neuroplasticity underlying the comorbidity of pain and depression. Neural Plast 2015:504691.
- Dostrovsky JO (2000) Role of thalamus in pain. Prog Brain Res 129:245-257.
- Dou Z, Su N, Zhou Z, Mi A, Xu L, Zhou J, Sun S, Liu Y, Hao M, Li Z (2023) Modulation of visceral pain by brain nuclei and brain circuits and the role of acupuncture: a narrative review. Front Neurosci 17:1243232. [CrossRef]
- Drissi I, Woods WA, Woods CG (2020) Understanding the genetic basis of congenital insensitivity to pain. Br Med Bull 133(1):65-78.
- Du H-J, Zhou S-Y (1990) Involvement of solitary tract nucleus in control of nociceptive transmission in cat spinal cord neurons. Pain 40(3):323-331.
- Duan B, Cheng L, Ma Q (2017) Spinal circuits transmitting mechanical pain and itch. Neurosci Bull 34(1):186-193.
- Dubin AE, Patapoutian A (2010) Nociceptors: the sensors of the pain pathway. J Clin Invest 120:3760-3772.
- Dum RP, Levinthal DJ, Strick PL (2016) Motor, cognitive, and affective areas of the cerebral cortex influence the adrenal medulla. Proc Natl Acad Sci U S A 113(35):9922-9927. [CrossRef]
- Edvinsson L (2001) Sensory nerves in man and their role in primary headaches. Cephalalgia 21(7):761-764.
- Ekman EF, Koman LA (2005) Acute pain following musculoskeletal injuries and orthopaedic surgery: machanisms and management. Instr Course Lect 54:21-33.
- Eliava M, Melchior M, Knobloch-Bollmann HS, Wahis J, da Silva Gouveia M, Tang Y, Ciobanu AC, Triana del Rio R, Roth LC, Althammer F, Chavant V, Goumon Y, Gruber T, Petit-Demoulière N, Busnelli M, Chini B, Tan LL, Mitre M, Froemke RC, Chao MV, Giese G, Sprengel R, Kuner R, Poisbeau P, Seeburg PH, Stoop R, Charlet A, Grinevich V (2016) A new population of parvocellular oxytocin neurons controlling magnocellular neuron activity and inflammatory pain processing. Neuron 89(6):1291-1304. [CrossRef]
- Emery EC, Wood JN (2019) Somatosensation a la mode: plasticity and polymodality in sensory neurons. Curr Opin Physiol 11:29-34.
- Evrard HC (2019) The organization of the primate insular cortex. Front Neuroanat13:43.
- Fasick V, Spengler RN, Samankan S, Nader ND, Ignatowski TA (2015) The hippocampus and TNF: Common links between chronic pain and depression. Neurosci Biobehav Rev 53:139-159.
- Fillingim RB (2017) Individual differences in pain: Understanding the mosaic that makes pain personal. Pain 158(Suppl 1):S11-S18.
- Finnerup NB, Kuner R, Jensen TS (2021) Neuropathic pain: from mechanisms to treatment. Physiol Rev 101(1):259-301.
- Fong H, Zheng J, Kurrasch D (2023) The structural and functional complexity of the integrative hypothalamus. Science 382(6669):388-394.
- Foo H, Mason P (2003a) Discharge of raphe magnus ON and OFF cells is predictive of the motor facilitation evoked by repeated laser stimulation. J Neurosci 23(5):1933-1940. [CrossRef]
- Foo H, Mason P (2003b) Brainstem modulation of pain during sleep and waking.Sleep Med Rev 7(2):145-154.
- Fore S, Palumbo F, Pelgrims R, Yaksi E (2018) Information processing in the vertebrate habenula. Semin Cell Dev Biol 8:130-139.
- Foote SL, Bloom FE, Aston-Jones G (1983) Nucleus locus ceruleus: new evidence of anatomical and physiological specificity. Physiol Rev 63(3):844-914.
- Franҫois A, Low SA, Sypek EI, Christensen AJ, Sotoudeh C, Beier KT, Ramakrishnan C, Ritola KD, Sharif-Naeini R, Deisseroth K, Delp SL, Malenka RC, Luo L, Hantman AW, Scherrer G (2017) Brainstem-spinal cord inhibitory circuit for mechanical pain modulation by GABA and enkephalins. Neuron 93(4):822-839.e6. [CrossRef]
- Frias B, Merighi A (2016) Capsaicin, nociception and pain. Molecules 21(6):797.
- Gamal-Eltrabily M, Espinosa de Los Monteros-Zúñiga A, Manzano-García A, Martínez-Lorenzana G, Condés-Lara M, González-Hernández A (2020) The rostral agranular insular cortex, a Nnw site of oxytocin to induce antinociception. J Neurosci 40(29):5669-5680.
- Gamal-Eltrabily M, Márquez-Morales C, Martínez-Lorenzana G, González-Hernández A, Condés-Lara M (2018) Peptidergic nature of nociception-related projections from the hypothalamic paraventricular nucleus to the dorsal horn of the spinal cord. Neurosci Lett 685:124-130.
- Gamal-Eltrabily M, Márquez-Morales C, Martínez-Lorenzana G, González-Hernández A, Condés-Lara M (2021) Cortical modulation of nociception. Neuroscience 15:458:256-270.
- Gamboa-Esteves FO, McWilliams PN, Batten TFC (2004) Substance P (NK1) and somatostatin (sst2A) receptor immunoreactivity in NTS-projecting rat dorsal horn neurones activated by nociceptive afferent input. J Chem Neuroanat 27(4):251-266. [CrossRef]
- Gamboa-Esteves FO, Tavares I, Almeida A, Batten TF, McWilliam PN, Lima D (2001) Projection sites of superficial and deep spinal dorsal horn cells in the nucleus tractus solitarii of the rat. Brain Res 921(1-2):195-205. [CrossRef]
- Gan Z, Gangadharan V, Liu S, Körber C, Tan LL, Li H, Oswald MJ, Kang J, Martin-Cortecero J, Männich D, Groh A, Kuner T, Wieland S, Kuner R (2022) Layer-specific pain relief pathways originating from primary motor cortex. Science 378(6626):1336-1343. [CrossRef]
- Gao DM, Hoffman D, Benabid AL (1996) Simultaneous recording of spontaneous activities and nociceptive responses from neurons in the pars compacta of substantia nigra and in the lateral habenula. Eur J Neurosci 8(7):1474-1478. [CrossRef]
- Gao DM, Jeaugey L, Pollak P, Benabid AL (1990) Intensity-dependent nociceptive responses from presumed dopaminergic neurons of the substantia nigra, pars compacta in the rat and their modification by lateral habenula inputs. Brain Res 529(1-2):315-319.
- Garcia AD, Buffalo EA (2020) Anatomy and function of the primate entorhinal cortex. Annu Rev Vis Sci 6:411-432.
- García-Boll E, Martínez-Lorenzana G, Condés-Lara M, González-Herández A (2018) Oxytocin inhibits the rat medullary dorsal horn Sp5c/C1 nociceptive transmission through OT but not V1A receptors. Neuropharmacology 129:109-117.
- Gasparini S, Howland JM, Thatcher AJ, Geering JC (2020) Central afferents to the nucleus of the solitary tract in rats and mice. J Comp Neurol 528(16):2708-2728.
- Gatto G, Smith KM, Ross SE, Goulding M (2019) Neuronal diversity in the somatosensory system: bridging the gap between cell type and function. Curr Opin Neurobiol 56:167-174. [CrossRef]
- Gear RW, Levine JD (2011) Nucleus accumbens facilitates nociception. Exp Neurol 229(2):502-506.
- Gebhart GF (2004) Descending modulation of pain. Neurosci Biobehav Rev 27:729-737.
- Gebhart GF (2009) Pain. In: Binder MD, Hirokawa N, Windhorst U (eds) Encyclopedia of neuroscience. Springer-Verlag, Berlin Heidelberg, pp 3063-3065.
- Gelnar PA, Krauss BR, Sheehe PR, Szeverenyi NM , Apkarian AV (1999) A comparative fMRI study of cortical representations for thermal painful, vibrotactile, and motor performance tasks. Neuroimage 10(4):460-482.
- Geuter S, Koban L, Wager TD (2017) The cognitive neuroscience of placebo effects: Concepts, predictions, and physiology. Annu Rev Neurosci 40:167-188.
- Godínez-Chaparro B, Martínez-Lorenzana G, Rodríguez-Jiménez R, Manzano-García A, Rojas-Piloni G, Condés-Lara M, González-Hernández A (2016) The potential role of serotonergic mechanisms in the spinal oxytocin-induced antinociception. Neuropeptides 60:51-60.
- Goodwin G, McMahon SB (2021) The physiological function of different voltage-gated sodium channels in pain. Nat Rev Neurosci 22:263–274.
- Grafe LA, Bhatnagar S (2018) Orexins and stress. Front Neuroendocrinol 51:132-145.
- Graham BA, Brichta AM, Callister RJ (2007) Moving from an averaged to specific view of spinal cord pain processing circuits. J Neurophysiol 98:1057-1063.
- Graven-Nielsen T, Mense S (2001) The peripheral apparatus of muscle pain: evidence from animal and human studies. Clin J Pain 2001 Mar;17(1):2-10.
- Gregory NS, Gautam M, Benson CJ, Sluka KA (2018) Acid sensing ion channel 1a (ASIC1a) mediates activity-induced pain by modulation of heteromeric ASIC channel kinetics. Neuroscience 386:166-174. [CrossRef]
- Grillner S, Robertson B (2016) The basal ganglia over 500 million years. Curr Biol 226(20):R1088-R1100.
- Groenewegen HJ (2003) The basal ganglia and motor control. Neural Plast 10:107-120.
- Gouveia FV, Baker PM, Mameli M, Germann J (2022) Editorial: The habenula and its role in neuropsychiatric symptoms. Front Behav Neurosci 16:929507.
- Gouveia FV, Ibrahim GM (2022) Habenula as a neural substrate for aggressive behavior. Front Psychiatry 13: 817302.
- Gu X, Zhang YZ, O´Malley JJ, De Preter CC, Penzo M, Hoon MA (2023) Neurons in the caudal ventrolateral medulla mediate descending pain control. Nat Neurosci 26(4):594-605.
- Gwak YS, Claire E. Hulsebosch CE, Joong Woo Leem JW (2017) Neuronal-glial interactions maintain chronic neuropathic pain after spinal cord Injury. Neural Plastic Volume 2017, Article ID 2480689. [CrossRef]
- Haber SN (2016) Corticostriatal circuitry. Dialogues Clin Neurosci 18(1):7-21.
- Häring M, Zeisel A, Hochgerner H, Rinwa1 P, Jakobsson JET, Lönnerberg P, La Manno G, Sharma1 N, Borgius L, Kiehn O, Lagerström MC, Linnarsson S, Ernfors P (2018) Neuronal atlas of the dorsal horn defines its architecture and links sensory input to transcriptional cell types. Nat Neurosci 21(6):869-880. [CrossRef]
- Hamani C, Saint-Cyr JA, Fraser J, Kaplitt M, Lozano AM (2004) The subthalamic nucleus in the context of movement disorders. Brain 127(Pt 1):4-20.
- Hammond C, Shibazaki T, Rouzaire-Dubois B (1983) Branched output neurons of the rat subthalamic nucleus: electrophysiological study of the synaptic effects on identified cells in the two main target nuclei, the entopeduncular nucleus and the substantia nigra. Neuroscience 9(3):511-520.
- Han Y, Yuan K, Zheng Y, Lu L (2020) Orexin receptor antagonists as emerging treatments for psychiatric disorders. Neurosci Bull 36(4):432–448.
- Haque T, Akhter F, Kato T, Sato F, Takeda R, Higashiyama K, Moritani M, Bae Y-C, Sessle BJ, Yoshida A (2012) Somatotopic direct projections from orofacial areas of secondary somatosensory cortex to trigeminal sensory nuclear complex in rats. Neuroscience 219:214-233. [CrossRef]
- Harding EK, Fung SW, Bonin RP (2020) Insights into spinal dorsal horn circuit function and dysfunction using optical approaches. Front Neural Circuits 14:31. [CrossRef]
- Harris HN, Peng YB (2020) Evidence and explanation for the involvement of the nucleus accumbens in pain processing. Neural Regen Res 15:597-605.
- Hawkins JL, Moore NJ, Miley D, Durham PL (2018) Secondary traumatic stress increases expression of proteins implicated in peripheral and central sensitization of trigeminal neurons. Brain Res 1687:162-172. [CrossRef]
- Heinricher MM, Cheng ZF, Fields HL (1987) Evidence for two classes of nociceptive modulating neurons in the periaqueductal gray. J Neurosci 7(1):271-278.
- Heinricher MM, Tavares I, Leith JL, Lumb BM (2009) Descending control of nociception: Specificity, recruitment and plasticity. Brain Res Rev 60:214-225.
- Helmer LML,Weijenberg RAF, de Vries R, Achterberg WP, Lautenbacher S, Sampson EL, Lobbezoo F (2020) Crying out in pain-A systematic review into the validity of vocalization as an indicator for pain. Eur J Pain 24(9):1703-1715.
- Henderson LA, Keay KA (2018) Imaging acute and chronic pain in the human brainstem and spinal cord. Neuroscientist 24(1):84-96.
- Herrero M-T, Barcia C, Navarro JM (2002) Functional anatomy of thalamus and basal ganglia. Childs Nerv Syst 18(8):386-404.
- Hikosaka O (2010) The habenula: from stress evasion to value-based decision-making. Nat Rev Neurosci 11(7):503-153.
- Hoegh M (2022) Pain Science in practice (part 3): peripheral sensitization. J Orthop Sports Phys Ther 52(6):303-306.
- Hökfelt T, Barde S, Xu Z-QD, Kuteeva E, Rüegg J, Le Maitre E, Risling M, Kehr J, Ihnatko R, Theodorsson E, Palkovits M, Deakin W, Bagdy G, Juhasz G, Prud´homme HJ, Mechawar N, Diaz-Heijtz R, Ögren SO (2018) Neuropeptide and small transmitter coexistence: Fundamental studies and relevance to mental illness. Front Neural Circuits 12:106.
- Holden JE, Naleway E (2001) Microinjection of carbachol in the lateral hypothalamus produces opposing actions on nociception mediated by alpha(1)- and alpha(2)-adrenoceptors. Brain Res 911(1):27-36. [CrossRef]
- Holden JE, Naleway Farah E, Jeong Y (2005) Stimulation of the lateral hypothalamus produces antinociception mediated by 5-HT1A, 5-HT1B and 5-HT3 receptors in the rat spinal cord dorsal horn. Neuroscience 135(4):1255-1268.
- Holt MK (2022) The ins and outs of the caudal nucleus of the solitary tract: An overview of cellular populations and anatomical connections. J Neuroendocrinol 34(6):e13132. [CrossRef]
- Holt MK, Rinaman L (2022) The role of nucleus of the solitary tract glucagon-like peptide-1 and prolactin-releasing peptide neurons in stress: anatomy, physiology and cellular interactions. Br J Pharmacol 179(4): 642–658.
- Hu H, Cui Y, Yang Y (2020) Circuits and functions of the lateral habenula in health and in disease. Nat Rev Neurosci 21(5):277-295.
- Hucho T, Levine JD (2007) Signaling pathways in sensitization: toward a nociceptor cell biology. Neuron 55:365-376.
- Hultborn H, Illert M, Santini M (1976) Convergence on interneurones mediating the reciprocal Ia inhibition of motoneurones. II. Effects from segmental flexor reflex pathways. Acta Physiol Scand 96(3):351-367.
- Huma Z, Ireland K, Maxwell DL (2015) The spino-bulbar-cerebellar pathway: Activation of neurons projecting to the lateral reticular nucleus in the rat in response to noxious mechanical stimuli. Neurosci Lett 591:197-201. [CrossRef]
- Humphries MD, Prescott TJ (2010) The ventral basal ganglia, a selection mechanism at the crossroads of space, strategy, and reward. Prog Neurobiol 90:385-417. [CrossRef]
- Hunt SP, Mantyh PW (2001) The molecular dynamics of pain control. Nat Rev Neurosci 2:83-91.
- Iovino M, Giagulli VA, Licchelli B, Iovino E, Guastamacchia E, Triggiani V (2016) Synaptic inputs of neural afferent pathways to vasopressin- and oxytocin-secreting neurons of supraoptic and paraventricular hypothalamic nuclei. Endocr Metab Immune Disord Drug Targets 16(4):276-287. [CrossRef]
- Jacobson LH, Hoyer D, de Lecea L (2022) Hypocretins (orexins): The ultimate translational neuropeptides. J Intern Med 291(5):533-556.
- Jänig W (2014) [Neurobiology of visceral pain] Schmerz 28(3):233-251 (in German).
- Jean A (1991) The nucleus tractus solitarius: neuroanatomic, neurochemical and functional aspects. Arch Int Physiol Biochim Biophys 99(5):A3-52 (in French).
- Jennings EM, Okine BN, Roche M, Finn DP (2014) Stress-induced hyperalgesia. Prog Neurobiol 121:1-18.
- Jensen TS, Finnerup NB (2009) Central pain. In: Binder MD, Hirokawa N, Windhorst U (eds) Encyclopedia of neuroscience. Springer-Verlag, Berlin Heidelberg, pp 647-650.
- Ji R-R, Donnelly CR, Nedergaard M (2019) Astrocytes in chronic pain and itch. 20:667-685.
- Ji Y, Onwukwe C, Smith J, Laub H, Posa L, Keller A, Masri R, Cramer N (2023) Noradrenergic input from nucleus of the solitary tract regulates parabrachial activity in mice. eNeuro 10(5):ENEURO.0412-22.2023. [CrossRef]
- Jia T, Wang Y-D, Chen J, Zhang X, Cao J-L, Xiao C, Zhou C (2022) A nigro-subthalamo-parabrachial pathway modulates pain-like behaviors. Nat Commun 13(1):7756.
- Johansson H, Djupsjöbacka M, Sjölander P (1993) Influences on the gamma-muscle spindle system from muscle afferents stimulated by KCl and lactic acid. Neurosci Res 6(1):49-57.
- Jiang M, Sun Y, Lei Y, Hu F, Xia Z, Liu Y, Ma Z, Gu X (2020) GPR30 receptor promotes anxiety-induced postoperative hyperalgesia by upregulating GABAA-α4β1δ subunits in periaqueductal gray in female rats. BMC Anesthesiol 20(1):93.
- Jürgens TP, May A (2014) Role of sphenopalatine ganglion stimulation in cluster headache. Curr Pain Headache Rep 18(7):433. [CrossRef]
- Jürgens U (2002) Neural pathways underlying vocal control. Neurosci Biobehav Rev 26(2):235-258.
- Julius D, Basbaum AI (2001) Molecular mechanisms of nociception. Nature 413:203-210.
- Jurek B, Neumann ID (2018) The oxytocin receptor: From intracellular signaling to behavior. Physiol Rev 98(3):1805-1908.
- Kalezic I, Bugaychenko LA, Kostyukov AI, PilyavskiiAI, Ljubisavljevid M, Windhorst U, Johansson H (2004) Fatigue-related depression of the feline monosynaptic gastrocnemius-soleus reflex. J Physiol 556(Pt 1):283-296.
- Kang X, Tang H, Liu Y, Yuan Y, Wang M (2021) Research progress on the mechanism of orexin in pain regulation in different brain regions. Open Life Sci 16(1):46–52. [CrossRef]
- Kanashiro A , Hiroji Hiroki C, Morais da Fonseca D, Birbrair A, Gomes Ferreira R, Shimizu Bassi G, Fonseca MD, Kusuda R, Martelossi Cebinelli GC, Pinho da Silva K, Wagner Wanderley C, Menezes GC, Alves-Fiho JC, Oliveira AG, Cunha TM, Sampaio Pupo A, Ulloa L, Queiroz Cunha F (2020) The role of neutrophils in neuro-immune modulation. Pharmacol Res 151: 104580. [CrossRef]
- Kawai Y (2018) Differential ascending projections from the male rat caudal nucleus of the tractus solitarius: An interface between local microcircuits and global macrocircuits. Front Neuroanat 24:12:63. [CrossRef]
- Kawasaki M, Sakai A, Ueta Y (2024) Pain modulation by oxytocin. Peptides 179:171263.
- Kelly EA, Fudge JL (2018) The neuroanatomic complexity of the CRF and DA systems and their interface: What we still don't know. Neurosci Biobehav Rev 90:247-259.
- Khalilzadeh E, Vafaei Saiah G (2017) The possible mechanisms of analgesia produced by microinjection of morphine into the lateral habenula in the acute model of trigeminal pain in rats. Res Pharm Sci 12(3):241-248.
- Khan A, Khan S, Kim YS (2019) Insight into pain modulation: nociceptors sensitization and therapeutic targets. Curr Drug Targets 20(7):775-788.
- Kim K, Mishina M, Kokubo R, Nakajima T, Moromoto D, Isu T, Kobayashi S, Teramoto A (2013) Ketamine for acute neuropathic pain in patients with spinal cord injury. J Clin Neurosci 20(6):804-807.
- King M, Carnahan H (2019) Revisiting the brain activity associated with innocuous and noxious cold exposure. Neurosci Biobehav Rev 104:197-208.
- Klein MO, Battagello DS, Cardoso AR, Hauser AR, Bittencourt JC, Correa RG (2019) Dopamine: Functions, signaling, and association with neurological diseases. Cell Mol Neurobiol 39(1):31-59.
- Kniffki KD, Schomburg ED, Steffens H (1981) Synaptic effects from chemically activated fine muscle afferents upon alpha-motoneurones in decerebrate and spinal cats. Brain Res 206(2):361-370.
- Kobayashi M, Nakaya Y, Kobayashi S (2024) Functional roles of descending projections from the cerebral cortex to the trigeminal spinal subnucleus caudalis in orofacial nociceptive information processing. J Oral Biosci 66(2):304-307. [CrossRef]
- Koch SC, Acton D, Goulding M (2018) Spinal circuits for touch, pain and itch. Annu Rev Physiol 80:189-217.
- Koshimizu T-a, Tsujimoto G (2009) New topics in vasopressin receptors and approach to novel drugs: vasopressin and pain perception. J Pharmacol Sci 109(1):33-37. [CrossRef]
- Kostyukov AI, Bugaychenko LA, Kalezic I, Pilyavskii AI, Windhorst U, Djupsjöbacka M (2005) Effects in feline gastrocnemius-soleus motoneurones induced by muscle fatigue. Exp Brain Res 163(3):284-294.
- Koutsikou S, Apps R, Lumb BM (2017) Top-down control of spinal sensorimotor circuits essential for survival. J Physiol (Lond) 595(13):4151-4158.
- Kumamoto E (2019) Cellular mechanisms for antinociception produced by oxytocin and orexins in the rat spinal lamina II - comparison with those of other endogenous pain modulators. Pharmaceuticals (Basel)12(3):136. [CrossRef]
- Kummer KK, Mitrić M, Kalpachidou T, Kress M (2020) The medial prefrontal cortex as a central hub for mental comorbidities associated with chronic pain. Int J Mol Sci 21(10):3440. [CrossRef]
- Kuner R, Flor H (2016) Structural plasticity and reorganisation in chronic pain. Nat Rev Neurosci 18:20-30.
- Kuner R, Kuner T (2021) Cellular circuits in the brain and their modulation in acute and chronic pain. Physiol Rev 101(1):213-258.
- Kunz M, Bär K-J, Karmann AJ, Wagner G, Lautenbacher S (2023) Facial expressions of pain: the role of the serotonergic system. Psychopharmacology (Berl) 240(12):2597-2605.
- Kunz M, Meixner D, Lautenbacher S (2019) Facial muscle movements encoding pain-a systematic review. Pain 60(3):535-549.
- Kvetnansky R, Sabban EL, Palkovits M (2009) Catecholaminergic systems in stress: structural and molecular genetic approaches. Physiol Rev 89:535-606.
- Kwiat GC, Basbaum AI (1992) The origin of brainstem noradrenergic and serotonergic projections to the spinal cord dorsal horn in the rat. Somatosensory & motor research 9.2:157-173. [CrossRef]
- Kwiat GC, Liu H, Williamson AM, Basbaum AI (1993) GABAergic regulation of noradrenergic spinal projection neurons of the A5 cell group in the rat: an electron microscopic analysis. J Comp Neurol 330(4):557-570. [CrossRef]
- Labrakakis C (2023) The role of the insular cortex in pain. Int J Mol Sci 24(6):5736.
- Lamotte G, Shouman K, Benarroch EE (2021) Stress and central autonomic network. Auton Neurosci 235:102870.
- Lau BK, Vaughan CW (2014) Descending modulation of pain: the GABA disinhibition hypothesis of analgesia. Curr Opin Neurobiol 29:159-164.
- Laurin J, Pertici V, Doucet E, Marqueste T, Decherchi P (2015) Group III and IV muscle afferents: role on central motor drive and clinical implications. Neuroscience 290:543-551. [CrossRef]
- Lavenex P, Amaral DG (2000) Hippocampal-neocortical interaction: a hierarchy of associativity. Hippocampus 10(4):420-430.
- Lay M, Dong X (2020) Neural mechanisms of itch. Annu Rev Neurosci 43:187-205.
- Lee SM, Jang HB, Fan Y, Lee B, Kim SC, Bills KB, Steffensen SC, Kim HY (2022) Nociceptive stimuli activate the hypothalamus-habenula circuit to inhibit the meso-limbic reward system and cocaine seeking-behaviors. J Neurosci 42(49):9180-9192.
- Lefevre A, Benusiglio D, Tang Y, Krabicher Q, Charlet A, Grinevich V (2021) Oxytocinergic feedback circuitries: An anatomical basis for neuromodulation of social behaviors. Front Neural Circuits 15:688234.
- Legrain V, Iannetti GD, Plaghki L, Mouraux A (2010) The pain matrix reloaded: A salience detection system for the body. Prog Neurobiol 93:111-124.
- Leite-Almeida H, Valle-Fernandes A, Almeida A (2006) Brain projections from the medullary dorsal reticular nucleus: an anterograde and retrograde tracing study in the rat. Neuroscience 140(2):577-595. [CrossRef]
- Leknes S, Tracey I (2008) A common neurobiology for pain and pleasure. Nat Rev Neurosci 9:314-320.
- Lewin GR, Lu Y, Park TJ (2004) A plethora of painful molecules. Curr Opin Neurobiol 14:443-449.
- Li C, Liu S, Lu X, Tao F (2019) Role of descending dopaminergic pathways in pain modulation. Curr Neuropharmacol 17(12):1176–1182.
- Li S, Cao X, Li Y, Tang Y, Cheng S, Zhang D (2024) Enhancing ventrolateral prefrontal cortex activation mitigates social pain and modifies subsequent social attitudes: Insights from TMS and fMRI. Neuroimage 292:120620. [CrossRef]
- Liang D, Labrakakis C (2024) Multiple posterior insula projections to the brainstem descending pain modulatory system. Int J Mol Sci 25(17):9185.
- Lim LE (1994) Psychogenic pain. Singapore Med J 35(5):519-522.
- Lima D, Almeida A (2002) The medullary dorsal reticular nucleus as a pronociceptive centre of the pain control system. Prog Neurobiol 66:81-108. [CrossRef]
- Lindvall O, Björklund A, Skagerberg G (1983) Dopamine-containing neurons in the spinal cord: anatomy and some functional aspects. Ann Neurol 14(3):255-260.
- Lischka A, Lassuthova P, Çakar A, Record CJ, Van Lent J, Baets J, Dohrn MF, Senderek J, Lampert A, Bennett DL, Wood JN, Timmerman V, Hornemann T, Auer-Grumbach M, Parman Y, Hübner CA, Elbracht M, Eggermann K, Woods CG, Cox JJ, Reilly MM, Kurth I (2022) Genetic pain loss disorders. Nat Rev Dis Primers 8(1):41.
- Liu Y, Latremoliere A, Li X, Zhang Z, Chen M, Wang X, Fang C, Zhu J, Alexandre C, Gao Z, Chen B, Ding X, Zhou J-Y, Zhang Y, Chen C, Wang KH, Woolf CJ, He Z (2018) Touch and tactile neuropathic pain sensitivity are set by corticospinal projections. Nature 561(7724):547-550. [CrossRef]
- Livneh Y, Andermann (2021) Cellular activity in insular cortex across seconds to hours: Sensations and predictions of bodily states. Neuron 109(22):3576-3593. [CrossRef]
- Loeser JD, Treede RD (2008) The Kyoto protocol of IASP basic pain terminology. Pain 137:473–477.
- Lu C, Yang T, Zhao H, Zhang M, Meng F, Fu H, Xie Y, Xu H (2016) Insular cortex is critical for the perception, modulation, and chronification of pain. Neurosci Bull 32(2):191-201.
- Lubejko ST, Livrizzi G, Buczynski SA, Patel J, Yung JC, Yaksh TL, Banghart MR (2024) Inputs to the locus coeruleus from the periaqueductal gray and rostroventral medulla shape opioid-mediated descending pain modulation. Sci Adv 10(17):eadj9581. [CrossRef]
- Luiz AP, MacDonald DI, Santana-Varela S, Millet Q, Sikander S, Wood JN, Emery EC (2019) Cold sensing by Nav1.8-positive and Nav1.8-negative sensory neurons. Proc Natl Acad Sci U S A 116(9):3811-3816.
- Lumb BM (2002) Inescapable and escapable pain is represented in distinct hypothalamic-midbrain circuits: specific roles for Adelta- and C-nociceptors. Exp Physiol 87(2):281-286.
- Luo J, Feng J, Liu S, Walters ET, Hu H (2015) Molecular and cellular mechanisms that initiate pain and itch. Cell Mol Life Sci 72:3201-3223.
- Lyubashina OA, Sivachenko IB, Sokolov AY (2019) Differential responses of neurons in the rat caudal ventrolateral medulla to visceral and somatic noxious stimuli and their alterations in colitis. Brain Res Bull 152:299-310. [CrossRef]
- Ma S, Hangya B, Leonard CS, Wisden W, Gundlach AL (2018) Dual-transmitter systems regulating arousal, attention, learning and memory. Neurosci Biobehav Rev 85:21-33.
- Maeda H, Mogenson GJ (1982) Effects of peripheral stimulation on the activity of neurons in the ventral tegmental area, substantia nigra and midbrain reticular formation of rats.Brain Res Bull 8(1):7-14.
- Maisky VA, Pilyavskii AI, Kalezic I, Ljubisavljevic M, Kostyukov AI, Windhorst U, Johansson H (2002) NADPH-diaphorase activity and c-fos expression in medullary neurons after fatiguing stimulation of hindlimb muscles in the rat. Auton Neurosci 101(1-2):1-12. [CrossRef]
- Makovac E, Venezia A, Hohenschurz-Schmidt D, Dipasquale O, Jackson JB, Medina S, O´Daly O, Williams SCR, McMahon SB, Howard MA (2021) The association between pain-induced autonomic reactivity and descending pain control is mediated by the periaqueductal grey. J Physiol 599(23):5243-5260.
- Maletic V, Raison CL (2009). Neurobiology of depression, fibromyalgia and neuropathic pain. Front Biosci (Landmark Ed) 14(14):5291-338.
- Marangoni S, Beatty J, Steagall PV (2023) An ethogram of acute pain behaviors in cats based on expert consensus. PLoS One 18(9):e0292224.
- Martins I, Tavares I (2017) Reticular formation and pain: The past and the future. Front Neuroanat 2017 Jul 5:11:51.
- Maruyama K (2021) Senso-immunology: crosstalk between nociceptive and immune systems. FEBS J. [CrossRef]
- Mason P (2001) Contributions of the medullary raphe and ventromedial reticular region to pain modulation and other homeostatic functions. Annu Rev Neurosci 24:737-777. [CrossRef]
- Mason P (2005) Deconstructing endogenous pain modulation. J Neurophysiol 94:1659-1663.
- Mavani GP, DeVita MV, Michelis MF (2015) A review of the nonpressor and nonantidiuretic actions of the hormone vasopressin. Front Med (Lausanne) 2:19.
- McGrawth PA, Williams T (2009) Pain in children. In: Binder MD, Hirokawa N, Windhorst U (eds) Encyclopedia of neuroscience. Springer-Verlag, Berlin Heidelberg, pp 3071-3074.
- McMahon SB, La Russa F, Bennett DLH (2015) Crosstalk between the nociceptive and immune systems in host defence and disease. Nat Rev Neurosci 16:389-402.
- Mense S (1993) Nociception from skeletal muscle in relation to clinical muscle pain. Pain 54:241-289.
- Mense S (2003) The pathogenesis of muscle pain. Curr Pain Headache Rep 7(6):419-425.
- Mense S (2008) Muscle pain: mechanisms and clinical significance Dtsch Ärztebl Int. 105(12): 214–219.
- Merighi A (2018) The histology, physiology, neurochemistry and circuitry of the substantia gelatinosa Rolandi (lamina II) in mammalian spinal cord. Prog Neurobiol 169:91-134. [CrossRef]
- Metzger M, Bueno D, Lima LB (2017) The lateral habenula and the serotonergic system. Pharmacol Biochem Behav 162:22-28.
- Michaelis M, Vogel C, Blenk KH, Arnarson A, Jänig W (1998) Inflammatory mediators sensitize acutely axotomized nerve fibers to mechanical stimulation in the rat. J Neurosci 18:7581-7587. [CrossRef]
- Millan MJ (2002) Descending control of pain. Prog Neurobiol 66(6):355-474.
- Mills EP, Keay KA, Henderson LA (2021) Brainstem pain-modulation circuitry and its plasticity in neuropathic pain: Insights from human brain imaging investigations. Front Pain Res (Lausanne) 2:705345.
- Miranda OC, Hegedüs K, Kis G, Antal M (2023) Synaptic targets of glycinergic neurons in laminae I-III of the spinal dorsal horn. Int J Mol Sci 24(8):6943.
- Mitsi V, Zachariou V (2016) Modulation of pain, nociception, and analgesia by the brain reward center. Neuroscience 338:81-92. [CrossRef]
- Mogil JS (2020) Qualitative sex differences in pain processing: emerging evidence of a biased literature. Nat Rev Neurosci 21:353-365. [CrossRef]
- Monhemius R, Green DL, Roberts MH, Azami J (2001) Periaquaeductal grey mediated inhibition of response to noxious stimulation is dynamically activated in a rat model of neuropathic pain. Neurosci Lett 298(1):70-74.
- Morales M, Margolis EB (2017) Ventral tegmental area: cellular heterogeneity, connectivity and behaviour. Nat Rev Neurosci 18:8573.
- Morgan MM, Whittier KL, Hegarty DM, Aicher SA (2008) Periaqueductal gray neurons project to spinally projecting GABAergic neurons in the rostral ventromedial medulla. Pain 140(2):376-386. [CrossRef]
- Moriya S, Yamashita A, Masukawa D, Sakaguchi J, Ikoma Y, Sameshima Y, Kambe Y, Yamanaka A, Kuwaki T (2022) Involvement of A5/A7 noradrenergic neurons and B2 serotonergic neurons in nociceptive processing: a fiber photometry study. Neural Regen Res 17(4):881-886.
- Moriya S, Yamashita A, Nishi R, Ikoma Y, Yamanaka A, Kuwaki T (2019) Acute nociceptive stimuli rapidly induce the activity of serotonin and noradrenalin neurons in the brain stem of awake mice. IBRO Rep 7:1–9. [CrossRef]
- Motzkin JC, Hiser J, Carroll J, Wolf R, Baskaya MK, Koenigs M, Atlas LY (2023) Human ventromedial prefrontal cortex lesions enhance the effect of expectations on pain perception. Cortex 166:188-206. [CrossRef]
- Moulton EA, Schmahmann JD, Becerra L, Borsook D (2010) The cerebellum and pain: Passive integrator or active participator? Brain Res Rev 65:14-27.
- Myers B, Scheimann JR, Franco-Villanueva A, Herman JP (2017) Ascending mechanisms of stress integration: implications for brainstem regulation of neuroendocrine and behavioral stress responses. Neurosci Biobehav Rev 74(Pt B): 366-375.
- Nadrigny F, Le Meur K, Schomburg ED, Safavi-Abbasi S, Dibaj P (2017) Two-photon laser-scanning microscopy for single and repetitive imaging of dorsal and lateral spinal white matter in vivo. Physiol Res 66(3):531-537. [CrossRef]
- Nagai M, Kishi K, Kato S (2007) Insular cortex and neuropsychiatric disorders: a review of recent literature. Eur Psychiatry 22(6):387-394.
- Nahorski MS, Chen Y-C, Woods CG (2015) New Mendelian disorders of painlessness. Trends Neurosci 38:712-724.
- Navratilova E, Qu C, Ji G, Neugebauer V, Guerrero M, Rosen H, Roberts E, Porreca F (2024) Opposing effects on descending control of nociception by µ and κ opioid receptors in the anterior cingulate cortex. Anesthesiology 40(2):272-283.
- Nelson AB, Kreitzer AC (2014) Reassessing models of basal ganglia function and dysfunction. Annu Rev Neurosci 37:117-135.
- Nencini S, Ivanusic JJ (2016) The physiology of bone pain. How much do we really know? Front Physiol 7:157. [CrossRef]
- Neugebauer V (2015) Amygdala pain mechanisms. Handb Exp Pharmacol 227:261-284.
- Neugebauer V, Galhardo V, Maione S, Mackey SC (2009) Forebrain pain mechanisms. Brain Res Rev 60:226-242.
- Neugebauer V, Mazzitelli M, Cragg B, Ji G, Navratilova E, Porreca F (2020) Amygdala, neuropeptides, and chronic pain-related affective behaviors. Neuropharmacology 170:108052. [CrossRef]
- Nguyen E, Grajales-Reyes JG, Gereau 4th RW, Ross SE (2023) Cell type-specific dissection of sensory pathways involved in descending modulation. Trends Neurosci 46(7):539-550.
- Nicol GD, Vasko MR (2007) Unraveling the story of NGF-mediated sensitization of nociceptive sensory neurons: ON or OFF the Trks? Mol Interv 7:26-41.
- Nieuwenhuys R, Voogd J, van Huijzen C (1978) The human central nervous system. A synopsis and atlas. Springer, Berlin Heidelberg New York.
- Oane I, Barborica A, Mindruta IR (2023) Cingulate cortex: anatomy, structural and functional connectivity. J Clin Neurophysiol 40(6):482-490.
- Ogawa SK, Watabe-Uchida M (2018) Organization of dopamine and serotonin system: Anatomical and functional mapping of monosynaptic inputs using rabies virus. Pharmacol Biochem Behav 174:9-22.
- O’Neill A, Lirk P (2022) Multimodal analgesia. Anesthesiol Clin 40(3):455-468.
- Ong WY, Stohler CS, Herr DR (2019) Role of the prefrontal cortex in pain processing. Mol Neurobiol 56:1137-1166.
- Ossipov MH, Dussor GO, Porreca F (2010) Central moduation of pain. J Clin Invest 120(11):3779-3787.
- Ossipov MH, Morimura K, Porreca F (2014) Descending pain modulation and chronification of pain. Curr Opin Support Palliat Care 8(2):143-151.
- Pagliusi M, Gomes FV (2023) The role of the rostral ventromedial medulla in stress responses. Brain Sci 13(5):776.
- Pan B, Castro-Lopes JM, Coimbra A (1999) Central afferent pathways conveying nociceptive input to the hypothalamic paraventricular nucleus as revealed by a combination of retrograde labeling and c-fos activation. J Comp Neurol 1999 413(1):129-145.
- Pan H, Wu Z, Zhou H, Chen S, Zhang H, Li D (2008) Modulation of pain transmission by G-protein-coupled receptors. Pharmacol Ther 117(1):141-161.
- Panneton WM, Gan Q, Ariel M (2015) Injections of algesic solutions into muscle activate the lateral reticular formation: A nociceptive relay of the spinoreticulothalamic tract. PLoS One 2015 Jul 8;10(7):e0130939. [CrossRef]
- Paulus MP, Stein MB (2006) An insular view of anxiety. Biol Psychiatry 60:383–387.
- Pautrat A, Rolland M, Barthelemy M, Baunez C, Sinniger V, Piallat B, Savasta M, Overton PG, David O, Coizet V (2018) Revealing a novel nociceptive network that links the subthalamic nucleus to pain processing. Elife 7:e36607. [CrossRef]
- Pay S, Barasi S (1982) A study of the connections of nociceptive substantia nigra neurones. Pain 12(1):75-89.
- Peng B, Jiao Y, Zhang Y, Li S, Chen S, Xu S, Gao P, Fan Y, Yu W (2023) Bulbospinal nociceptive ON and OFF cells related neural circuits and transmitters. Front Pharmacol 14:1159753.
- Peng K, Steele SC, Becerra L, Borsook D (2018) Brodmann area 10: Collating, integrating and high level processing of nociception and pain. Prog Neurobiol 161:1-22. [CrossRef]
- Pertovaara A (2006) NA pain modulation. Prog Neurobiol 80:53-83.
- Pertovaara A (2013) The NA pain regulation system: a potential target for pain therapy. Eur J Pharmacol 716(1-3):2-7.
- Petrides M (2005) Lateral prefrontal cortex: architectonic an functional organization. Phil Trans R Soc B 360:781-795.
- Peyron R, Quesada C, Fauchon C (2019) Cingulate-mediated approaches to treating chronic pain. Handb Clin Neurol 166:317-326.
- Pezet S, McMahon SB (2006) Neurotrophins: mediators and modulators of pain. Annu Rev Neurosci 29:507-538.
- Pilyavskii AI, Maznychenko AV, Maisky VA, Kostyukov AI, Hellström F, Windhorst U (2005) Capsaicin-induced effects on c-fos expression and NADPH-diaphorase activity in the feline spinal cord. Eur J Pharmacol 521(1-3):70-78. [CrossRef]
- Pinho-Ribeiro FA, Verri WA, Chiu IM (2017) Nociceptor sensory neuron-immune interactions in pain and inflammation. Trends Immunol 38(1):5-19.
- Pinto-Ribeiro F, Ansah OB, Almeido A, Pertovaara A (2011) Response properties of nociceptive neurons in the caudal ventrolateral medulla (CVLM) in monoarthritic and healthy control rats: modulation of responses by the paraventricular nucleus of the hypothalamus (PVN). Brain Res Bull 86(1-2):82-90. [CrossRef]
- Ploner M, Gross J, Timmermann L, Schnitzler A (2002) Cortical representation of first and second pain sensation in humans. Proc Natl Acad Sci USA 99:12444-12448.
- Ploski JE, Vaidya VA (2021) The neurocircuitry of posttraumatic stress disorder and major depression: Insights into overlapping and distinct circuit dysfunction – A tribute to Ron Duman. Biol Psychiatry 90(2):109-117. [CrossRef]
- Poe GR, Foote S, Eschenko O, Johansen JP, Bouret S, Aston-Jones G, Harley CW, Manahan-Vaughan D, Weinshenker D, Valentino R, Berridge C, Chandler DJ, Waterhouse B, Sara SJ (2020) Locus coeruleus: a new look at the blue spot. Nat Rev Neurosci 21(11):644-659.
- Poisbeau P, Grinevich V, Charlet A (2018) Oxytocin signaling in pain: Cellular, circuit, system, and behavioral levels. Curr Top Behav Neurosci 35:193-211.
- Porreca F, Ossipov MH, Gebhart GF (2002) Chronic pain and medullary descending facilitation. Trends Neurosci 25:319-325.
- Prescott SA (2015) Synaptic inhibition and disinhibition in the spinal dorsal horn. Prog Mol Biol Trans Sci 131:359-383 .
- Presto P, Mazzitelli M, Junell R, Griffin Z, Neugebauer V (2022) Sex differences in pain along the neuraxis. Neuropharmacology 210:109030.
- Price TJ, Ray PR (2019) Recent advances toward understanding the mysteries of the acute to chronic pain transition. Curr Opin Physiol 11:42-50. [CrossRef]
- Puntillo F, Giglio M, Paladini A, Perchiazzi G, Viswanath O, Urits I, Sabba C, Varrassi G, Brienza N (2021) Pathophysiology of musculoskeletal pain: a narrative review. Ther Adv Musculoskelet Dis 13:1759720X21995067.
- Puopolo M (2019) The hypothalamic-spinal dopaminergic system: a target for pain modulation. Neural Regen Res 14(6):925–930.
- Qian K, Zhou J, Xiong J, Wang Q, Chen L, Zhuang T, Jin J, Zhang G, Hao C, Huang L, Chen Y (2024) Discovery of a novel Kv7.2/7.3 channels agonist for the treatment of neuropathic pain. Eur J Med Chem 280:116953. [CrossRef]
- Queme LF, Jankowski MP (2019) Sex differences and mechanisms of muscle pain. Curr Opin Physiol 11:1-6.
- Quevedo JN (2009) Presynaptic inhibition. In: Binder MD, Hirokawa N, Windhorst U (eds) Encyclopedia of neuroscience. Springer-Verlag, Berlin Heidelberg, pp 3266-3270.
- Reichling DB, Levine JD (2009) Critical role of nociceptor plasticity in chronic pain. Trends Neurosci 32:611-618.
- Reid AR, Côté PD, McDougall JJ (2022) Long-term blockade of nociceptive Nav1.7 channels is analgesic in rat models of knee arthritis. Biomolecules 12(11):1571.
- Ren K, Dubner R (2007) Pain facilitation and activity-dependent plasticity in pain modulatory circuitry: role of BNDF-TrkB signaling and NMDA receptors. Mol Neurobiol 35:224-235.
- Ren K, Randich A, Gebhart GF (1990) Modulation of spinal nociceptive transmission from nuclei tractus solitarii: a relay for effects of vagal afferent stimulation. J Neurophysiol 63(5):971-986. [CrossRef]
- Ren S, Wang S, Lv S, Gao J, Mao Y, Liu Y, Xie Q, Zhang T, Zhao L, Shi J (2024) The nociceptive inputs of the paraventricular hypothalamic nucleus in formalin stimulated mice. Neurosci Lett 841:137948.
- Revand R, Singh SK, Muthu MS (2023) Subthreshold doses of inflammatory mediators potentiate one another to elicit reflex cardiorespiratory responses in anesthetized rats. Cardiovasc Hematol Agents Med Chem 2023 Apr 7. [CrossRef]
- Rezaee L, Karimi-Haghighi S, Fazli-Tabaei S, Haghparast A (2018) Effects of intrathecal administration of orexin-1 receptor antagonist on antinociceptive responses induced by chemical stimulation of lateral hypothalamus in an animal model of tonic nociception. Neuropeptides 69:19-25. [CrossRef]
- Robinson DA (1992) Implications of neural networks for how we think about brain function. Behav Brain Sci 15:644-655.
- Rocha I, González-García M, Carillo-Franco L, Dawid-Milner MS, López-González MV (2024) Influence of brainstem’s area A5 on sympathetic outflow and cardiorespiratory dynamics. Biology (Basel) 13(3):161.
- Roelofs K (2017) Freeze for action: neurobiological mechanisms in animal and human freezing. Phil Trans R Soc B 372:20160206.
- Ross JA, van Bockstaele EJ (2020) The locus coeruleus-norepinephrine system in stress and arousal: Unraveling historical, current, and future perspectives. Front Psychiatry 11:601519.
- Ross SE (2011) Pain and itch: insights into the neural circuits of aversive somatosensation in health and disease.Curr Opin Neurobiol 21(6):880-887. [CrossRef]
- Rossi A, Mazzocchio R, Decchi B (2003) Effect of chemically activated fine muscle afferents on spinal recurrent inhibition in humans. Clin Neurophysiol 114(2):279-287. [CrossRef]
- Roth RH, Ding JB (2024) Cortico-basal ganglia plasticity in motor learning. Neuron 112(15):2486-2502.
- Roy N, Parhar I (2022) Habenula orphan G-protein coupled receptors in the pathophysiology of fear and anxiety. Neurosci Biobehav Rev 132:870-883. [CrossRef]
- Saab CY, Waxman SG, Hains BC (2008) Alarm or curse? The pain of neuroinflammation. Brain Res Rev 58:226-235.
- Saab CY, Willis WD (2003) The cerebellum: organization, functions and its role in nociception. Brain Res Rev 42:85-95.
- Saadé NE, Jabbur SJ (2008) Nociceptive behavior in animal models for peripheral neuropathy: Spinal and supraspinal mechanisms. Prog Neurobiol 86:22-47. [CrossRef]
- Saman Y, Arshad Q, Dutia M, Rea P (2020) Stress and the vestibular system. Int Rev Neurobiol 2020;152:221-236.
- Sandkühler J (2009) Models and mechanisms of hyperalgesia and allodynia. Physiol Rev 89:707-758.
- Sandri A, Cecchini MP, Riello M, Zanini A, Nocini R, Fiorio M, Tinazzi M (2021) Pain, smell, and taste in adults: a narrative review of multisensory perception and interaction. Pain Ther 10(1):245-268. [CrossRef]
- Sandrini G, Serrao M, Rossi P, Romaniello A, Cruccu G, Willer JC (2005) The lower limb flexion reflex in humans. Prog Neurobiol 77:353-395.
- Sartori SB, Singewald N (2019) Novel pharmacological targets in drug development for the treatment of anxiety and anxiety-related disorders. Pharmacology&Therapeutics 204:107402. [CrossRef]
- Sato A, Sato Y, Schmidt RF (1997) The impact of somatosensory input on autonomic functions. Rev Physiol Biochem Pharmacol 130:1-328.
- Sato F, Akhter F, Haque T, Kato T, Takeda R, Nagase Y, Sessle BJ, Yoshida A (2013) Projections from the insular cortex to pain-receptive trigeminal caudal subnucleus (medullary dorsal horn) and other lower brainstem areas in rats. Neuroscience 233:9-27. [CrossRef]
- Savadipour A, Palmer D, Ely EV, Collins KH, Garcia-Castorena JM, Harissa Z, Kim YS, Oestrich A, Qu F, Rashidi N, Guilak F (2023) The role of PIEZO ion channels in the musculoskeletal system. Am J Physiol Cell Physiol 324(3):C728-C740.
- Sawchenko PE, Swanson LW (1982) Immunohistochemical identification of neurons in the paraventricular nucleus of the hypothalamus that project to the medulla or to the spinal cord in the rat. J Comp Neurol ;205(3):260-272. [CrossRef]
- Schaible H-G (2009) Joint pain. In: Binder MD, Hirokawa N, Windhorst U (eds) Encyclopedia of neuroscience. Springer-Verlag, Berlin Heidelberg, pp 2072-2076.
- Schmidt RF, Struppler A (1983) Der Schmerz. Ursachen, Diagnose, Therapie. Piper Verlag, München - Zürich.
- Scholz J, Woolf CJ (2002) Can we conquer pain? Nature Neurosci Suppl 5:1062-1067.
- Schomburg ED, Dibaj P, Steffens H (2011a) Differentiation between Aδ and C fibre evoked nociceptive reflexes by TTX resistance and opioid sensitivity in the cat. Neurosci Res 69(3):241-245.
- Schomburg ED, Dibaj P, Steffens H (2011b) Role of L-DOPA in spinal nociceptive reflex activity: higher sensitivity of Aδ versus C fibre-evoked nociceptive reflexes to L-DOPA. Physiol Res 60(4):701-703. [CrossRef]
- Schomburg ED, Kalezic I, Dibaj P, Steffens H (2013) Reflex transmission to lumbar α-motoneurones in the mouse similar and different to those in the cat. Neurosci Res 76:133-140.
- Schomburg ED, Steffens H, Dibaj P, Sears TA (2012) Major contribution of Aδ-fibres to increased reflex transmission in the feline spinal cord during acute muscle inflammation.Neurosci Res 72(2):155-162. [CrossRef]
- Schomburg ED, Steffens H, Pilyavskii AI, Maisky VA, Brück W, Dibaj P, Sears TA (2015) Long lasting activity of nociceptive muscular afferents facilitates bilateral flexion pattern in the feline spinal cord. Neurosci Res 95:51-58 . [CrossRef]
- Schouenborg J (2002) Modular organisation and spinal somatosensory imprinting, Brain Res Rev 40:80-91.
- Schouenborg J (2003) Somatosensory imprinting in spinal reflex modules. J Rehabil Med Suppl. 41:73–80 .
- Seminowicz DA, Moayedi M (2017) The dorsolateral prefrontal cortex in acute and chronic pain. J Pain 18(9):1027–1035.
- Seminowicz DA, Remeniuk B, Krimmel SR, Smith MT, Barrett FS, Wulff AB, Furman AJ, Geuter S, Lindquist MA, Irwin MR, Finan PH (2019) Pain-related nucleus accumbens function: modulation by reward and sleep disruption. Pain 160(5):1196-1207.
- Seymour B (2019) Pain: a precision signal for reinforcement learning and control. Neuron 101:1029-1041.
- Sliz D, Hayley S (2012) Major depressive disorder and alterations in insular cortical activity: A review of current functional magnetic imaging research. Front Hum Neurosci 6: 323. [CrossRef]
- Smith N, Juresic S, Lor K, Schmidt C, Howrigon AB, Olatoye O (2024) Intravenous magnesium sulfate as an adjunct to intravenous ketamine for acute pain: a case serious. J Pain Palliat Care Pharmacother 13:1-8.
- Sprenger C, Finsterbusch J, Büchel C (2015) Spinal cord-midbrain functional connectivity is related to perceived pain intensity: a combined spino-cortical FMRI study. J Neurosci 35(10):4248-4257. [CrossRef]
- Spuz C, Tomaszycki ML, Borszcz GS (2014) N-methyl-D-aspartate receptor agonism and antagonism within the amygdaloid central nucleus suppresses pain affect: differential contribution of the ventrolateral periaqueductal gray. J Pain 15(12):1305-1318. [CrossRef]
- Starr CJ, Sawaki L, Wittenberg GF, Burdette JH, Oshiro Y, Quevedo AS, McHaffie JG, Coghill RC (2011) The contribution of the putamen to sensory aspects of pain: insights from structural connectivity and brain lesions. Brain 134(Pt 7):1987-2004. [CrossRef]
- Strand LI, Gundrosen KF, Küfner Lein R, Laekeman M, Lobbezoo F, Defrin R, Husebo BS (2019) Body movements as pain indicators in older people with cognitive impairment: A systematic review. Eur J Pain 23(4):669-685. [CrossRef]
- Sugimura YK, Takahashi Y, Watabe AM, Kato F (2016) Synaptic and network consequences of monosynaptic nociceptive inputs of parabrachial nucleus origin in the central amygdala. J Neurophysiol 115(6):2721-2739. [CrossRef]
- Svensson P, Minoshima S, Beydoun A, Morrow TJ, Casey KL (1997) Cerebral processing of acute skin and muscle pain in humans. J Neurophysiol 78(1):450-460.
- Takayanagi Y, Onaka T (2021) Roles of oxytocin in stress responses, allostasis and resilience. Int J Mol Sci 23(1):150.
- Tan S, Faull RLM, Curtis MA (2023) The tracts, cytoarchitecture, and neurochemistry of the spinal cord. Anat Rec (Hoboken) 306(4):777-819.
- Tanaka M, Török N, Tóth F, Szabó A, Vécsei L (2021) Co-players in chronic pain: neuroinflammation and the tryptophan-kynurenine metabolic pathway. Biomedicines 9(8):897. [CrossRef]
- Taniguchi W, Nakatsuka T, Miyazaki N, Yamada H, Takeda D, Fujita T, Kumamoto E, Yoshida M (2011) In vivo patch-clamp analysis of dopaminergic antinociceptive actions on substantia gelatinosa neurons in the spinal cord. Pain 152(1):95-105. [CrossRef]
- Tang J-S, Qu C-L, Huo F-Q (2009) The thalamic nucleus submedius and ventrolateral orbital cortex are involved in nociceptive modulation: a novel pain modulation pathway. Prog Neurobiol 89(4):383-389.
- Taub DG, Jiang Q, Pietrafesa F, Su J, Carroll A, Greene C, Blanchard MR, Jain A, El-Rifai M, Callen A, Yager K, Chung C, He Z, Chen C, Woolf CJ (2024) The secondary somatosensory cortex gates mechanical and heat sensitivity. Nat Commun 2024 Feb 12;15(1):1289.
- Tavares I, Costa-Pereira JT, Martins I (2021) Monoaminergic and opioidergic modulation of brainstem circuits: New insights into the clinical challenges of pain treatment? Front Pain Res (Lausanne) 2:696515.
- Tavares I, Lima D (2002) The caudal ventrolateral medulla as an important inhibitory modulator of pain transmission in the spinal cord. J Pain 3(5):337-346. [CrossRef]
- Tavares I, Lima D (2007) From neuroanatomy to gene therapy: searching for new ways to manipulate the supraspinal endogenous pain modulatory system. J Anat 211:261-268. [CrossRef]
- Tavares I, Lima D, Coimbra A (1997) The pontine A5 noradrenergic cells which project to the spinal cord dorsal horn are reciprocally connected with the caudal ventrolateral medulla in the rat. Eur J Neurosci 9(11):2452-2461. [CrossRef]
- Tewari A, Jog R, Jog MS (2016) The striatum and subthalamic nucleus as independent and collaborative structures in motor control. Front Syst Neurosci 10:17. [CrossRef]
- Thomaidou MA, Peerdeman KJ, Koppeschaar MI, Evers AWM, Veldhuijzen DS (2021) How negative experience influences the brain: A comprehensive review of the neurobiological underpinnings of nocebo hyperalgesia. Front Neurosci 15:652552. [CrossRef]
- Thompson JM, Neugebauer V (2019) Cortico-limbic pain mechanisms. Neurosci Lett 702:15-23.
- Timmers I, Quaedflieg CWEM, Hsu C, Heathcote LC, Rovnaghi CR, Simons LE (2019) The interaction between stress and chronic pain through the lens of threat learning. Neurosci Biobehav Rev 107:641-655.
- Todd AJ (2010) Neuronal circuitry for pain processing in the dorsal horn. Nat Rev Neurosci 11(12):823-836.
- Todd AJ (2017) Identifying functional populations among the interneurons in laminae I-III of the spinal dorsal horn. Mol Pain 1744806917693003. [CrossRef]
- Todd AJ, Spike RC (1993) The localization of classical transmitters and neuropeptides withinh neurons in laminae I-III of the mammalian spinal dorsal horn. Prog Neurobiol 41:609-645.
- Toledo F, Carson F (2022) Neurobiological features of posttraumatic stress disorder (PTSD) and their role in understanding adaptive behavior and stress resilience. Int J Environ Res Public Health 19(16):10258. [CrossRef]
- Toney GM, Mifflin SW (2000) Sensory modalities conveyed in the hindlimb somatic afferent input to nucleus tractus solitarius. J Appl Physiol (1985) 88(6):2062-2073. [CrossRef]
- Tracey I (2011) Can neuroimaging studies identify pain endophenotypes in humans? Nat Rev Neurol 7:173-181.
- Tracey I, Mantyh PW (2007) The cerebral signature for pain perception and its modulation. Neuron 55:377-391.
- Tracy LM, Georgiou-Karistianis N, Gibson SJ, Giummarra MJ (2015) Oxytocin and the modulation of pain experience: Implications for chronic pain management. Neurosci Biobehav Rev 55:53-67.
- Tremblay R, Lee S, Rudy B (2016) GABAergic interneurons in the neocortex: From cellular properties to circuits. Neuron 91(2):260-292.
- Urban MO, Gebhart GF (1999) Supraspinal contributions to hyperalgesia. Proc Natl Acad Sci 96:7687-7692.
- Valentino RJ, Van Bockstaele E (2015) Endogenous opioids: The downside of opposing stress. Neurobiol Stress 1: 23–32.
- Van den Pol AN (1999) Hypothalamic hypocretin (orexin): robust innervation of the spinal cord. J Neurosci 19(8):3171-3182.
- Vander Weele CM, Siciliano CA, Tye KM (2019) Dopamine tunes prefrontal outputs to orchestrate aversive processing. Brain Res 1713:16-31.
- Vanegas H, Schaible H-G (2004) Descending control of persistent pain: inhibitory or facilitatory? Brain Res Rev 46:295-309.
- Vangeel L, Benoit M, Miron Y, Miller PE, De Clercq K, Chaltin P, Verfaillie C, Vriens J, Voets T (2020) Functional expression and pharmacological modulation of TRPM3 in human sensory neurons. Br J Pharmacol 177(12):2683-2695.
- Vázquez-León P, Miranda-Páez A, Chávez-Reyes J, Allende G, Barragán-Iglesias P, Marichal-Cancino BA (2021) The periaqueductal gray and its extended participation in drug addiction phenomena. Neurosci Bull 37(10):1493–1509.
- Verriotis M, Chang P, Fitzgerald M, Fabrizi L (2016) The development of the nociceptive brain. Neuroscience 338:207-219.
- Villanueva L, Le Bars D (1995) The activation of bulbo-spinal controls by peripheral nociceptive inputs: diffuse noxious inhibitory controls. Biol Res 28(1):113-125.
- Villemure C, Schweinhardt P (2010) Supraspinal pain processing: distinct roles of emotion and attention. Neuroscientist 16:276-284.
- Vincent K, Tracey I (2010) Sex hormones and pain: the evidence from functional imaging. Curr Pain Headache Rep 14:396-403.
- Vogt BA (2005) Pain and emotion interactions in subregions of the cingulate gyrus. Nat Rev Neurosci 6(7):533-544.
- Vogt BA (2016) Midcingulate cortex: Structure, connections, homologies, functions and diseases. J Chem Neuroanat 74:28-46.
- Vogt BA (2019) Cingulate cortex in the three limbic subsystems. Handb Clin Neurol 166:39-51.
- Wager TD, Atlas LY, Lindquist MA, Roy M, Woo C-W, Kross E (2013) An fMRI-based neurologic signature of physical pain. N Engl J Med 368(15):1388-1397.
- Wagner KM, Roeder Z, Desrocher K, Buhler AV, Heinricher MM, Cleary DR (2013) The dorsomedial hypothalamus mediates stress-induced hyperalgesia and is the source of the pronociceptive peptide cholecystokinin in the rostral ventromedial medulla. Neuroscience 238:29-38. [CrossRef]
- Wallace DM, Magnuson DJ, Gray TS (1992) Organization of amygdaloid projections to brainstem dopaminergic, noradrenergic, and adrenergic cell groups in the rat. Brain Res Bull 28(3):447-454.
- Wang H, Ehnert C, Brenner GJ, Woolf CJ (2006) Bradykinin and peripheral sensitization. Biol Chem 387:11-14.
- Wang J, Li Z-H, Feng B, Zhang T, Zhang H, Li H, Chen T, Cui J, Zang W-.D, Li Y-Q (2015) Corticotrigeminal projections from the insular cortex to the trigeminal caudal subnucleus regulate orofacial pain after nerve injury via extracellular signal-regulated kinase activation in insular cortex neurons. Front Cell Neurosci 9:493. [CrossRef]
- Wang M, Tutt JO, Dorricott NO, Parker KL, Russo AF, Sowers LP (2022) Involvement of the cerebellum in migraine. Front Syst Neurosci 16: 984406.
- Wang QP, Nakai Y (1994) The dorsal raphe: an important nucleus in pain modulation. Brain Res Bull 34(6):575-785.
- Wang S, Lim J, Joseph J, Wang S, Wei F, Ro JY, Chung M (2017) Spontaneous and bite-evoked muscle pain are mediated by a common nociceptive pathway with differential contribution by TRPV1. J Pain 18(11):1333-1345. [CrossRef]
- Wang X-Q, Mokhtari T, Zeng Y-X, Yue L-P, Hu L (2021) The distinct functions of dopaminergic receptors on pain modulation: A narrative review. Neural Plast 2021:6682275.
- Waterhouse BD, Predale HK, Plummer NW, Jensen P, Chandler DJ (2022) Probing the structure and function of locus coeruleus projections to CNS motor centers. Front Neural Circuits 16: 895481. [CrossRef]
- Wei H-H, Yuan X-S, Chen Z-K, Chen P-P, Xiang Z, Qu W-M, Li R-X, Zhou G-M, Huang Z-L (2021) Presynaptic inputs to vasopressin neurons in the hypothalamic supraoptic nucleus and paraventricular nucleus in mice. Exp Neurol 343:113784.
- Welman FHSM, Smit AE, Jongen JLM, Tibboel D, van der Geest JN, Holstege JC (2018) Pain experience is somatotopically organized and overlaps with pain anticipation in the human cerebellum. Cerebellum 17(4):447-460. [CrossRef]
- Wen Z, Wu Z, Huang S, Chou T, Cheng H, Lo Y, Jean Y, Sung C (2024) Local magnesium sulfate administration ameliorates nociception, peripheral inflammation, and spinal sensitization in a rat model of incisional pain. Neuroscience 547:98-107.
- Wercberger R, Basbaum AI (2019) Spinal cord projection neurons: a superficial, and also deep analysis. Curr Opin Physiol 11:109-1153.
- West SJ, Bannister K, Dickenson AH, Bennett DL (2015) Circuitry and plasticity of the dorsal horn – toward a better understanding of neuropathic pain. Neuroscience 300:254-275.
- Westlund KN, Craig AD (1996) Association of spinal lamina I projections with brainstem catecholamine neurons in the monkey. Exp Brain Res 110(2):151-162.
- Wichmann T, DeLong MR (2016) Deep brain stimulation for movement disorders of basal ganglia origin: Restoring function or functionality? Neurotherapeutics 13(2):264-283.
- Wiech K, Edwards R, Moseley GL, Berna C, Ploner M, Tracey I (2014) Dissociable neural mechanisms underlying the modulation of pain and anxiety? An FMRI pilot study. PLoS One 9(12):e110654. [CrossRef]
- Willis WD (1996) Temperature perception and pain. In Greger R, Windhorst U (eds) Comprehensive human physiology. From cellular mechanisms to integration, vol 1. Springer-Verlag, Berlin Heidelberg New York, pp 677-696.
- Willis WD, Westlund KN (1997) Neuroanatomy of the pain system and of the pathways that modulate pain. J Clin Neurophysiol 14(1):2-31.
- Windhorst U (2021) Spinal cord circuits: models and reality. Neurophysiology 53(3-6):142-222 .
- Windhorst U, Dibaj P (2023) Plastic spinal motor circuits in health and disease. J Integr Neurosci 22(6):167 .
- Windhorst U, Kirmayer D, Soibelman F, Misri A, Rose R (1997a) Effects of neurochemically excited group III-IV muscle afferents on motoneuron afterhyperpolarization. Neurosci. 76, 915-929 .
- Windhorst U, Meyer-Lohmann J, Kirmayer D, Zochodne D (1997b) Renshaw cell responses to intra-arterial injection of muscle metabolites into cat calf muscles. Neurosci Res 27(3):235-247 .
- Witter MP (2009) Hippocampal formation. In: Binder MD, Hirokawa N, Windhorst U (eds) Encyclopedia of neuroscience. Springer-Verlag, Berlin Heidelberg, pp 1840-1845 .
- Witter MP, Doan TP, Jacobsen B, Nilssen ES, Ohara S (2017) Architecture of the entorhinal cortex. A review of entorhinal anatomy in rodents with some comparative notes. Front Syst Neurosci 11:46 . [CrossRef]
- Woolf CJ, Ma Q (2007) Nociceptors--noxious stimulus detectors. Neuron 2007 Aug 2;55(3):353-364.
- Woulfe JM, Flumerfelt BA, Hrycyshyn AW (1990) Efferent connections of the A1 noradrenergic cell group: a DBH immunohistochemical and PHA-L anterograde tracing study. Exp Neurol 109(3):308-322. [CrossRef]
- Wu S-X, Wang W, Li H, Wang Y-Y, Feng Y-P, Li Y-Q (2010) The synaptic connectivity that underlies the noxious transmission and modulation within the superficial dorsal horn of the spinal cord. Prog Neurobiol 91:38-54 .
- Xu A, Larsen B, Baller EB, Scott JC, Sharma V, Adebimpe A, Basbaum AI, Dworkin RH, Edwards RR, Woolf CJ, Eickhoff SB, Eickhoff CR, Satterwaite TD (2020) Convergent neural representations of experimentally-induced acute pain in healthy volunteers: A large-scale fMRI meta-analysis. Neurosci Biobehav Rev 112:300-323. [CrossRef]
- Yang H, de Jong JW, Cerniauskas I, Peck JR, Lim BK, Gong H, Fields HL, Lammel S (2021) Pain modulates dopamine neurons via a spinal-parabrachial-mesencephalic circuit. Nat Neurosci 24(10):1402-1413 .
- Yang S, Chang MC (2019) Chronic pain: Structural and functional changes in brain structures and associated negative affective states. Int J Mol Sci 20(13): 3130 . [CrossRef]
- Ye D-W, Liu C, Liu T-T, Tian X-B, Xiang H-B (2014) Motor cortex-periaqueductal gray-spinal cord neuronal circuitry may involve in modulation of nociception: a virally mediated transsynaptic tracing study in spinally transected transgenic mouse model. PLoS One 9(2):e89486 . [CrossRef]
- Yen C-T, Lu P-L (2013) Thalamus and pain. Acta Anaethesiologica Taiwanica 51:73-80.
- Yetnikoff L, Lavezzi HN, Reichard RA, Zahm DS (2014) An update on the connections of the ventral mesencephalic dopaminergic complex. Neuroscience 282:23-48 .
- Yin J-B, Liang S-H, Li F, Zhao W-J, Bai Y, Sun Y, Wu Z-Y, Ding T, Sun Y, Liu H-X, Lu Y-C, Zhang T, Huang J, Chen T, Li H, Chen Z-F, Cao J, Ren R, Peng Y-N, Yang J, Zang W-D, Li X, Dong Y-L, Li Y-Q (2020) tdmPFC-vlPAG projection neurons contribute to pain threshold maintenance and antianxiety behaviors. J Clin Invest 130(12):6555-6570.
- Yoshii T (2021) The role of the thalamus in post-traumatic stress disorder. Int J Mol Sci 22(4):1730 .
- Yoshimura M, Furue H (2006) Mechanisms for the anti-nociceptive actions of the descending NA and serotonergic systems in the spinal cord. J Pharmacol Sci 101:107-117 .
- You H-J, Lei J, Pertovaara A (2022) Thalamus: The 'promoter' of endogenous modulation of pain and potential therapeutic target in pathological pain. Neurosci Biobehav Rev 139:104745.
- Zeilhofer HU, Wildner H, Yévenes GE (2012) Fast synaptic inhibition in spinal sensory processing and pain control. Physiol Rev 92:193-235 .
- Zhang G, Cui M, Ji R, Zou S, Song L, Fan B, Yang L, WangD, Hu S, Zhang X, Fang T, Yu X, Yang J-X, Chaudhury D, Liu H, Hu A, Ding H-L, Cao J-L, Zhang H (2023) Neural and molecular investigation into the paraventricular thalamic-nucleus accumbens circuit for pain sensation and non-opioid analgesia. Pharmacol Res 191:106776 .
- Zhang H, Li L, Zhang X, Ru G, Zang W (2024) Role of the Dorsal Raphe Nucleus in Pain Processing. Brain Sci 14(10):982.
- Zhang L, Zhang Y, Zhao Z-Q (2005) Anterior cingulate cortex contributes to the descending facilitatory modulation of pain via dorsal reticular nucleus. Eur J Neurosci 22(5):1141-1148.
- Zheng H Lim JY, Kim Y, Jung ST, Hwang SW (2021) The role of oxytocin, vasopressin, and their receptors at nociceptors in peripheral pain modulation. Front Neuroendocrinol 100942.
- Zheng H, Lim JY, Seong JY, Hwang SW (2020) The role of corticotropin-releasing hormone at peripheral nociceptors: Implications for pain modulation. Biomedicines 8(12):623. [CrossRef]
- Zimmermann AL, Kovatsis EM, Pozsgai RY, Tasnim A, Zhang Q, Ginty DD (2019) Distinct modes of presynaptic inhibition of cutaneous afferents and their functions in behavior. Neuron 102(2):420-434.e8.
- Zouikr I, Bartholomeusz MD, Hodgson DM (2016) Early life programming of pain: focus on neuroimmune to endocrine communication. J Transl Med 14:123.
- Zhou K, Zhu L, Hou G, Chen X, Chen B, Yang C, Zhu Y (2021) The contribution of thalamic nuclei in salience processing. Front Behav Neurosci 15:634618.
- Zhu J-N, Yung W-H, Chow BK-C, Chan Y-S, Wang J-J (2006) The cerebellar-hypothalamic circuits: potential pathways underlying cerebellar involvement in somatic-visceral integration. Brain Res Rev 52(1):93-106.
- Zhuo M, Gebhart GF (1997) Biphasic modulation of spinal nociceptive transmission from the medullary raphe nuclei in the rat. J Neurophysiol 78(2):746-758. [CrossRef]
- Zyuzin J, Jendzjowsky N (2022) Neuroanatomical and neurophysiological evidence of pulmonary nociceptor and carotid chemoreceptor convergence in the nucleus tractus solitarius and nucleus ambiguus. J Neurophysiol 127(6):1511-1518. [CrossRef]
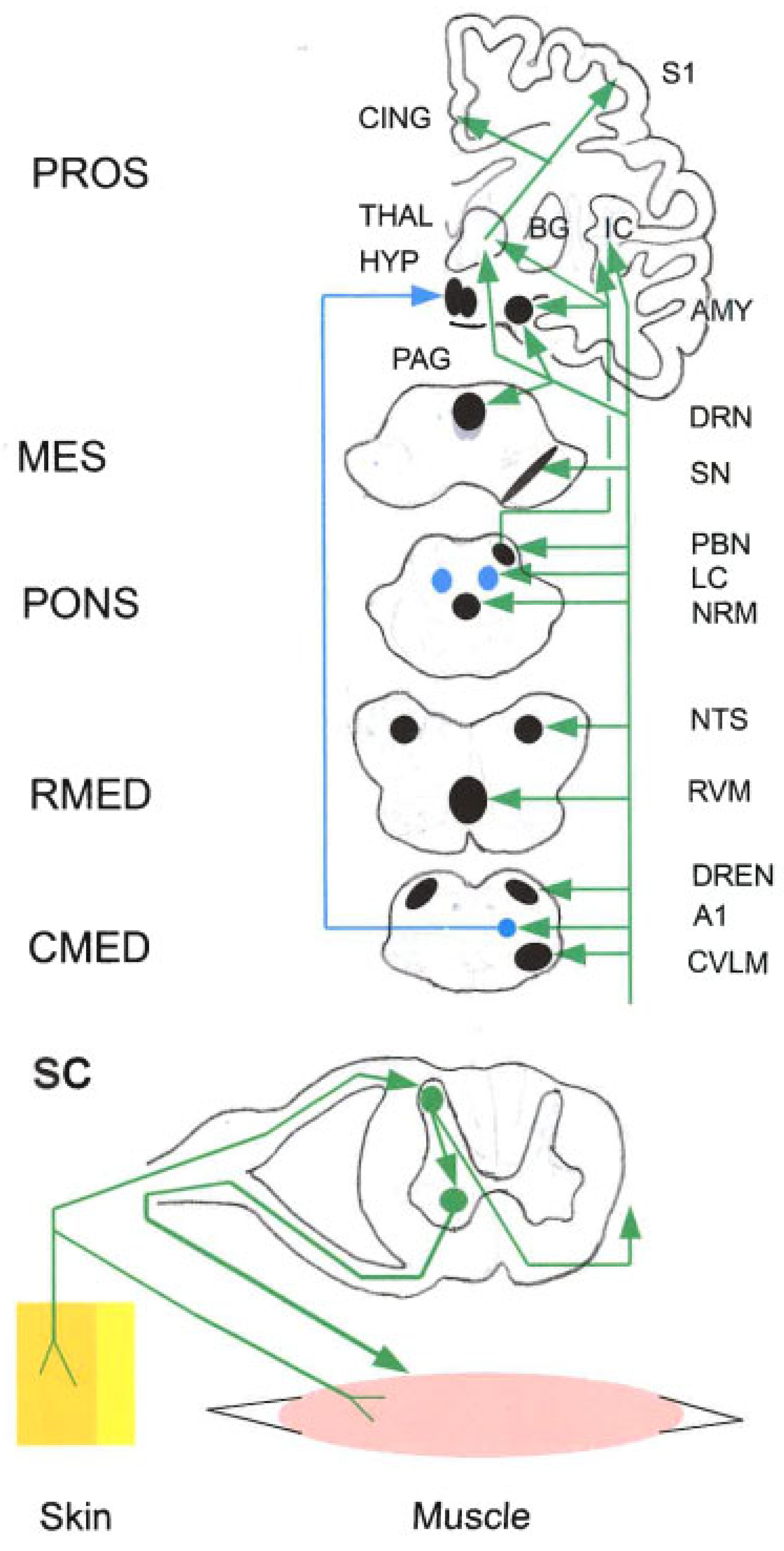
| Primary motor cortex (M1) → THAL (MD) → NAc (Gan et al. 2022) (not shown) |
| Primary motor cortex (M1) → zona incerta and PAG (Gan et al. 2022) |
| mPFC → `medullary DH´ (Akhter et al. 2014). |
| mPFC ↔ AMY → PAG (Ong et al. 2019; Ossipov et al. 2010) |
| mPFC → PAG (Akhter et al. 2014) |
| mPFC → NTS (Akhter et al. 2014) |
| dmPFC → vlPAG (Yin et al. 2020) |
| ACC ↔ AMY (Harris and Peng 2020) |
| ACC ↔ NAc (Harris and Peng 2020) |
| ACC → PAG (Heinricher et al. 2009) |
| ACC → DH (Chen et al. 2014) |
| IC → DH (Sato et al. 2013; Wang et al. 2015) |
| AMY →LC (Wallace et al. 1992) |
| THAL (MD) and (VM) → cingulate cortex (CC), cortico-spinal tract (CST) or dlPAG → DH (You et al. 2022) |
| THAL (VM) → insular cortex (IC) and vlPAG → DH. (You et al. 2022) |
| SNr → STN → PBN (inh) (Jia et al. 2022) |
| HYP → DH (DA; Gamal-Eltrabily et al. 2018; Kelly and Fudge 2018; Lindvall et al. 1983). |
| HYP → DH (5-HT; Condés-Lara et al. 2024; Gamal-Eltrabily et al. 2018) |
| lHYP → DH (ORX; Van den Pol 1999) |
| HYP DMH → PAG (Heinricher et al. 2009) |
| HYP DMH → RVM → DH (Amorim et al. 2015; Heinricher et al. 2009; Wagner et al. 2013) |
| HYP DMH → DReN → DH (Amorim et al. 2015) |
| lHYP → A7 → DH (Holden and Naleway 2001) |
| M1 → medio-dorsal THAL → NAc (Gan et al. 2022) |
| M1 → PAG (Gan et al. 2022) |
| PAG → AMY (Millan 2002) |
| PAG → HYP (Millan 2002) |
| PAG → LC (Ossipov et al. 2010; Willis and Westlund 1997) |
| PAG ↔ NTS (Millan 2002) |
| PAG → NRM (Willis and Westlund 1997) |
| PAG → RVM (De Preter and Heinricher 2024; Lau and Vaughan 2014; Millan 2002; Ossipov et al. 2010) |
| PAG → DReN (Millan 2002) |
| PBN → RVM (De Preter and Heinricher 2024) |
| PBN → DH (Millan 2002) |
| DRN → DH (Kuner and Kuner 2021) |
| NTS → HYP (Millan 2002) |
| NTS → RVM (Millan 2002) |
| NTS → DH (Millan 2002) |
| LC ↔ RVM (Ossipov et al. 2010) |
| LC (A6 cells) → DH (Pertovaara 2006, 2013; Tavares et al. 2021; Willis and Westlund 1997) |
| NA A1 cells → DH (Pertovaara 2006, 2013) |
| NA A5 cells → CVLM (Tavares and Lima 2007) |
| NA A5 cells → DH (inh) (Dostrovsky 2000; Pertovaara 2006, 2013; Tavares and Lima 2007) |
| NA A7 → DH (inh) (Dostrosky 2000; Pertovaara 2006, 2013) |
| VLM → DH (inh) (Lau and Vaughan 2014; Ossipov et al. 2010) |
| CVLM → DH (Martins and Tavares 2017; Tavares and Lima 2002). |
| CVLM → LC → SC (Gu et al. 2023) |
| CVLM → A5-→ DH (Tavares et al. 1997) |
| DReN ↔ DH (exc) (Heinricher et al. 2009; Millan 2002; Martins and Tavares 2017; Tavares and Lima 2007) |
| RVM → DH (exc or inh) (Heinricher et al. 2009; Millan 2002; Martins and Tavares 2017; Ossipov et al. 2010) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




