Submitted:
06 May 2025
Posted:
07 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Sample Preparation
2.3. Sample Characterization
2.4. Non-Isothermal Crystallization
2.5. Isothermal Crystallization
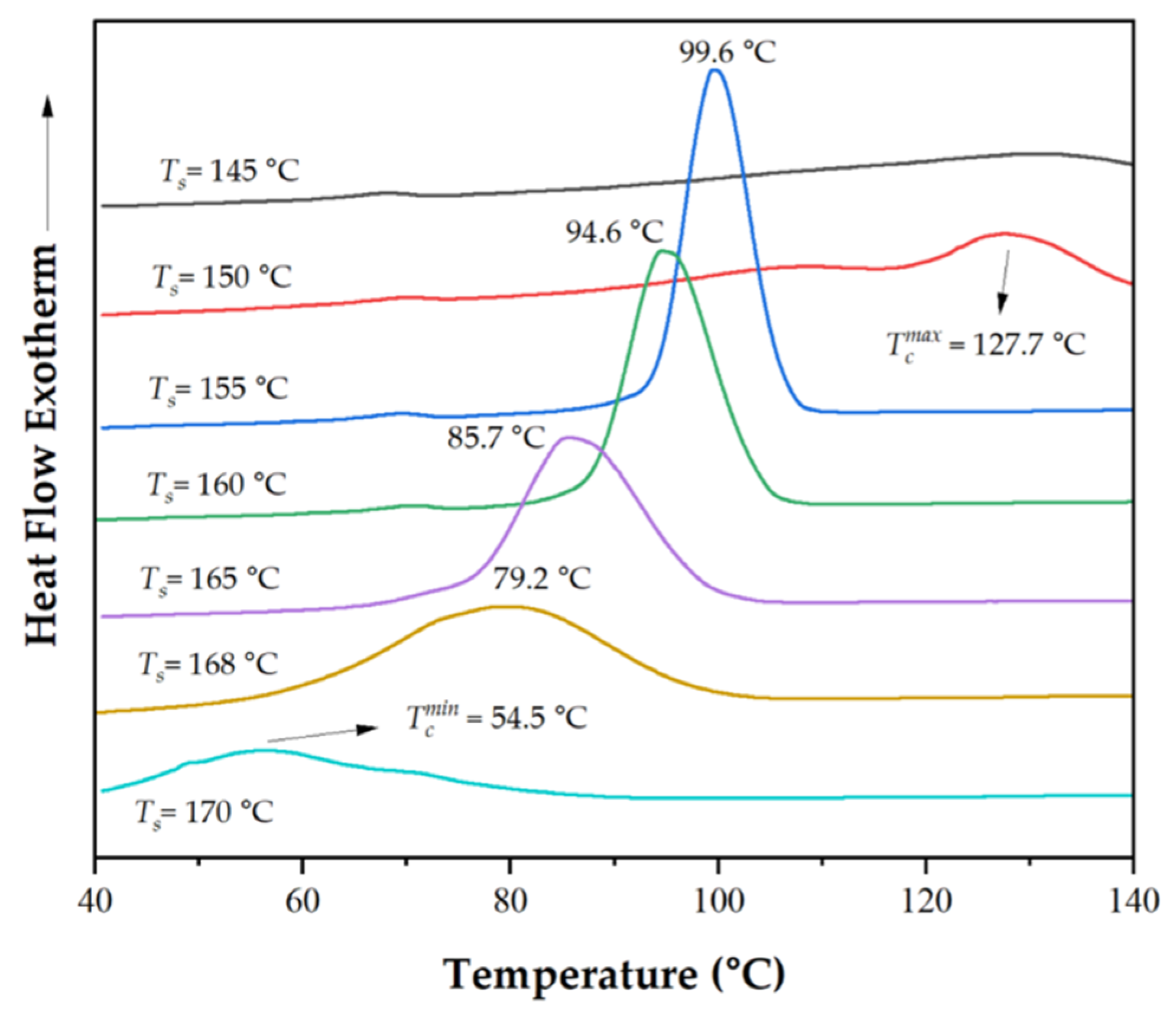
2.6. Self-Nucleation Experiments
3. Results and Discussion
3.1. Non-Isothermal Crystallization Behavior of P3HBHHx. Effect of Nucleating Agents
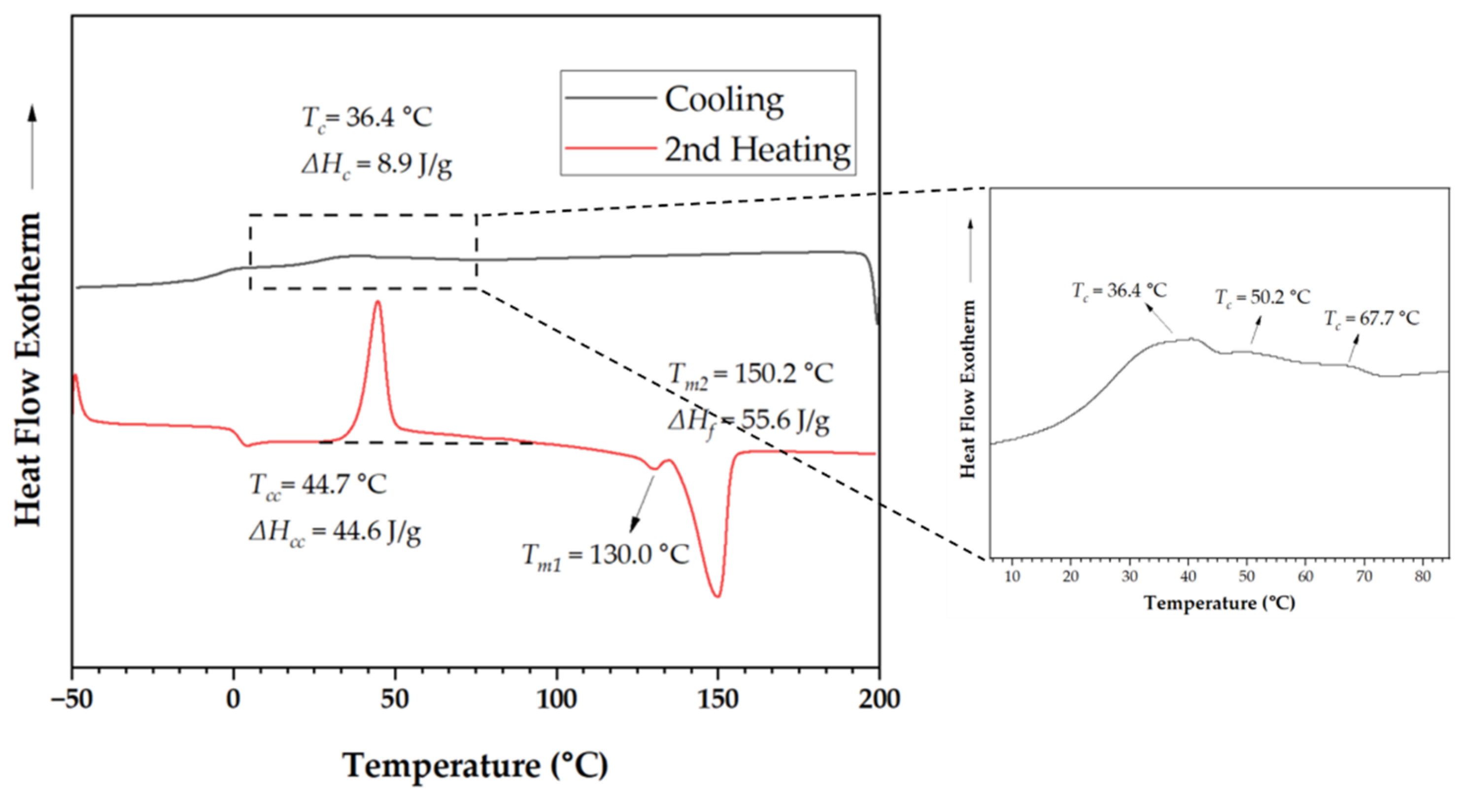
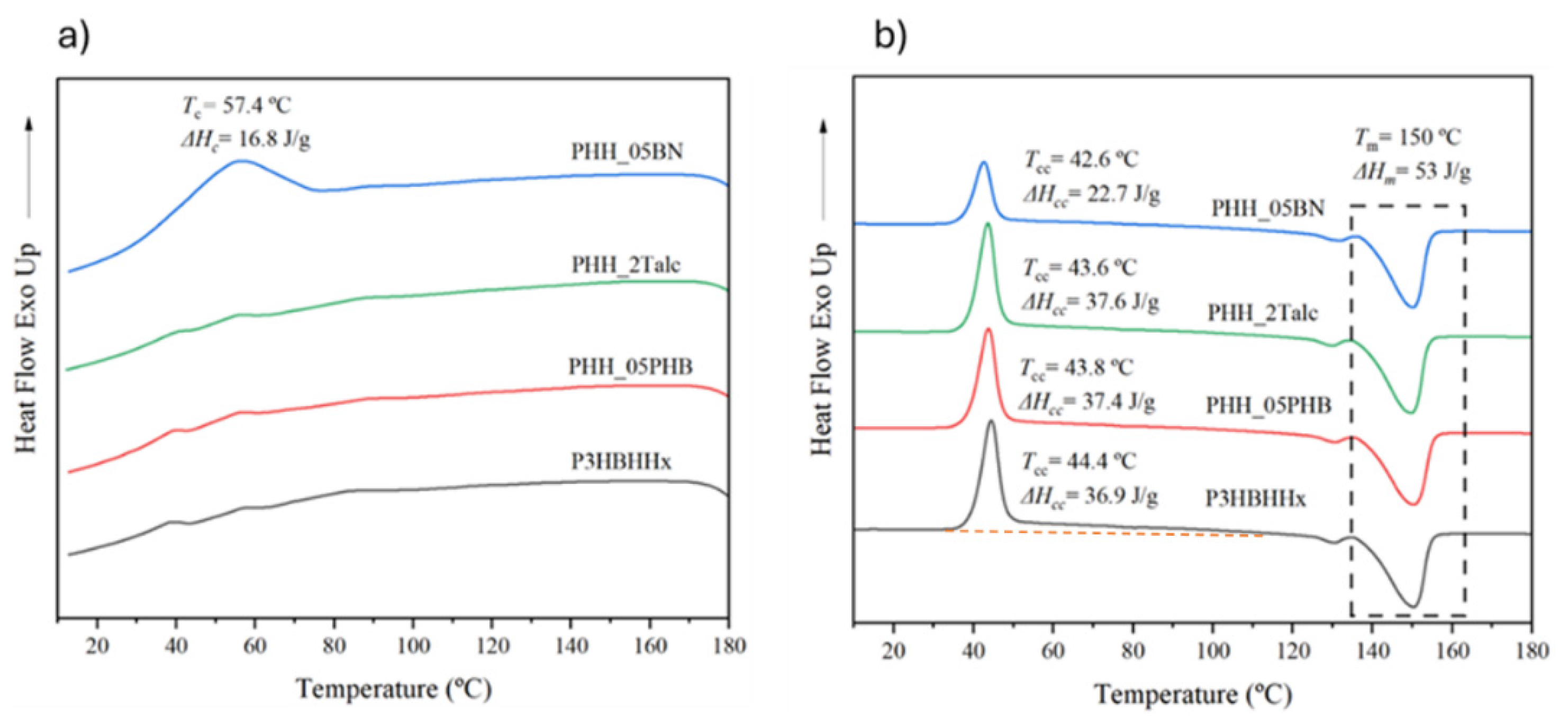
3.1.1. Thermal Behavior of neat P3HBHHx
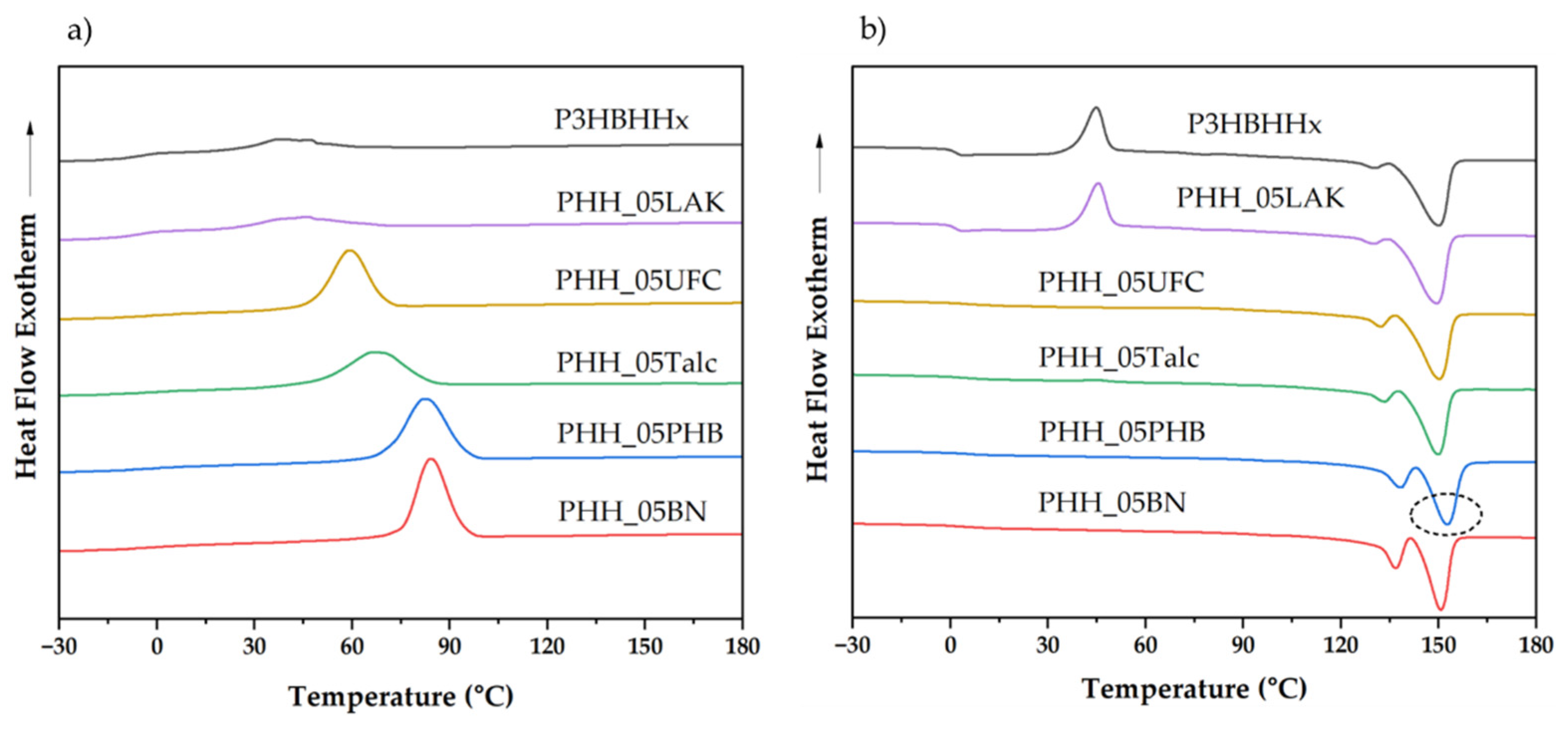
3.1.2. Effect of Nucleating Agents on Non-Isothermal Crystallization of P3HBHHx
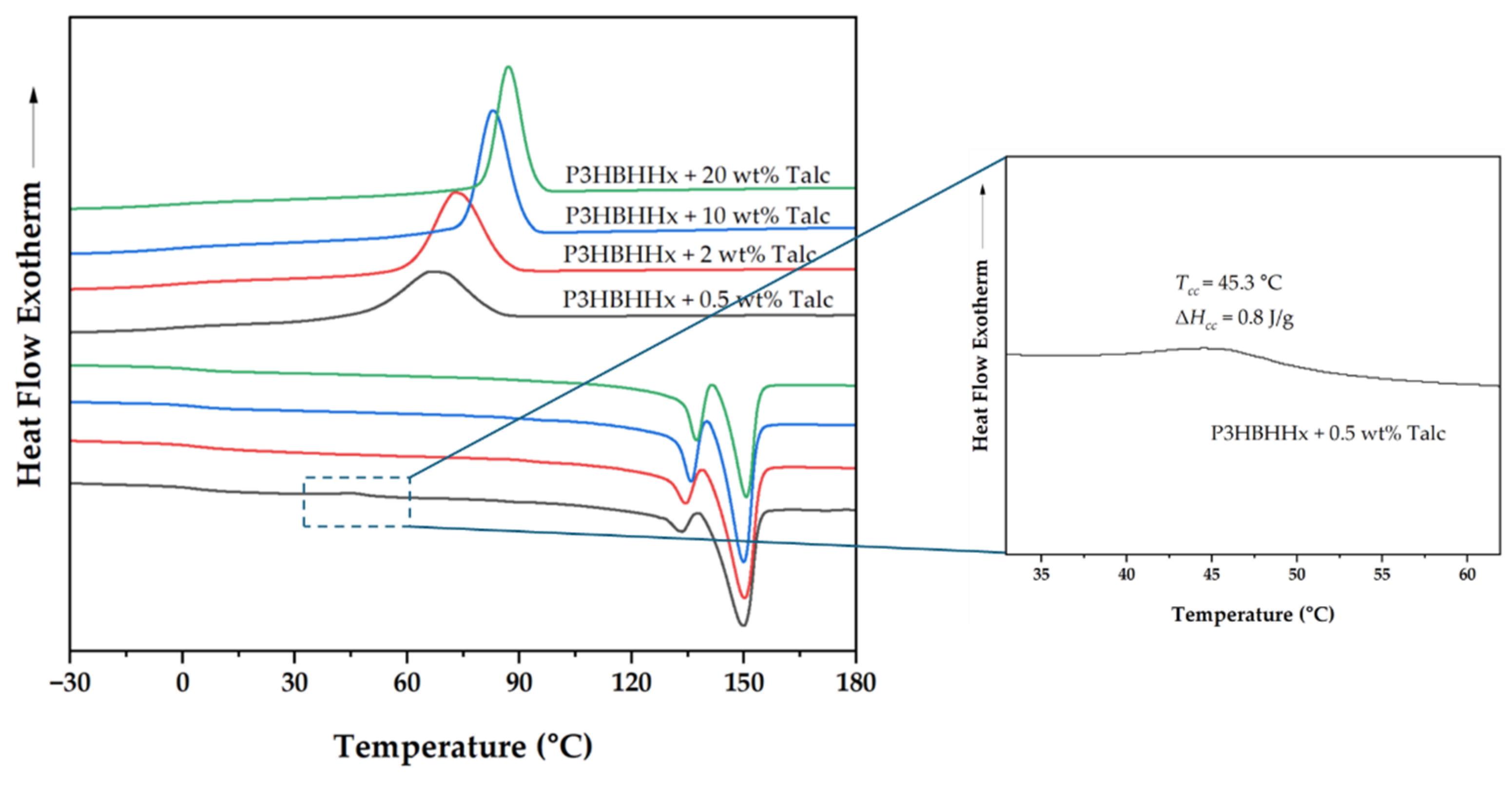
3.1.3. Effect of Increased Nucleating Agent Concentration on the Crystallization Behavior of P3HBHHx
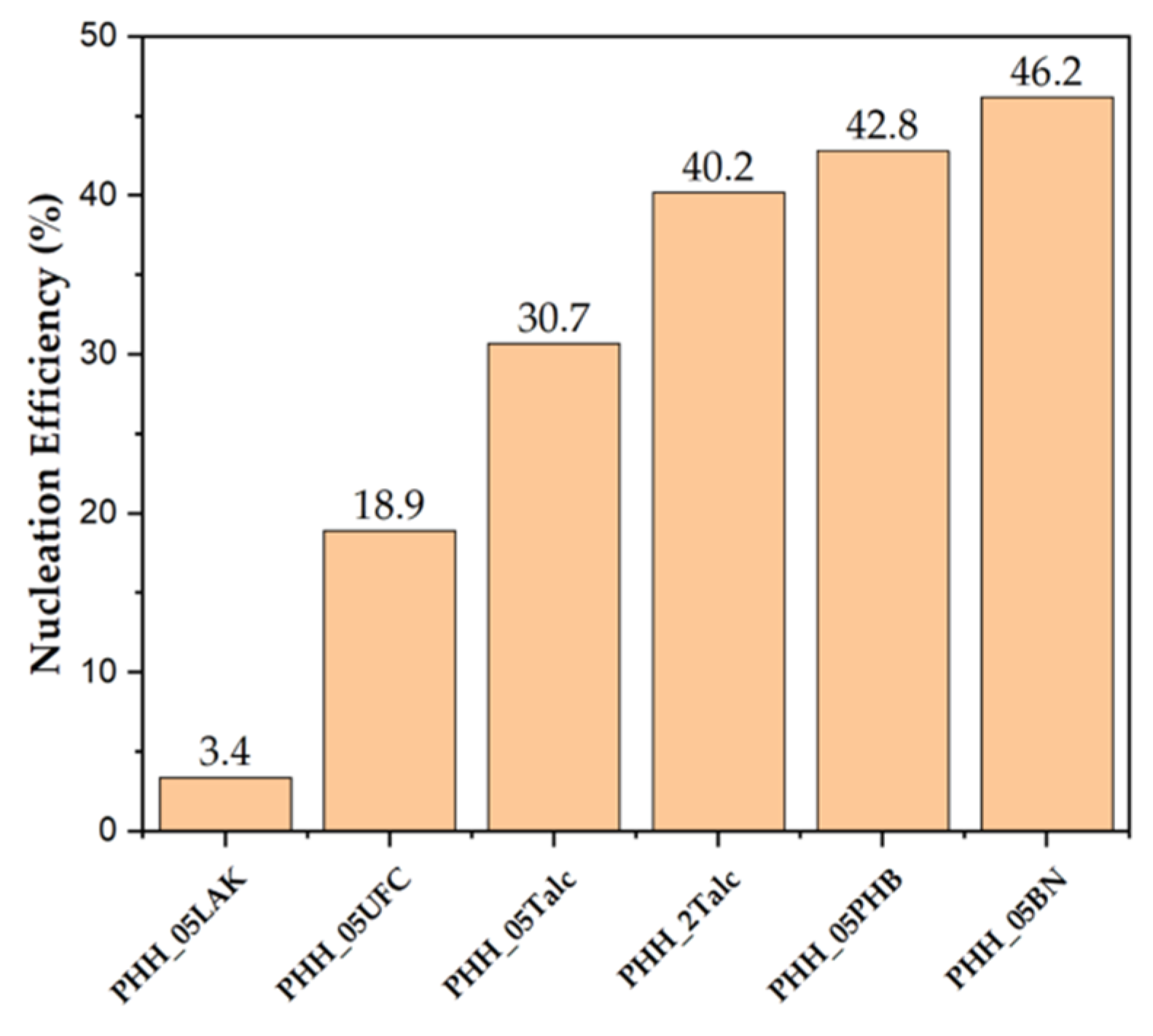
3.1.4. Nucleation Efficiency
3.2. Structural Comparison of P3HBHHx with Different Nucleating Agents
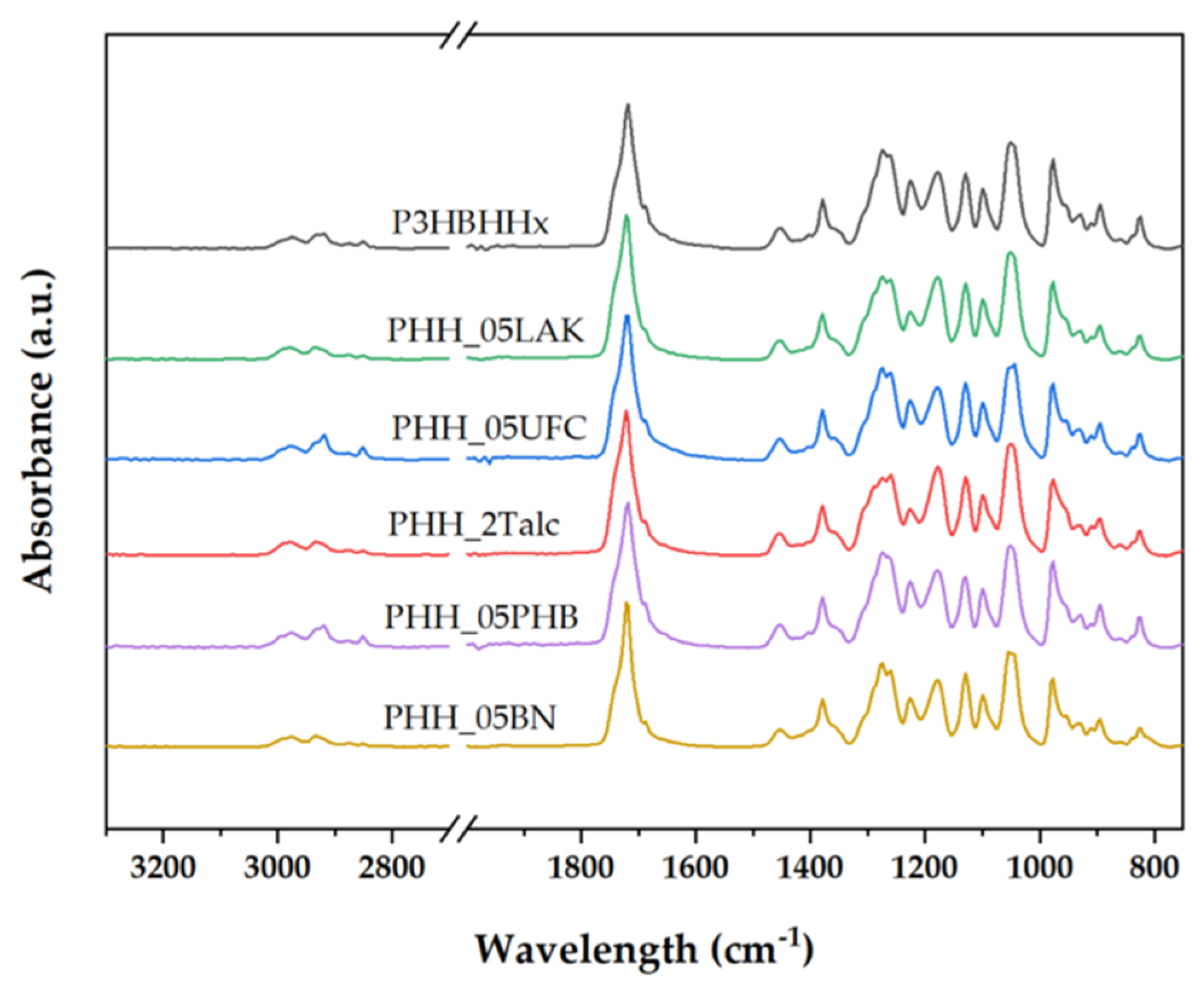
3.2.1. FTIR Analysis
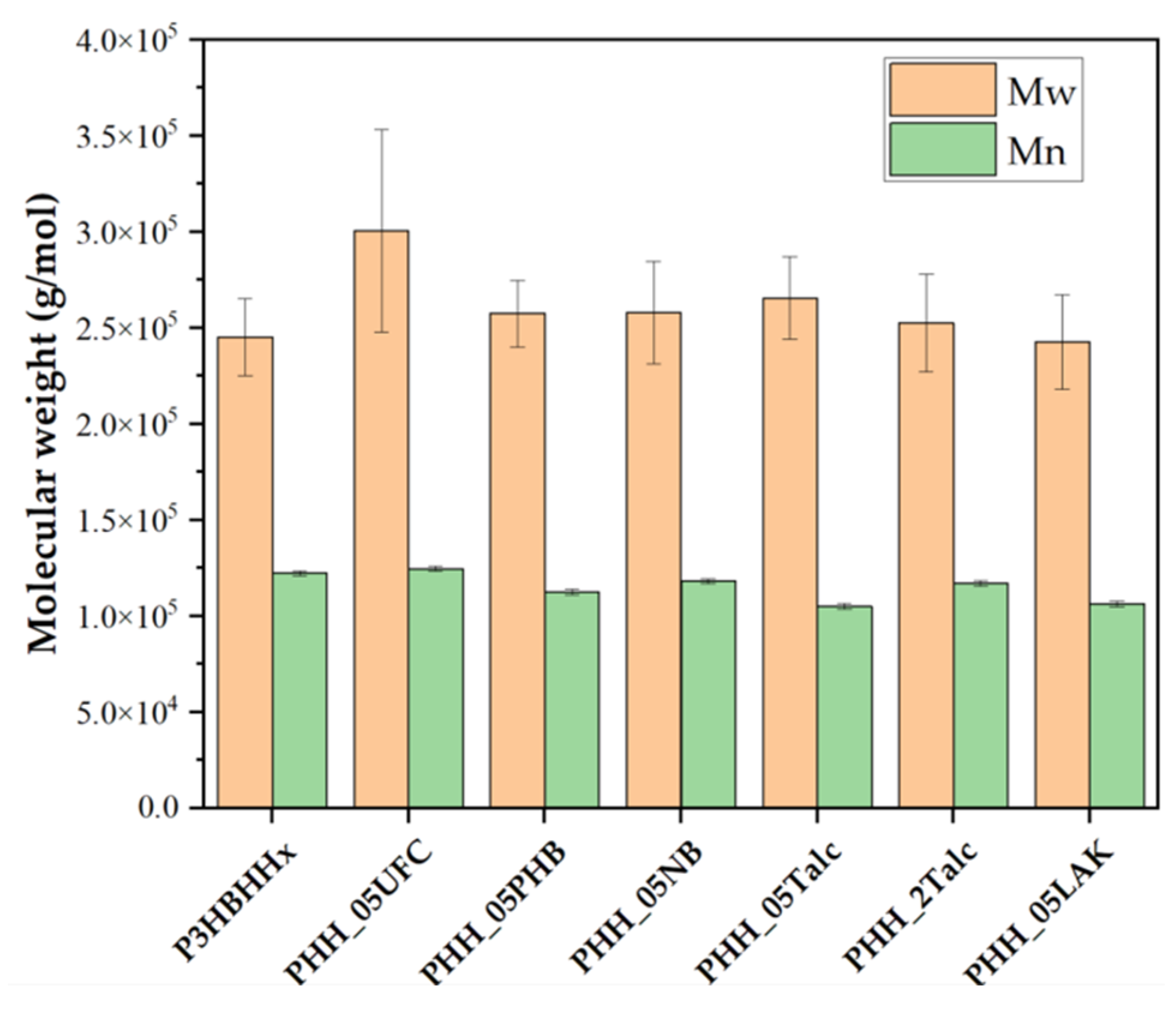
3.2.2. Degradation of Nucleated Samples During Processing
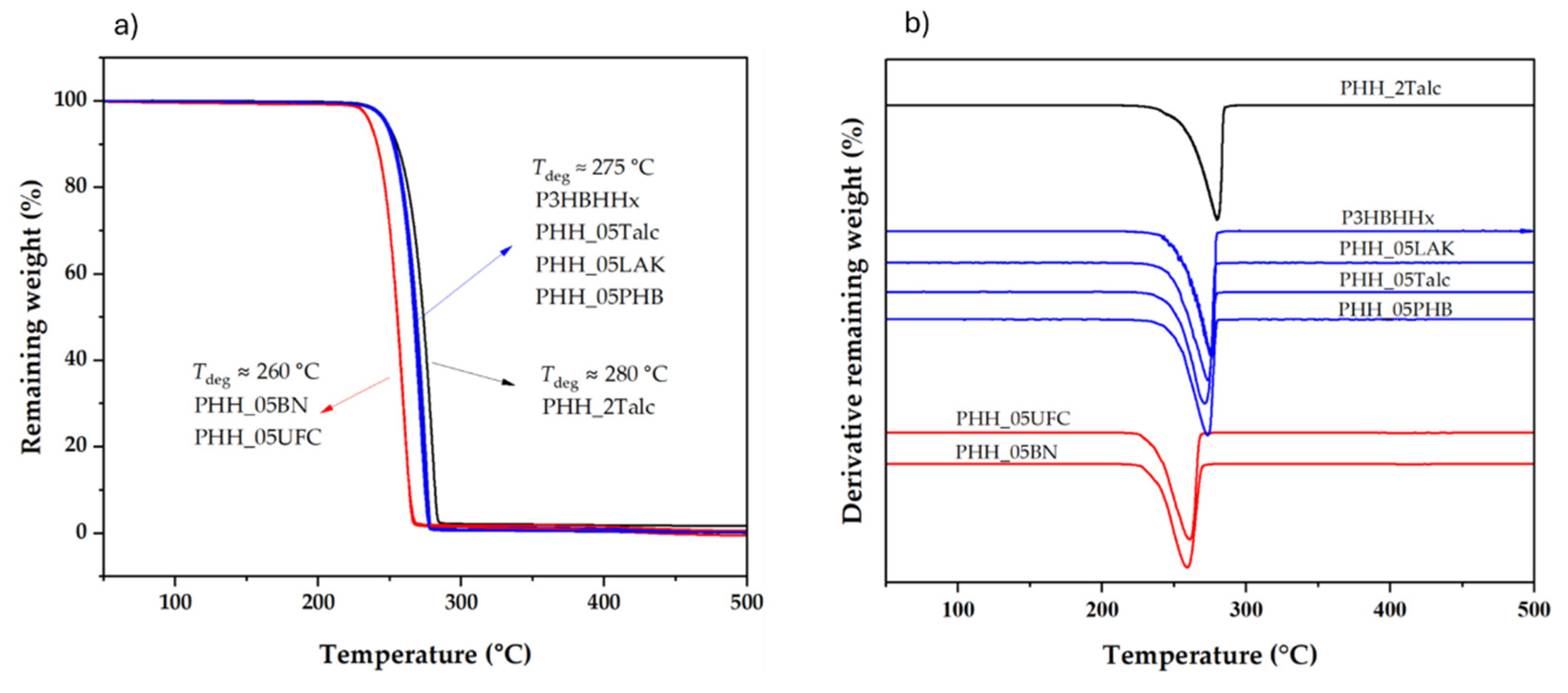
3.2.3. Influence of Nucleating Agents on the Thermal Stability of P3HBHHx
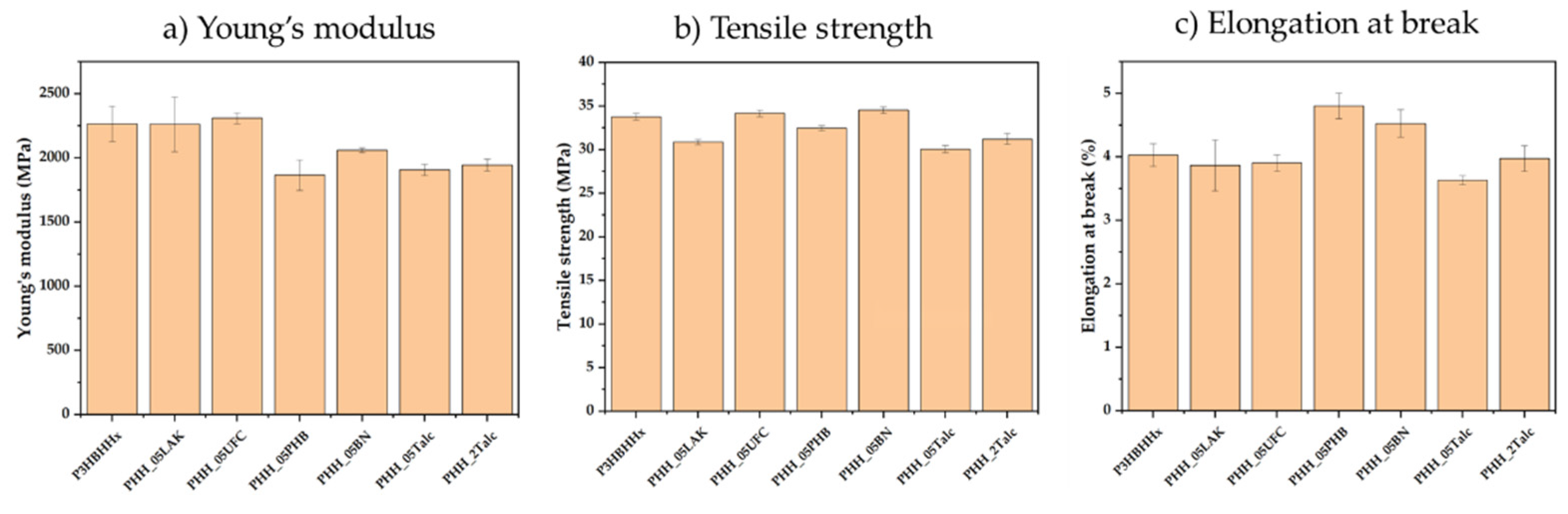
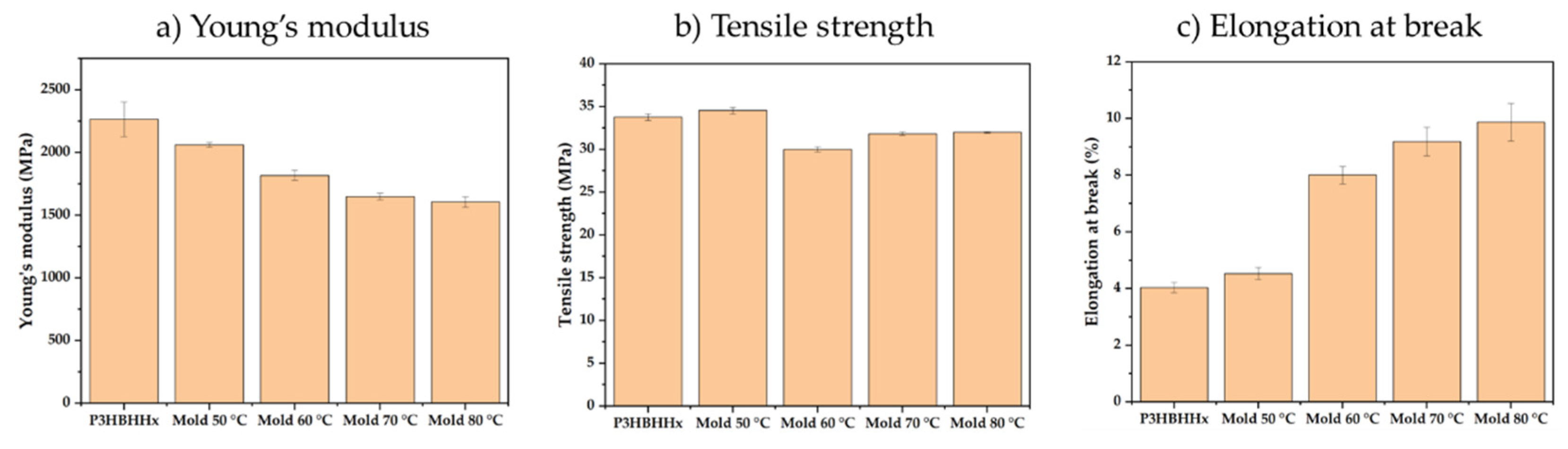
3.2.4. Mechanical Testing
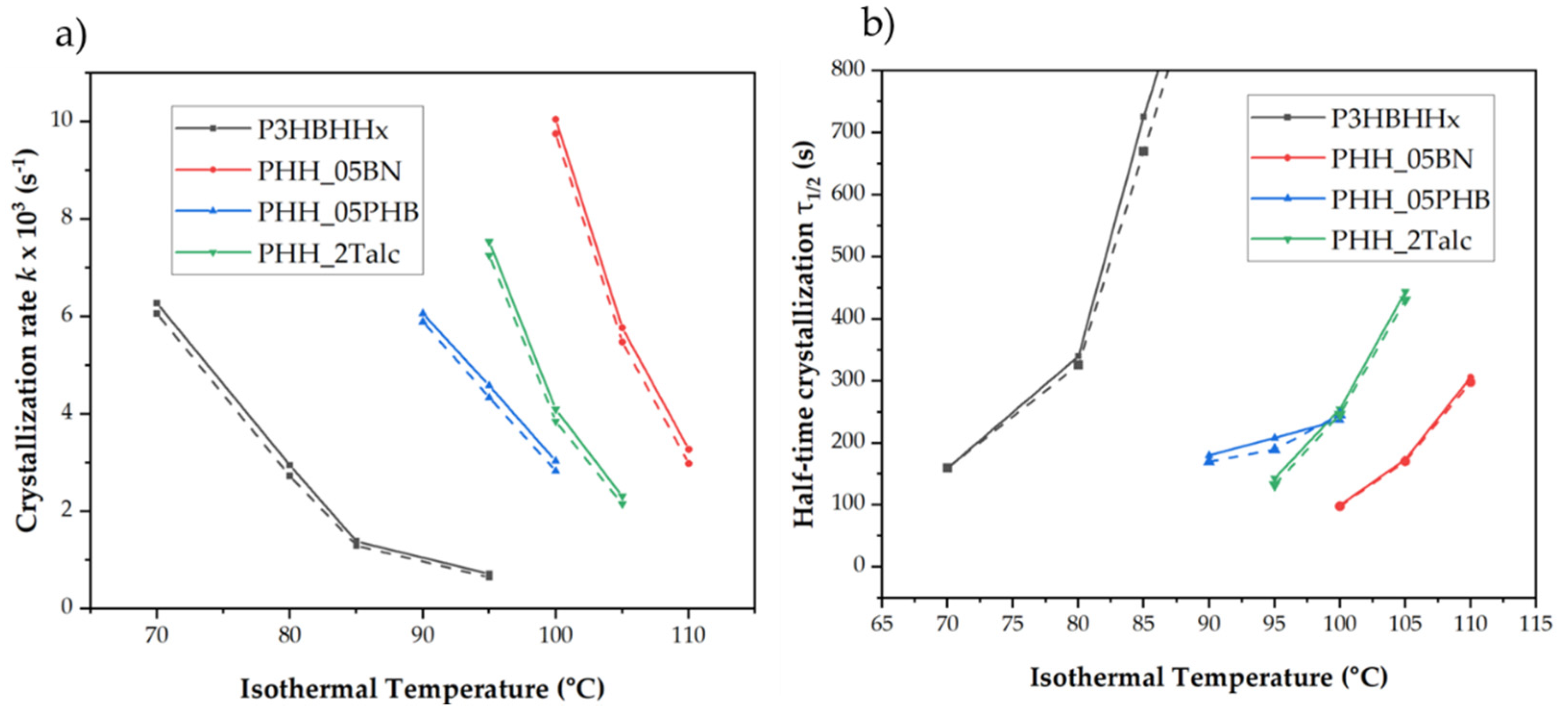
3.3. Isothermal Crystallization and Kinetic Analysis
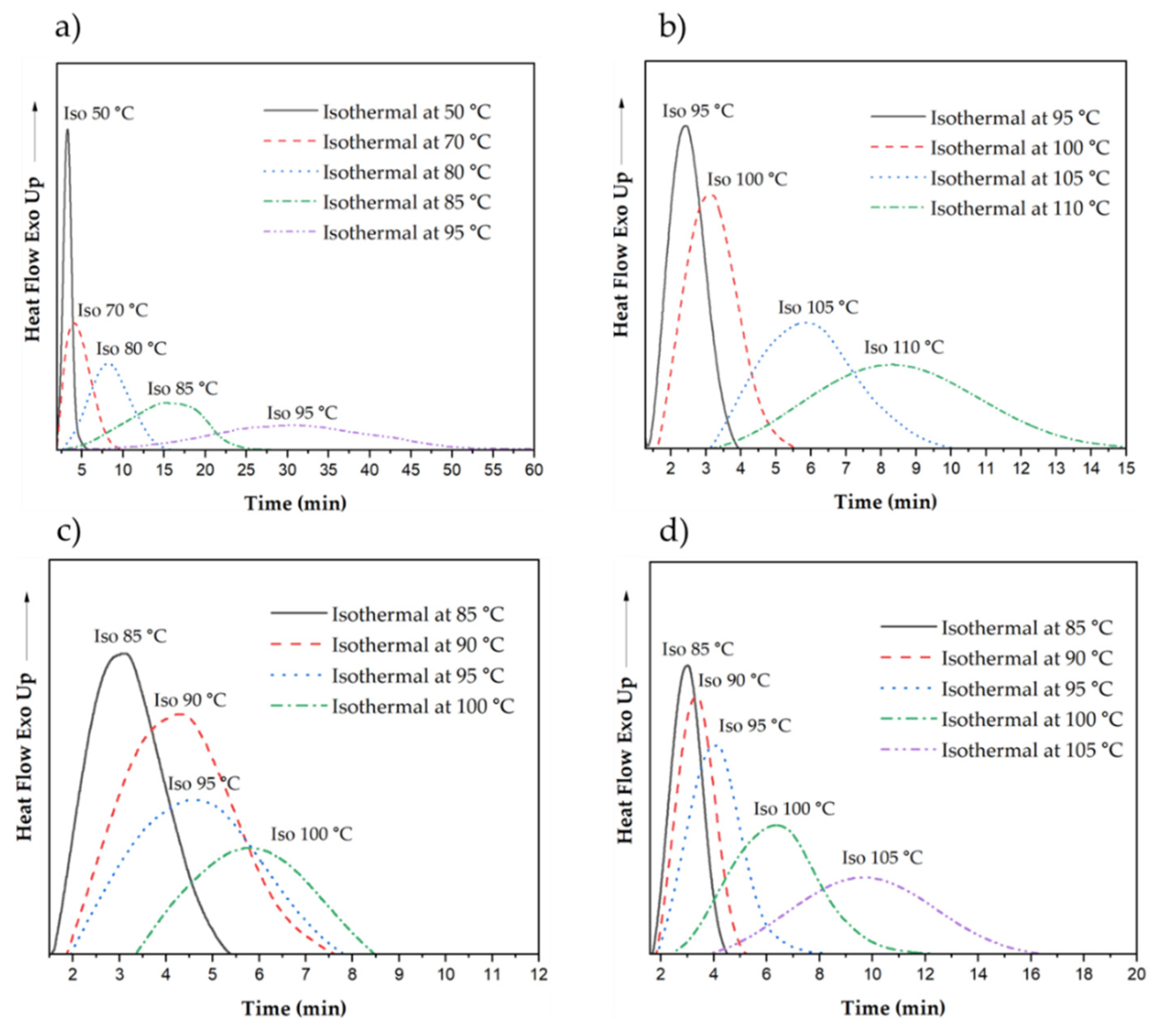
3.3.1. Crystallization of Neat and Nucleated P3HBHHx at Different Isothermal Temperatures
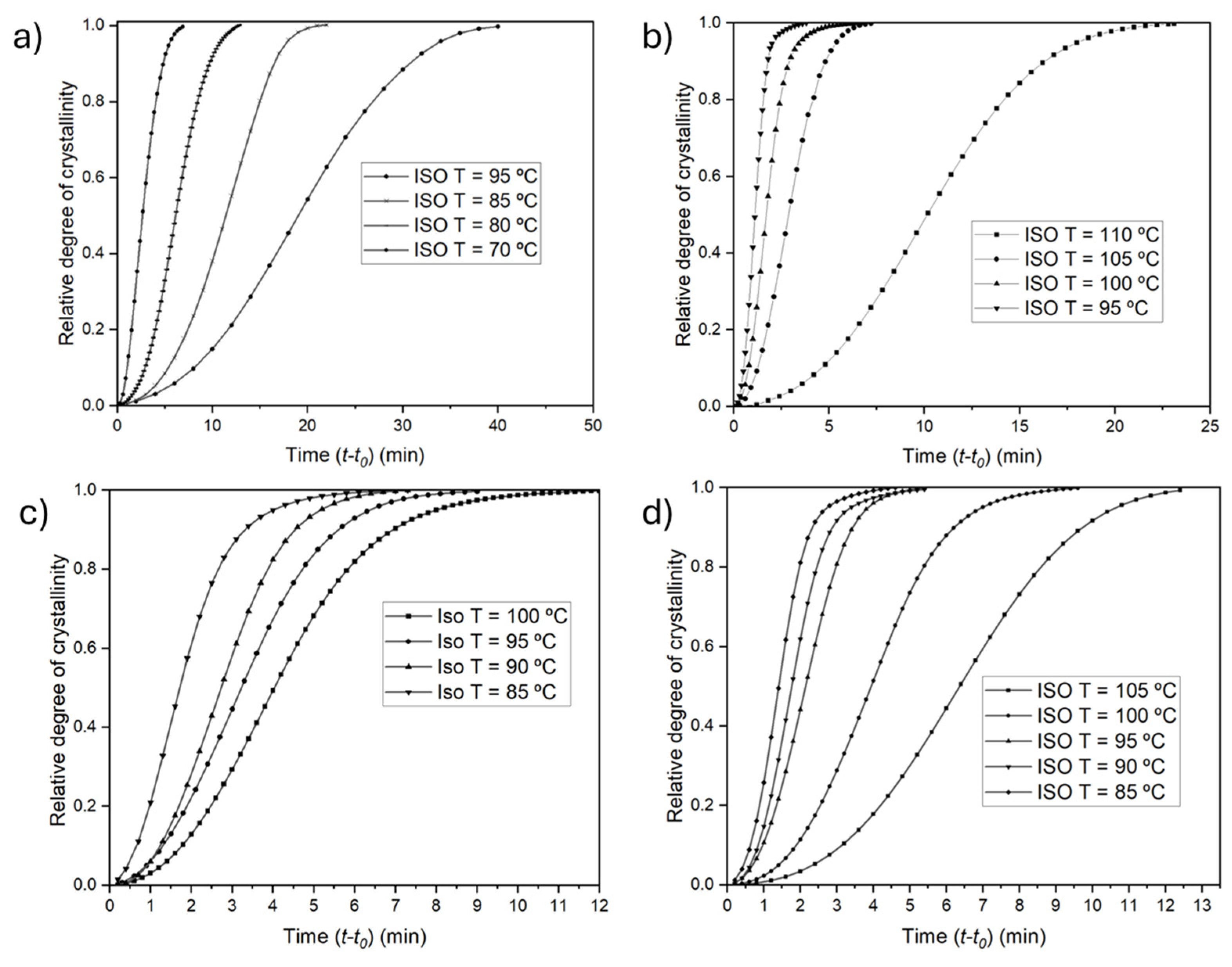
3.3.2. Evolution of the Relative Crystallinity over Time
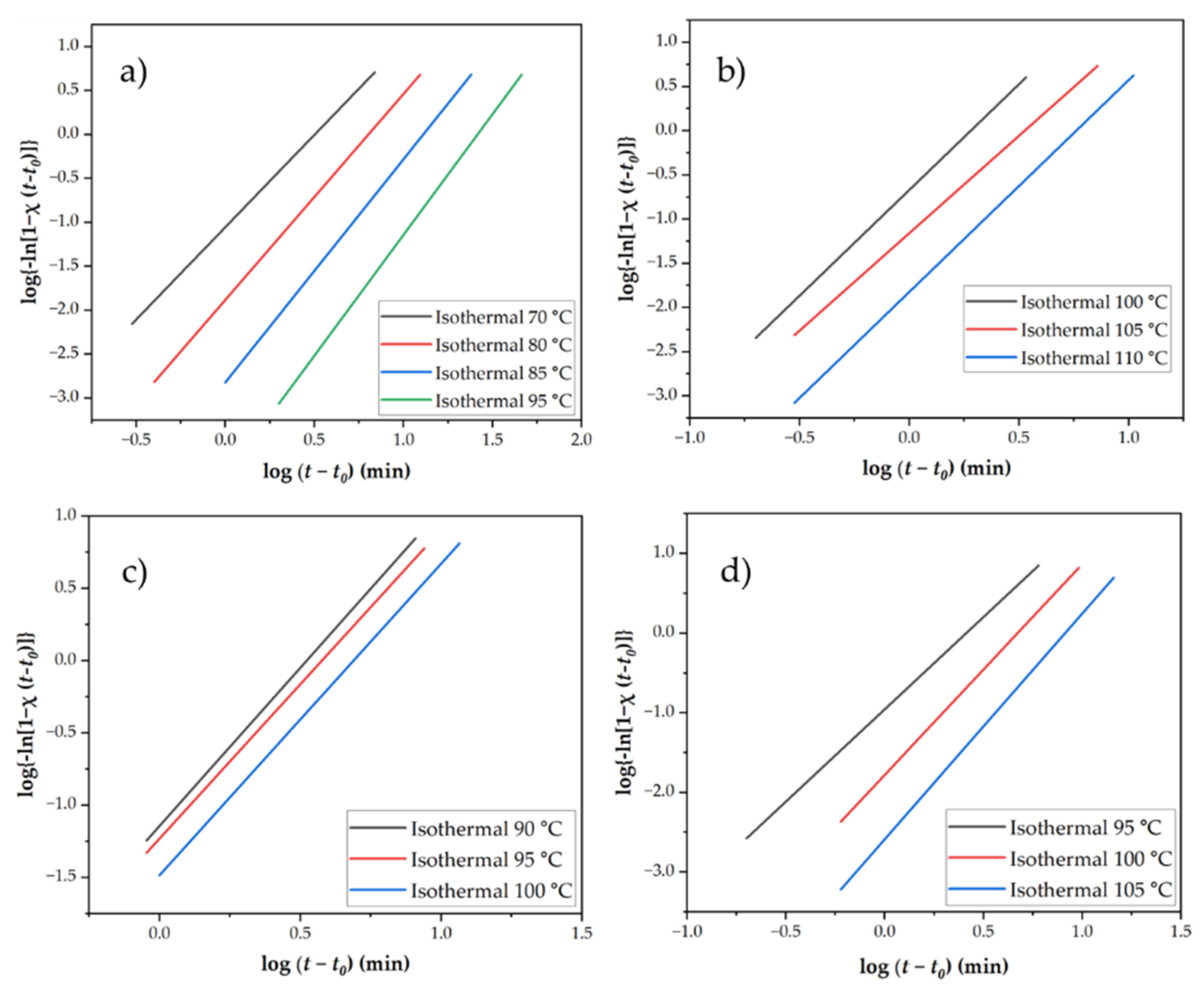
3.3.3. Avrami Modeling of Crystallization Kinetics
3.3.4. Analysis of Avrami plots
3.4. Spherulitic Morphology and Crystal Growth of P3HBHHx Samples
3.4.1. Spherulitic Morphology
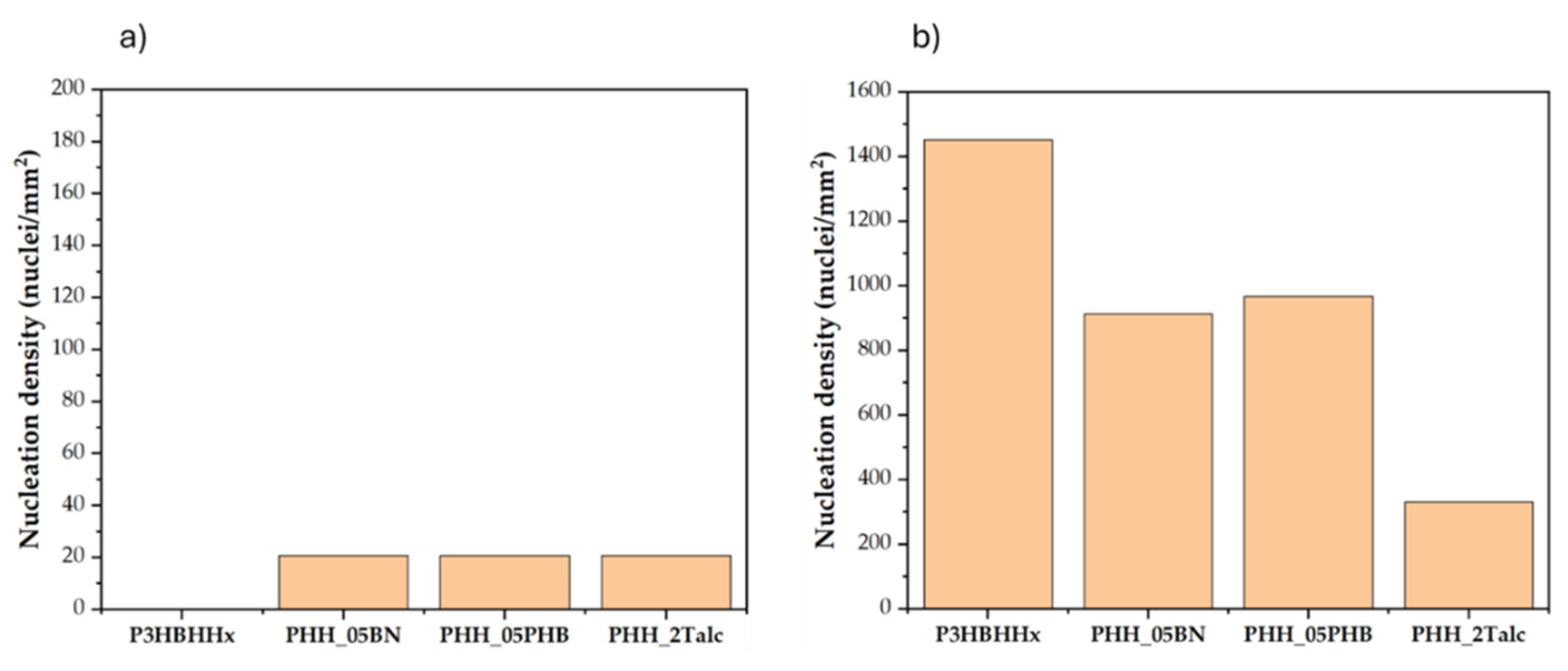
3.4.2. Nucleation Density
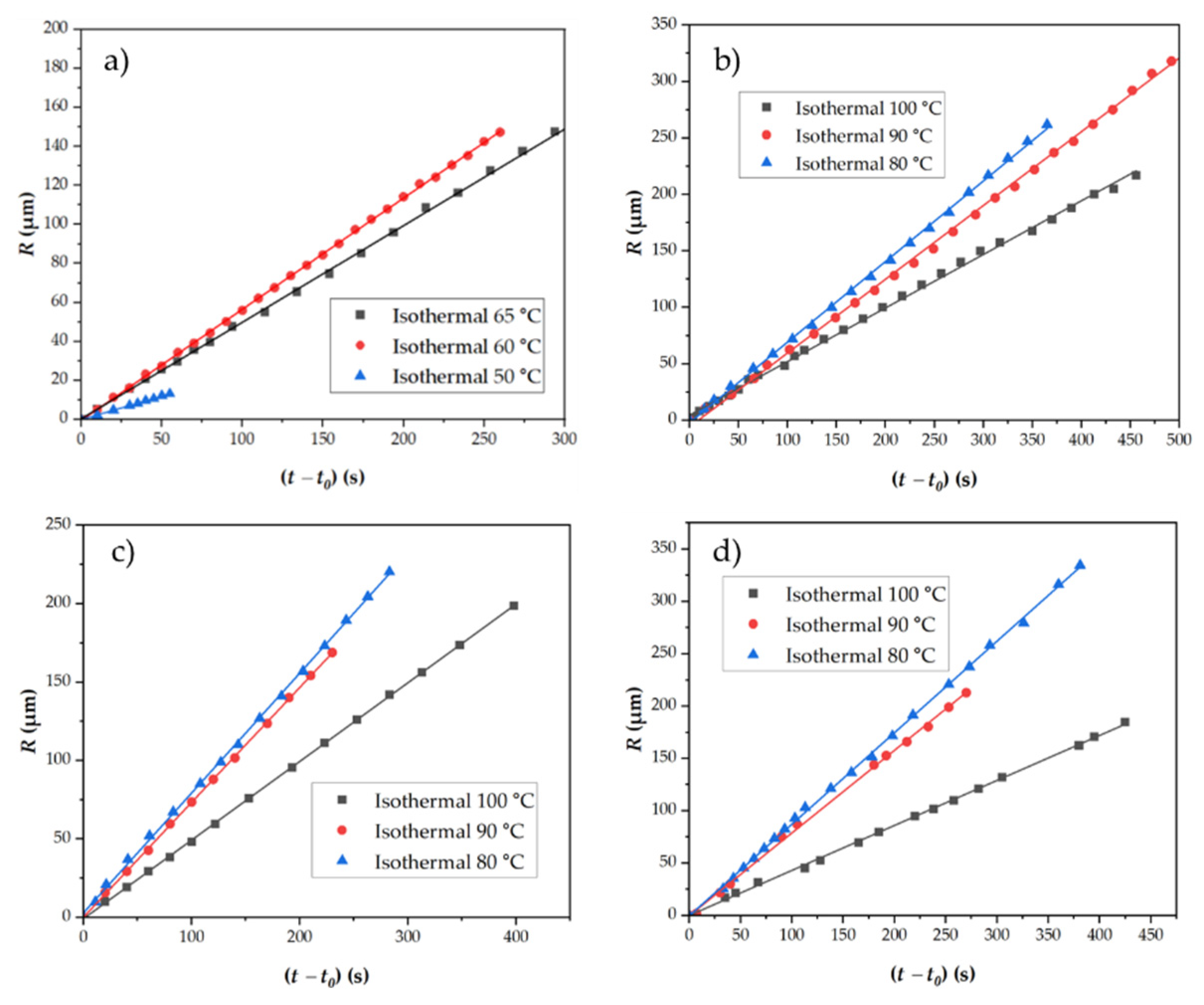
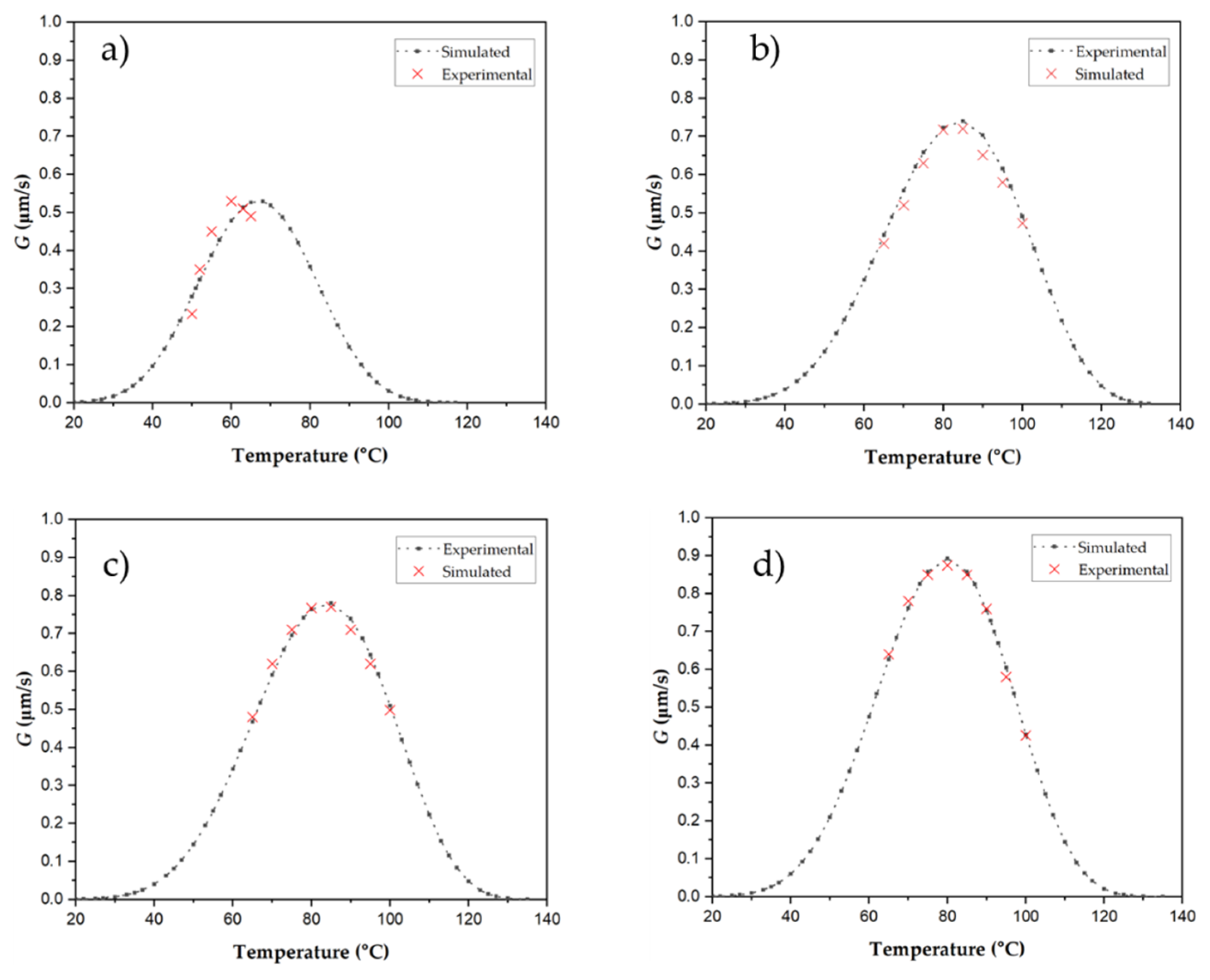
3.4.3. Crystal Growth Rates Determined from POM
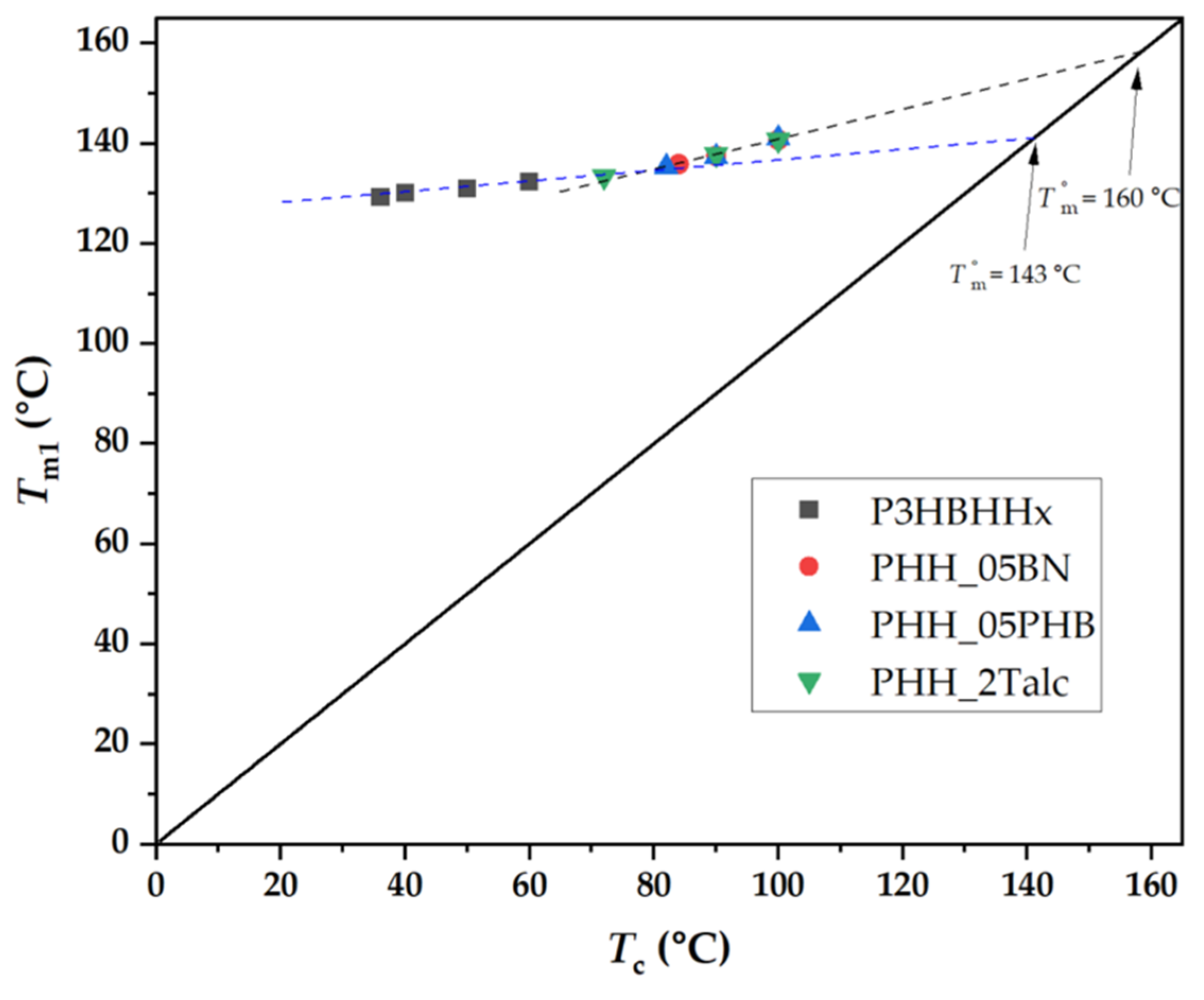
3.4.4. Determination of Equilibrium Melting Temperature ()
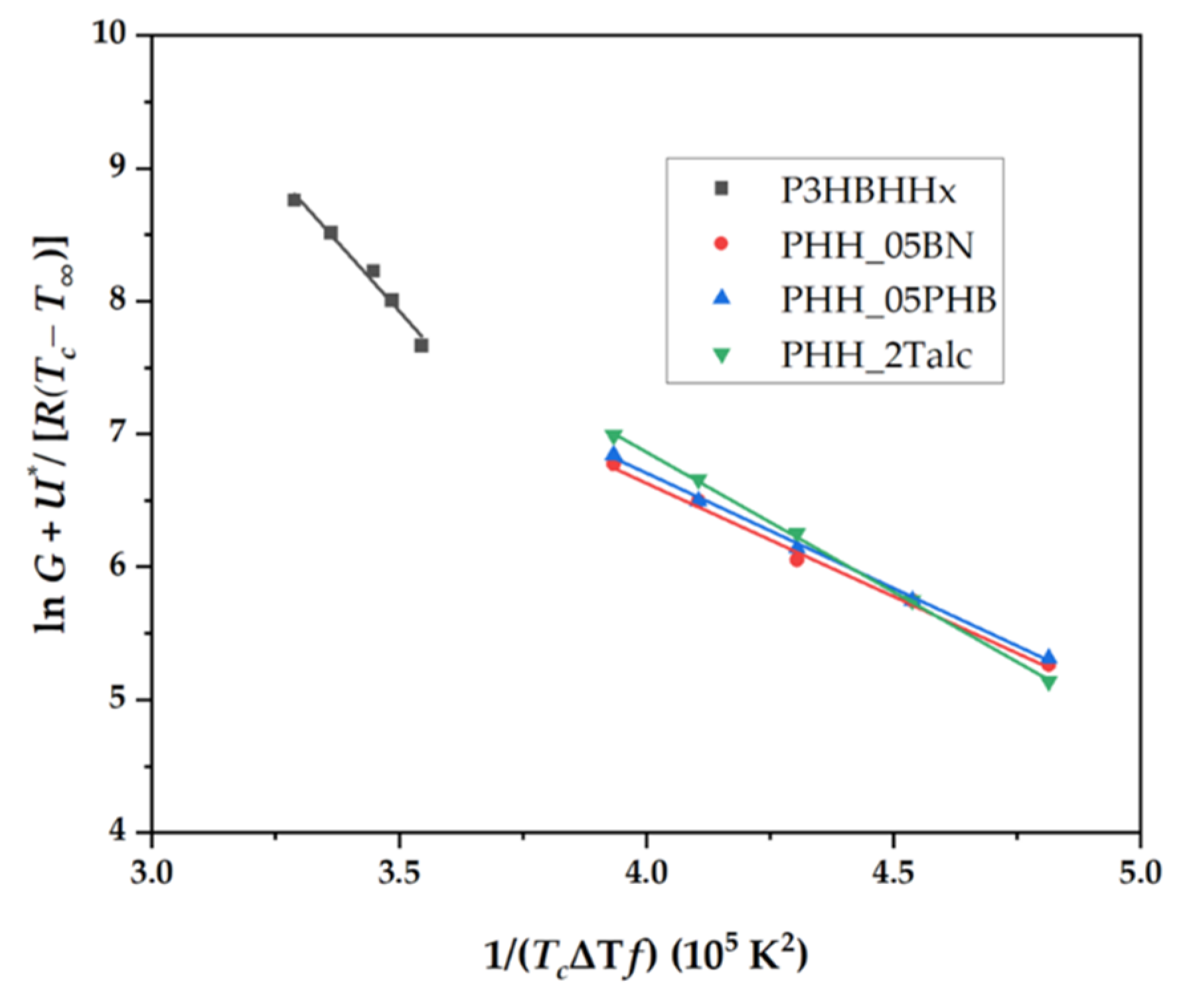
3.4.5. Kinetic Analysis Based on the Lauritzen-Hoffman Model
3.5. Fast Cooling Behavior of P3HBHHx and Nucleated Samples
3.6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| P3HBHHx | Poly(3-hydroxybutyrate-co-3-hydroxyhexanoate) |
| BN | Boron nitride |
| PHB | Poly(3-hydroxybutyrate) |
| DSC | Differential scanning calorimetry |
| POM | Polarized optical microscopy |
| TGA | Thermogravimetric analysis |
| PHAs | Polyhydroxyalkanoates |
| 3HHx | 3-hydroxyhexanoate |
| PLA | Polylactic acid |
| UFC | Ultrafine cellulose |
| GPC | Gel permeation chromatography |
| FTIR | Fourier-transform infrared |
References
- Chodak, I. Polyhydroxyalkanoates: Origin, Properties and Applications. In Monomers, Polymers and Composites from Renewable Resources; Belgacem, M.N., Gandini, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2008; pp. 451–477.
- Muthuraj, R.; Valerio, O.; Mekonnen, T.H. Recent Developments in Short- and Medium-Chain-Length Polyhydroxyalkanoates: Production, Properties, and Applications. Int. J. Biol. Macromol. 2021, 187, 422–440. [CrossRef]
- Raho, S.; Carofiglio, V.E.; Montemurro, M.; Miceli, V.; Centrone, D.; Stufano, P.; Schioppa, M.; Pontonio, E.; Rizzello, C.G. Production of the Polyhydroxyalkanoate PHBV from Ricotta Cheese Exhausted Whey by Haloferax Mediterranei Fermentation. Foods 2020, 9(10), 1459. [CrossRef]
- Shen, M.Y.; Chu, C.Y.; Sawatdeenarunat, C.; Bhuyar, P. Production, Downstream Processing, and Characterization of Polyhydroxyalkanoates (PHAs) Boosted by Pyruvate Supplement Using Mixed Microbial Culture (MMC) and Organic Wastewater. Biomass Convers. Biorefin. 2023, 13(17), 15861–15869. [CrossRef]
- Sabapathy, P.C.; Devaraj, S.; Meixner, K.; Anburajan, P.; Kathirvel, P.; Ravikumar, Y.; Zabed, H.M.; Qi, X. Recent Developments in Polyhydroxyalkanoates (PHAs) Production – A Review. Bioresour Technol. 2020, 306, 123132. [CrossRef]
- Serafim, L.S.; Lemos, P.C.; Albuquerque, M.G.E.; Reis, M.A.M. Strategies for PHA Production by Mixed Cultures and Renewable Waste Materials. Appl Microbiol Biotechnol 2008, 81, 615–628. [CrossRef]
- Salehizadeh, H.; Van Loosdrecht, M.C.M. Production of Polyhydroxyalkanoates by Mixed Culture: Recent Trends and Biotechnological Importance. Biotechnol Adv 2004, 22, 261–279. [CrossRef]
- Sandhya, M.; Aravind, J.; Kanmani, P. Production of Polyhydroxyalkanoates from Ralstonia Eutropha Using Paddy Straw as Cheap Substrate. International Journal of Environmental Science and Technology 2013, 10, 47–54. [CrossRef]
- Palmeiro-Sánchez, T., O’Flaherty, V., & Lens, P. N. Polyhydroxyalkanoate Bio-Production and Its Rise as Biomaterial of the Future. J Biotechnol. 2022, 348, 10–25.
- Poltronieri, P.; Kumar, P. Polyhydroxyalkanoates (PHAs) in Industrial Applications. In Handbook of Ecomaterials ; Martínez, L.M.T., Kharissova, O.V., Kharisov, B.I., Eds.; Springer: Cham, Switzerland, 2017; Volume 4, pp. 2843–2872.
- Yean, O. S., Yee, C. J., & Kumar, S. Degradation of Polyhydroxyalkanoate (PHA): A Review. Journal of Siberian Federal University. Biology 2017, 10, 21–225. [CrossRef]
- Li, Z., Yang, J., & Loh, X. J. Polyhydroxyalkanoates: Opening Doors for a Sustainable Future. NPG Asia Mater. 2016, 8(4), e265–e265. [CrossRef]
- Tang, H. J., Neoh, S. Z., & Sudesh, K. A Review on Poly(3-Hydroxybutyrate-Co-3-Hydroxyhexanoate) [P(3HB-Co-3HHx)] and Genetic Modifications That Affect Its Production. Front Bioeng Biotechnol. 2022, 10, 1057067. [CrossRef]
- Cai, H.; Qiu, Z. Effect of Comonomer Content on the Crystallization Kinetics and Morphology of Biodegradable Poly(3-Hydroxybutyrate-Co-3-Hydroxyhexanoate). Physical Chemistry Chemical Physics 2009, 11(41), 9569–9577. [CrossRef]
- Dong, T.; Mori, T.; Aoyama, T.; Inoue, Y. Rapid Crystallization of Poly(3-Hydroxybutyrate-Co-3-Hydroxyhexanoate) Copolymer Accelerated by Cyclodextrin-Complex as Nucleating Agent. Carbohydr Polym. 2010, 80(2), 387–393. [CrossRef]
- Yu, H.-Y.; Qin, Z.-Y.; Zhou, Z. Cellulose Nanocrystals as Green Fillers to Improve Crystallization and Hydrophilic Property of Poly(3-Hydroxybutyrate-Co-3-Hydroxyvalerate). Prog. Nat. Sci. Mater. Int. 2011, 21(6), 478–484. [CrossRef]
- Xu, P.; Cao, Y.; Lv, P.; Ma, P.; Dong, W.; Bai, H.; Wang, W.; Du, M.; Chen, M. Enhanced Crystallization Kinetics of Bacterially Synthesized Poly(3-Hydroxybutyrate-Co-3-Hydroxyhexanate) with Structural Optimization of Oxalamide Compounds as Nucleators. Polym Degrad Stab 2018, 154, 170–176. [CrossRef]
- Jacquel, N.; Tajima, K.; Nakamura, N.; Kawachi, H.; Pan, P.; Inoue, Y. Nucleation Mechanism of Polyhydroxybutyrate and Poly(Hydroxybutyrate-Co- Hydroxyhexanoate) Crystallized by Orotic Acid as a Nucleating Agent. J Appl Polym Sci 2010, 115(2), 709–715. [CrossRef]
- Qian, J.; Zhu, L.; Zhang, J.; Whitehouse, R.S. Comparison of Different Nucleating Agents on Crystallization of Poly(3-Hydroxybutyrate-Co-3-Hydroxyvalerates). J. Polym. Sci. B Polym. Phys. 2007, 45(13), 1564–1577. [CrossRef]
- Feng, Y.; Ma, P.; Xu, P.; Wang, R.; Dong, W.; Chen, M.; Joziasse, C. The Crystallization Behavior of Poly(Lactic Acid) with Different Types of Nucleating Agents. Int J Biol Macromol 2018, 106, 955–962. [CrossRef]
- Zou, G.X.; Jiao, Q.W.; Zhang, X.; Zhao, C.X.; Li, J.C. Crystallization Behavior and Morphology of Poly(Lactic Acid) with a Novel Nucleating Agent. J Appl Polym Sci 2015, 132(5). [CrossRef]
- Fillon, B., Wittmann, J. C., Lotz, B., & Thierry, A. Self-nucleation and recrystallization of isotactic polypropylene (α phase) investigated by differential scanning calorimetry. J. Polym. Sci. B Polym. Phys. 1993, 31(10), 1383-1393. [CrossRef]
- Fillon, B., Lotz, B., Thierry, A., & Wittmann, J. C. Self-nucleation and enhanced nucleation of polymers. Definition of a convenient calorimetric “efficiency scale” and evaluation of nucleating additives in isotactic polypropylene (α phase). J. Polym. Sci. B Polym. Phys. 1993, 31(10), 1395-1405. [CrossRef]
- Yang, H.X.; Sun, M.; Zhou, P. Investigation of Water Diffusion in Poly(3-Hydroxybutyrate-Co-3-Hydroxyhexanoate) by Generalized Two-Dimensional Correlation ATR-FTIR Spectroscopy. Polymer 2009, 50(6), 1533–1540. [CrossRef]
- Cichosz, S.; Masek, A. Cellulose Fibers Hydrophobization via a Hybrid Chemical Modification. Polymers (Basel) 2019, 11(7), 1174. [CrossRef]
- Wu, X.; Han, Q. Thermal Conductivity of Monolayer Hexagonal Boron Nitride: From Defective to Amorphous. Comput Mater Sci 2020, 184, 109938. [CrossRef]
- Zhang, L.; Hsieh, Y. Lo Ultrafine Cellulose Acetate Fibers with Nanoscale Structural Features. J Nanosci Nanotechnol 2008, 8(9), 4461–4469. [CrossRef]
- Avrami, M. Kinetics of Phase Change. I: General Theory. J Chem Phys 1939, 7(12), 1103–1112. [CrossRef]
- Avrami, M. Kinetics of Phase Change. II Transformation-Time Relations for Random Distribution of Nuclei. J Chem Phys 1940, 8(2), 212–224. [CrossRef]
- Crist, B.; Schultz, J.M. Polymer Spherulites: A Critical Review. Prog Polym Sci 2016, 56, 1–63. [CrossRef]
- Xu, J.; Ye, H.; Zhang, S.; Guo, B. Organization of Twisting Lamellar Crystals in Birefringent Banded Polymer Spherulites: A Mini-Review. Crystals (Basel) 2017, 7(8), 241. [CrossRef]
- Nagarajan, S.; Widyantari, R.; Chuang, W.T.; Lin, J.M.; Woo, E.M. Grating Harmony of Orderly-Banded Spherulites of Poly(3-Hydroxybutyrate-Co-3-Hydroxyvalerate) with Tunable Structures. Polymer (Guildf) 2025, 317, 127892. [CrossRef]
- Lotz, B.; Cheng, S.Z.D. A Critical Assessment of Unbalanced Surface Stresses as the Mechanical Origin of Twisting and Scrolling of Polymer Crystals. Polymer (Guildf) 2005, 46(3), 577–610. [CrossRef]
- Owen, A. J., Heinzel, J., Škrbić, Ž., & Divjaković, V. Crystallization and melting behaviour of PHB and PHB/HV copolymer. Polymer 1992, 33(7), 1563-1567. [CrossRef]
- Keridou, I.; Del Valle, L.J.; Funk, L.; Turon, P.; Franco, L.; Puiggalí, J. Non-Isothermal Crystallization Kinetics of Poly(4-Hydroxybutyrate) Biopolymer. Molecules 2019, 24. [CrossRef]
- Lauritzen, J.I.; Hoffman, J.D. Extension of Theory of Growth of Chain-Folded Polymer Crystals to Large Undercoolings. J Appl Phys 1973, 44, 4340–4352. [CrossRef]
- Chen, C.; Cheung, M.K.; Yu, P.H.F. Crystallization Kinetics and Melting Behaviour of Microbial Poly(3-Hydroxybutyrate-Co-3-Hydroxyhexanoate). Polym Int 2005, 54, 1055–1064. [CrossRef]
- Suzuki, T.; Kovacs, A.J. Temperature dependence of spherulitic growth rate of isotactic polystyrene. A critical comparison with the kinetic theory of surface nucleation. Polymer Journal 1970, 1(1), 82-100. [CrossRef]

| Sample code | Nucleating agent | Concentration (wt%) |
|---|---|---|
| P3HBHHx | None | 0 |
| PHH_05BN / PHH_2BN | Boron nitride | 0.5 / 2 |
| PHH_05PHB / PHH_2PHB | P3HB | 0.5 / 2 |
| PHH_05Talc / PHH_2Talc | Talc | 0.5 / 2 |
| PHH_05LAK / PHH_2LAK | Organic potassium salt | 0.5 / 2 |
| PHH_05UFC / PHH_2UFC | Ultrafine cellulose | 0.5 / 2 |
| Samples | Tc (˚C) | ΔHc (J/g) | Tg (˚C) | Tcc (˚C) | ΔHcc (˚C) | Tm1 (˚C) | Tm2 (˚C) | ΔHf (J/g) |
|---|---|---|---|---|---|---|---|---|
| P3HBHHx | 36.4 | 8.9 | 1.4 | 44.7 | 44.6 | 130.0 | 150.2 | 55.6 |
| PHH_05LAK | 45.6 | 18.3 | 1.4 | 45.5 | 38.4 | 129.8 | 149.5 | 58.0 |
| PHH_05UFC | 58.9 | 48.5 | 4.8 | - | - | 132.1 | 150.2 | 56.5 |
| PHH_05Talc | 66.5 | 46.3 | 3.7 | 45.3 | 0.8 | 133.2 | 149.9 | 49.7 |
| PHH_05PHB | 81.9 | 59.5 | 4.4 | - | - | 138.1 | 152.7 | 60.0 |
| PHH_05BN | 84.1 | 56.5 | 3.3 | - | - | 136.6 | 150.7 | 56.0 |
| Tc (˚C)a | NE (%)b | |
| P3HBHHx | 54.5 | - |
| PHH_05LAK | 57.0 | 3.4 |
| PHH_05UFC | 68.3 | 18.9 |
| PHH_05Talc | 76.9 | 30.7 |
| PHH_2Talc | 83.9 | 40.2 |
| PHH_05PHB | 85.8 | 42.8 |
| PHH_05BN | 88.3 | 46.2 |
|
Tdeg (˚C) |
T5% (˚C) |
T50% (˚C) |
T90% (˚C) |
Residue at 450 ˚C (wt%) | |
|---|---|---|---|---|---|
| P3HBHHx | 275.7 | 246.9 | 269.1 | 276.7 | 0 |
| PHH_05LAK | 273.5 | 246.8 | 267.3 | 275.1 | 0.57 |
| PHH_05UFC | 261.1 | 236.3 | 255.4 | 263.6 | 0.50 |
| PHH_05PHB | 273.6 | 247.1 | 267.8 | 275.3 | 0.52 |
| PHH_05BN | 258.3 | 236.1 | 255.2 | 263.8 | 0.58 |
| PHH_05Talc | 271.4 | 246.7 | 266.9 | 274.6 | 0.53 |
| PHH_2Talc | 279.5 | 247.9 | 264.3 | 281.7 | 1.76 |
| Sample | Young’s modulus (MPa) |
Tensile Strength (MPa) | Elongation at Break (%) |
|---|---|---|---|
| P3HBHHx | 2264 ± 138 | 33.7 ± 0.4 | 4.0 ± 0.2 |
| PHH_05LAK | 2260 ± 213 | 30.8 ± 0.2 | 3.8 ± 0.1 |
| PHH_05UFC | 2309 ± 42 | 34.1 ± 0.4 | 3.9 ± 0.1 |
| PHH_05PHB | 1866 ± 119 | 32.4 ± 0.3 | 4.8 ± 0.2 |
| PHH_05BN | 2060 ± 19 | 34.5 ± 0.4 | 4.5 ± 0.2 |
| PHH_05Talc | 1906 ± 43 | 30.0 ± 0.4 | 3.6 ± 0.1 |
| PHH_2Talc | 1943 ± 46 | 31.2 ± 0.6 | 3.9 ± 0.2 |
| Sample | Isothermal Temperature (˚C) | Enthalpy of Crystallization (J/g) | Half-Time of Crystallization τ(1/2) (s) | Crystallization rate k x103(s-1) |
|---|---|---|---|---|
| P3HBHHx | 50 | 41.5 | 77 | 13.06 |
| 70 | 55.1 | 159 | 6.27 | |
| 80 | 60.3 | 340 | 2.94 | |
| 85 | 60.1 | 726 | 1.38 | |
| 95 | 60.8 | 1404 | 0.71 | |
| PHH_05BN | 95 | 48.1 | 68 | 14.67 |
| 100 | 64.3 | 99 | 10.05 | |
| 105 | 66.5 | 173 | 5.77 | |
| 110 | 66.7 | 306 | 3.27 | |
| PHH_05PHB | 85 | 46.9 | 104 | 9.66 |
| 90 | 59.4 | 165 | 6.06 | |
| 95 | 60.2 | 193 | 5.18 | |
| 100 | 60.5 | 242 | 4.13 | |
| PHH_2Talc | 85 | 44.5 | 84 | 11.84 |
| 90 | 55.6 | 106 | 9.47 | |
| 95 | 58.0 | 133 | 7.53 | |
| 100 | 58.4 | 244 | 4.09 | |
| 105 | 60.3 | 434 | 2.31 |
| Sample | Tc (˚C) | n | Z x106 (s-n) |
k x103(s-1) Avrami | τ(1/2) (s) Avrami |
|---|---|---|---|---|---|
| P3HBHHx | 70 | 2.10 | 16.11 | 5.26 | 160 |
| 80 | 2.34 | 0.890 | 2.63 | 326 | |
| 85 | 2.54 | 0.046 | 1.29 | 670 | |
| 95 | 2.75 | 0.002 | 0.64 | 1366 | |
| PHH_05BN | 100 | 2.39 | 11.98 | 8.76 | 98 |
| 105 | 2.20 | 8.38 | 4.98 | 170 | |
| 110 | 2.39 | 0.87 | 2.88 | 298 | |
| PHH_05PHB | 90 | 2.29 | 5.21 | 4.95 | 172 |
| 95 | 2.06 | 14.43 | 4.47 | 187 | |
| 100 | 2.12 | 5.79 | 3.43 | 246 | |
| PHH_2Talc | 95 | 2.32 | 8.42 | 6.45 | 132 |
| 100 | 2.68 | 0.27 | 3.54 | 246 | |
| 105 | 2.88 | 0.02 | 2.04 | 431 |
| Sample | P3HBHHx | G (µm/s) | Induction time (s) |
|---|---|---|---|
| P3HBHHx | 50 | 0.23 | 1 |
| 60 | 0.57 | 45 | |
| 65 | 0.49 | 50 | |
| PHH_05BN | 80 | 0.71 | 1 |
| 90 | 0.65 | 71 | |
| 100 | 0.48 | 150 | |
| PHH_05PHB | 80 | 0.76 | 1 |
| 90 | 0.73 | 22 | |
| 100 | 0.50 | 80 | |
| PHH_2Talc | 80 | 0.87 | 1 |
| 90 | 0.79 | 75 | |
| 100 | 0.43 | 100 |
| Sample | Linear plot | R2 | Kg 10-5 (K2) |
| P3HBHHx | y = -4.097x + 22.295 | 0.9946 | 4.10 |
| PHH_05BN | y = -1.707x + 13.463 | 0.9924 | 1.71 |
| PHH_05PHB | y = -1.734x + 13.644 | 0.9957 | 1.73 |
| PHH_2Talc | y = -2.107x + 15.293 | 0.9996 | 2.11 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




