Submitted:
30 April 2025
Posted:
02 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. What Is the Role of Microglia in the Adult Brain?
3. Comparing Histological Methods for Interrogating Microglia Function After Pharmacological and Environmental Exposure
4. Microglia Mediate the Effects of Chronic Stress on the Brain
5. Microglia Respond to Psychostimulant Exposure
6. Chronic Stress Effects on Drug Seeking: Potential Microglia Involvement
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Ross, S.; Peselow, E. Co-occurring psychotic and addictive disorders: neurobiology and diagnosis. Clin Neuropharmacol 2012, 35, 235–243. [Google Scholar] [CrossRef]
- Sinha, R. Chronic Stress, Drug Use, and Vulnerability to Addiction. Annals of the New York Academy of Sciences 2008, 1141, 105–130. [Google Scholar] [CrossRef] [PubMed]
- Turner, R.J.; Lloyd, D.A. Cumulative adversity and drug dependence in young adults: racial/ethnic contrasts. Addiction 2003, 98, 305–315. [Google Scholar] [CrossRef] [PubMed]
- Mantsch, J.R.; Baker, D.A.; Funk, D.; Lê, A.D.; Shaham, Y. Stress-Induced Reinstatement of Drug Seeking: 20 Years of Progress. Neuropsychopharmacology 2016, 41, 335–356. [Google Scholar] [CrossRef] [PubMed]
- Shaham, Y.; Wit, H.d. Lost in Translation: CRF1 Receptor Antagonists and Addiction Treatment. Neuropsychopharmacology 2016, 41. [Google Scholar] [CrossRef]
- Kwako, L.E.; Spagnolo, P.A.; Schwandt, M.L.; Thorsell, A.; George, D.T.; Momenan, R.; Rio, D.E.; Huestis, M.; Anizan, S.; Concheiro, M.; et al. The corticotropin releasing hormone-1 (CRH1) receptor antagonist pexacerfont in alcohol dependence: a randomized controlled experimental medicine study. Neuropsychopharmacology 2015, 40, 1053–1063. [Google Scholar] [CrossRef]
- Askew, K.; Li, K.; Olmos-Alonso, A.; Garcia-Moreno, F.; Liang, Y.; Richardson, P.; Tipton, T.; Chapman, M.A.; Riecken, K.; Beccari, S.; et al. Coupled Proliferation and Apoptosis Maintain the Rapid Turnover of Microglia in the Adult Brain. Cell Reports 2017, 18, 391–405. [Google Scholar] [CrossRef]
- Ginhoux, F.; Greter, M.; Leboeuf, M.; Nandi, S.; See, P.; Gokhan, S.; Mehler, M.F.; Conway, S.J.; Ng, L.G.; Stanley, E.R.; et al. Fate Mapping Analysis Reveals That Adult Microglia Derive from Primitive Macrophages. Science 2010, 330, 841–845. [Google Scholar] [CrossRef]
- Van Hove, H.; Martens, L.; Scheyltjens, I.; De Vlaminck, K.; Pombo Antunes, A.R.; De Prijck, S.; Vandamme, N.; De Schepper, S.; Van Isterdael, G.; Scott, C.L.; et al. A single-cell atlas of mouse brain macrophages reveals unique transcriptional identities shaped by ontogeny and tissue environment. Nature Neuroscience 2019 22:6 2019, 22. [Google Scholar] [CrossRef]
- Prinz, M.; Priller, J. Microglia and brain macrophages in the molecular age: from origin to neuropsychiatric disease. Nature Reviews Neuroscience 2014 15:5 2014, 15. [Google Scholar] [CrossRef]
- Paolicelli, R.C.; Sierra, A.; Stevens, B.; Tremblay, M.-E.; Aguzzi, A.; Ajami, B.; Amit, I.; Audinat, E.; Bechmann, I.; Bennett, M.; et al. Microglia states and nomenclature: A field at its crossroads. Neuron 2022, 110, 3458–3483. [Google Scholar] [CrossRef] [PubMed]
- Paolicelli, R.C.; Bolasco, G.; Pagani, F.; Maggi, L.; Scianni, M.; Panzanelli, P.; Giustetto, M.; Ferreira, T.A.; Guiducci, E.; Dumas, L.; et al. Synaptic Pruning by Microglia Is Necessary for Normal Brain Development. Science 2011, 333, 1456–1458. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, M.-È.; Lowery, R.L.; Majewska, A.K. Microglial Interactions with Synapses Are Modulated by Visual Experience. PLOS Biology, 2010; 8. [Google Scholar] [CrossRef]
- Schafer, D.P.; Lehrman, E.K.; Kautzman, A.G.; Koyama, R.; Mardinly, A.R.; Yamasaki, R.; Ransohoff, R.M.; Greenberg, M.E.; Barres, B.A.; Stevens, B. Microglia sculpt postnatal neural circuits in an activity and complement-dependent manner. Neuron 2012, 74, 691–705. [Google Scholar] [CrossRef]
- Stevens, B.; Allen, N.J.; Vazquez, L.E.; Howell, G.R.; Christopherson, K.S.; Nouri, N.; Micheva, K.D.; Mehalow, A.K.; Huberman, A.D.; Stafford, B.; et al. The classical complement cascade mediates CNS synapse elimination. Cell 2007, 131, 1164–1178. [Google Scholar] [CrossRef] [PubMed]
- Franklin, T.C.; Xu, C.; Duman, R.S. Depression and sterile inflammation: Essential role of danger associated molecular patterns. Brain, Behavior, and Immunity 2018, 72, 2–13. [Google Scholar] [CrossRef]
- Badimon, A.; Strasburger, H.J.; Ayata, P.; Chen, X.; Nair, A.; Ikegami, A.; Hwang, P.; Chan, A.T.; Graves, S.M.; Uweru, J.O.; et al. Negative feedback control of neuronal activity by microglia. Nature 2020, 586, 417–423. [Google Scholar] [CrossRef]
- Hendrickx, D.A.E.; van Eden, C.G.; Schuurman, K.G.; Hamann, J.; Huitinga, I. Staining of HLA-DR, Iba1 and CD68 in human microglia reveals partially overlapping expression depending on cellular morphology and pathology. J Neuroimmunol 2017, 309, 12–22. [Google Scholar] [CrossRef]
- Patel, S.; Player, M.R. Colony-stimulating factor-1 receptor inhibitors for the treatment of cancer and inflammatory disease. Curr Top Med Chem 2009, 9, 599–610. [Google Scholar] [CrossRef]
- Chitu, V.; Biundo, F.; Shlager, G.G.L.; Park, E.S.; Wang, P.; Gulinello, M.E.; Gokhan, S.; Ketchum, H.C.; Saha, K.; DeTure, M.A.; et al. Microglial Homeostasis Requires Balanced CSF-1/CSF-2 Receptor Signaling. Cell Rep 2020, 30, 3004–3019. [Google Scholar] [CrossRef]
- Chitu, V.; Gokhan, S.; Nandi, S.; Mehler, M.F.; Stanley, E.R. Emerging Roles for CSF-1 Receptor and its Ligands in the Nervous System. Trends Neurosci 2016, 39, 378–393. [Google Scholar] [CrossRef]
- Erblich, B.; Zhu, L.; Etgen, A.M.; Dobrenis, K.; Pollard, J.W. Absence of colony stimulation factor-1 receptor results in loss of microglia, disrupted brain development and olfactory deficits. PLoS One 2011, 6, e26317. [Google Scholar] [CrossRef] [PubMed]
- Arreola, M.A.; Soni, N.; Crapser, J.D.; Hohsfield, L.A.; Elmore, M.R.P.; Matheos, D.P.; Wood, M.A.; Swarup, V.; Mortazavi, A.; Green, K.N. Microglial dyshomeostasis drives perineuronal net and synaptic loss in a CSF1R(+/-) mouse model of ALSP, which can be rescued via CSF1R inhibitors. Sci Adv 2021, 7. [Google Scholar] [CrossRef] [PubMed]
- Wicherska-Pawlowska, K.; Wrobel, T.; Rybka, J. Toll-Like Receptors (TLRs), NOD-Like Receptors (NLRs), and RIG-I-Like Receptors (RLRs) in Innate Immunity. TLRs, NLRs, and RLRs Ligands as Immunotherapeutic Agents for Hematopoietic Diseases. Int J Mol Sci 2021, 22. [Google Scholar] [CrossRef]
- Nayak, D.; Roth, T.L.; McGavern, D.B. Microglia development and function. Annu Rev Immunol 2014, 32, 367–402. [Google Scholar] [CrossRef]
- Kemp, G.M.; Altimimi, H.F.; Nho, Y.; Heir, R.; Klyczek, A.; Stellwagen, D. Sustained TNF signaling is required for the synaptic and anxiety-like behavioral response to acute stress. Mol Psychiatry 2022, 27, 4474–4484. [Google Scholar] [CrossRef]
- Patterson, S.L. Immune dysregulation and cognitive vulnerability in the aging brain: Interactions of microglia, IL-1β, BDNF and synaptic plasticity. Neuropharmacology 2015, 96. [Google Scholar] [CrossRef]
- Nguyen, P.T.; Dorman, L.C.; Pan, S.; Vainchtein, I.D.; Han, R.T.; Nakao-Inoue, H.; Taloma, S.E.; Barron, J.J.; Molofsky, A.B.; Kheirbek, M.A.; Molofsky, A.V. Microglial Remodeling of the Extracellular Matrix Promotes Synapse Plasticity. Cell 2020, 182, 388–403.e315. [Google Scholar] [CrossRef] [PubMed]
- Parkhurst, Christopher N. ; Yang, G.; Ninan, I.; Savas, Jeffrey N.; Yates, John R.; Lafaille, Juan J.; Hempstead, Barbara L.; Littman, Dan R.; Gan, W.-B. Microglia Promote Learning-Dependent Synapse Formation through Brain-Derived Neurotrophic Factor. Cell 2013, 155. [Google Scholar] [CrossRef]
- Woodburn, S.C.; Asrat, H.S.; Flurer, J.K.; Schwierling, H.C.; Bollinger, J.L.; Vollmer, L.L.; Wohleb, E.S. Depletion of microglial BDNF increases susceptibility to the behavioral and synaptic effects of chronic unpredictable stress. Brain, Behavior, and Immunity 2023, 109, 127–138. [Google Scholar] [CrossRef]
- Haynes, S.E.; Hollopeter, G.; Yang, G.; Kurpius, D.; Dailey, M.E.; Gan, W.-B.; Julius, D.; Haynes, S.E.; Hollopeter, G.; Yang, G.; et al. The P2Y12 receptor regulates microglial activation by extracellular nucleotides. Nature Neuroscience 2006 9:12 2006, 9. [Google Scholar] [CrossRef]
- Ma, C.; Li, B.; Silverman, D.; Ding, X.; Li, A.; Xiao, C.; Huang, G.; Worden, K.; Muroy, S.; Chen, W.; et al. Microglia regulate sleep through calcium-dependent modulation of norepinephrine transmission. Nat Neurosci 2024, 27, 249–258. [Google Scholar] [CrossRef] [PubMed]
- Lalo, U.; Pankratov, Y. ATP-mediated signalling in the central synapses. Neuropharmacology 2023, 229, 109477. [Google Scholar] [CrossRef]
- Lalo, U.; Palygin, O.; Verkhratsky, A.; Grant, S.G.N.; Pankratov, Y.; Lalo, U.; Palygin, O.; Verkhratsky, A.; Grant, S.G.N.; Pankratov, Y. ATP from synaptic terminals and astrocytes regulates NMDA receptors and synaptic plasticity through PSD-95 multi-protein complex. Scientific Reports 2016 6:1 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Ho, T.; Jobling, A.I.; Greferath, U.; Chuang, T.; Ramesh, A.; Fletcher, E.L.; Vessey, K.A. Vesicular expression and release of ATP from dopaminergic neurons of the mouse retina and midbrain. Frontiers in Cellular Neuroscience 2015, 9. [Google Scholar] [CrossRef]
- Witting, A.; Walter, L.; Wacker, J.; Möller, T.; Stella, N. P2X7 receptors control 2-arachidonoylglycerol production by microglial cells. Proc Natl Acad Sci U S A 2004, 101, 3214–3219. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Leak, R.K.; Hu, X. Neurotransmitter receptors on microglia. Stroke Vasc Neurol 2016, 1, 52–58. [Google Scholar] [CrossRef]
- Acosta-Martinez, M. Shaping Microglial Phenotypes Through Estrogen Receptors: Relevance to Sex-Specific Neuroinflammatory Responses to Brain Injury and Disease. J Pharmacol Exp Ther 2020, 375, 223–236. [Google Scholar] [CrossRef]
- Villa, A.; Gelosa, P.; Castiglioni, L.; Cimino, M.; Rizzi, N.; Pepe, G.; Lolli, F.; Marcello, E.; Sironi, L.; Vegeto, E.; Maggi, A. Sex-Specific Features of Microglia from Adult Mice. Cell Rep 2018, 23, 3501–3511. [Google Scholar] [CrossRef]
- Guneykaya, D.; Ivanov, A.; Hernandez, D.P.; Haage, V.; Wojtas, B.; Meyer, N.; Maricos, M.; Jordan, P.; Buonfiglioli, A.; Gielniewski, B.; et al. Transcriptional and Translational Differences of Microglia from Male and Female Brains. Cell Rep 2018, 24, 2773–2783. [Google Scholar] [CrossRef]
- Ball, J.B.; Mcnulty, C.J.; Green-Fulgham, S.M.; Dragavon, J.M.; Correia Rocha, I.R.; Finch, M.R.; Prévost, E.D.; Siddique, I.I.; Woodall, B.J.; Watkins, L.R.; et al. Combining RNAscope and immunohistochemistry to visualize inflammatory gene products in neurons and microglia. Frontiers in Molecular Neuroscience 2023, 16. [Google Scholar] [CrossRef]
- Schmid, C.D.; Melchior, B.; Masek, K.; Puntambekar, S.S.; Danielson, P.E.; Lo, D.D.; Gregor Sutcliffe, J.; Carson, M.J. Differential gene expression in LPS/IFNγ activated microglia and macrophages: <i>in vitro</i> versus <i>in vivo</i>. Journal of Neurochemistry 2009, 109, 117–125. [Google Scholar] [CrossRef]
- Liu, Q.-R.; Canseco-Alba, A.; Liang, Y.; Ishiguro, H.; Onaivi, E.S. Low Basal CB2R in Dopamine Neurons and Microglia Influences Cannabinoid Tetrad Effects. International Journal of Molecular Sciences 2020, 21, 9763. [Google Scholar] [CrossRef] [PubMed]
- Olah, M.; Menon, V.; Habib, N.; Taga, M.F.; Ma, Y.; Yung, C.J.; Cimpean, M.; Khairallah, A.; Coronas-Samano, G.; Sankowski, R.; et al. Single cell RNA sequencing of human microglia uncovers a subset associated with Alzheimer’s disease. Nature Communications 2020, 11. [Google Scholar] [CrossRef]
- Dissing-Olesen, L.; Walker, A.J.; Feng, Q.; Barr, H.J.; Walker, A.C.; Xie, L.; Wilton, D.K.; Das, I.; Benowitz, L.I.; Stevens, B. FEAST: A flow cytometry-based toolkit for interrogating microglial engulfment of synaptic and myelin proteins. Nature Communications 2023, 14. [Google Scholar] [CrossRef] [PubMed]
- Martin, E.; El-Behi, M.; Fontaine, B.; Delarasse, C. Analysis of Microglia and Monocyte-derived Macrophages from the Central Nervous System by Flow Cytometry. Journal of Visualized Experiments 2017. [Google Scholar] [CrossRef]
- Walker, D.; Lue, L.-F.; Beach, T.; Tooyama, I. Microglial Phenotyping in Neurodegenerative Disease Brains: Identification of Reactive Microglia with an Antibody to Variant of CD105/Endoglin. Cells 2019, 8, 766. [Google Scholar] [CrossRef]
- Ashton, J.; Glass, M. The Cannabinoid CB2 Receptor as a Target for Inflammation-Dependent Neurodegeneration. Current Neuropharmacology 2007, 5, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Bollinger, J.L.; Dadosky, D.T.; Flurer, J.K.; Rainer, I.L.; Woodburn, S.C.; Wohleb, E.S. Microglial P2Y12 mediates chronic stress-induced synapse loss in the prefrontal cortex and associated behavioral consequences. Neuropsychopharmacology 2023, 48, 1347–1357. [Google Scholar] [CrossRef]
- Scofield, M.D.; Heinsbroek, J.A.; Gipson, C.D.; Kupchik, Y.M.; Spencer, S.; Smith, A.C.W.; Roberts-Wolfe, D.; Kalivas, P.W. The Nucleus Accumbens: Mechanisms of Addiction across Drug Classes Reflect the Importance of Glutamate Homeostasis. Pharmacological Reviews 2016, 68, 816–871. [Google Scholar] [CrossRef]
- Turner, B.D.; Kashima, D.T.; Manz, K.M.; Grueter, C.A.; Grueter, B.A. Synaptic Plasticity in the Nucleus Accumbens: Lessons Learned from Experience. ACS Chemical Neuroscience 2018, 9, 2114–2126. [Google Scholar] [CrossRef]
- Everitt, B.J.; Robbins, T.W. Neural systems of reinforcement for drug addiction: from actions to habits to compulsion. Nature Neuroscience 2005, 8, 1481–1489. [Google Scholar] [CrossRef] [PubMed]
- Mulholland, P.J.; Chandler, L.J.; Kalivas, P.W. Signals from the Fourth Dimension Regulate Drug Relapse. Trends in Neurosciences 2016, 39, 472–485. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Keller, C.; Carter, J.S.; Kruyer, A.; Kearns, A.M.; Hopkins, J.L.; Hodebourg, R.; Kalivas, P.W.; Reichel, C.M. Behavioral and accumbens synaptic plasticity induced by cues associated with restraint stress. Neuropsychopharmacology 2021, 46, 1848–1856. [Google Scholar] [CrossRef]
- Jurga, A.M.; Paleczna, M.; Kuter, K.Z. Overview of General and Discriminating Markers of Differential Microglia Phenotypes. Frontiers in Cellular Neuroscience 2020, 14. [Google Scholar] [CrossRef]
- Jung, S.; Aliberti, J.; Graemmel, P.; Sunshine, M.J.; Kreutzberg, G.W.; Sher, A.; Littman, D.R. Analysis of Fractalkine Receptor CX<sub>3</sub>CR1 Function by Targeted Deletion and Green Fluorescent Protein Reporter Gene Insertion. Molecular and Cellular Biology 2000, 20, 4106–4114. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, T.; Feng, G. Tmem119-EGFP and Tmem119-CreERT2 Transgenic Mice for Labeling and Manipulating Microglia. eneuro 2019, 6, ENEURO.0448–0418. [Google Scholar] [CrossRef] [PubMed]
- Buttgereit, A.; Lelios, I.; Yu, X.; Vrohlings, M.; Krakoski, N.R.; Gautier, E.L.; Nishinakamura, R.; Becher, B.; Greter, M. Sall1 is a transcriptional regulator defining microglia identity and function. Nature Immunology 2016, 17, 1397–1406. [Google Scholar] [CrossRef]
- Sharma, K.; Bisht, K.; Eyo, U.B. A Comparative Biology of Microglia Across Species. Frontiers in Cell and Developmental Biology 2021, 9. [Google Scholar] [CrossRef]
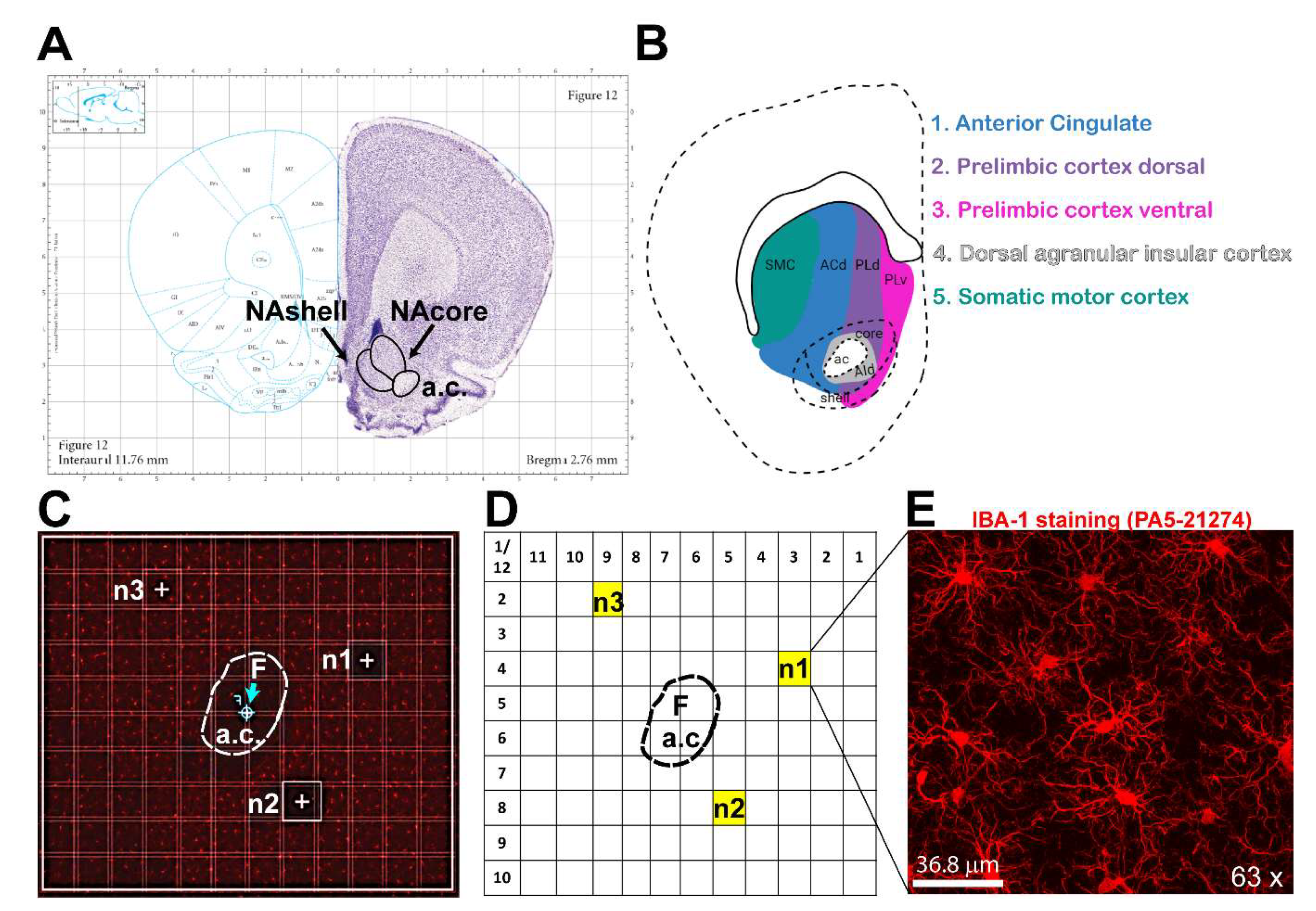
- Paxinos, G.; Watson, C. Paxinos and Watson's the rat brain in stereotaxic coordinates, Seventh ed.; Academic Press/Elsevier, 2014.: Amsterdam, 2014. [Google Scholar]
- Voorn, P.; Vanderschuren, L.J.M.J.; Groenewegen, H.J.; Robbins, T.W.; Pennartz, C.M.A. Putting a spin on the dorsal–ventral divide of the striatum. Trends in Neurosciences 2004, Volume 27, 468–474. [Google Scholar] [CrossRef]
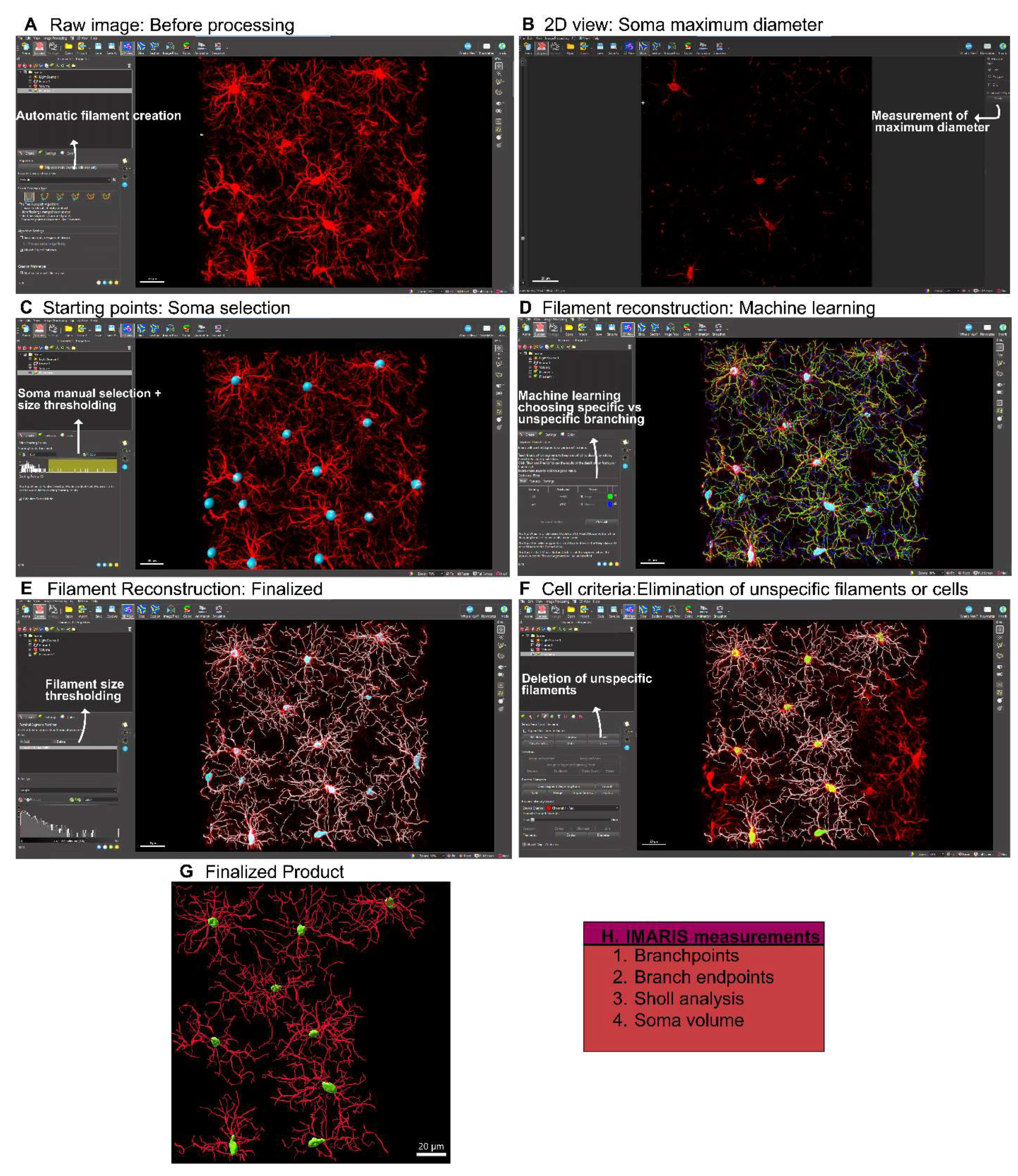
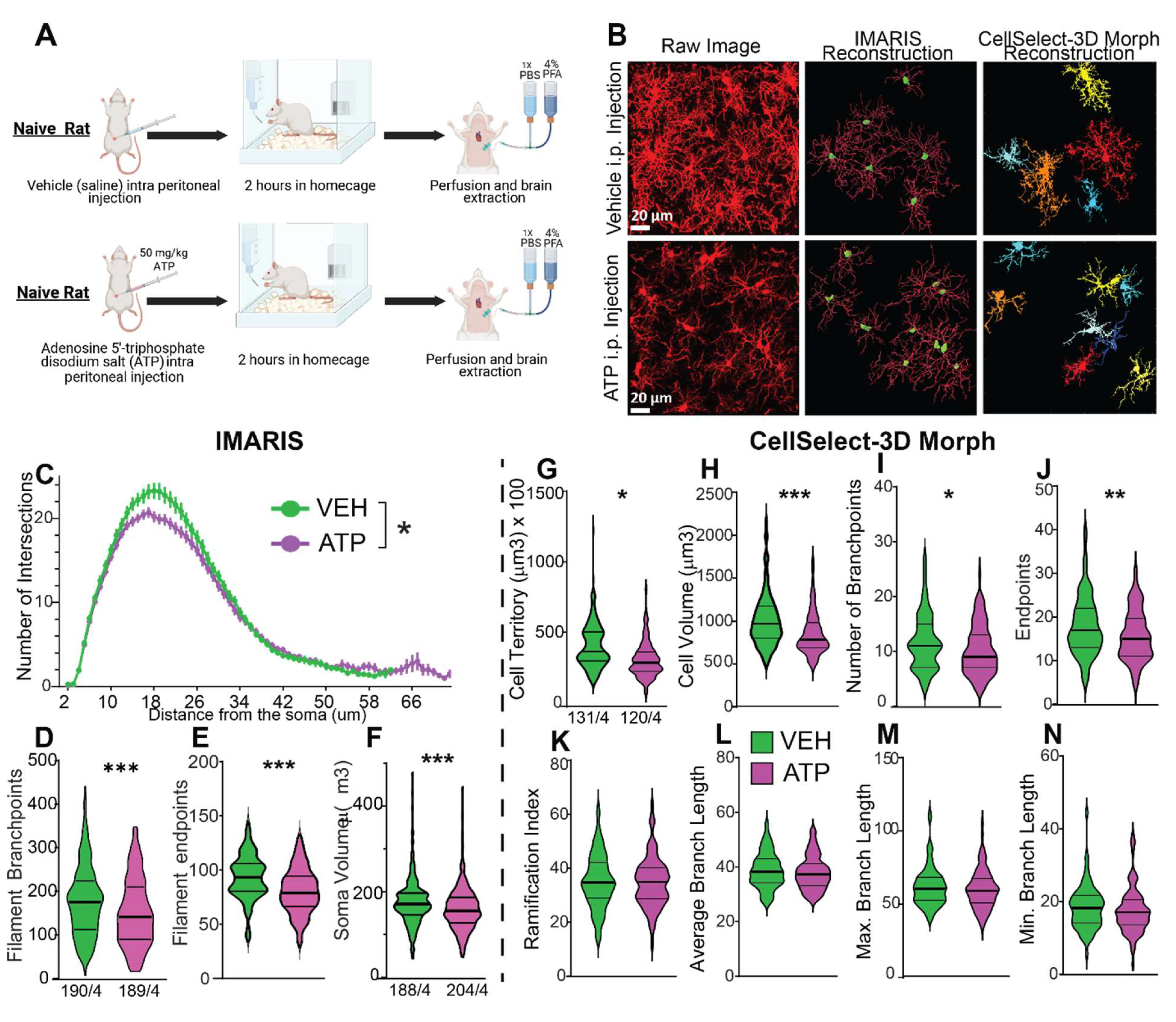
- York, E.M.; Ledue, J.M.; Bernier, L.-P.; Macvicar, B.A. 3DMorph Automatic Analysis of Microglial Morphology in Three Dimensions from<i>Ex Vivo</i>and<i>In Vivo</i>Imaging. eneuro 2018, 5, ENEURO.0266–0218. [Google Scholar] [CrossRef]
- Tanaka, K.; Furuyashiki, T.; Kitaoka, S.; Senzai, Y.; Imoto, Y.; Segi-Nishida, E.; Deguchi, Y.; Breyer, R.M.; Breyer, M.D.; Narumiya, S. Prostaglandin E2-mediated attenuation of mesocortical dopaminergic pathway is critical for susceptibility to repeated social defeat stress in mice. J Neurosci 2012, 32, 4319–4329. [Google Scholar] [CrossRef] [PubMed]
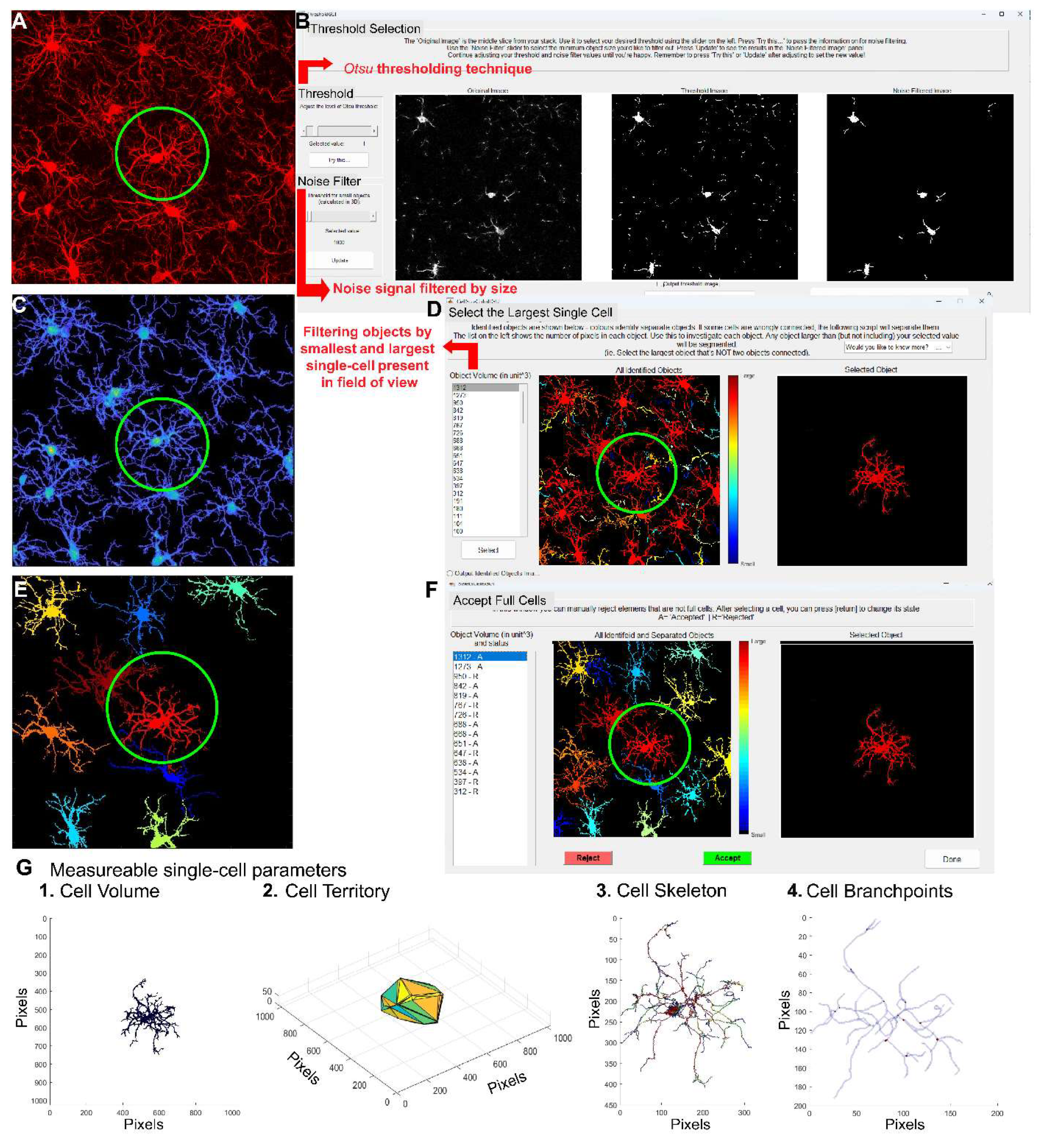
- Reddaway, J.; Richardson, P.E.; Bevan, R.J.; Stoneman, J.; Palombo, M. Microglial morphometric analysis: so many options, so little consistency. Frontiers in Neuroinformatics 2023, 17. [Google Scholar] [CrossRef] [PubMed]
- Sierra, A.; Paolicelli, R.C.; Kettenmann, H. Cien Años de Microglía: Milestones in a Century of Microglial Research. Trends in Neurosciences 2019, 42, 778–792. [Google Scholar] [CrossRef] [PubMed]
- Chaure, F., J. Taborda-Bejarano, E.M. York, and C. Garcia-Keller. CellSelect-3Dmorph. [Github Repository] 2023 november 11, 2024.
- Otsu, N. A Threshold Selection Method from Gray-Level Histograms. IEEE Transactions on Systems, Man, and Cybernetics 1979, 9, 62–66. [Google Scholar] [CrossRef]
- Sáez, P.J.; Shoji, K.F.; Retamal, M.A.; Harcha, P.A.; Ramírez, G.; Jiang, J.X.; Von Bernhardi, R.; Sáez, J.C. ATP Is Required and Advances Cytokine-Induced Gap Junction Formation in Microglia In Vitro. Mediators of Inflammation 2013, 2013, 1–16. [Google Scholar] [CrossRef]
- Radley, J.J.; Herman, J.P. Preclinical Models of Chronic Stress: Adaptation or Pathology? Biol Psychiatry 2023, 94, 194–202. [Google Scholar] [CrossRef]
- Liston, C.; Miller, M.M.; Goldwater, D.S.; Radley, J.J.; Rocher, A.B.; Hof, P.R.; Morrison, J.H.; McEwen, B.S. Stress-induced alterations in prefrontal cortical dendritic morphology predict selective impairments in perceptual attentional set-shifting. J Neurosci 2006, 26, 7870–7874. [Google Scholar] [CrossRef]
- Johnson, J.D.; Campisi, J.; Sharkey, C.M.; Kennedy, S.L.; Nickerson, M.; Greenwood, B.N.; Fleshner, M. Catecholamines mediate stress-induced increases in peripheral and central inflammatory cytokines. Neuroscience 2005, 135. [Google Scholar] [CrossRef]
- Maier, S.F. Bi-directional immune–brain communication: Implications for understanding stress, pain, and cognition. Brain, Behavior, and Immunity 2003, 17. [Google Scholar] [CrossRef]
- Frank, M.G.; Baratta, M.V.; Sprunger, D.B.; Watkins, L.R.; Maier, S.F. Microglia serve as a neuroimmune substrate for stress-induced potentiation of CNS pro-inflammatory cytokine responses. Brain Behav Immun 2007, 21, 47–59. [Google Scholar] [CrossRef]
- Schramm, E.; Waisman, A. Microglia as Central Protagonists in the Chronic Stress Response. Neurol Neuroimmunol Neuroinflamm 2022, 9, e200023. [Google Scholar] [CrossRef] [PubMed]
- Wohleb, E.S.; Franklin, T.; Iwata, M.; Duman, R.S. Integrating neuroimmune systems in the neurobiology of depression. Nat Rev Neurosci 2016, 17, 497–511. [Google Scholar] [CrossRef] [PubMed]
- Bollinger, J.L.; Collins, K.E.; Patel, R.; Wellman, C.L. Behavioral stress alters corticolimbic microglia in a sex- and brain region-specific manner. PLoS One 2017, 12, e0187631. [Google Scholar] [CrossRef]
- Wang, H.; He, Y.; Sun, Z.; Ren, S.; Liu, M.; Wang, G.; Yang, J. Microglia in depression: an overview of microglia in the pathogenesis and treatment of depression. Journal of Neuroinflammation 2022, 19, 132. [Google Scholar] [CrossRef] [PubMed]
- Afridi, R.; Suk, K. Microglial Responses to Stress-Induced Depression: Causes and Consequences. Cells 2023, 12, 1521. [Google Scholar] [CrossRef]
- Woodburn, S.C.; Bollinger, J.L.; Wohleb, E.S. Synaptic and behavioral effects of chronic stress are linked to dynamic and sex-specific changes in microglia function and astrocyte dystrophy. Neurobiology of Stress 2021, 14, 100312. [Google Scholar] [CrossRef]
- Yuan, T.; Orock, A.; Greenwood-Van Meerveld, B. Amygdala microglia modify neuronal plasticity via complement C1q/C3-CR3 signaling and contribute to visceral pain in a rat model. Am J Physiol Gastrointest Liver Physiol 2021, 320, G1081–G1092. [Google Scholar] [CrossRef]
- Biltz, R.G.; Swanson, S.P.; Draime, N.; Davis, A.C.; Yin, W.; Goodman, E.J.; Gallagher, N.R.; Bhattacharya, A.; Sheridan, J.F.; Godbout, J.P. Antagonism of the brain P2X7 ion channel attenuates repeated social defeat induced microglia reactivity, monocyte recruitment and anxiety-like behavior in male mice. Brain, Behavior, and Immunity 2024, 115, 356–373. [Google Scholar] [CrossRef]
- Hinwood, M.; Morandini, J.; Day, T.A.; Walker, F.R. Evidence that Microglia Mediate the Neurobiological Effects of Chronic Psychological Stress on the Medial Prefrontal Cortex. Cerebral Cortex 2012, 22, 1442–1454. [Google Scholar] [CrossRef]
- Milior, G.; Lecours, C.; Samson, L.; Bisht, K.; Poggini, S.; Pagani, F.; Deflorio, C.; Lauro, C.; Alboni, S.; Limatola, C.; et al. Fractalkine receptor deficiency impairs microglial and neuronal responsiveness to chronic stress. Brain, Behavior, and Immunity 2016, 55, 114–125. [Google Scholar] [CrossRef]
- Horchar, M.J.; Wohleb, E.S. Glucocorticoid receptor antagonism prevents microglia-mediated neuronal remodeling and behavioral despair following chronic unpredictable stress. Brain, Behavior, and Immunity 2019, 81, 329–340. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Chen, H.-S.; Li, H.-H.; Wang, H.-J.; Zou, R.-S.; Lu, X.-J.; Wang, J.; Nie, B.-B.; Wu, J.-F.; Li, S.; et al. Microglia-dependent excessive synaptic pruning leads to cortical underconnectivity and behavioral abnormality following chronic social defeat stress in mice. Brain, Behavior, and Immunity 2023, 109, 23–36. [Google Scholar] [CrossRef]
- Chen, H.; Fu, S.; Li, X.; Shi, M.; Qian, J.; Zhao, S.; Yuan, P.; Ding, L.; Xia, X.; Zheng, J.C. Microglial glutaminase 1 mediates chronic restraint stress-induced depression-like behaviors and synaptic damages. Signal Transduct Target Ther 2023, 8, 452. [Google Scholar] [CrossRef]
- Han, Q.-Q.; Shen, S.-Y.; Chen, X.-R.; Pilot, A.; Liang, L.-F.; Zhang, J.-R.; Li, W.-H.; Fu, Y.; Le, J.-M.; Chen, P.-Q.; Yu, J. Minocycline alleviates abnormal microglial phagocytosis of synapses in a mouse model of depression. Neuropharmacology 2022, 220, 109249. [Google Scholar] [CrossRef] [PubMed]
- Bassett, B.; Subramaniyam, S.; Fan, Y.; Varney, S.; Pan, H.; Carneiro, A.M.D.; Chung, C.Y. Minocycline alleviates depression-like symptoms by rescuing decrease in neurogenesis in dorsal hippocampus via blocking microglia activation/phagocytosis. Brain, Behavior, and Immunity 2021, 91, 519–530. [Google Scholar] [CrossRef]
- Avalos, M.P.; Guzman, A.S.; Rigoni, D.; Gorostiza, E.A.; Sanchez, M.A.; Mongi-Bragato, B.; Garcia-Keller, C.; Perassi, E.M.; Virgolini, M.B.; Peralta Ramos, J.M.; et al. Minocycline prevents chronic restraint stress-induced vulnerability to developing cocaine self-administration and associated glutamatergic mechanisms: a potential role of microglia. Brain, Behavior, and Immunity 2022, 101, 359–376. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Huang, X.; Pan, X.; Zhang, T.; Hou, C.; Su, W.-J.; Liu, L.-L.; Li, J.-M.; Wang, Y.-X. Minocycline prevents the depressive-like behavior through inhibiting the release of HMGB1 from microglia and neurons. Brain, Behavior, and Immunity 2020, 88, 132–143. [Google Scholar] [CrossRef]
- Qiao, H.; Li, M.-X.; Xu, C.; Chen, H.-B.; An, S.-C.; Ma, X.-M. Dendritic Spines in Depression: What We Learned from Animal Models. Neural Plasticity 2016, 2016. [Google Scholar] [CrossRef]
- Wohleb, E.S.; Terwilliger, R.; Duman, C.H.; Duman, R.S. Stress-Induced Neuronal Colony Stimulating Factor 1 Provokes Microglia-Mediated Neuronal Remodeling and Depressive-like Behavior. Biol Psychiatry 2018, 83, 38–49. [Google Scholar] [CrossRef]
- Elmore, M.R.; Najafi, A.R.; Koike, M.A.; Dagher, N.N.; Spangenberg, E.E.; Rice, R.A.; Kitazawa, M.; Matusow, B.; Nguyen, H.; West, B.L.; Green, K.N. Colony-stimulating factor 1 receptor signaling is necessary for microglia viability, unmasking a microglia progenitor cell in the adult brain. Neuron 2014, 82, 380–397. [Google Scholar] [CrossRef]
- Berglund, R.; Cheng, Y.; Piket, E.; Z. Adzemovic, M.; Zeitelhofer, M.; Olsson, T.; Ortlieb Guerreiro-Cacais, A.; Jagodic, M.; Berglund, R.; Cheng, Y.; et al. The aging mouse CNS is protected by an autophagy-dependent microglia population promoted by IL-34. Nature Communications 2024 15:1 2024, 15. [Google Scholar] [CrossRef]
- Eyo, U.; Molofsky, A.V. Defining microglial-synapse interactions. Science 2023, 381. [Google Scholar] [CrossRef] [PubMed]
- Tillmon, H.; Soteros, B.M.; Shen, L.; Cong, Q.; Wollet, M.; General, J.; Chin, H.; Lee, J.B.; Carreno, F.R.; Morilak, D.A.; et al. Complement and microglia activation mediate stress-induced synapse loss in layer 2/3 of the medial prefrontal cortex in male mice. Nat Commun 2024, 15, 9803. [Google Scholar] [CrossRef]
- Klawonn, A.M.; Malenka, R.C. Nucleus Accumbens Modulation in Reward and Aversion. Cold Spring Harb Symp Quant Biol 2018, 83, 119–129. [Google Scholar] [CrossRef]
- Bessa, J.M.; Morais, M.; Marques, F.; Pinto, L.; Palha, J.A.; Almeida, O.F.; Sousa, N. Stress-induced anhedonia is associated with hypertrophy of medium spiny neurons of the nucleus accumbens. Transl Psychiatry 2013, 3, e266. [Google Scholar] [CrossRef]
- Gaspar, R.; Soares-Cunha, C.; Domingues, A.V.; Coimbra, B.; Baptista, F.I.; Pinto, L.; Ambrosio, A.F.; Rodrigues, A.J.; Gomes, C.A. The Duration of Stress Determines Sex Specificities in the Vulnerability to Depression and in the Morphologic Remodeling of Neurons and Microglia. Front Behav Neurosci 2022, 16, 834821. [Google Scholar] [CrossRef] [PubMed]
- Douma, E.H.; de Kloet, E.R. Stress-induced plasticity and functioning of ventral tegmental dopamine neurons. Neuroscience & Biobehavioral Reviews 2020, 108, 48–77. [Google Scholar] [CrossRef]
- Hill, A.S.; Sahay, A.; Hen, R. Increasing Adult Hippocampal Neurogenesis is Sufficient to Reduce Anxiety and Depression-Like Behaviors. Neuropsychopharmacology 2015, 40, 2368–2378. [Google Scholar] [CrossRef]
- Du Preez, A.; Onorato, D.; Eiben, I.; Musaelyan, K.; Egeland, M.; Zunszain, P.A.; Fernandes, C.; Thuret, S.; Pariante, C.M. Chronic stress followed by social isolation promotes depressive-like behaviour, alters microglial and astrocyte biology and reduces hippocampal neurogenesis in male mice. Brain Behav Immun 2021, 91, 24–47. [Google Scholar] [CrossRef]
- Fang, S.; Wu, Z.; Guo, Y.; Zhu, W.; Wan, C.; Yuan, N.; Chen, J.; Hao, W.; Mo, X.; Guo, X.; et al. Roles of microglia in adult hippocampal neurogenesis in depression and their therapeutics. Front Immunol 2023, 14, 1193053. [Google Scholar] [CrossRef]
- Wohleb, E.S.; Hanke, M.L.; Corona, A.W.; Powell, N.D.; Stiner, L.M.; Bailey, M.T.; Nelson, R.J.; Godbout, J.P.; Sheridan, J.F. beta-Adrenergic receptor antagonism prevents anxiety-like behavior and microglial reactivity induced by repeated social defeat. J Neurosci 2011, 31, 6277–6288. [Google Scholar] [CrossRef] [PubMed]
- Poggini, S.; Lopez, M.B.; Albanese, N.C.; Golia, M.T.; Ibanez, F.G.; Limatola, C.; Furhmann, M.; Lalowski, M.; Tremblay, M.E.; Maggi, L.; et al. Minocycline treatment improves cognitive and functional plasticity in a preclinical mouse model of major depressive disorder. Behav Brain Res 2023, 441, 114295. [Google Scholar] [CrossRef]
- Wohleb, E.S.; Powell, N.D.; Godbout, J.P.; Sheridan, J.F. Stress-induced recruitment of bone marrow-derived monocytes to the brain promotes anxiety-like behavior. J Neurosci 2013, 33, 13820–13833. [Google Scholar] [CrossRef] [PubMed]
- Brevet, M.; Kojima, H.; Asakawa, A.; Atsuchi, K.; Ushikai, M.; Ataka, K.; Inui, A.; Kimura, H.; Sevestre, H.; Fujimiya, M. Chronic foot-shock stress potentiates the influx of bone marrow-derived microglia into hippocampus. J Neurosci Res 2010, 88, 1890–1897. [Google Scholar] [CrossRef] [PubMed]
- da Silva, M.C.M.; Iglesias, L.P.; Candelario-Jalil, E.; Khoshbouei, H.; Moreira, F.A.; de Oliveira, A.C.P. Role of Microglia in Psychostimulant Addiction. Curr Neuropharmacol 2023, 21, 235–259. [Google Scholar] [CrossRef]
- Ahearn, O.C.; Watson, M.N.; Rawls, S.M. Chemokines, cytokines and substance use disorders. Drug Alcohol Depend 2021, 220, 108511. [Google Scholar] [CrossRef]
- Kohno, M.; Link, J.; Dennis, L.E.; McCready, H.; Huckans, M.; Hoffman, W.F.; Loftis, J.M. Neuroinflammation in addiction: A review of neuroimaging studies and potential immunotherapies. Pharmacology Biochemistry and Behavior 2019, 179, 34–42. [Google Scholar] [CrossRef]
- Lacagnina, M.J.; Rivera, P.D.; Bilbo, S.D. Glial and Neuroimmune Mechanisms as Critical Modulators of Drug Use and Abuse. Neuropsychopharmacology 2017, 42, 156–177. [Google Scholar] [CrossRef]
- Goldstein, R.Z.; Volkow, N.D. Dysfunction of the prefrontal cortex in addiction: neuroimaging findings and clinical implications. Nat Rev Neurosci 2011, 12, 652–669. [Google Scholar] [CrossRef]
- Angarita, G.A.; Worhunsky, P.D.; Naganawa, M.; Toyonaga, T.; Nabulsi, N.B.; Li, C.R.; Esterlis, I.; Skosnik, P.D.; Radhakrishnan, R.; Pittman, B.; et al. Lower prefrontal cortical synaptic vesicle binding in cocaine use disorder: An exploratory (11) C-UCB-J positron emission tomography study in humans. Addict Biol 2022, 27, e13123. [Google Scholar] [CrossRef]
- Golden, S.A.; Russo, S.J. Mechanisms of Psychostimulant-Induced Structural Plasticity. Cold Spring Harb Perspect Med 2012, 2, a011957. [Google Scholar] [CrossRef]
- DePoy, L.M.; Gourley, S.L. Synaptic Cytoskeletal Plasticity in the Prefrontal Cortex Following Psychostimulant Exposure. Traffic 2015, 16, 919–940. [Google Scholar] [CrossRef]
- Radley, J.J.; Anderson, R.M.; Cosme, C.V.; Glanz, R.M.; Miller, M.C.; Romig-Martin, S.A.; LaLumiere, R.T. The Contingency of Cocaine Administration Accounts for Structural and Functional Medial Prefrontal Deficits and Increased Adrenocortical Activation. J Neurosci 2015, 35, 11897–11910. [Google Scholar] [CrossRef]
- Selemon, L.D.; Begović, A.; Goldman-Rakic, P.S.; Castner, S.A. Amphetamine sensitization alters dendritic morphology in prefrontal cortical pyramidal neurons in the non-human primate. Neuropsychopharmacology 2007, 32, 919–931. [Google Scholar] [CrossRef] [PubMed]
- Siemsen, B.M.; Giannotti, G.; McFaddin, J.A.; Scofield, M.D.; McGinty, J.F. Biphasic effect of abstinence duration following cocaine self-administration on spine morphology and plasticity-related proteins in prelimbic cortical neurons projecting to the nucleus accumbens core. Brain Struct Funct 2019, 224, 741–758. [Google Scholar] [CrossRef] [PubMed]
- Anderson, E.M.; Self, D.W. It's only a matter of time: longevity of cocaine-induced changes in dendritic spine density in the nucleus accumbens. Curr Opin Behav Sci 2017, 13, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Hutchinson, M.R.; Zhang, Y.; Brown, K.; Coats, B.D.; Shridhar, M.; Sholar, P.W.; Patel, S.J.; Crysdale, N.Y.; Harrison, J.A.; Maier, S.F.; et al. Non-stereoselective reversal of neuropathic pain by naloxone and naltrexone: involvement of toll-like receptor 4 (TLR4). Eur J Neurosci 2008, 28, 20–29. [Google Scholar] [CrossRef]
- Hutchinson, M.R.; Zhang, Y.; Shridhar, M.; Evans, J.H.; Buchanan, M.M.; Zhao, T.X.; Slivka, P.F.; Coats, B.D.; Rezvani, N.; Wieseler, J.; et al. Evidence that opioids may have toll-like receptor 4 and MD-2 effects. Brain Behav Immun 2010, 24, 83–95. [Google Scholar] [CrossRef]
- Wang, X.; Loram, L.C.; Ramos, K.; de Jesus, A.J.; Thomas, J.; Cheng, K.; Reddy, A.; Somogyi, A.A.; Hutchinson, M.R.; Watkins, L.R.; Yin, H. Morphine activates neuroinflammation in a manner parallel to endotoxin. Proc Natl Acad Sci U S A 2012, 109, 6325–6330. [Google Scholar] [CrossRef]
- Brown, K.T.; Levis, S.C.; O'Neill, C.E.; Northcutt, A.L.; Fabisiak, T.J.; Watkins, L.R.; Bachtell, R.K. Innate immune signaling in the ventral tegmental area contributes to drug-primed reinstatement of cocaine seeking. Brain Behav Immun 2018, 67, 130–138. [Google Scholar] [CrossRef]
- Northcutt, A.L.; Hutchinson, M.R.; Wang, X.; Baratta, M.V.; Hiranita, T.; Cochran, T.A.; Pomrenze, M.B.; Galer, E.L.; Kopajtic, T.A.; Li, C.M.; et al. DAT isn't all that: cocaine reward and reinforcement require Toll-like receptor 4 signaling. Mol Psychiatry 2015, 20, 1525–1537. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Gao, S.Q.; Liu, Z.C.; Chen, X.; He, J.G.; Hu, Z.L. The HMGB1-RAGE axis in nucleus accumbens facilitates cocaine-induced conditioned place preference via modulating microglial activation. Brain Behav 2024, 14, e3457. [Google Scholar] [CrossRef]
- Chivero, E.T.; Thangaraj, A.; Tripathi, A.; Periyasamy, P.; Guo, M.L.; Buch, S. NLRP3 Inflammasome Blockade Reduces Cocaine-Induced Microglial Activation and Neuroinflammation. Mol Neurobiol 2021, 58, 2215–2230. [Google Scholar] [CrossRef] [PubMed]
- Cotto, B.; Li, H.; Tuma, R.F.; Ward, S.J.; Langford, D. Cocaine-mediated activation of microglia and microglial MeCP2 and BDNF production. Neurobiol Dis 2018, 117, 28–41. [Google Scholar] [CrossRef] [PubMed]
- Burkovetskaya, M.E.; Small, R.; Guo, L.; Buch, S.; Guo, M.L. Cocaine self-administration differentially activates microglia in the mouse brain. Neurosci Lett 2020, 728, 134951. [Google Scholar] [CrossRef]
- Kays, J.S.; Yamamoto, B.K. Evaluation of Microglia/Macrophage Cells from Rat Striatum and Prefrontal Cortex Reveals Differential Expression of Inflammatory-Related mRNA after Methamphetamine. Brain Sci 2019, 9. [Google Scholar] [CrossRef]
- Chen, H.; Uz, T.; Manev, H. Minocycline affects cocaine sensitization in mice. Neurosci Lett 2009, 452, 258–261. [Google Scholar] [CrossRef]
- Lewitus, G.M.; Konefal, S.C.; Greenhalgh, A.D.; Pribiag, H.; Augereau, K.; Stellwagen, D. Microglial TNF-α Suppresses Cocaine-Induced Plasticity and Behavioral Sensitization. Neuron 2016, 90, 483–491. [Google Scholar] [CrossRef]
- Lewitus, G.M.; Pribiag, H.; Duseja, R.; St-Hilaire, M.; Stellwagen, D. An Adaptive Role of TNFα in the Regulation of Striatal Synapses. J. Neurosci. 2014, 34, 6146–6155. [Google Scholar] [CrossRef]
- Linker, K.E.; Gad, M.; Tawadrous, P.; Cano, M.; Green, K.N.; Wood, M.A.; Leslie, F.M. Microglial activation increases cocaine self-administration following adolescent nicotine exposure. Nat Commun 2020, 11, 306. [Google Scholar] [CrossRef]
- Alajaji, M.; Lazenka, M.F.; Kota, D.; Wise, L.E.; Younis, R.M.; Carroll, F.I.; Levine, A.; Selley, D.E.; Sim-Selley, L.J.; Damaj, M.I. Early adolescent nicotine exposure affects later-life cocaine reward in mice. Neuropharmacology 2016, 105, 308–317. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.T.; Levis, S.C.; O'Neill, C.E.; Levy, C.; Rice, K.C.; Watkins, L.R.; Bachtell, R.K. Toll-like receptor 4 antagonists reduce cocaine-primed reinstatement of drug seeking. Psychopharmacology (Berl) 2023, 240, 1587–1600. [Google Scholar] [CrossRef]
- Wisor, J.P.; Schmidt, M.A.; Clegern, W.C. Cerebral microglia mediate sleep/wake and neuroinflammatory effects of methamphetamine. Brain Behav Immun 2011, 25, 767–776. [Google Scholar] [CrossRef] [PubMed]
- Sinha, R. How does stress increase risk of drug abuse and relapse? Psychopharmacology (Berl) 2001, 158, 343–359. [Google Scholar] [CrossRef]
- Antelman, S.M.; Eichler, A.J.; Black, C.A.; Kocan, D. Interchangeability of stress and amphetamine in sensitization. Science 1980, 207, 329–331. [Google Scholar] [CrossRef] [PubMed]
- Piazza, P.V.; Deminiere, J.M.; Le Moal, M.; Simon, H. Factors that predict individual vulnerability to amphetamine self-administration. Science 1989, 245, 1511–1513. [Google Scholar] [CrossRef]
- Piazza, P.V.; Deminiere, J.M.; le Moal, M.; Simon, H. Stress- and pharmacologically-induced behavioral sensitization increases vulnerability to acquisition of amphetamine self-administration. Brain Res 1990, 514, 22–26. [Google Scholar] [CrossRef]
- Piazza, P.V.; Maccari, S.; Deminiere, J.M.; Le Moal, M.; Mormede, P.; Simon, H. Corticosterone levels determine individual vulnerability to amphetamine self-administration. Proc Natl Acad Sci U S A 1991, 88, 2088–2092. [Google Scholar] [CrossRef]
- Mantsch, J.R.; Ho, A.; Schlussman, S.D.; Kreek, M.J. Predictable individual differences in the initiation of cocaine self-administration by rats under extended-access conditions are dose-dependent. Psychopharmacology (Berl) 2001, 157, 31–39. [Google Scholar] [CrossRef]
- Deroche, V.; Piazza, P.V.; Casolini, P.; Maccari, S.; Le Moal, M.; Simon, H. Stress-induced sensitization to amphetamine and morphine psychomotor effects depend on stress-induced corticosterone secretion. Brain Res 1992, 598, 343–348. [Google Scholar] [CrossRef]
- Deroche, V.; Piazza, P.V.; Maccari, S.; Le Moal, M.; Simon, H. Repeated corticosterone administration sensitizes the locomotor response to amphetamine. Brain Res 1992, 584, 309–313. [Google Scholar] [CrossRef] [PubMed]
- Goeders, N.E.; Guerin, G.F. Non-contingent electric footshock facilitates the acquisition of intravenous cocaine self-administration in rats. Psychopharmacology 1994, 114, 63–70. [Google Scholar] [CrossRef] [PubMed]
- De Vry, J.; Donselaar, I.; Van Ree, J.M. Food deprivation and acquisition of intravenous cocaine self-administration in rats: effect of naltrexone and haloperidol. J Pharmacol Exp Ther 1989, 251, 735–740. [Google Scholar] [CrossRef]
- Haney, M.; Maccari, S.; Le Moal, M.; Simon, H.; Piazza, P.V. Social stress increases the acquisition of cocaine self-administration in male and female rats. Brain Res 1995, 698, 46–52. [Google Scholar] [CrossRef]
- Ramsey, N.F.; Van Ree, J.M. Emotional but not physical stress enhances intravenous cocaine self-administration in drug-naive rats. Brain Res 1993, 608, 216–222. [Google Scholar] [CrossRef]
- Rigoni, D.; Avalos, M.P.; Boezio, M.J.; Guzmán, A.S.; Calfa, G.D.; Perassi, E.M.; Pierotti, S.M.; Bisbal, M.; Garcia-Keller, C.; Cancela, L.M.; Bollati, F. Stress-induced vulnerability to develop cocaine addiction depends on cofilin modulation. Neurobiology of Stress 2021, 15, 100349. [Google Scholar] [CrossRef]
- Schenk, S.; Lacelle, G.; Gorman, K.; Amit, Z. Cocaine self-administration in rats influenced by environmental conditions: implications for the etiology of drug abuse. Neurosci Lett 1987, 81, 227–231. [Google Scholar] [CrossRef] [PubMed]
- Mantsch, J.R.; Saphier, D.; Goeders, N.E. Corticosterone facilitates the acquisition of cocaine self-administration in rats: opposite effects of the type II glucocorticoid receptor agonist dexamethasone. J Pharmacol Exp Ther 1998, 287, 72–80. [Google Scholar] [CrossRef]
- Covington, H.E., 3rd; Miczek, K.A. Repeated social-defeat stress, cocaine or morphine. Effects on behavioral sensitization and intravenous cocaine self-administration "binges". Psychopharmacology (Berl) 2001, 158, 388–398. [Google Scholar] [CrossRef]
- Miczek, K.A.; Yap, J.J.; Covington, H.E. , 3rd. Social stress, therapeutics and drug abuse: preclinical models of escalated and depressed intake. Pharmacol Ther 2008, 120, 102–128. [Google Scholar] [CrossRef]
- Engeln, M.; Fox, M.E.; Lobo, M.K. Housing conditions during self-administration determine motivation for cocaine in mice following chronic social defeat stress. Psychopharmacology (Berl) 2021, 238, 41–54. [Google Scholar] [CrossRef]
- Mantsch, J.R.; Katz, E.S. Elevation of glucocorticoids is necessary but not sufficient for the escalation of cocaine self-administration by chronic electric footshock stress in rats. Neuropsychopharmacology 2007, 32, 367–376. [Google Scholar] [CrossRef] [PubMed]
- McReynolds, J.R.; Wolf, C.P.; Starck, D.M.; Mathy, J.C.; Schaps, R.; Krause, L.A.; Hillard, C.J.; Mantsch, J.R. Role of mesolimbic cannabinoid receptor 1 in stress-driven increases in cocaine self-administration in male rats. Neuropsychopharmacology 2023, 48, 1121–1132. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.H.; Koob, G.F. Transition from moderate to excessive drug intake: change in hedonic set point. Science 1998, 282, 298–300. [Google Scholar] [CrossRef]
- Mantsch, J.R.; Yuferov, V.; Mathieu-Kia, A.M.; Ho, A.; Kreek, M.J. Neuroendocrine alterations in a high-dose, extended-access rat self-administration model of escalating cocaine use. Psychoneuroendocrinology 2003, 28, 836–862. [Google Scholar] [CrossRef] [PubMed]
- Yu, G.; Chen, H.; Sharp, B.M. Amplified reacquisition of nicotine self-administration in rats by repeated stress during abstinence. Psychopharmacology (Berl) 2014, 231, 3189–3195. [Google Scholar] [CrossRef]
- Glynn, R.M.; Rosenkranz, J.A.; Wolf, M.E.; Caccamise, A.; Shroff, F.; Smith, A.B.; Loweth, J.A. Repeated restraint stress exposure during early withdrawal accelerates incubation of cue-induced cocaine craving. Addiction Biology 2018, 23, 80–89. [Google Scholar] [CrossRef]
- Munshi, S.; Rosenkranz, J.A.; Caccamise, A.; Wolf, M.E.; Corbett, C.M.; Loweth, J.A. Cocaine and chronic stress exposure produce an additive increase in neuronal activity in the basolateral amygdala. Addiction Biology 2021, 26, e12848. [Google Scholar] [CrossRef]
- Reverte, I.; Marchetti, C.; Pezza, S.; Zenoni, S.F.; Scaringi, G.; Ferrucci, L.; D'Ottavio, G.; Pignataro, A.; Andolina, D.; Raspa, M.; et al. Microglia-mediated calcium-permeable AMPAR accumulation in the nucleus accumbens drives hyperlocomotion during cocaine withdrawal. Brain, Behavior, and Immunity 2024, 115, 535–542. [Google Scholar] [CrossRef]
- Kelly, K.A.; Miller, D.B.; Bowyer, J.F.; O'Callaghan, J.P. Chronic exposure to corticosterone enhances the neuroinflammatory and neurotoxic responses to methamphetamine. J Neurochem 2012, 122, 995–1009. [Google Scholar] [CrossRef]
- Lo Iacono, L.; Catale, C.; Martini, A.; Valzania, A.; Viscomi, M.T.; Chiurchiu, V.; Guatteo, E.; Bussone, S.; Perrone, F.; Di Sabato, P.; et al. From Traumatic Childhood to Cocaine Abuse: The Critical Function of the Immune System. Biol Psychiatry 2018, 84, 905–916. [Google Scholar] [CrossRef] [PubMed]
- Kettenmann, H.; Hanisch, U.K.; Noda, M.; Verkhratsky, A. Physiology of microglia. Physiol Rev 2011, 91, 461–553. [Google Scholar] [CrossRef] [PubMed]
- Kettenmann, H.; Kirchhoff, F.; Verkhratsky, A. Microglia: new roles for the synaptic stripper. Neuron 2013, 77, 10–18. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



