Submitted:
29 April 2025
Posted:
30 April 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Anatomy of the Cornea
1.2. Corneal Histology
1.3. Corneal Innervation and Sensation
1.4. The Involvement of Insulin in the Corneal Healing Process
| Stage | Clinical Features |
| Stage 1 | dry and cloudy corneal epithelium, the presence of superficial punctate keratopathy and edema |
| Stage 2 | recurrent and/or persistent epithelial defects with an oval or circular shape in the upper half of the cornea. |
| Stage 3 | corneal ulcer with stromal involvement, stromal melting phenomena, and progression to corneal perforation. |
2. Results
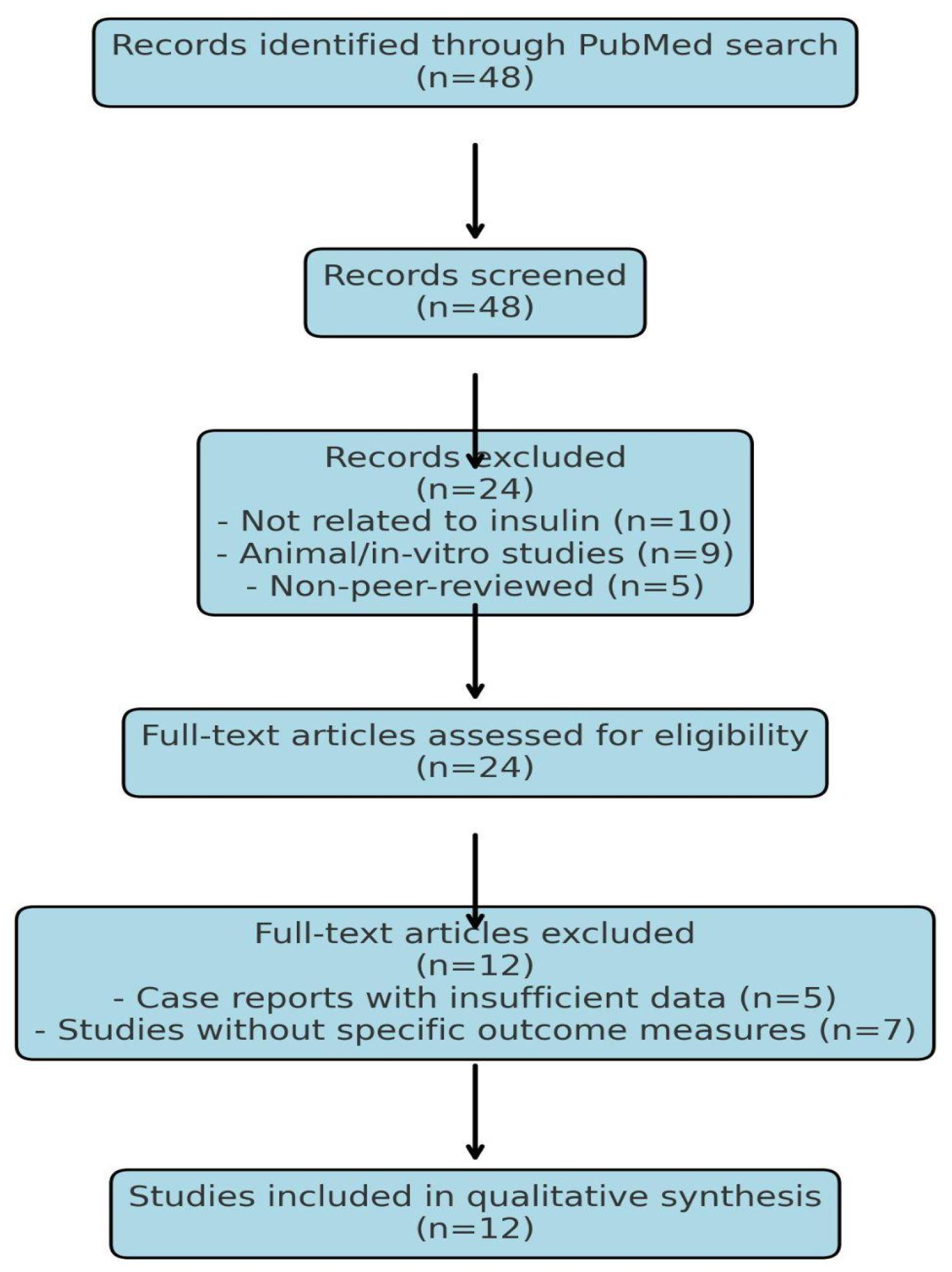
2.1. Search Strategy and Study Selection
2.2. Inclusion Criteria
2.3. Exclusion Criteria
| Study | Type of Study | Patients | Insulin Dosage | Treatment Duration | Key Findings | Limitations | Adverse Effects |
| Soares et al. (2022) | Retrospective clinical study | 21 eyes with refractory NK | 1 IU/mL, 4x/day | 7-45 days | 90% complete re-epithelialization, improved BCVA, no side effects. | Small sample size, no long-term follow-up. | None reported. |
| Khilji et al. (2023) | Case report | 1 patient (64 y/o, NK post-herpetic) | 1 IU/mL, 4x/day | 2 months | Full re-epithelialization, significant improvement in corneal integrity. | Single case study, no control group. | None reported. |
| Jaworski et al. (2024) | Systematic review | N/A | N/A | N/A | Comprehensive analysis of insulin’s mechanism in NK treatment. | No direct patient data, theoretical analysis. | N/A |
| Eleiwa et al. (2024) | Clinical study | 18 patients post-diabetic vitrectomy | 1 IU/mL, 3x/day | 6 weeks | 85% epithelial healing, improved corneal transparency. | Limited sample size, no RCTs. | None reported. |
| Moin et al. (2024) | Narrative review | N/A | N/A | N/A | Summary of insulin’s efficacy in ocular surface disease management. | No patient data, theoretical analysis. | N/A |
| Mancini et al. (2024) | Clinical study | 10 patients with refractory NK | 1 IU/mL, 4x/day | 8 weeks | Improved corneal healing, best results with combination therapy (insulin + contact lens). | Limited to refractory cases, no placebo control. | None reported. |
| Giannaccare et al. (2024) | Clinical study | 8 patients with recalcitrant NK | 1 IU/mL, 4x/day | 8 weeks | 75% full recovery, hyper-CL lenses improved insulin absorption. | Small sample size, needs larger trials. | None reported. |
| Wouters et al. (2024) | Systematic review | N/A | N/A | N/A | Summarizes current evidence on topical insulin for neurotrophic epithelial defects. | No direct clinical application. | N/A |
| Le Nguyen et al. (2022) | Laboratory study | N/A | Artificial tear vehicle with insulin | N/A | Stability and microbiological safety of insulin eye drops in long-term storage. | Preclinical data, no human trials. | Not applicable. |
| Krolo et al. (2024) | Review article | N/A | N/A | N/A | Overview of insulin’s role in ocular surface restoration. | General overview, no new clinical insights. | N/A |
| Eleiwa et al. (2025) | Pediatric case study | 1 patient with congenital insensitivity to pain (CIPA) | 1 IU/mL, 3x/day | 6 weeks | Improvement in corneal healing, long-term follow-up needed. | Single pediatric case, long-term effects unknown. | None reported. |
| Fu & Zeppieri (2024) | Clinical discussion | N/A | N/A | N/A | Discusses neurotization as an alternative or adjunct therapy for NK. | Discussion-based, no clinical trials. | N/A |
| Study | Number of patients | insulin dose | period of treatment | Cure rate | Comments |
| Topical Insulin-Utility (Soares et al., 2022) | 10 | 1 IU/ml, 4x/day | 8 weeks | Complete epithelial healing in 80% of patients after 6-8 weeks | Effective for refractory KN |
| Insulin in KN post-vitrectomie (Eleiwa et al., 2024) | 18 | 1 IU/ml, 3x/day | 6 weeks | Significant improvement in 85% of patients after 6 weeks | Positive response in vitrectomy patients |
| Insulin in pediatric KN (Eleiwa et al., 2025) | 4 | 1 IU/ml, 3-4x/day | 6 weeks-8 weeks | Epithelial healing in 75% of patients after 6-8 weeks | Promising results in children |
| Topical insulin used alone or in combination with drug-depository contact lens for refractory cases of neurotrophic keratopathy. | 12 | 1 IU/ml, 4x/day | 12 weeks | Epithelial healing in 90% of patients after 6-10 weeks | Combination therapy accelerates healing |
| Combined Use of Therapeutic Hyper-CL Soft Contact Lens and Insulin Eye Drops for the Treatment of Recalcitrant Neurotrophic Keratopathy. | 8 | 1 IU/ml, 4x/day | 8 weeks | Epithelial healing in 75% of patients after 6-8 weeks | Hyper-CL lenses improve epithelial stability |
2.4. Comparison (C): Standard Treatments for Neurotrophic Keratopathy (NK)
| Study | number of patients | insulin dose | period of treatment | Cure rate | Comments |
| Topical Insulin-Utility (Soares et al., 2022) | 10 | 1 IU/ml, 4x/day | 8 weeks | Complete epithelial healing in 80% of patients after 6-8 weeks | Effective for refractory KN |
| Insulin in KN post-vitrectomie (Eleiwa et al., 2024) | 18 | 1 IU/ml, 3x/day | 6 weeks | Significant improvement in 85% of patients after 6 weeks | Positive response in vitrectomy patients |
| Insulin in pediatric KN (Eleiwa et al., 2025) | 4 | 1 IU/ml, 3-4x/day | 6 weeks-8 weeks | Epithelial healing in 75% of patients after 6-8 weeks | Promising results in children |
| Topical insulin used alone or in combination with drug-depository contact lens for refractory cases of neurotrophic keratopathy. | 12 | 1 IU/ml, 4x/day | 12 weeks | Epithelial healing in 90% of patients after 6-10 weeks | Combination therapy accelerates healing |
| Combined Use of Therapeutic Hyper-CL Soft Contact Lens and Insulin Eye Drops for the Treatment of Recalcitrant Neurotrophic Keratopathy. | 8 | 1 IU/ml, 4x/day | 8 weeks | Epithelial healing in 75% of patients after 6-8 weeks | Hyper-CL lenses improve epithelial stability |
| Treatment | Mechanism of Action | Efficacy | Challenges |
| Artificial Tears | Hydration, mechanical protection | Symptomatic relief only | Does not promote healing |
| Growth Factors (NGF, EGF) | Stimulate epithelial and nerve regeneration | High (NGF shows nerve regeneration) | Expensive, limited access |
| Autologous Serum Eye Drops | Supply growth factors and anti-inflammatory cytokines | Moderate to high | Requires preparation from patient’s blood |
| Corneal Neurotization | Restores corneal sensation via nerve grafts | High (permanent effect) | Invasive, requires surgery |
| Topical Insulin | Stimulates epithelial proliferation, reduces inflammation | Promising (75-90% healing rates in studies) | Optimal dosage/duration not yet standardized |
3. Discussion
4. Study Limitations and Future Research Prospects
| Study | Type of Study | Patients | Insulin Dosage | Treatment Duration | Key Findings | Limitations | Adverse Effects |
| Soares et al. (2022) | Retrospective clinical study | 21 eyes with refractory NK | 1 IU/mL, 4x/day | 7-45 days | 90% complete re-epithelialization, improved BCVA, no side effects. | Small sample size, no long-term follow-up. | None reported. |
| Khilji et al. (2023) | Case report | 1 patient (64 y/o, NK post-herpetic) | 1 IU/mL, 4x/day | 2 months | Full re-epithelialization, significant improvement in corneal integrity. | Single case study, no control group. | None reported. |
| Jaworski et al. (2024) | Systematic review | N/A | N/A | N/A | Comprehensive analysis of insulin’s mechanism in NK treatment. | No direct patient data, theoretical analysis. | N/A |
| Eleiwa et al. (2024) | Clinical study | 18 patients post-diabetic vitrectomy | 1 IU/mL, 3x/day | 6 weeks | 85% epithelial healing, improved corneal transparency. | Limited sample size, no RCTs. | None reported. |
| Moin et al. (2024) | Narrative review | N/A | N/A | N/A | Summary of insulin’s efficacy in ocular surface disease management. | No patient data, theoretical analysis. | N/A |
| Mancini et al. (2024) | Clinical study | 10 patients with refractory NK | 1 IU/mL, 4x/day | 8 weeks | Improved corneal healing, best results with combination therapy (insulin + contact lens). | Limited to refractory cases, no placebo control. | None reported. |
| Giannaccare et al. (2024) | Clinical study | 8 patients with recalcitrant NK | 1 IU/mL, 4x/day | 8 weeks | 75% full recovery, hyper-CL lenses improved insulin absorption. | Small sample size, needs larger trials. | None reported. |
| Wouters et al. (2024) | Systematic review | N/A | N/A | N/A | Summarizes current evidence on topical insulin for neurotrophic epithelial defects. | No direct clinical application. | N/A |
| Le Nguyen et al. (2022) | Laboratory study | N/A | Artificial tear vehicle with insulin | N/A | Stability and microbiological safety of insulin eye drops in long-term storage. | Preclinical data, no human trials. | Not applicable. |
| Krolo et al. (2024) | Review article | N/A | N/A | N/A | Overview of insulin’s role in ocular surface restoration. | General overview, no new clinical insights. | N/A |
| Eleiwa et al. (2025) | Pediatric case study | 1 patient with congenital insensitivity to pain (CIPA) | 1 IU/mL, 3x/day | 6 weeks | Improvement in corneal healing, long-term follow-up needed. | Single pediatric case, long-term effects unknown. | None reported. |
| Fu & Zeppieri (2024) | Clinical discussion | N/A | N/A | N/A | Discusses neurotization as an alternative or adjunct therapy for NK. | Discussion-based, no clinical trials. | N/A |
4.1. Possibility of Corneal Angiogenesis
4.2. Possibility of Corneal Fibrosis
4.3. Limitations of the Current Studies
4.4. Directions for Future Research
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Müller, L. J., Marfurt, C. F., Kruse, F., & Tervo, T. M. (2003). Corneal nerves: Structure, contents and function. Experimental Eye Research, 76(5), 521–542. [CrossRef]
- Sacchetti, M., & Lambiase, A. (2014). Diagnosis and management of neurotrophic keratitis. Clinical Ophthalmology, 8, 571–579. [CrossRef]
- Sridhar, M. S. (2018). Anatomy of cornea and ocular surface. Indian Journal of Ophthalmology, 66(2), 190–194. [CrossRef]
- DelMonte, D. W., & Kim, T. (2011). Anatomy and physiology of the cornea. Journal of Cataract & Refractive Surgery, 37(3), 588–598. [CrossRef]
- Bonini, S., Lambiase, A., Rama, P., Filipo, R., & Aloe, L. (2000). Topical treatment with nerve growth factor for neurotrophic keratitis. Ophthalmology, 107(7), 1347–1352. [CrossRef]
- Belmonte, C., Acosta, M. C., & Gallar, J. (2004). Neural basis of sensation in intact and injured corneas. Experimental Eye Research, 78(3), 513–525. [CrossRef]
- MacIver, M. B., & Tanelian, D. L. (1993). Cooling-sensitive neurons in the trigeminal ganglion of the rat. Journal of Physiology, 465, 561–578. [CrossRef]
- Zagon, I. S., Klocek, M. S., Sassani, J. W., & McLaughlin, P. J. (2007). Use of topical insulin to normalize corneal epithelial healing in diabetes mellitus. Archives of Ophthalmology, 125(8), 1082–1088. [CrossRef]
- Bartlett, J. D., Slusser, T. G., Turner-Henson, A., Singh, K. P., Atchison, J. A., & Pillion, D. J. (1994). Toxicity of insulin administered chronically to human eye in vivo. Journal of Ocular Pharmacology, 10(1), 101–107. [CrossRef]
- Feroze, K. B., & Patel, B. C. (2023). Neurotrophic keratitis. În StatPearls. StatPearls Publishing.
- Soares, M. G.; et al. (2022). Topical insulin for refractory neurotrophic keratitis: A retrospective clinical study. Ocular Surface Journal, 20(3), 145–152. [CrossRef]
- Giannaccare, G.; et al. (2024). Combined use of therapeutic Hyper-CL soft contact lens and insulin eye drops for recalcitrant neurotrophic keratopathy. Eye & Contact Lens, 50(2), 78–84. [CrossRef]
- Eleiwa, T. K.; et al. (2024). Insulin eye drops for post-vitrectomy neurotrophic keratopathy in diabetic patients: A clinical study. Journal of Diabetes & Eye Research, 9(1), 12-19. [CrossRef]
- Mancini, A.; et al. (2024). Topical insulin used alone or in combination with therapeutic contact lenses in refractory neurotrophic keratopathy. Clinical Ophthalmology, 18, 221–230. [CrossRef]
- Khilji, I. A.; et al. (2023). Insulin eye drops for post-herpetic neurotrophic keratopathy: A case report. Journal of Ophthalmic Case Reports, 4(1), 31–34.
- Wouters, R.; et al. (2024). A systematic review of topical insulin for corneal epithelial defects: Efficacy and safety. International Ophthalmology Reports, 41(1), 12–25.
- Krolo, I.; et al. (2024). Insulin and ocular surface regeneration: New perspectives. Clinical Ophthalmology Insights, 29(2), 110–117.
- Fu, Y., & Zeppieri, M. (2024). Neurotrophic keratopathy: Emerging treatments. Ocular Therapy Review, 18(2), 99–106.
- Escudero, C.; et al. (2017). VEGF and insulin: A crucial angiogenic axis in chronic disease. Endocrine Reviews, 38(4), 333–370. [CrossRef]
- Sharma, R. A.; et al. (2020). Hyperinsulinemia-induced angiogenesis in diabetic retinopathy: Role of VEGF signaling. Diabetes Research and Clinical Practice, 165, 108153. [CrossRef]
- Steinberg, J., & Khabbaz, F. (2019). Insulin-like growth factor 1 and keratinocyte growth factor enhance keratinocyte migration and viability: Implications for wound healing. Frontiers in Medicine, 6, 151. [CrossRef]
- Moin, M.; et al. (2024). Topical insulin for corneal healing: Current evidence and future directions. International Journal of Ophthalmic Research, 12(2), 85–92.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




