Submitted:
18 April 2025
Posted:
23 April 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
3. Results and Discussion
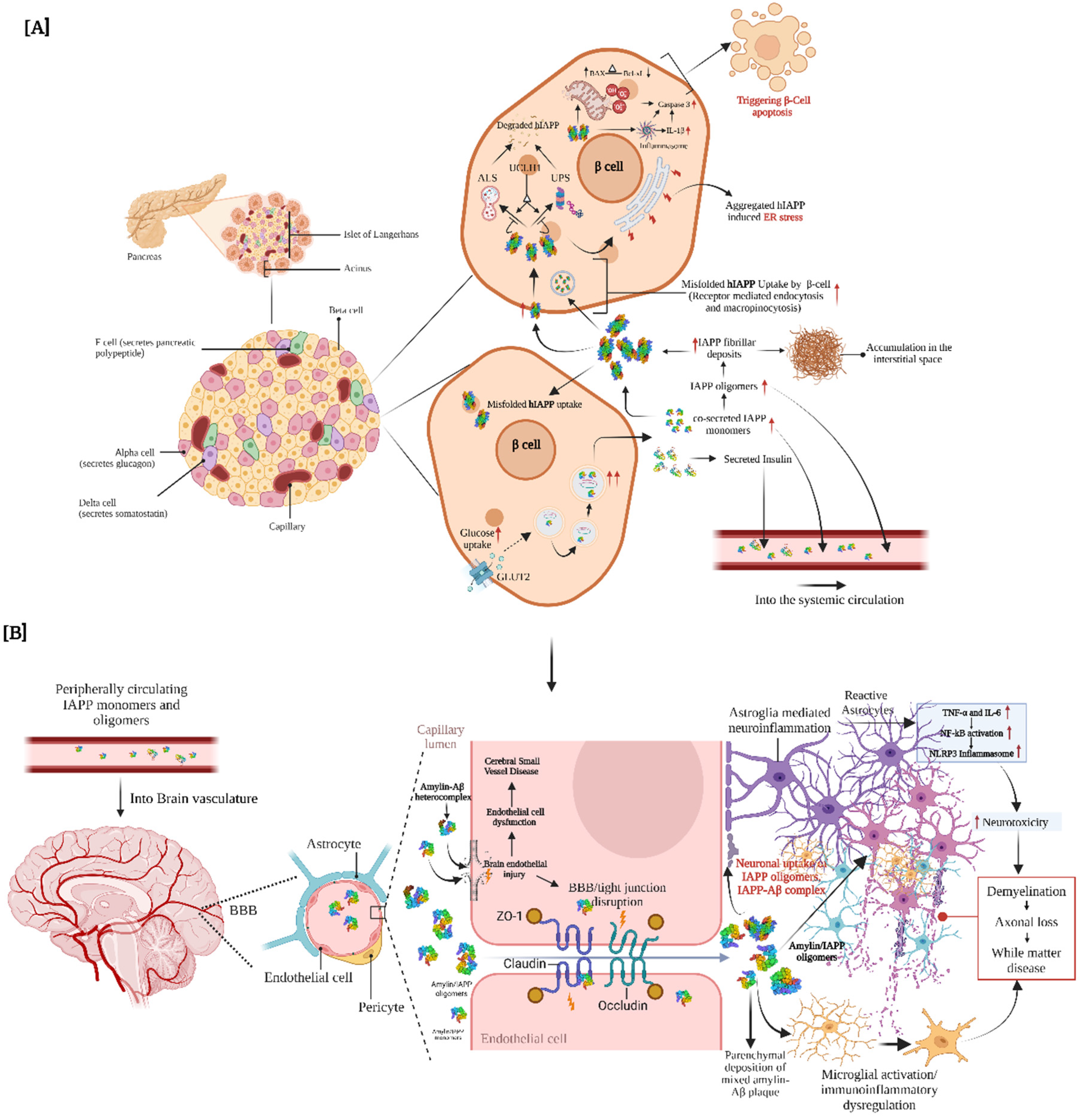
3.1. Molecular Insights into Amylin Aggregation and Proteostasis Impairment
3.2. hIAPP-Mediated Proteotoxic Stress: Impact on Ubiquitin-Proteasome and Autophagy-Lysosome Systems
3.3. hIAPP Accumulation in the Neurovascular System: A Possible Origin of Metabolic Dementia
3.4. Biophysical Links Between Pancreatic Amylin and Cerebral Amyloid-β
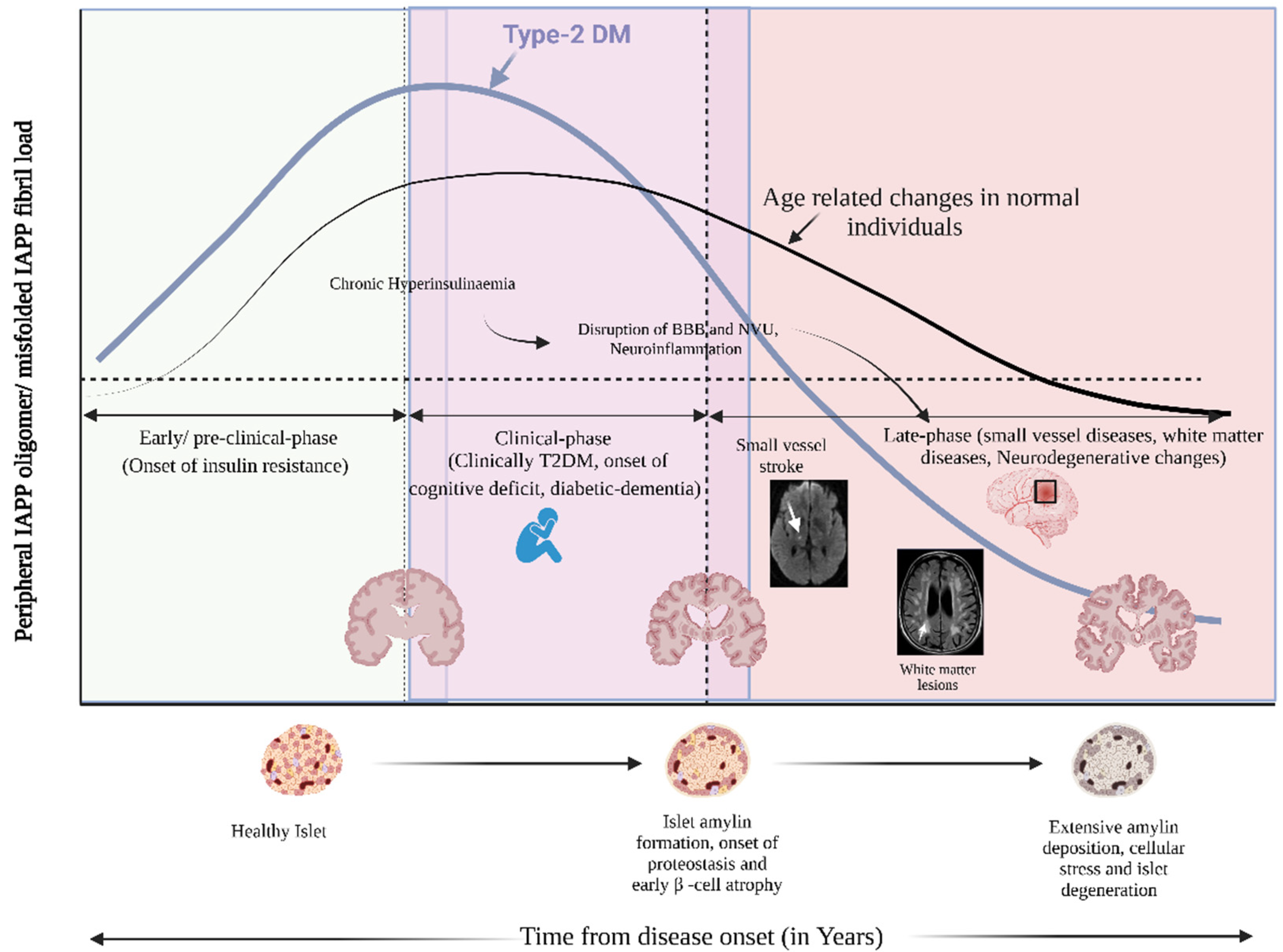
- Early Preclinical Phase (Onset of Insulin Resistance): Chronic hyperinsulinemia drives excessive hIAPP secretion, leading to early islet amyloid formation before the onset of clinical diabetes. As β-cell stress intensifies, proteostatic imbalances result in intra- and extracellular hIAPP accumulation, promoting inflammation and cellular degeneration.
- Clinical Phase (Overt T2DM, Onset of Cognitive Deficits, and Diabetic Dementia): Circulating hIAPP aggregates compromise the blood-brain barrier (BBB) and disrupt the neurovascular unit (NVU), contributing to neuroinflammation and cerebral small vessel pathology.
- Late Phase (Small Vessel Disease, White Matter Lesions, and Neurodegenerative Changes): This cascade progresses to cerebral small vessel disease, white matter lesions, and advanced neurodegeneration, including diabetic dementia.
4. Conclusions and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Aguzzi, A.; O'Connor, T. Protein aggregation diseases: pathogenicity and therapeutic perspectives. Nature reviews. Drug discovery 2010, 9, 237–248. [Google Scholar] [CrossRef] [PubMed]
- Eisele, Y.S.; Monteiro, C.; Fearns, C.; Encalada, S.E.; Wiseman, R.L.; Powers, E.T.; Kelly, J.W. Targeting protein aggregation for the treatment of degenerative diseases. Nature reviews. Drug discovery 2015, 14, 759–780. [Google Scholar] [CrossRef] [PubMed]
- Marzban, L.; Park, K.; Verchere, C.B. Islet amyloid polypeptide and type 2 diabetes. Experimental gerontology 2003, 38, 347–351. [Google Scholar] [CrossRef]
- Sanke, T.; Bell, G.I.; Sample, C.; Rubenstein, A.H.; Steiner, D.F. An islet amyloid peptide is derived from an 89-amino acid precursor by proteolytic processing. The Journal of biological chemistry 1988, 263, 17243–17246. [Google Scholar] [CrossRef] [PubMed]
- Jurgens, C.A.; Toukatly, M.N.; Fligner, C.L.; Udayasankar, J.; Subramanian, S.L.; Zraika, S.; Aston-Mourney, K.; Carr, D.B.; Westermark, P.; Westermark, G.T.; et al. β-cell loss and β-cell apoptosis in human type 2 diabetes are related to islet amyloid deposition. The American journal of pathology 2011, 178, 2632–2640. [Google Scholar] [CrossRef]
- Roberts, A.N.; Leighton, B.; Todd, J.A.; Cockburn, D.; Schofield, P.N.; Sutton, R.; Holt, S.; Boyd, Y.; Day, A.J.; Foot, E.A.; et al. Molecular and functional characterization of amylin, a peptide associated with type 2 diabetes mellitus. Proceedings of the National Academy of Sciences of the United States of America 1989, 86, 9662–9666. [Google Scholar] [CrossRef]
- Abedini, A.; Schmidt, A.M. Mechanisms of islet amyloidosis toxicity in type 2 diabetes. FEBS letters 2013, 587, 1119–1127. [Google Scholar] [CrossRef]
- Mukherjee, A.; Morales-Scheihing, D.; Butler, P.C.; Soto, C. Type 2 diabetes as a protein misfolding disease. Trends in molecular medicine 2015, 21, 439–449. [Google Scholar] [CrossRef]
- Akter, R.; Cao, P.; Noor, H.; Ridgway, Z.; Tu, L.H.; Wang, H.; Wong, A.G.; Zhang, X.; Abedini, A.; Schmidt, A.M.; et al. Islet Amyloid Polypeptide: Structure, Function, and Pathophysiology. Journal of diabetes research 2016, 2016, 2798269. [Google Scholar] [CrossRef]
- Jeong, H.R.; An, S.S. Causative factors for formation of toxic islet amyloid polypeptide oligomer in type 2 diabetes mellitus. Clinical interventions in aging 2015, 10, 1873–1879. [Google Scholar] [CrossRef]
- Caillon, L.; Hoffmann, A.R.; Botz, A.; Khemtemourian, L. Molecular Structure, Membrane Interactions, and Toxicity of the Islet Amyloid Polypeptide in Type 2 Diabetes Mellitus. Journal of diabetes research 2016, 2016, 5639875. [Google Scholar] [CrossRef]
- Ott, A.; Stolk, R.P.; Hofman, A.; van Harskamp, F.; Grobbee, D.E.; Breteler, M.M. Association of diabetes mellitus and dementia: the Rotterdam Study. Diabetologia 1996, 39, 1392–1397. [Google Scholar] [CrossRef]
- Li, L.; Hölscher, C. Common pathological processes in Alzheimer disease and type 2 diabetes: a review. Brain research reviews 2007, 56, 384–402. [Google Scholar] [CrossRef]
- Butterfield, D.A.; Di Domenico, F.; Barone, E. Elevated risk of type 2 diabetes for development of Alzheimer disease: a key role for oxidative stress in brain. Biochimica et biophysica acta 2014, 1842, 1693–1706. [Google Scholar] [CrossRef] [PubMed]
- Mondal, R.; Deb, S.; Chowdhury, D.; Sarkar, S.; Guha Roy, A.; Shome, G.; Sarkar, V.; Lahiri, D.; Benito-León, J. Neurometabolic substrate transport across brain barriers in diabetes mellitus: Implications for cognitive function and neurovascular health. Neuroscience letters 2024, 843, 138028. [Google Scholar] [CrossRef] [PubMed]
- de la Monte, S.M. Contributions of brain insulin resistance and deficiency in amyloid-related neurodegeneration in Alzheimer's disease. Drugs 2012, 72, 49–66. [Google Scholar] [CrossRef] [PubMed]
- de la Monte, S.M.; Tong, M. Brain metabolic dysfunction at the core of Alzheimer's disease. Biochemical pharmacology 2014, 88, 548–559. [Google Scholar] [CrossRef]
- Johnson, K.H.; O'Brien, T.D.; Betsholtz, C.; Westermark, P. Islet amyloid polypeptide: mechanisms of amyloidogenesis in the pancreatic islets and potential roles in diabetes mellitus. Laboratory investigation; a journal of technical methods and pathology 1992, 66, 522–535. [Google Scholar]
- Jaikaran, E.T.; Higham, C.E.; Serpell, L.C.; Zurdo, J.; Gross, M.; Clark, A.; Fraser, P.E. Identification of a novel human islet amyloid polypeptide beta-sheet domain and factors influencing fibrillogenesis. Journal of molecular biology 2001, 308, 515–525. [Google Scholar] [CrossRef]
- Westermark, P.; Engström, U.; Johnson, K.H.; Westermark, G.T.; Betsholtz, C. Islet amyloid polypeptide: pinpointing amino acid residues linked to amyloid fibril formation. Proceedings of the National Academy of Sciences of the United States of America 1990, 87, 5036–5040. [Google Scholar] [CrossRef]
- Engel, M.F. Membrane permeabilization by Islet Amyloid Polypeptide. Chemistry and physics of lipids 2009, 160, 1–10. [Google Scholar] [CrossRef]
- Janson, J.; Ashley, R.H.; Harrison, D.; McIntyre, S.; Butler, P.C. The mechanism of islet amyloid polypeptide toxicity is membrane disruption by intermediate-sized toxic amyloid particles. Diabetes 1999, 48, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Mattson, M.P. Neuroprotective signaling and the aging brain: take away my food and let me run. Brain research 2000, 886, 47–53. [Google Scholar] [CrossRef]
- Schubert, D. Serpins inhibit the toxicity of amyloid peptides. The European journal of neuroscience 1997, 9, 770–777. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Liu, H.; Yu, H.; Cooper, G.J. Fas-associated death receptor signaling evoked by human amylin in islet beta-cells. Diabetes 2008, 57, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Zhu, Z.; Chen, P. [Community investigation of the activities of daily living (ADL) and medical conditions of the elderly in Shanghai]. Zhonghua yi xue za zhi 1998, 78, 124–127. [Google Scholar]
- Casas, S.; Gomis, R.; Gribble, F.M.; Altirriba, J.; Knuutila, S.; Novials, A. Impairment of the ubiquitin-proteasome pathway is a downstream endoplasmic reticulum stress response induced by extracellular human islet amyloid polypeptide and contributes to pancreatic beta-cell apoptosis. Diabetes 2007, 56, 2284–2294. [Google Scholar] [CrossRef]
- Masters, S.L.; O'Neill, L.A. Disease-associated amyloid and misfolded protein aggregates activate the inflammasome. Trends in molecular medicine 2011, 17, 276–282. [Google Scholar] [CrossRef]
- Rivera, E.; Straub, J.; Thirumalai, D. Sequence and crowding effects in the aggregation of a 10-residue fragment derived from islet amyloid polypeptide. Biophysical journal 2009, 96, 4552–4560. [Google Scholar] [CrossRef]
- Anguiano, M.; Nowak, R.J.; Lansbury, P.T., Jr. Protofibrillar islet amyloid polypeptide permeabilizes synthetic vesicles by a pore-like mechanism that may be relevant to type II diabetes. Biochemistry 2002, 41, 11338–11343. [Google Scholar] [CrossRef]
- Imai, J.; Maruya, M.; Yashiroda, H.; Yahara, I.; Tanaka, K. The molecular chaperone Hsp90 plays a role in the assembly and maintenance of the 26S proteasome. The EMBO journal 2003, 22, 3557–3567. [Google Scholar] [CrossRef]
- CONCONI, M.; PETROPOULOS, I.; EMOD, I.; TURLIN, E.; BIVILLE, F.; FRIGUET, B. Protection from oxidative inactivation of the 20S proteasome byheat-shock protein 90. Biochemical Journal 1998, 333, 407–415. [Google Scholar] [CrossRef] [PubMed]
- Costes, S.; Huang, C.-j.; Gurlo, T.; Daval, M.; Matveyenko, A.V.; Rizza, R.A.; Butler, A.E.; Butler, P.C. β-Cell Dysfunctional ERAD/Ubiquitin/Proteasome System in Type 2 Diabetes Mediated by Islet Amyloid Polypeptide–Induced UCH-L1 Deficiency. Diabetes 2010, 60, 227–238. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Trikha, S.; Sarkar, A.; Jeremic, A.M. Proteasome regulates turnover of toxic human amylin in pancreatic cells. The Biochemical journal 2016, 473, 2655–2670. [Google Scholar] [CrossRef]
- Rivera, J.F.; Costes, S.; Gurlo, T.; Glabe, C.G.; Butler, P.C. Autophagy defends pancreatic β cells from human islet amyloid polypeptide-induced toxicity. The Journal of clinical investigation 2014, 124, 3489–3500. [Google Scholar] [CrossRef] [PubMed]
- Morita, S.; Sakagashira, S.; Shimajiri, Y.; Eberhardt, N.L.; Kondo, T.; Kondo, T.; Sanke, T. Autophagy protects against human islet amyloid polypeptide-associated apoptosis. Journal of diabetes investigation 2011, 2, 48–55. [Google Scholar] [CrossRef]
- Rivera, J.F.; Gurlo, T.; Daval, M.; Huang, C.J.; Matveyenko, A.V.; Butler, P.C.; Costes, S. Human-IAPP disrupts the autophagy/lysosomal pathway in pancreatic β-cells: protective role of p62-positive cytoplasmic inclusions. Cell Death & Differentiation 2011, 18, 415–426. [Google Scholar] [CrossRef]
- Shigihara, N.; Fukunaka, A.; Hara, A.; Komiya, K.; Honda, A.; Uchida, T.; Abe, H.; Toyofuku, Y.; Tamaki, M.; Ogihara, T.; et al. Human IAPP–induced pancreatic β cell toxicity and its regulation by autophagy. The Journal of clinical investigation 2014, 124, 3634–3644. [Google Scholar] [CrossRef]
- Oskarsson, M.E.; Paulsson, J.F.; Schultz, S.W.; Ingelsson, M.; Westermark, P.; Westermark, G.T. In vivo seeding and cross-seeding of localized amyloidosis: a molecular link between type 2 diabetes and Alzheimer disease. The American journal of pathology 2015, 185, 834–846. [Google Scholar] [CrossRef]
- Jackson, K.; Barisone, G.A.; Diaz, E.; Jin, L.W.; DeCarli, C.; Despa, F. Amylin deposition in the brain: A second amyloid in Alzheimer disease? Annals of neurology 2013, 74, 517–526. [Google Scholar] [CrossRef]
- Fawver, J.N.; Ghiwot, Y.; Koola, C.; Carrera, W.; Rodriguez-Rivera, J.; Hernandez, C.; Dineley, K.T.; Kong, Y.; Li, J.; Jhamandas, J.; et al. Islet amyloid polypeptide (IAPP): a second amyloid in Alzheimer's disease. Current Alzheimer research 2014, 11, 928–940. [Google Scholar] [CrossRef] [PubMed]
- Srodulski, S.; Sharma, S.; Bachstetter, A.B.; Brelsfoard, J.M.; Pascual, C.; Xie, X.S.; Saatman, K.E.; Van Eldik, L.J.; Despa, F. Neuroinflammation and neurologic deficits in diabetes linked to brain accumulation of amylin. Molecular neurodegeneration 2014, 9, 30. [Google Scholar] [CrossRef] [PubMed]
- Lim, Y.A.; Ittner, L.M.; Lim, Y.L.; Götz, J. Human but not rat amylin shares neurotoxic properties with Abeta42 in long-term hippocampal and cortical cultures. FEBS letters 2008, 582, 2188–2194. [Google Scholar] [CrossRef]
- Yan, L.M.; Velkova, A.; Tatarek-Nossol, M.; Andreetto, E.; Kapurniotu, A. IAPP mimic blocks Abeta cytotoxic self-assembly: cross-suppression of amyloid toxicity of Abeta and IAPP suggests a molecular link between Alzheimer's disease and type II diabetes. Angewandte Chemie (International ed. in English) 2007, 46, 1246–1252. [Google Scholar] [CrossRef] [PubMed]
- Yan, L.M.; Velkova, A.; Kapurniotu, A. Molecular characterization of the hetero-assembly of β-amyloid peptide with islet amyloid polypeptide. Current pharmaceutical design 2014, 20, 1182–1191. [Google Scholar] [CrossRef]
- Andreetto, E.; Yan, L.M.; Tatarek-Nossol, M.; Velkova, A.; Frank, R.; Kapurniotu, A. Identification of hot regions of the Abeta-IAPP interaction interface as high-affinity binding sites in both cross- and self-association. Angewandte Chemie (International ed. in English) 2010, 49, 3081–3085. [Google Scholar] [CrossRef]
- Butterfield, D.A.; Drake, J.; Pocernich, C.; Castegna, A. Evidence of oxidative damage in Alzheimer's disease brain: central role for amyloid beta-peptide. Trends in molecular medicine 2001, 7, 548–554. [Google Scholar] [CrossRef]
- Dyrks, T.; Dyrks, E.; Hartmann, T.; Masters, C.; Beyreuther, K. Amyloidogenicity of beta A4 and beta A4-bearing amyloid protein precursor fragments by metal-catalyzed oxidation. The Journal of biological chemistry 1992, 267, 18210–18217. [Google Scholar] [CrossRef]
- Palmblad, M.; Westlind-Danielsson, A.; Bergquist, J. Oxidation of methionine 35 attenuates formation of amyloid beta -peptide 1-40 oligomers. The Journal of biological chemistry 2002, 277, 19506–19510. [Google Scholar] [CrossRef]
- Lustbader, J.W.; Cirilli, M.; Lin, C.; Xu, H.W.; Takuma, K.; Wang, N.; Caspersen, C.; Chen, X.; Pollak, S.; Chaney, M.; et al. ABAD directly links Abeta to mitochondrial toxicity in Alzheimer's disease. Science (New York, N.Y.) 2004, 304, 448–452. [Google Scholar] [CrossRef]
- Martins, R.N.; Harper, C.G.; Stokes, G.B.; Masters, C.L. Increased cerebral glucose-6-phosphate dehydrogenase activity in Alzheimer's disease may reflect oxidative stress. Journal of neurochemistry 1986, 46, 1042–1045. [Google Scholar] [CrossRef]
- Yaar, M.; Zhai, S.; Pilch, P.F.; Doyle, S.M.; Eisenhauer, P.B.; Fine, R.E.; Gilchrest, B.A. Binding of beta-amyloid to the p75 neurotrophin receptor induces apoptosis. A possible mechanism for Alzheimer's disease. The Journal of clinical investigation 1997, 100, 2333–2340. [Google Scholar] [CrossRef]
- Wei, W.; Norton, D.D.; Wang, X.; Kusiak, J.W. Abeta 17-42 in Alzheimer's disease activates JNK and caspase-8 leading to neuronal apoptosis. Brain : a journal of neurology 2002, 125, 2036–2043. [Google Scholar] [CrossRef]
- Pereira, C.; Ferreiro, E.; Cardoso, S.M.; de Oliveira, C.R. Cell degeneration induced by amyloid-beta peptides: implications for Alzheimer's disease. Journal of molecular neuroscience : MN 2004, 23, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Bhaskar, K.; Miller, M.; Chludzinski, A.; Herrup, K.; Zagorski, M.; Lamb, B.T. The PI3K-Akt-mTOR pathway regulates Abeta oligomer induced neuronal cell cycle events. Molecular neurodegeneration 2009, 4, 14. [Google Scholar] [CrossRef]
- Fuentealba, R.A.; Farias, G.; Scheu, J.; Bronfman, M.; Marzolo, M.P.; Inestrosa, N.C. Signal transduction during amyloid-beta-peptide neurotoxicity: role in Alzheimer disease. Brain research. Brain research reviews 2004, 47, 275–289. [Google Scholar] [CrossRef] [PubMed]
- Mattson, M.P.; Tomaselli, K.J.; Rydel, R.E. Calcium-destabilizing and neurodegenerative effects of aggregated beta-amyloid peptide are attenuated by basic FGF. Brain research 1993, 621, 35–49. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, H.; Eckert, A.; Müller, W.E. Disturbances of the neuronal calcium homeostasis in the aging nervous system. Life sciences 1994, 55, 2011–2018. [Google Scholar] [CrossRef]
- Muller, W.E.; Kirsch, C.; Eckert, G.P. Membrane-disordering effects of beta-amyloid peptides. Biochemical Society transactions 2001, 29, 617–623. [Google Scholar] [CrossRef]
- Verdier, Y.; Zarándi, M.; Penke, B. Amyloid beta-peptide interactions with neuronal and glial cell plasma membrane: binding sites and implications for Alzheimer's disease. Journal of peptide science : an official publication of the European Peptide Society 2004, 10, 229–248. [Google Scholar] [CrossRef]
- Giulian, D.; Haverkamp, L.J.; Yu, J.H.; Karshin, W.; Tom, D.; Li, J.; Kirkpatrick, J.; Kuo, L.M.; Roher, A.E. Specific domains of beta-amyloid from Alzheimer plaque elicit neuron killing in human microglia. The Journal of neuroscience : the official journal of the Society for Neuroscience 1996, 16, 6021–6037. [Google Scholar] [CrossRef] [PubMed]
- Haass, C.; Selkoe, D.J. Soluble protein oligomers in neurodegeneration: lessons from the Alzheimer's amyloid beta-peptide. Nature reviews. Molecular cell biology 2007, 8, 101–112. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, S.T.; Vieira, M.N.; De Felice, F.G. Soluble protein oligomers as emerging toxins in Alzheimer's and other amyloid diseases. IUBMB life 2007, 59, 332–345. [Google Scholar] [CrossRef] [PubMed]
- Glabe, C.G.; Kayed, R. Common structure and toxic function of amyloid oligomers implies a common mechanism of pathogenesis. Neurology 2006, 66, S74–78. [Google Scholar] [CrossRef]
- Bharadwaj, P.R.; Dubey, A.K.; Masters, C.L.; Martins, R.N.; Macreadie, I.G. Abeta aggregation and possible implications in Alzheimer's disease pathogenesis. Journal of cellular and molecular medicine 2009, 13, 412–421. [Google Scholar] [CrossRef]
- Mondal, R.; Deb, S.; Shome, G.; Sarkar, V.; Lahiri, D.; Datta, S.S.; Benito-León, J. Molecular dynamics of amyloid-β transport in Alzheimer's disease: Exploring therapeutic plasma exchange with albumin replacement – Current insights and future perspectives. Neurologia (Barcelona, Spain) 2025, 40, 306–328. [Google Scholar] [CrossRef]
- May, P.C.; Boggs, L.N.; Fuson, K.S. Neurotoxicity of human amylin in rat primary hippocampal cultures: similarity to Alzheimer's disease amyloid-beta neurotoxicity. Journal of neurochemistry 1993, 61, 2330–2333. [Google Scholar] [CrossRef]
- Lim, Y.A.; Rhein, V.; Baysang, G.; Meier, F.; Poljak, A.; Raftery, M.J.; Guilhaus, M.; Ittner, L.M.; Eckert, A.; Götz, J. Abeta and human amylin share a common toxicity pathway via mitochondrial dysfunction. Proteomics 2010, 10, 1621–1633. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



