Submitted:
21 March 2025
Posted:
28 March 2025
You are already at the latest version
Abstract
Understanding the molecular causes of complex diseases remains one of the most pressing challenges in biomedicine. Despite large-scale genome-wide association studies (GWAS) mapping thousands of risk loci, identifying which genetic variants truly drive disease remains difficult. Traditional statistical genetics has laid a strong foundation for variant discovery, but it often struggles to capture non-linear interactions and cannot fully integrate the breadth of the interconnected multi-omics data. In recent years, deep learning approaches have shown promise in bridging these gaps: modeling high-order genetic interactions, uncovering latent biological structure, and enabling multi-layered data integration. However, issues like overfitting, lack of interpretability, and limited statistical rigor have slowed their adoption in causal inference and therapeutic target discovery. In this review, we explore how traditional statistical and deep learning methods can be applied to uncover causal mechanisms in complex disease. We critically examine the strengths and blind spots of traditional GWAS, post-GWAS fine-mapping, and Mendelian randomization, and contrast them with emerging deep learning frameworks for epistasis detection and multi-omics integration. Finally, we propose a future direction centered around hybrid models that blend the scalability of deep learning with the inferential power of statistical genetics. Our goal is to guide researchers in developing next-generation computational tools to uncover the molecular basis of complex diseases and accelerate the translation of genetic findings into effective treatments.
Keywords:
1. Introduction
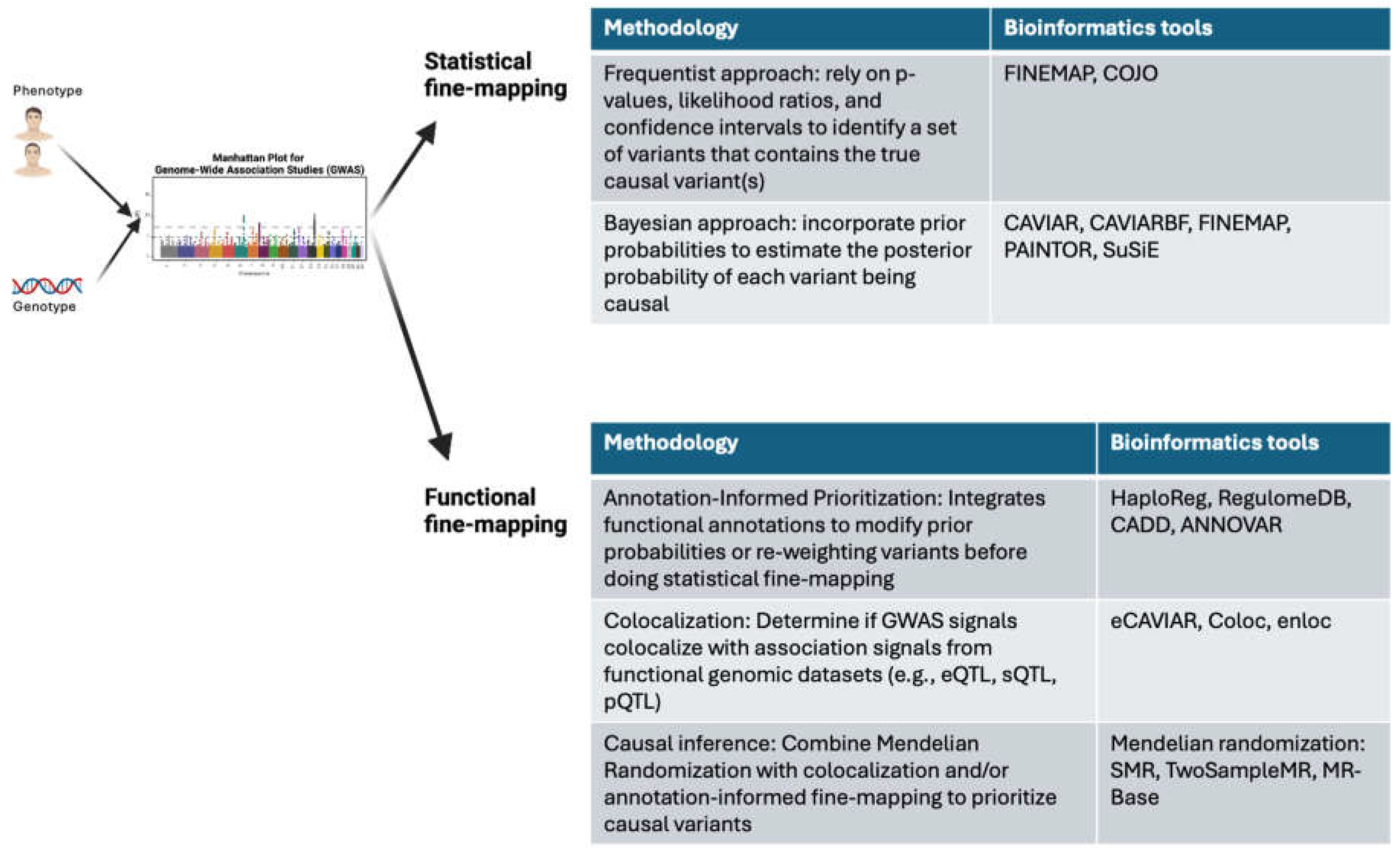
2. Established Statistical Genetics Approaches
3. Why are Traditional Statistical Genetics Methods Alone Insufficient?
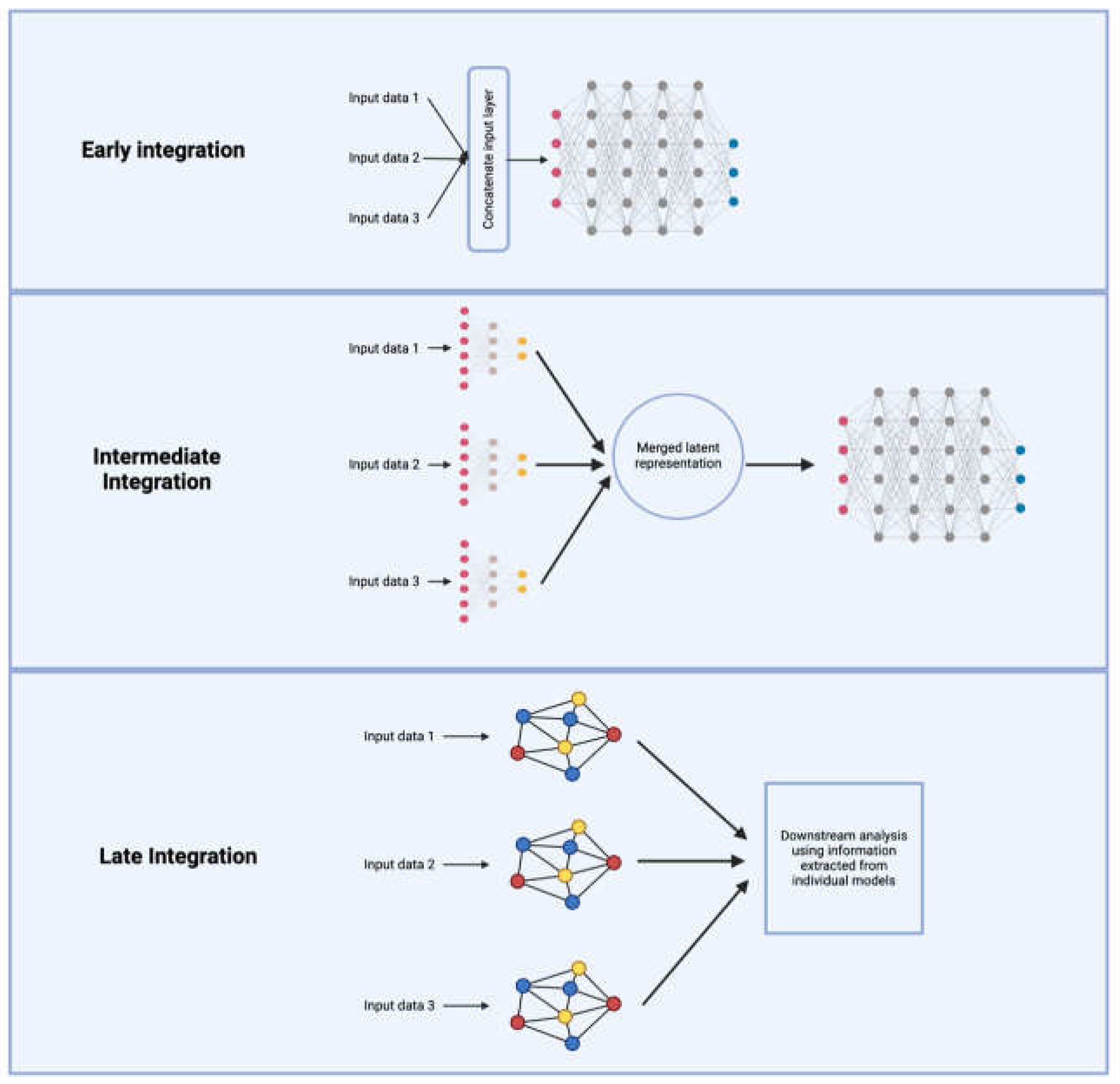
4. Emerging Deep Learning Approaches for Causal Learning with Population Genetic and Multi-Omics Data
5. Challenges of Deep Learning Models in Genomics Research
6. Future Directions: Integrating Deep Learning with Statistical Principles
References
- Anton, N., Geamănu, A., Iancu, R., Pîrvulescu, R.A., Popa-Cherecheanu, A., Barac, R.I., Bandol, G., Bogdănici, C.M., 2024. A Mini-Review on Gene Therapy in Glaucoma and Future Directions. IJMS 25, 11019. [CrossRef]
- Ashuach, T., Gabitto, M.I., Koodli, R.V., Saldi, G.-A., Jordan, M.I., Yosef, N., 2023. MultiVI: deep generative model for the integration of multimodal data. Nat Methods 20, 1222–1231. [CrossRef]
- Athaya, T., Ripan, R.C., Li, X., Hu, H., 2023. Multimodal deep learning approaches for single-cell multi-omics data integration. Briefings in Bioinformatics 24, bbad313. [CrossRef]
- Ausmees, K., Nettelblad, C., 2022. A deep learning framework for characterization of genotype data. G3 Genes|Genomes|Genetics 12, jkac020. [CrossRef]
- Baird, D.A., Liu, J.Z., Zheng, J., Sieberts, S.K., Perumal, T., Elsworth, B., Richardson, T.G., Chen, C.-Y., Carrasquillo, M.M., Allen, M., Reddy, J.S., De Jager, P.L., Ertekin-Taner, N., Mangravite, L.M., Logsdon, B., Estrada, K., Haycock, P.C., Hemani, G., Runz, H., Smith, G.D., Gaunt, T.R., AMP-AD eQTL working group, 2021. Identifying drug targets for neurological and psychiatric disease via genetics and the brain transcriptome. PLoS Genet 17, e1009224. [CrossRef]
- Baker, E., Leonenko, G., Schmidt, K.M., Hill, M., Myers, A.J., Shoai, M., De Rojas, I., Tesi, N., Holstege, H., Van Der Flier, W.M., Pijnenburg, Y.A.L., Ruiz, A., Hardy, J., Van Der Lee, S., Escott-Price, V., 2023. What does heritability of Alzheimer’s disease represent? PLoS ONE 18, e0281440. [CrossRef]
- Bazzari, F.H., Bazzari, A.H., 2022. BACE1 Inhibitors for Alzheimer’s Disease: The Past, Present and Any Future? Molecules 27, 8823. [CrossRef]
- Benkirane, H., Pradat, Y., Michiels, S., Cournède, P.-H., 2023. CustOmics: A versatile deep-learning based strategy for multi-omics integration. PLoS Comput Biol 19, e1010921. [CrossRef]
- Benner, C., Spencer, C.C.A., Havulinna, A.S., Salomaa, V., Ripatti, S., Pirinen, M., 2016. FINEMAP: efficient variable selection using summary data from genome-wide association studies. Bioinformatics 32, 1493–1501. [CrossRef]
- Brand, J.E., Zhou, X., Xie, Y., n.d. Recent Developments in Causal Inference and Machine Learning.
- Brombacher, E., Hackenberg, M., Kreutz, C., Binder, H., Treppner, M., 2022. The performance of deep generative models for learning joint embeddings of single-cell multi-omics data. Front. Mol. Biosci. 9, 962644. [CrossRef]
- Cano-Gamez, E., Trynka, G., 2020. From GWAS to Function: Using Functional Genomics to Identify the Mechanisms Underlying Complex Diseases. Front. Genet. 11, 424. [CrossRef]
- Chai, H., Zhou, X., Zhang, Z., Rao, J., Zhao, H., Yang, Y., 2021. Integrating multi-omics data through deep learning for accurate cancer prognosis prediction. Computers in Biology and Medicine 134, 104481. [CrossRef]
- Chaudhary, K., Poirion, O.B., Lu, L., Garmire, L.X., 2018. Deep Learning–Based Multi-Omics Integration Robustly Predicts Survival in Liver Cancer. Clinical Cancer Research 24, 1248–1259. [CrossRef]
- Dai, H., Zhao, Y., Qian, C., Cai, M., Zhang, R., Chu, M., Dai, J., Hu, Z., Shen, H., Chen, F., 2013. Weighted SNP Set Analysis in Genome-Wide Association Study. PLoS ONE 8, e75897. [CrossRef]
- Diaz-Ortiz, M.E., Seo, Y., Posavi, M., Carceles Cordon, M., Clark, E., Jain, N., Charan, R., Gallagher, M.D., Unger, T.L., Amari, N., Skrinak, R.T., Davila-Rivera, R., Brody, E.M., Han, N., Zack, R., Van Deerlin, V.M., Tropea, T.F., Luk, K.C., Lee, E.B., Weintraub, D., Chen-Plotkin, A.S., 2022. GPNMB confers risk for Parkinson’s disease through interaction with α-synuclein. Science 377, eabk0637. [CrossRef]
- Dibaeinia, P., Ojha, A., Sinha, S., 2025. Interpretable AI for inference of causal molecular relationships from omics data. Sci. Adv. 11, eadk0837. [CrossRef]
- Dinu, I., Mahasirimongkol, S., Liu, Q., Yanai, H., Sharaf Eldin, N., Kreiter, E., Wu, X., Jabbari, S., Tokunaga, K., Yasui, Y., 2012. SNP-SNP Interactions Discovered by Logic Regression Explain Crohn’s Disease Genetics. PLoS ONE 7, e43035. [CrossRef]
- Dong, X., Liu, C., Dozmorov, M., 2021. Review of multi-omics data resources and integrative analysis for human brain disorders. Briefings in Functional Genomics 20, 223–234. [CrossRef]
- Elmaleh, D.R., Downey, M.A., Kundakovic, L., Wilkinson, J.E., Neeman, Z., Segal, E., 2021. New Approaches to Profile the Microbiome for Treatment of Neurodegenerative Disease. JAD 82, 1373–1401. [CrossRef]
- Fang, H., Chen, L., Knight, J.C., 2020. From genome-wide association studies to rational drug target prioritisation in inflammatory arthritis. The Lancet Rheumatology 2, e50–e62. [CrossRef]
- Gallagher, M.D., Chen-Plotkin, A.S., 2018. The Post-GWAS Era: From Association to Function. The American Journal of Human Genetics 102, 717–730. [CrossRef]
- Genetic Investigation of ANthropometric Traits (GIANT) Consortium, DIAbetes Genetics Replication And Meta-analysis (DIAGRAM) Consortium, Yang, J., Ferreira, T., Morris, A.P., Medland, S.E., Madden, P.A.F., Heath, A.C., Martin, N.G., Montgomery, G.W., Weedon, M.N., Loos, R.J., Frayling, T.M., McCarthy, M.I., Hirschhorn, J.N., Goddard, M.E., Visscher, P.M., 2012. Conditional and joint multiple-SNP analysis of GWAS summary statistics identifies additional variants influencing complex traits. Nat Genet 44, 369–375. [CrossRef]
- Giambartolomei, C., Vukcevic, D., Schadt, E.E., Franke, L., Hingorani, A.D., Wallace, C., Plagnol, V., 2014. Bayesian Test for Colocalisation between Pairs of Genetic Association Studies Using Summary Statistics. PLoS Genet 10, e1004383. [CrossRef]
- Gupta, V., Chitranshi, N., Gupta, V.B., 2024. Genetic Risk, Inflammation, and Therapeutics: An Editorial Overview of Recent Advances in Aging Brains and Neurodegeneration. Aging and disease 15, 1989. [CrossRef]
- Hao, X., Zeng, P., Zhang, S., Zhou, X., 2018. Identifying and exploiting trait-relevant tissues with multiple functional annotations in genome-wide association studies. PLoS Genet 14, e1007186. [CrossRef]
- Hauptmann, T., Kramer, S., 2023. A fair experimental comparison of neural network architectures for latent representations of multi-omics for drug response prediction. BMC Bioinformatics 24, 45. [CrossRef]
- Heilbron, K., Mozaffari, S.V., Vacic, V., Yue, P., Wang, W., Shi, J., Jubb, A.M., Pitts, S.J., Wang, X., 2021. Advancing drug discovery using the power of the human genome. The Journal of Pathology 254, 418–429. [CrossRef]
- Hill-Burns, E.M., Factor, S.A., Zabetian, C.P., Thomson, G., Payami, H., 2011. Evidence for More than One Parkinson’s Disease-Associated Variant within the HLA Region. PLoS ONE 6, e27109. [CrossRef]
- Hingorani, A.D., Kuan, V., Finan, C., Kruger, F.A., Gaulton, A., Chopade, S., Sofat, R., MacAllister, R.J., Overington, J.P., Hemingway, H., Denaxas, S., Prieto, D., Casas, J.P., 2019. Improving the odds of drug development success through human genomics: modelling study. Sci Rep 9, 18911. [CrossRef]
- Hoffmann, M., Poschenrieder, J.M., Incudini, M., Baier, S., Fritz, A., Maier, A., Hartung, M., Hoffmann, C., Trummer, N., Adamowicz, K., Picciani, M., Scheibling, E., Harl, M.V., Lesch, I., Frey, H., Kayser, S., Wissenberg, P., Schwartz, L., Hafner, L., Acharya, A., Hackl, L., Grabert, G., Lee, S.-G., Cho, G., Cloward, M.E., Jankowski, J., Lee, H.K., Tsoy, O., Wenke, N., Pedersen, A.G., Bønnelykke, K., Mandarino, A., Melograna, F., Schulz, L., Climente-González, H., Wilhelm, M., Iapichino, L., Wienbrandt, L., Ellinghaus, D., Van Steen, K., Grossi, M., Furth, P.A., Hennighausen, L., Di Pierro, A., Baumbach, J., Kacprowski, T., List, M., Blumenthal, D.B., 2024a. Network medicine-based epistasis detection in complex diseases: ready for quantum computing. Nucleic Acids Research 52, 10144–10160. [CrossRef]
- Hoffmann, M., Poschenrieder, J.M., Incudini, M., Baier, S., Fritz, A., Maier, A., Hartung, M., Hoffmann, C., Trummer, N., Adamowicz, K., Picciani, M., Scheibling, E., Harl, M.V., Lesch, I., Frey, H., Kayser, S., Wissenberg, P., Schwartz, L., Hafner, L., Acharya, A., Hackl, L., Grabert, G., Lee, S.-G., Cho, G., Cloward, M.E., Jankowski, J., Lee, H.K., Tsoy, O., Wenke, N., Pedersen, A.G., Bønnelykke, K., Mandarino, A., Melograna, F., Schulz, L., Climente-González, H., Wilhelm, M., Iapichino, L., Wienbrandt, L., Ellinghaus, D., Van Steen, K., Grossi, M., Furth, P.A., Hennighausen, L., Di Pierro, A., Baumbach, J., Kacprowski, T., List, M., Blumenthal, D.B., 2024b. Network medicine-based epistasis detection in complex diseases: ready for quantum computing. Nucleic Acids Research 52, 10144–10160. [CrossRef]
- Hormozdiari, F., Kostem, E., Kang, E.Y., Pasaniuc, B., Eskin, E., n.d. Identifying Causal Variants at Loci with Multiple Signals of Association.
- Hormozdiari, F., van de Bunt, M., Segrè, A.V., Li, X., Joo, J.W.J., Bilow, M., Sul, J.H., Sankararaman, S., Pasaniuc, B., Eskin, E., 2016. Colocalization of GWAS and eQTL Signals Detects Target Genes. The American Journal of Human Genetics 99, 1245–1260. [CrossRef]
- Hu, P., Jiao, R., Jin, L., Xiong, M., 2018. Application of Causal Inference to Genomic Analysis: Advances in Methodology. Front. Genet. 9, 238. [CrossRef]
- Hu, X., Chu, L., Pei, J., Liu, W., Bian, J., 2021. Model complexity of deep learning: a survey. Knowl Inf Syst 63, 2585–2619. [CrossRef]
- Huang, W., Richards, S., Carbone, M.A., Zhu, D., Anholt, R.R.H., Ayroles, J.F., Duncan, L., Jordan, K.W., Lawrence, F., Magwire, M.M., Warner, C.B., Blankenburg, K., Han, Y., Javaid, M., Jayaseelan, J., Jhangiani, S.N., Muzny, D., Ongeri, F., Perales, L., Wu, Y.-Q., Zhang, Y., Zou, X., Stone, E.A., Gibbs, R.A., Mackay, T.F.C., 2012. Epistasis dominates the genetic architecture of Drosophila quantitative traits. Proc. Natl. Acad. Sci. U.S.A. 109, 15553–15559. [CrossRef]
- Islam, Md.M., Wang, Y., Hu, P., 2018. Deep Learning Models for Predicting Phenotypic Traits and Diseases from Omics Data, in: Aceves-Fernandez, M.A. (Ed.), Artificial Intelligence - Emerging Trends and Applications. InTech. [CrossRef]
- Kang, M., Ko, E., Mersha, T.B., 2022. A roadmap for multi-omics data integration using deep learning. Briefings in Bioinformatics 23, bbab454. [CrossRef]
- Kelly, J., Berzuini, C., Keavney, B., Tomaszewski, M., Guo, H., 2022. A review of causal discovery methods for molecular network analysis. Molec Gen & Gen Med 10, e2055. [CrossRef]
- King, E.A., Davis, J.W., Degner, J.F., 2019. Are drug targets with genetic support twice as likely to be approved? Revised estimates of the impact of genetic support for drug mechanisms on the probability of drug approval. PLoS Genet 15, e1008489. [CrossRef]
- Kong, Y., Yu, T., 2018. A graph-embedded deep feedforward network for disease outcome classification and feature selection using gene expression data. Bioinformatics 34, 3727–3737. [CrossRef]
- Lagemann, K., Lagemann, C., Taschler, B., Mukherjee, S., 2023. Deep learning of causal structures in high dimensions under data limitations. Nat Mach Intell 5, 1306–1316. [CrossRef]
- Li, Y., Wu, F.-X., Ngom, A., 2016a. A review on machine learning principles for multi-view biological data integration. Brief Bioinform bbw113. [CrossRef]
- Li, Y., Wu, F.-X., Ngom, A., 2016b. A review on machine learning principles for multi-view biological data integration. Brief Bioinform bbw113. [CrossRef]
- Liu, B., Montgomery, S.B., 2020. Identifying causal variants and genes using functional genomics in specialized cell types and contexts. Hum Genet 139, 95–102. [CrossRef]
- Liu, B., Wei, Y., Zhang, Y., Yang, Q., 2017. Deep Neural Networks for High Dimension, Low Sample Size Data, in: Proceedings of the Twenty-Sixth International Joint Conference on Artificial Intelligence. Presented at the Twenty-Sixth International Joint Conference on Artificial Intelligence, International Joint Conferences on Artificial Intelligence Organization, Melbourne, Australia, pp. 2287–2293. [CrossRef]
- Lu, Q., Yao, X., Hu, Y., Zhao, H., 2016. GenoWAP: GWAS signal prioritization through integrated analysis of genomic functional annotation. Bioinformatics 32, 542–548. [CrossRef]
- Ma, T., Zhang, A., 2019. Integrate multi-omics data with biological interaction networks using Multi-view Factorization AutoEncoder (MAE). BMC Genomics 20, 944. [CrossRef]
- Milne, R.L., Herranz, J., Michailidou, K., Dennis, J., Tyrer, J.P., Zamora, M.P., Arias-Perez, J.I., González-Neira, A., Pita, G., Alonso, M.R., Wang, Q., Bolla, M.K., Czene, K., Eriksson, M., Humphreys, K., Darabi, H., Li, J., Anton-Culver, H., Neuhausen, S.L., Ziogas, A., Clarke, C.A., Hopper, J.L., Dite, G.S., Apicella, C., Southey, M.C., Chenevix-Trench, G., kConFab Investigators, Australian Ovarian Cancer Study Group, Swerdlow, A., Ashworth, A., Orr, N., Schoemaker, M., Jakubowska, A., Lubinski, J., Jaworska-Bieniek, K., Durda, K., Andrulis, I.L., Knight, J.A., Glendon, G., Mulligan, A.M., Bojesen, S.E., Nordestgaard, B.G., Flyger, H., Nevanlinna, H., Muranen, T.A., Aittomäki, K., Blomqvist, C., Chang-Claude, J., Rudolph, A., Seibold, P., Flesch-Janys, D., Wang, X., Olson, J.E., Vachon, C., Purrington, K., Winqvist, R., Pylkäs, K., Jukkola-Vuorinen, A., Grip, M., Dunning, A.M., Shah, M., Guénel, P., Truong, T., Sanchez, M., Mulot, C., Brenner, H., Dieffenbach, A.K., Arndt, V., Stegmaier, C., Lindblom, A., Margolin, S., Hooning, M.J., Hollestelle, A., Collée, J.M., Jager, A., Cox, A., Brock, I.W., Reed, M.W.R., Devilee, P., Tollenaar, R.A.E.M., Seynaeve, C., Haiman, C.A., Henderson, B.E., Schumacher, F., Le Marchand, L., Simard, J., Dumont, M., Soucy, P., Dörk, T., Bogdanova, N.V., Hamann, U., Försti, A., Rüdiger, T., Ulmer, H.-U., Fasching, P.A., Häberle, L., Ekici, A.B., Beckmann, M.W., Fletcher, O., Johnson, N., Dos Santos Silva, I., Peto, J., Radice, P., Peterlongo, P., Peissel, B., Mariani, P., Giles, G.G., Severi, G., Baglietto, L., Sawyer, E., Tomlinson, I., Kerin, M., Miller, N., Marme, F., Burwinkel, B., Mannermaa, A., Kataja, V., Kosma, V.-M., Hartikainen, J.M., Lambrechts, D., Yesilyurt, B.T., Floris, G., Leunen, K., Alnæs, G.G., Kristensen, V., Børresen-Dale, A.-L., García-Closas, M., Chanock, S.J., Lissowska, J., Figueroa, J.D., Schmidt, M.K., Broeks, A., Verhoef, S., Rutgers, E.J., Brauch, H., Brüning, T., Ko, Y.-D., The GENICA Network, Couch, F.J., Toland, A.E., The TNBCC, Yannoukakos, D., Pharoah, P.D.P., Hall, P., Benítez, J., Malats, N., Easton, D.F., 2014. A large-scale assessment of two-way SNP interactions in breast cancer susceptibility using 46 450 cases and 42 461 controls from the breast cancer association consortium. Human Molecular Genetics 23, 1934–1946. [CrossRef]
- Minikel, E.V., Painter, J.L., Dong, C.C., Nelson, M.R., 2024. Refining the impact of genetic evidence on clinical success. Nature 629, 624–629. [CrossRef]
- Moore, J.H., Gilbert, J.C., Tsai, C.-T., Chiang, F.-T., Holden, T., Barney, N., White, B.C., 2006. A flexible computational framework for detecting, characterizing, and interpreting statistical patterns of epistasis in genetic studies of human disease susceptibility. Journal of Theoretical Biology 241, 252–261. [CrossRef]
- Mortezaei, Z., Tavallaei, M., 2021. Recent innovations and in-depth aspects of post-genome wide association study (Post-GWAS) to understand the genetic basis of complex phenotypes. Heredity 127, 485–497. [CrossRef]
- Nicholls, H.L., John, C.R., Watson, D.S., Munroe, P.B., Barnes, M.R., Cabrera, C.P., 2020. Reaching the End-Game for GWAS: Machine Learning Approaches for the Prioritization of Complex Disease Loci. Front. Genet. 11, 350. [CrossRef]
- Niel, C., Sinoquet, C., Dina, C., Rocheleau, G., 2015. A survey about methods dedicated to epistasis detection. Front. Genet. 6. [CrossRef]
- Nott, A., Holtman, I.R., Coufal, N.G., Schlachetzki, J.C.M., Yu, M., Hu, R., Han, C.Z., Pena, M., Xiao, J., Wu, Y., Keulen, Z., Pasillas, M.P., O’Connor, C., Nickl, C.K., Schafer, S.T., Shen, Z., Rissman, R.A., Brewer, J.B., Gosselin, D., Gonda, D.D., Levy, M.L., Rosenfeld, M.G., McVicker, G., Gage, F.H., Ren, B., Glass, C.K., 2019. Brain cell type–specific enhancer–promoter interactome maps and disease - risk association. Science 366, 1134–1139. [CrossRef]
- Park, S., Soh, J., Lee, H., 2021. Super.FELT: supervised feature extraction learning using triplet loss for drug response prediction with multi-omics data. BMC Bioinformatics 22, 269. [CrossRef]
- Passeri, E., Elkhoury, K., Morsink, M., Broersen, K., Linder, M., Tamayol, A., Malaplate, C., Yen, F.T., Arab-Tehrany, E., 2022. Alzheimer’s Disease: Treatment Strategies and Their Limitations. IJMS 23, 13954. [CrossRef]
- Pérez-Granado, J., Piñero, J., Furlong, L.I., 2022. Benchmarking post-GWAS analysis tools in major depression: Challenges and implications. Front. Genet. 13, 1006903. [CrossRef]
- Poirion, O.B., Jing, Z., Chaudhary, K., Huang, S., Garmire, L.X., 2021. DeepProg: an ensemble of deep-learning and machine-learning models for prognosis prediction using multi-omics data. Genome Med 13, 112. [CrossRef]
- Ritchie, M.D., Hahn, L.W., Roodi, N., Bailey, L.R., Dupont, W.D., Parl, F.F., Moore, J.H., 2001. Multifactor-Dimensionality Reduction Reveals High-Order Interactions among Estrogen-Metabolism Genes in Sporadic Breast Cancer. The American Journal of Human Genetics 69, 138–147. [CrossRef]
- Ronen, J., Hayat, S., Akalin, A., 2019. Evaluation of colorectal cancer subtypes and cell lines using deep learning. Life Sci. Alliance 2, e201900517. [CrossRef]
- Sapkota, Y., Mackey, J.R., Lai, R., Franco-Villalobos, C., Lupichuk, S., Robson, P.J., Kopciuk, K., Cass, C.E., Yasui, Y., Damaraju, S., 2013. Assessing SNP-SNP Interactions among DNA Repair, Modification and Metabolism Related Pathway Genes in Breast Cancer Susceptibility. PLoS ONE 8, e64896. [CrossRef]
- Schmid, A.W., Fauvet, B., Moniatte, M., Lashuel, H.A., 2013. Alpha-synuclein Post-translational Modifications as Potential Biomarkers for Parkinson Disease and Other Synucleinopathies. Molecular & Cellular Proteomics 12, 3543–3558. [CrossRef]
- Scholkopf, B., Locatello, F., Bauer, S., Ke, N.R., Kalchbrenner, N., Goyal, A., Bengio, Y., 2021. Toward Causal Representation Learning. Proc. IEEE 109, 612–634. [CrossRef]
- Sturma, N., Squires, C., Drton, M., Uhler, C., 2023. Unpaired Multi-Domain Causal Representation Learning. [CrossRef]
- Sun, Q., Crowley, C.A., Huang, L., Wen, J., Chen, J., Bao, E.L., Auer, P.L., Lettre, G., Reiner, A.P., Sankaran, V.G., Raffield, L.M., Li, Y., 2022. From GWAS variant to function: A study of ∼148,000 variants for blood cell traits. Human Genetics and Genomics Advances 3, 100063. [CrossRef]
- Tam, V., Patel, N., Turcotte, M., Bossé, Y., Paré, G., Meyre, D., 2019. Benefits and limitations of genome-wide association studies. Nat Rev Genet 20, 467–484. [CrossRef]
- Tanvir, R.B., Islam, M.M., Sobhan, M., Luo, D., Mondal, A.M., 2024. MOGAT: A Multi-Omics Integration Framework Using Graph Attention Networks for Cancer Subtype Prediction. IJMS 25, 2788. [CrossRef]
- Taylor-King, J.P., Bronstein, M., Roblin, D., 2024. The Future of Machine Learning Within Target Identification: Causality, Reversibility, and Druggability. Clin Pharma and Therapeutics 115, 655–657. [CrossRef]
- Tong, L., Mitchel, J., Chatlin, K., Wang, M.D., 2020. Deep learning based feature-level integration of multi-omics data for breast cancer patients survival analysis. BMC Med Inform Decis Mak 20, 225. [CrossRef]
- Uffelmann, E., Huang, Q.Q., Munung, N.S., De Vries, J., Okada, Y., Martin, A.R., Martin, H.C., Lappalainen, T., Posthuma, D., 2021. Genome-wide association studies. Nat Rev Methods Primers 1, 59. [CrossRef]
- UK10K Consortium, Iotchkova, V., Ritchie, G.R.S., Geihs, M., Morganella, S., Min, J.L., Walter, K., Timpson, N.J., Dunham, I., Birney, E., Soranzo, N., 2019. GARFIELD classifies disease-relevant genomic features through integration of functional annotations with association signals. Nat Genet 51, 343–353. [CrossRef]
- Upton, A., Trelles, O., Cornejo-García, J.A., Perkins, J.R., 2016. Review: High-performance computing to detect epistasis in genome scale data sets. Brief Bioinform 17, 368–379. [CrossRef]
- Vahabi, N., Michailidis, G., 2022. Unsupervised Multi-Omics Data Integration Methods: A Comprehensive Review. Front. Genet. 13, 854752. [CrossRef]
- Valous, N.A., Popp, F., Zörnig, I., Jäger, D., Charoentong, P., 2024. Graph machine learning for integrated multi-omics analysis. Br J Cancer 131, 205–211. [CrossRef]
- Verma, S.S., Cooke Bailey, J.N., Lucas, A., Bradford, Y., Linneman, J.G., Hauser, M.A., Pasquale, L.R., Peissig, P.L., Brilliant, M.H., McCarty, C.A., Haines, J.L., Wiggs, J.L., Vrabec, T.R., Tromp, G., Ritchie, M.D., eMERGE Network, NEIGHBOR Consortium, 2016. Epistatic Gene-Based Interaction Analyses for Glaucoma in eMERGE and NEIGHBOR Consortium. PLoS Genet 12, e1006186. [CrossRef]
- Viaud, G., Mayilvahanan, P., Cournede, P.-H., 2022. Representation Learning for the Clustering of Multi-Omics Data. IEEE/ACM Trans. Comput. Biol. and Bioinf. 19, 135–145. [CrossRef]
- Wang, T., Shao, W., Huang, Z., Tang, H., Zhang, J., Ding, Z., Huang, K., 2021. MOGONET integrates multi-omics data using graph convolutional networks allowing patient classification and biomarker identification. Nat Commun 12, 3445. [CrossRef]
- Wei, W.-H., Hemani, G., Haley, C.S., 2014. Detecting epistasis in human complex traits. Nat Rev Genet 15, 722–733. [CrossRef]
- Wen, X., Pique-Regi, R., Luca, F., 2017. Integrating molecular QTL data into genome-wide genetic association analysis: Probabilistic assessment of enrichment and colocalization. PLoS Genet 13, e1006646. [CrossRef]
- Wu, J., Chen, Z., Xiao, S., Liu, G., Wu, W., Wang, S., 2024. DeepMoIC: multi-omics data integration via deep graph convolutional networks for cancer subtype classification. BMC Genomics 25, 1209. [CrossRef]
- Wu, Y., Xie, L., 2025. AI-driven multi-omics integration for multi-scale predictive modeling of causal genotype-environment-phenotype relationships. Computational and Structural Biotechnology Journal 27, 265–277. [CrossRef]
- Xu, C., Liu, Q., Zhou, J., Xie, M., Feng, J., Jiang, T., 2020. Quantifying functional impact of non-coding variants with multi-task Bayesian neural network. Bioinformatics 36, 1397–1404. [CrossRef]
- Yan, H., Weng, D., Li, D., Gu, Y., Ma, W., Liu, Q., 2024. Prior knowledge-guided multilevel graph neural network for tumor risk prediction and interpretation via multi-omics data integration. Briefings in Bioinformatics 25, bbae184. [CrossRef]
- Zhang, X., Xing, Y., Sun, K., Guo, Y., 2021. OmiEmbed: A Unified Multi-Task Deep Learning Framework for Multi-Omics Data. Cancers 13, 3047. [CrossRef]
- Zhang, X., Yang, F., Yang, X., Li, Q., Li, N., Zhao, Y., 2024. MoCaGCN: Cancer Subtype Classification by Developing Causal Graph Structure Learning, in: 2024 IEEE International Conference on Medical Artificial Intelligence (MedAI). Presented at the 2024 IEEE International Conference on Medical Artificial Intelligence (MedAI), IEEE, Chongqing, China, pp. 617–625. [CrossRef]
- Zhang, Y., Liu, J.S., 2007. Bayesian inference of epistatic interactions in case-control studies. Nat Genet 39, 1167–1173. [CrossRef]
- Zhao, Y., Chen, F., Zhai, R., Lin, X., Diao, N., Christiani, D.C., 2012. Association Test Based on SNP Set: Logistic Kernel Machine Based Test vs. Principal Component Analysis. PLoS ONE 7, e44978. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



