Submitted:
11 March 2025
Posted:
14 March 2025
Read the latest preprint version here
Abstract
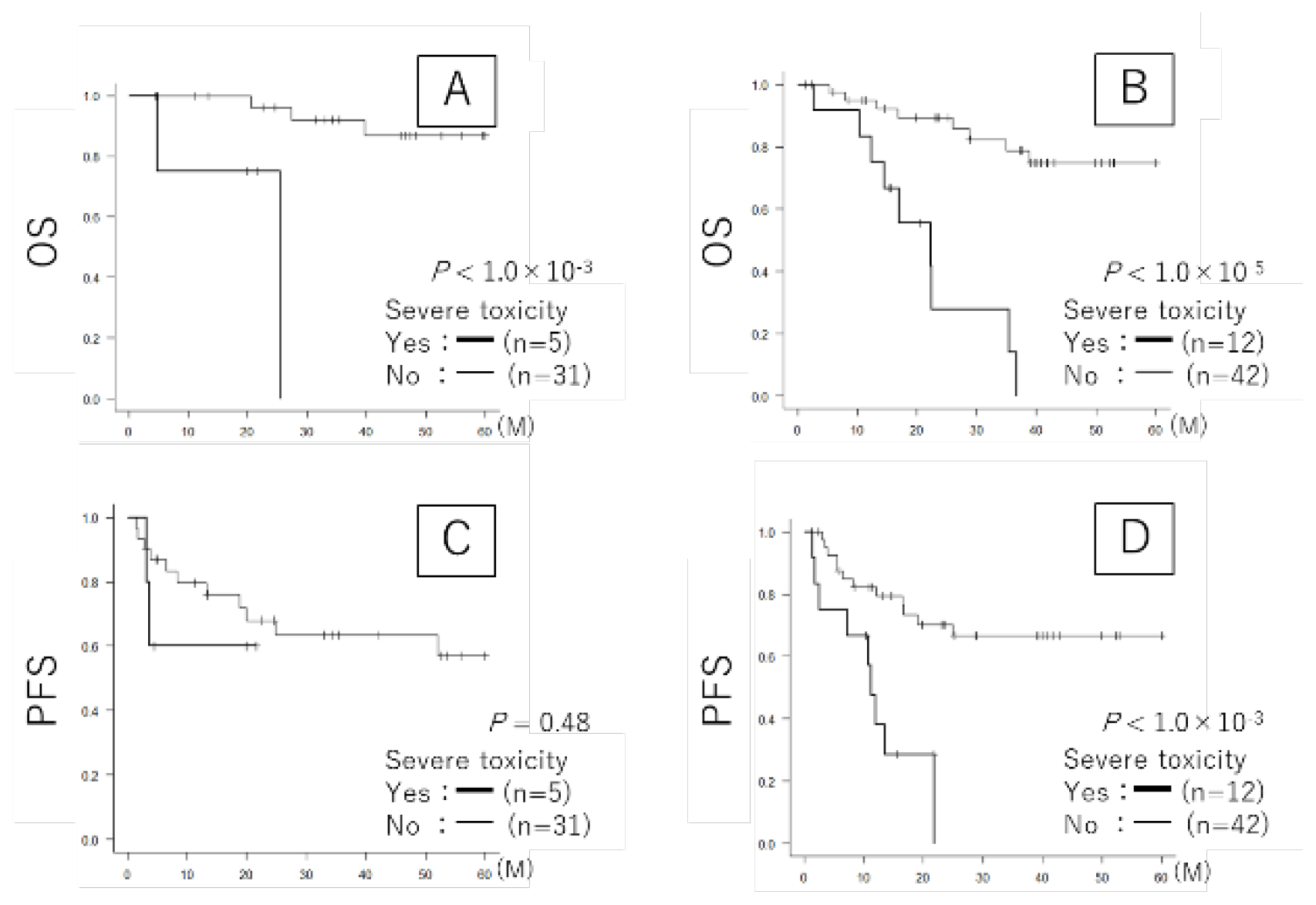
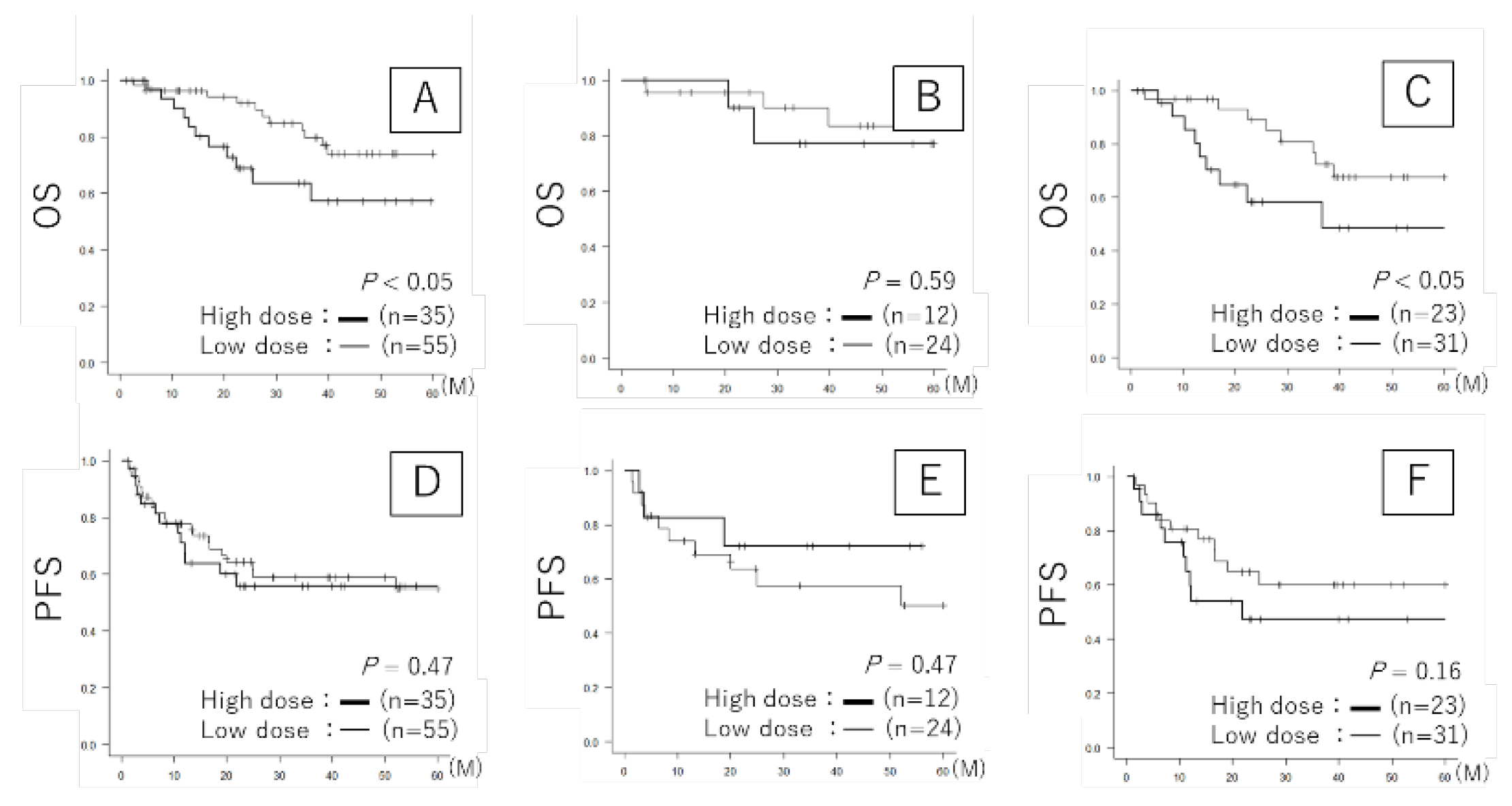
This study aimed to investigate the impact of cisplatin-based chemoradiotherapy dos-age per cycle on prognosis for elderly patients. This retrospective study included 90 patients with head and neck squamous cell carcinoma who received cisplatin - based chemoradiotherapy. Those who received triweekly cisplatin (100) regimen for defini-tive chemoradiotherapy and triweekly cisplatin (80) regimen for postoperative chemoradiotherapy were allocated to the high-dose group. Meanwhile, those who re-ceived tri-weekly cisplatin (80) regimen for definitive chemoradiotherapy and weekly cisplatin (40) regimen for postoperative chemoradiotherapy were allocated to the low-dose group. The outcomes in elderly and non-elderly patients following chemora-diotherapy were compared between the groups. As a result, the patients in the high-dose group had a significantly higher incidence of severe toxicity than that in low-the groups (P < 0.05), and the elderly patients in the high-dose group demonstrat-ed the highest rate of severe toxicity (34.8%) compared to the other groups. Further-more, only in the elderly patients, overall survival became significantly shorter in the high-dose group than that in the low-dose group (P < 0.05). In elderly patients, the total dosage of cisplatin administered to those who developed severe toxicity was signifi-cantly lower than that for patients who did not experience toxicity (P < 0.01). Fur-thermore, these patients demonstrated significantly short overall survival (P < 1.0×10-5). In conclusion, current cisplatin dosage per cycle may exhibit excessive for el-derly patients.
Keywords:
1. Introduction
2. Materials and Methods
Participants and Treatments
Evaluation
Statistical Analysis
3. Results
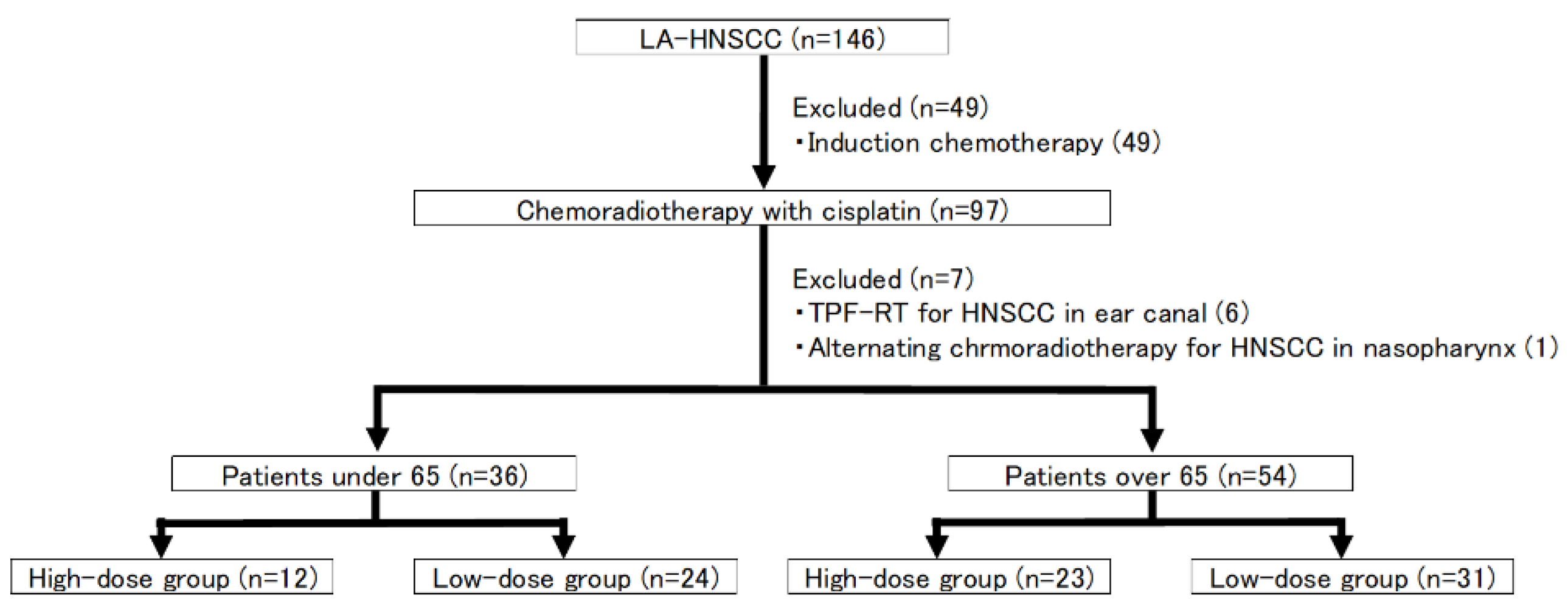
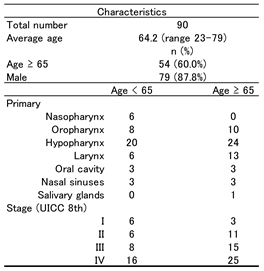
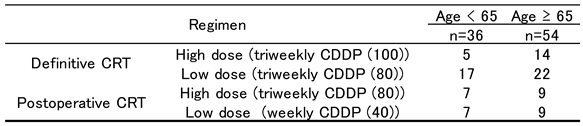
3.1. Patient Characteristics (Figure 1)
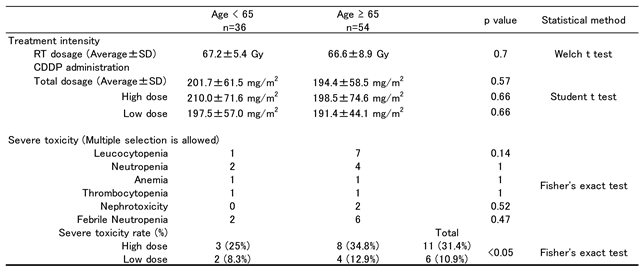
3.2. Treatment
3.3. Prognosis
3.4. Severe Toxicity and Prognosis (Table 4 and Figure 3)
 |
 |
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HNSCC | Head and neck squamous cell carcinoma |
| CRT | Chemoradiotherapy |
| ICT | Induction chemotherapy |
| OS | Overall survival |
| PFS | Progression free survival |
| JCOG | Japan Clinical Oncology Group |
| CTCAE | Common Terminology Criteria for Adverse Events |
| CRP | C-reactive protein |
| AST | Aspartate aminotransferase |
| ALT | Alanine aminotransferase |
| NLR | Neutrophil-to-lymphocyte ratio |
| mGPS | modified Glasgow Prognostic Score |
References
- Grénman, R.; Chevalier, D.; Gregoire, V.; Myers, E.; Rogers, S. Treatment of head and neck cancer in the elderly: European consensus (panel 6) at the EUFOS congress in Vienna 2007. Eur Arch Otorhinolaryngol. 2010, 267, 1619–1621. [Google Scholar] [PubMed]
- Szturz, P.; Bossi, P.; Vermorken, J.B. Systemic treatment in elderly head and neck cancer patients: recommendations for clinical practice. Curr Opin Otolaryngol Head Neck Surg. 2019, 27, 142–150. [Google Scholar] [PubMed]
- Vigneswaran, N.; Williams, M.D. Epidemiologic trends in head and neck cancer and aids in diagnosis. Oral Maxillofac Surg Clin North Am. 2014, 26, 123–141. [Google Scholar]
- Okuda, H.; Shibata, H.; Watanabe, T.; Terazawa, K.; Mori, K.; Ueda, N.; Ohashi, T.; Ogawa, T. Nonsurgical treatment strategies for elderly head and neck cancer patients: an emerging subject worldwide. Cancers. 2022, 14, 5689. [Google Scholar] [PubMed]
- Makovec, T. Cisplatin and beyond: molecular mechanisms of action and drug resistance development in cancer chemotherapy. Radiol Oncol. 2019, 53, 148–158. [Google Scholar]
- Dasari, S.; Tchounwou, P.B. Cisplatin in cancer therapy: molecular mechanisms of action. Eur J Pharmacol. 2014, 740, 364–378. [Google Scholar]
- Marty, M.; Pouillart, P.; Scholl, S.; Droz, J.P.; Azab, M.; Brion, N.; Pujade-Lauraine, E.; Paule, B.; Paes, D.; Bons, J. Comparison of the 5-hydroxytryptamine3 (serotonin) antagonist ondansetron (GR 38032F) with high-dose metoclopramide in the control of cisplatin-induced emesis. N Engl J Med. 1990, 322, 816–821. [Google Scholar] [CrossRef]
- Zenda, S.; Ishi, S.; Kawashima, M.; Arahira, S.; Tahara, M.; Hayashi, R.; Kishimoto, S.; Ichihashi, T. A dermatitis control program (DeCoP) for head and neck cancer patients receiving radiotherapy: a prospective phase II study. Int J Clin Oncol. 2013, 18, 350–355. [Google Scholar]
- Zenda, S.; Matsuura, K.; Tachibana, H.; Homma, A.; Kirita, T.; Monden, N.; Iwae, S.; Ota, Y.; Akimoto, T.; Otsuru, H.; Tahara, M.; Kato, K.; Asai, M. Multicenter phase II study of an opioid-based pain control program for head and neck cancer patients receiving chemoradiotherapy. Radiother Oncol. 2011, 101, 410–414. [Google Scholar] [CrossRef]
- Elting, L.S.; Cooksley, C.; Chambers, M.; Cantor, S.B.; Manzullo, E.; Rubenstein, E.B. The burdens of cancer therapy. Clinical and economic outcomes of chemotherapy-induced mucositis. Cancer. 2003, 98, 1531–1539. [Google Scholar]
- Neve, M.; Jameson, M.B.; Govender, S.; Hartopeanu, C. Impact of geriatric assessment on the management of older adults with head and neck cancer: a pilot study. J Geriatr Oncol. 2016, 7, 457–462. [Google Scholar] [PubMed]
- Pottel L, Lycke M, Boterberg T, Pottel H, Goethals L, Duprez F, Rottey S, Lievens Y, Van Den Noortgate N, Geldhof K, Buyse V, Kargar-Samani K, Ghekiere V, Debruyne PR. G-8 indicates overall and quality-adjusted survival in older head and neck cancer patients treated with curative radiochemotherapy. BMC Cancer. 2015, 15, 875.
- Ishii, R.; Ogawa, T.; Ohkoshi, A.; Nakanome, A.; Takahashi, M.; Katori, Y. Use of the Geriatric-8 screening tool to predict prognosis and complications in older adults with head and neck cancer: a prospective, observational study. J Geriatr Oncol. 2021, 12, 1039–1043. [Google Scholar] [PubMed]
- Mei, Z.; Shi, L.; Wang, B.; Yang, J.; Xiao, Z.; Du, P.; Wang, Q.; Yang, W. Prognostic role of pretreatment blood neutrophil-to-lymphocyte ratio in advanced cancer survivors: a systematic review and meta-analysis of 66 cohort studies. Cancer Treat Rev. 2017, 58, 1–53. [Google Scholar] [PubMed]
- Chua, W.; Charles, K.A.; Baracos, V.E.; Clarke, S.J. Neutrophil/lymphocyte ratio predicts chemotherapy outcomes in patients with advanced colorectal cancer. Br J Cancer. 2011, 104, 1288–1295. [Google Scholar]
- Piciucchi, M.; Stigliano, S.; Archibugi, L.; Zerboni, G.; Signoretti, M.; Barucca, V.; Valente, R.; Fave, G.; Capurso, G. The neutrophil/lymphocyte ratio at diagnosis is significantly associated with survival in metastatic pancreatic cancer patients. Int J Mol Sci. 2017, 18, 730. [Google Scholar]
- Toiyama, Y.; Miki, C.; Inoue, Y.; Tanaka, K.; Mohri, Y.; Kusunoki, M. Evaluation of an inflammation-based prognostic score for the identification of patients requiring postoperative adjuvant chemotherapy for stage II colorectal cancer. Exp Ther Med. 2011, 2, 95–101. [Google Scholar]
- Abe, S.; Nozawa, H.; Kawai, K.; Sasaki, K.; Murono, K.; Emoto, S.; Kishikawa, J.; Ozawa, T.; Yokoyama, Y.; Nagai, Y.; Anzai, H.; Sonoda, H.; Ishihara, S. Poor nutrition and sarcopenia are related to systemic inflammatory response in patients with rectal cancer undergoing preoperative chemoradiotherapy. Int J Colorectal Dis. 2022, 37, 189–200. [Google Scholar]
- Kiyota, N.; Tahara, M.; Mizusawa, J.; Kodaira, T.; Fujii, H.; Yamazaki, T. Weekly cisplatin plus radiation for postoperative head and neck cancer (JCOG1008): a multicenter, noninferiority, Phase II/III randomized controlled trial. J Clin Oncol. 2022, 40, 1980–1990. [Google Scholar]
- Boukovala, M.; Modest, D.P.; Ricard, I.; Fischer Von Weikersthal, L.; Decker, T.; Vehling-Kaiser, U.; Uhlig, J.; Schenk, M.; Freiberg-Richter, J.; Peuser, B.; Denzlinger, C.; Peveling Genannt Reddemann, C.; Graeven, U.; Schuch, G.; Schwaner, I.; Heinrich, K.; Neumann, J.; Jung, A.; Held, S.; Stintzing, S.; Heinemann, V.; Michl, M. Evaluation of the inflammation-based modified Glasgow Prognostic Score (mGPS) as a prognostic and predictive biomarker in patients with metastatic colorectal cancer receiving first-line chemotherapy: a post hoc analysis of the randomized phase III XELAVIRI trial (AIO KRK0110). ESMO Open. 2024, 9, 103374. [Google Scholar]
- Kanda, Y. Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transplant. 2013, 48, 452–458. [Google Scholar]
- Arlene A Forastiere, Qiang Zhang, Randal S Weber, Moshe H Maor, Helmuth Goepfert, Thomas F Pajak, et al. Long-term results of RTOG 91-11: a comparison of three nonsurgical treatment strategies to preserve the larynx in patients with locally advanced larynx cancer. J Clin Oncol. 2013, 31, 845–852. [Google Scholar]
- Adelstein, D.J.; Li, Y.; Adams, G.L.; Wagner, H.; Kish, J.A.; Ensley, J.F.; Schuller, D.E.; Forastiere, A.A. An intergroup phase III comparison of standard radiation therapy and two schedules of concurrent chemoradiotherapy in patients with unresectable squamous cell head and neck cancer. J Clin Oncol. 2003, 21, 92–98. [Google Scholar]
- Forastiere, A.A.; Zhang, Q.; Weber, R.S.; Maor, M.H.; Goepfert, H.; Pajak, T.F.; Morrison, W.; Glisson, B.; Trotti, A.; Ridge, J.A.; Thorstad, W.; Wagner, H.; Ensley, J.F.; Cooper, J.S. Long-term results of RTOG 91-11: a comparison of three nonsurgical treatment strategies to preserve the larynx in patients with locally advanced larynx cancer. J Clin Oncol. 2013, 31, 845–852. [Google Scholar] [PubMed]
- Sasaki, Y.; Tamura, T.; Eguchi, K.; Shinkai, T.; Fujiwara, Y.; Fukuda, M.; Ohe, Y.; Bungo, M.; Horichi, N.; Niimi, S.; Minato, K. Pharmacokinetics of (glycolate-0,0′)-diammine platinum (II), a new platinum derivative, in comparison with cisplatin and carboplatin. Cancer Chemother Pharmacol. 1989, 23, 243–246. [Google Scholar] [PubMed]
- Ando, Y.; Nishiyama, H.; Shimodaira, H.; Takano, N.; Sakaida, E.; Matsumoto, K.; Nakanishi, K.; Sakai, H.; Tsukamoto, S.; Komine, K.; Yasuda, Y. Chapter 3: Management of kidney injury caused by cancer drug therapy, from clinical practice guidelines for the management of kidney injury during anticancer drug therapy 2022. Int J Clin Oncol. 2023, 28, 1315–1332. [Google Scholar] [PubMed]
- Frederic Jungbauer, Lena Huber, Sonja Ludwig, Nicole Rotter, Beatrice Walter, Lena Zaubitzer, et al. Prognostic Factors for the Therapeutic Performance of Cisplatin in Head and Neck Malignancies. Front Oncol. 2022, 12, e778380.
- Chatterjee, S.; Kiyota, N.; Vaish, R.; Sharma, A.; Tahara, M.; Noronha, V.; Prabhash, K.; D’Cruz, A. Weekly versus 3-weekly cisplatin along with radiotherapy for locoregionally advanced non-nasopharyngeal head and neck cancers: is the equipoise in literature addressed yet? Head Neck. 2023, 45, 1594–1603. [Google Scholar]
- Mohamed, A.; Twardy, B.; Zordok, M.A.; Ashraf, K.; Alkhoder, A.; Schrapp, K.; Steuer, C.; Chen, Z.; Pakkala, S.; Pillai, R.; Trad Wadsworth, J.; Higgins, K.; Beitler, J.J.; Ramalingam, S.S.; Owonikoko, T.K.; Khuri, F.R.; Shin, D.M.; Behera, M.; Saba, N.F. Concurrent chemoradiotherapy with weekly versus triweekly cisplatin in locally advanced squamous cell carcinoma of the head and neck: comparative analysis. Head Neck. 2019, 41, 1490–1498. [Google Scholar]
- De Felice, F.; Belgioia, L.; Alterio, D.; Bonomo, P.; Maddalo, M.; Paiar, F.; Denaro, N.; Corvo, R.; Merlotti, A.; Bossi, P.; Pappagallo, G.L. Survival and toxicity of weekly cisplatin chemoradiotherapy versus three-weekly cisplatin chemoradiotherapy for head and neck cancer: a systematic review and meta-analysis endorsed by the Italian Association of Radiotherapy and Clinical Oncology (AIRO). Crit Rev Oncol Hematol. 2021, 162, 103345. [Google Scholar]
 |
 |
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




