Submitted:
12 March 2025
Posted:
14 March 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Literature Search and Selection Methodology
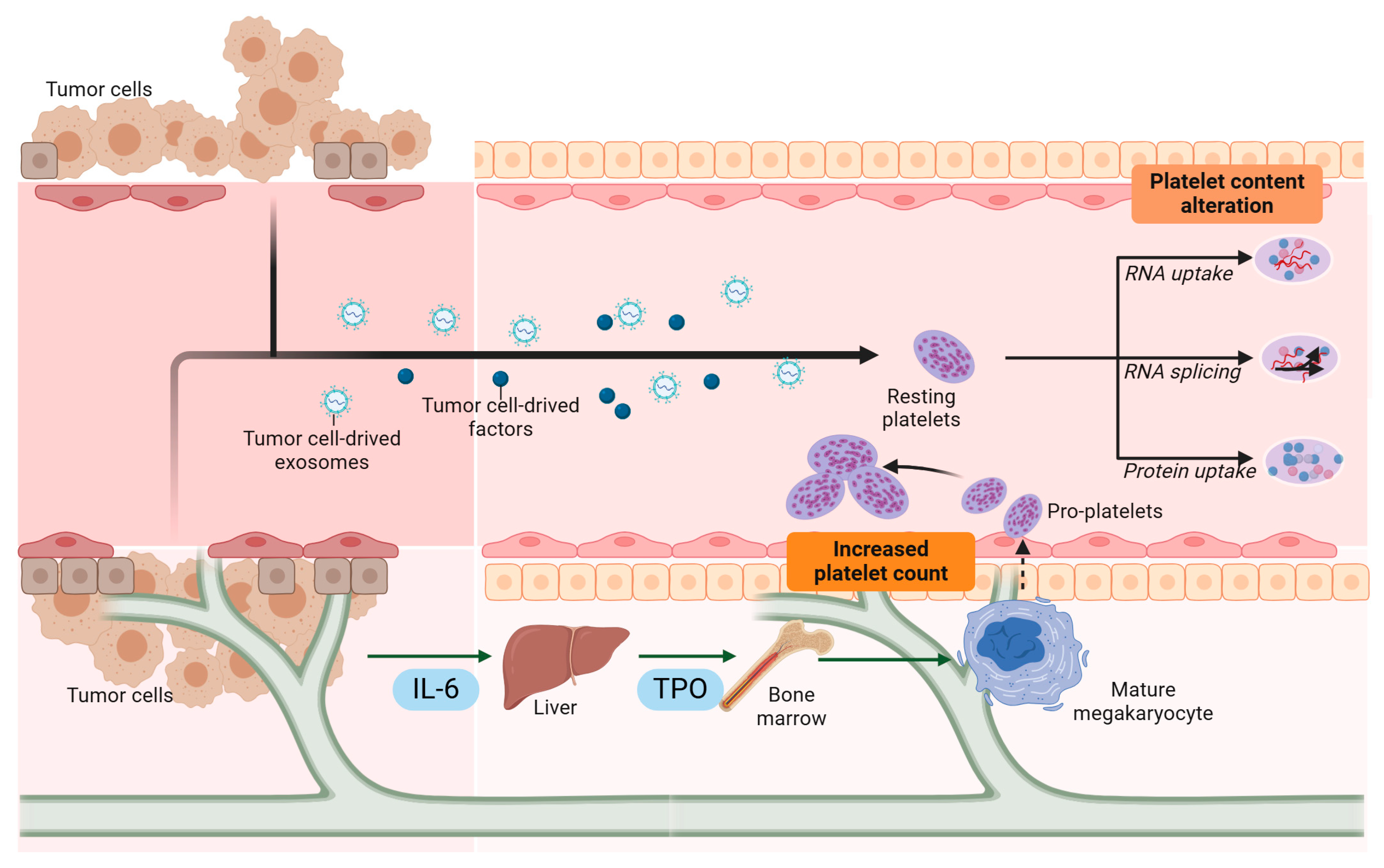
3. Tumor-Driven Platelet Education and Modulation
3.1. Sequestration of Tumor-Derived Biomolecules
3.2. Modulation of Platelet RNA Content
3.3. Megakaryocyte Education and Thrombopoiesis Alteration
3.4. Increased Platelet Levels (Thrombocytosis)
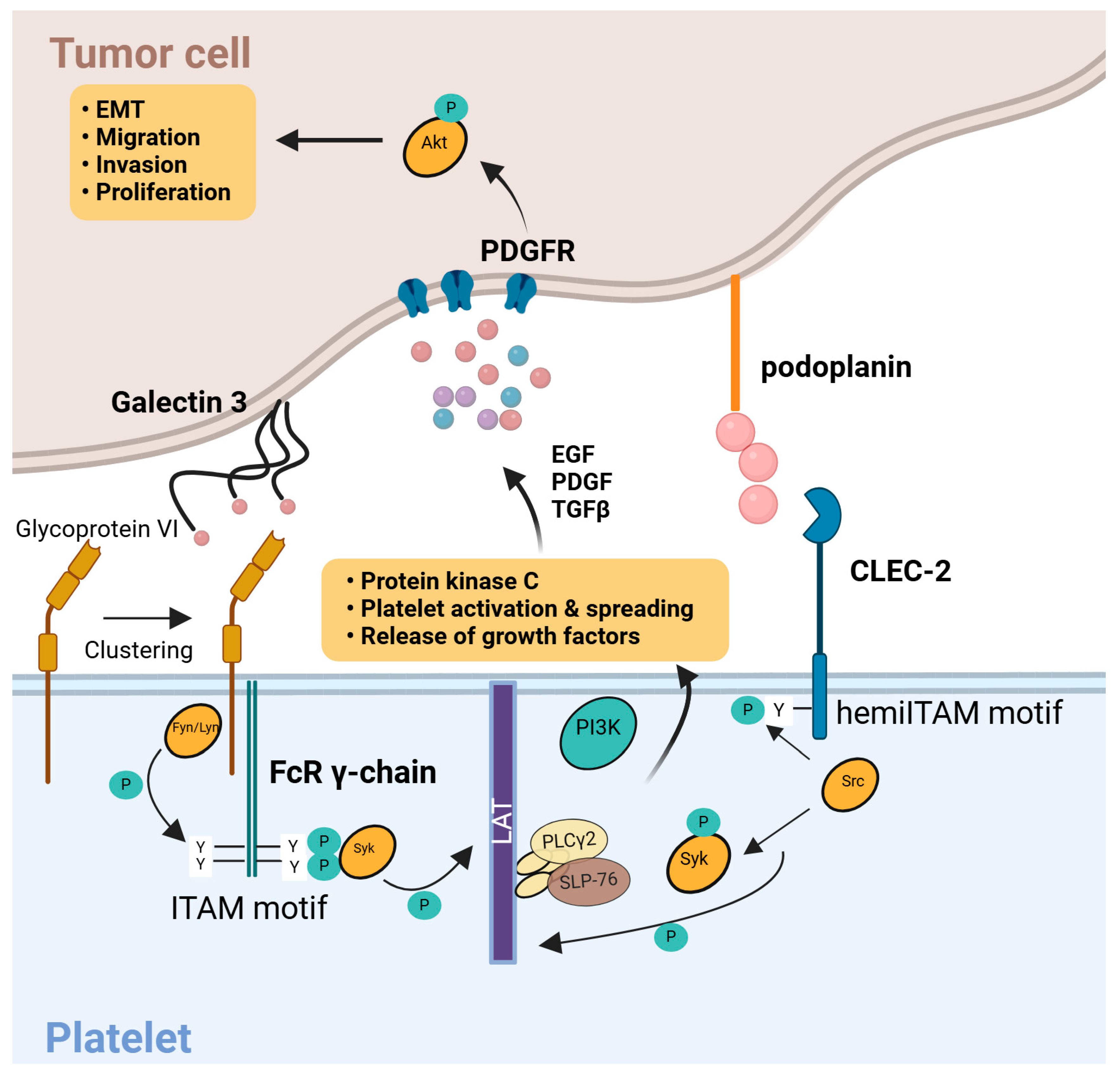
3.5. Tumor Cells Activate Platelets
3.6. Molecular Pathways of Platelet–Tumor Interactions
3.7. Clinical Implications
4. Diagnostic Applications of Tumor-Educated Platelets
4.1. Platelet Count and Volume as Diagnostic Clues
4.2. Platelet RNA Signatures for Cancer Detection
4.2.1. Specific mRNA Biomarkers
4.2.2. Whole Transcriptome Signatures and Machine Learning
4.2.3. Pan-Cancer vs. Cancer-Specific Models
4.2.4. Non-Coding RNAs
4.3. Platelet-Derived Protein Markers for Cancer
4.4. Diagnostic Performance and Comparative Analysis
5. Prognostic and Monitoring Applications of TEPs
5.1. Platelet Count and Indices as Prognostic Markers
5.2. Platelet RNA Signatures and Gene Expression for Prognosis
5.3. Platelets in Disease Monitoring and Therapeutic Decision-Making
6. Consistency of Findings and Comparative Analysis of Studies
7. Limitations and Challenges in TEP Research
7.1. Technical and Methodological Challenges
7.1.1. Platelet Isolation and Purity
7.1.2. Low Input and Analytical Sensitivity
7.1.3. Data Processing and Algorithms
7.1.4. Reproducibility and Standardization
7.2. Biological and Clinical Challenges
7.2.1. Specificity to Cancer
7.2.2. Heterogeneity Among Cancers
7.2.3. Dynamic Changes and Timing
7.2.4. Unknown Mechanisms
7.3. Validation and Clinical Trial Challenges
7.3.1. Need for Large-Scale Validation
7.3.2. Regulatory Approval and Standardization
7.3.3. Integration with Existing Workflows
8. Future Directions and Potential Solutions
8.1. Multi-Modal Liquid Biopsy Approaches
8.2. Refinement of Biomarker Panels
8.3. Automation and Clinical Laboratory Development
8.4. Standardizing Pre-Analytical Variables
8.5. Understanding Mechanisms
8.6. Clinical Trials and Real-World Evidence
8.7. Interdisciplinary Collaboration
9. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lone, S.N.; Nisar, S.; Masoodi, T.; Singh, M.; Rizwan, A.; Hashem, S.; El-Rifai, W.; Bedognetti, D.; Batra, S.K.; Haris, M.; et al. Liquid biopsy: a step closer to transform diagnosis, prognosis and future of cancer treatments. Molecular Cancer 2022, 21, 79. [Google Scholar] [CrossRef]
- Pantel, K.; Alix-Panabières, C. Liquid biopsy and minimal residual disease — latest advances and implications for cure. Nature Reviews Clinical Oncology 2019, 16, 409–424. [Google Scholar] [CrossRef]
- Best, M.G.; Vancura, A.; Wurdinger, T. Platelet RNA as a circulating biomarker trove for cancer diagnostics. J Thromb Haemost 2017, 15, 1295–1306. [Google Scholar] [CrossRef]
- Ding, S.; Dong, X.; Song, X. Tumor educated platelet: the novel BioSource for cancer detection. Cancer Cell International 2023, 23, 91. [Google Scholar] [CrossRef]
- Trivanović, D.; Mojsilović, S.; Bogosavljević, N.; Jurišić, V.; Jauković, A. Revealing profile of cancer-educated platelets and their factors to foster immunotherapy development. Translational Oncology 2024, 40, 101871. [Google Scholar] [CrossRef]
- Best, M.G.; Wesseling, P.; Wurdinger, T. Tumor-Educated Platelets as a Noninvasive Biomarker Source for Cancer Detection and Progression Monitoring. Cancer Res 2018, 78, 3407–3412. [Google Scholar] [CrossRef]
- Best, M.G.; Sol, N.; Kooi, I.; Tannous, J.; Westerman, B.A.; Rustenburg, F.; Schellen, P.; Verschueren, H.; Post, E.; Koster, J.; et al. RNA-Seq of Tumor-Educated Platelets Enables Blood-Based Pan-Cancer, Multiclass, and Molecular Pathway Cancer Diagnostics. Cancer Cell 2015, 28, 666–676. [Google Scholar] [CrossRef]
- Gu, M.; Zhai, Z.; Huang, L.; Zheng, W.; Zhou, Y.; Zhu, R.; Shen, F.; Yuan, C. Pre-treatment mean platelet volume associates with worse clinicopathologic features and prognosis of patients with invasive breast cancer. Breast Cancer 2016, 23, 752–760. [Google Scholar] [CrossRef]
- Schlesinger, M. Role of platelets and platelet receptors in cancer metastasis. Journal of Hematology & Oncology 2018, 11, 125. [Google Scholar] [CrossRef]
- Haemmerle, M.; Stone, R.L.; Menter, D.G.; Afshar-Kharghan, V.; Sood, A.K. The Platelet Lifeline to Cancer: Challenges and Opportunities. Cancer Cell 2018, 33, 965–983. [Google Scholar] [CrossRef]
- Antunes-Ferreira, M.; D’Ambrosi, S.; Arkani, M.; Post, E.; In ‘t Veld, S.G.J.G.; Ramaker, J.; Zwaan, K.; Kucukguzel, E.D.; Wedekind, L.E.; Griffioen, A.W.; et al. Tumor-educated platelet blood tests for Non-Small Cell Lung Cancer detection and management. Scientific Reports 2023, 13, 9359. [Google Scholar] [CrossRef]
- Zhuang, T.; Wang, S.; Yu, X.; He, X.; Guo, H.; Ou, C. Current status and future perspectives of platelet-derived extracellular vesicles in cancer diagnosis and treatment. Biomarker Research 2024, 12, 88. [Google Scholar] [CrossRef]
- Figueroa, J.M.; Skog, J.; Akers, J.; Li, H.; Komotar, R.; Jensen, R.; Ringel, F.; Yang, I.; Kalkanis, S.; Thompson, R.; et al. Detection of wild-type EGFR amplification and EGFRvIII mutation in CSF-derived extracellular vesicles of glioblastoma patients. Neuro Oncol 2017, 19, 1494–1502. [Google Scholar] [CrossRef]
- D'Ambrosi, S.; Nilsson, R.J.; Wurdinger, T. Platelets and tumor-associated RNA transfer. Blood 2021, 137, 3181–3191. [Google Scholar] [CrossRef]
- Xiang, Y.; Xiang, P.; Zhang, L.; Li, Y.; Zhang, J. A narrative review for platelets and their RNAs in cancers: New concepts and clinical perspectives. Medicine (Baltimore) 2022, 101, e32539. [Google Scholar] [CrossRef]
- Zhou, L.; Wu, D.; Zhou, Y.; Wang, D.; Fu, H.; Huang, Q.; Qin, G.; Chen, J.; Lv, J.; Lai, S.; et al. Tumor cell-released kynurenine biases MEP differentiation into megakaryocytes in individuals with cancer by activating AhR–RUNX1. Nature Immunology 2023, 24, 2042–2052. [Google Scholar] [CrossRef]
- Gaertner, F.; Ishikawa-Ankerhold, H.; Stutte, S.; Fu, W.; Weitz, J.; Dueck, A.; Nelakuditi, B.; Fumagalli, V.; van den Heuvel, D.; Belz, L.; et al. Plasmacytoid dendritic cells control homeostasis of megakaryopoiesis. Nature 2024, 631, 645–653. [Google Scholar] [CrossRef]
- Giannakeas, V.; Narod, S.A. Incidence of Cancer Among Adults With Thrombocytosis in Ontario, Canada. JAMA Network Open 2021, 4, e2120633–e2120633. [Google Scholar] [CrossRef]
- Hufnagel, D.H.; Cozzi, G.D.; Crispens, M.A.; Beeghly-Fadiel, A. Platelets, Thrombocytosis, and Ovarian Cancer Prognosis: Surveying the Landscape of the Literature. International Journal of Molecular Sciences 2020, 21, 8169. [Google Scholar] [CrossRef]
- Amer, H.; Kampan, N.C.; Itsiopoulos, C.; Flanagan, K.L.; Scott, C.L.; Kartikasari, A.E.R.; Plebanski, M. Interleukin-6 Modulation in Ovarian Cancer Necessitates a Targeted Strategy: From the Approved to Emerging Therapies. Cancers 2024, 16, 4187. [Google Scholar] [CrossRef]
- Kumar, A.; Taghi Khani, A.; Sanchez Ortiz, A.; Swaminathan, S. GM-CSF: A Double-Edged Sword in Cancer Immunotherapy. Front Immunol 2022, 13, 901277. [Google Scholar] [CrossRef]
- Oncul, S.; Cho, M.S. Interactions between Platelets and Tumor Microenvironment Components in Ovarian Cancer and Their Implications for Treatment and Clinical Outcomes. Cancers (Basel) 2023, 15. [Google Scholar] [CrossRef]
- Dudiki, T.; Veleeparambil, M.; Zhevlakova, I.; Biswas, S.; Klein, E.A.; Ford, P.; Podrez, E.A.; Byzova, T.V. Mechanism of Tumor-Platelet Communications in Cancer. Circulation Research 2023, 132, 1447–1461. [Google Scholar] [CrossRef]
- Chen, L.; Zhu, C.; Pan, F.; Chen, Y.; Xiong, L.; Li, Y.; Chu, X.; Huang, G. Platelets in the tumor microenvironment and their biological effects on cancer hallmarks. Front Oncol 2023, 13, 1121401. [Google Scholar] [CrossRef]
- Chen, L.; Zhu, C.; Pan, F.; Chen, Y.; Xiong, L.; Li, Y.; Chu, X.; Huang, G. Platelets in the tumor microenvironment and their biological effects on cancer hallmarks. Frontiers in Oncology 2023, 13. [Google Scholar] [CrossRef]
- Eslami-S, Z.; Cortés-Hernández, L.E.; Glogovitis, I.; Antunes-Ferreira, M.; D’Ambrosi, S.; Kurma, K.; Garima, F.; Cayrefourcq, L.; Best, M.G.; Koppers-Lalic, D.; et al. In vitro cross-talk between metastasis-competent circulating tumor cells and platelets in colon cancer: a malicious association during the harsh journey in the blood. Frontiers in Cell and Developmental Biology 2023, 11. [Google Scholar] [CrossRef]
- Morales-Pacheco, M.; Valenzuela-Mayen, M.; Gonzalez-Alatriste, A.M.; Mendoza-Almanza, G.; Cortés-Ramírez, S.A.; Losada-García, A.; Rodríguez-Martínez, G.; González-Ramírez, I.; Maldonado-Lagunas, V.; Vazquez-Santillan, K.; et al. The role of platelets in cancer: from their influence on tumor progression to their potential use in liquid biopsy. Biomark Res 2025, 13, 27. [Google Scholar] [CrossRef]
- Zhang, Q.; Song, X.; Song, X. Contents in tumor-educated platelets as the novel biosource for cancer diagnostics. Frontiers in Oncology 2023, 13. [Google Scholar] [CrossRef]
- Chen, M.; Hou, L.; Hu, L.; Tan, C.; Wang, X.; Bao, P.; Ran, Q.; Chen, L.; Li, Z. Platelet detection as a new liquid biopsy tool for human cancers. Frontiers in Oncology 2022, 12. [Google Scholar] [CrossRef]
- Detopoulou, P.; Panoutsopoulos, G.I.; Mantoglou, M.; Michailidis, P.; Pantazi, I.; Papadopoulos, S.; Rojas Gil, A.P. Relation of Mean Platelet Volume (MPV) with Cancer: A Systematic Review with a Focus on Disease Outcome on Twelve Types of Cancer. Curr Oncol 2023, 30, 3391–3420. [Google Scholar] [CrossRef]
- Jopek, M.A.; Pastuszak, K.; Sieczczyński, M.; Cygert, S.; Żaczek, A.J.; Rondina, M.T.; Supernat, A. Improving platelet-RNA-based diagnostics: a comparative analysis of machine learning models for cancer detection and multiclass classification. Mol Oncol 2024, 18, 2743–2754. [Google Scholar] [CrossRef]
- Hajjar, M.; Albaradei, S.; Aldabbagh, G. Machine Learning Approaches in Multi-Cancer Early Detection. Information 2024, 15, 627. [Google Scholar] [CrossRef]
- Xiao, R.; Liu, C.; Zhang, B.; Ma, L. Tumor-Educated Platelets as a Promising Biomarker for Blood-Based Detection of Renal Cell Carcinoma. Front Oncol 2022, 12, 844520. [Google Scholar] [CrossRef]
- In 't Veld, S.; Arkani, M.; Post, E.; Antunes-Ferreira, M.; D'Ambrosi, S.; Vessies, D.C.L.; Vermunt, L.; Vancura, A.; Muller, M.; Niemeijer, A.N.; et al. Detection and localization of early- and late-stage cancers using platelet RNA. Cancer Cell 2022, 40, 999–1009.e1006. [Google Scholar] [CrossRef]
- Yuan, M.; Jia, Y.; Xing, Y.; Wang, Y.; Liu, Y.; Liu, X.; Liu, D. Screening and validation of platelet activation-related lncRNAs as potential biomarkers for prognosis and immunotherapy in gastric cancer patients. Frontiers in Genetics 2022, 13. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Wei, X.; Wu, B.; Su, J.; Tan, W.; Yang, K. Tumor-educated platelet miR-34c-3p and miR-18a-5p as potential liquid biopsy biomarkers for nasopharyngeal carcinoma diagnosis. Cancer Manag Res 2019, 11, 3351–3360. [Google Scholar] [CrossRef] [PubMed]
- D'Ambrosi, S.; Visser, A.; Antunes-Ferreira, M.; Poutsma, A.; Giannoukakos, S.; Sol, N.; Sabrkhany, S.; Bahce, I.; Kuijpers, M.J.E.; Oude Egbrink, M.G.A.; et al. The Analysis of Platelet-Derived circRNA Repertoire as Potential Diagnostic Biomarker for Non-Small Cell Lung Cancer. Cancers (Basel) 2021, 13. [Google Scholar] [CrossRef]
- Tavukcuoglu, Z.; Butt, U.; Faria, A.V.S.; Oesterreicher, J.; Holnthoner, W.; Laitinen, S.; Palviainen, M.; Siljander, P.R. Platelet-derived extracellular vesicles induced through different activation pathways drive melanoma progression by functional and transcriptional changes. Cell Commun Signal 2024, 22, 601. [Google Scholar] [CrossRef]
- Yu, L.; Guo, Y.; Chang, Z.; Zhang, D.; Zhang, S.; Pei, H.; Pang, J.; Zhao, Z.J.; Chen, Y. Bidirectional Interaction Between Cancer Cells and Platelets Provides Potential Strategies for Cancer Therapies. Frontiers in Oncology 2021, 11. [Google Scholar] [CrossRef]
- Yang, L.; Jiang, Q.; Li, D.Z.; Zhou, X.; Yu, D.S.; Zhong, J. TIMP1 mRNA in tumor-educated platelets is diagnostic biomarker for colorectal cancer. Aging (Albany NY) 2019, 11, 8998–9012. [Google Scholar] [CrossRef]
- Hu, Y.; Zeng, C.; Li, J.; Ren, S.; Shao, M.; Lei, W.; Yi, J.; Han, W.; Cao, J.; Zou, J.; et al. TRIM27 revealing by tumor educated platelet RNA-sequencing, as a potential biomarker for malignant ground-glass opacities diagnosis mediates glycolysis of non-small cell lung cancer cells partially through HOXM1. Transl Lung Cancer Res 2024, 13, 2307–2325. [Google Scholar] [CrossRef]
- Nicolò, E.; Gianni, C.; Pontolillo, L.; Serafini, M.S.; Munoz-Arcos, L.S.; Andreopoulou, E.; Curigliano, G.; Reduzzi, C.; Cristofanilli, M. Circulating tumor cells et al.: towards a comprehensive liquid biopsy approach in breast cancer. Translational Breast Cancer Research 2024, 5. [Google Scholar] [CrossRef]
- Mondal, D.; Shinde, S.; Sinha, V.; Dixit, V.; Paul, S.; Gupta, R.K.; Thakur, S.; Vishvakarma, N.K.; Shukla, D. Prospects of liquid biopsy in the prognosis and clinical management of gastrointestinal cancers. Frontiers in Molecular Biosciences 2024, 11. [Google Scholar] [CrossRef]
- She, W.; Garitaonaindia, Y.; Lin, Y. The latest advances in liquid biopsy for lung cancer-a narrative review. Transl Lung Cancer Res 2024, 13, 3241–3251. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Xu, Y.; Tong, L.; Zhang, X.; Zhang, S. Recent progress of exosomal lncRNA/circRNA–miRNA–mRNA axis in lung cancer: implication for clinical application. Frontiers in Molecular Biosciences 2024, 11. [Google Scholar] [CrossRef]
- Xu, Y.; Sun, Y.; Song, X.; Ren, J. The mechanisms and diagnostic potential of lncRNAs, miRNAs, and their related signaling pathways in cervical cancer. Frontiers in Cell and Developmental Biology 2023, 11. [Google Scholar] [CrossRef]
- Du, X.; Zang, X.; Zhang, H.; Liu, L.; Xu, Y.; Li, X.; Mou, R.; Xu, H.; Zhu, J.; Xie, R. Mean platelet volume/platelet count ratio can predict the recurrence-free survival rate of patients after complete resection of gastrointestinal stromal tumors. Frontiers in Oncology 2024, 14. [Google Scholar] [CrossRef]
- Yoshikawa, N.; Matsukawa, T.; Hattori, S.; Iyoshi, S.; Yoshida, K.; Yoshihara, M.; Tamauchi, S.; Shimizu, Y.; Ikeda, Y.; Yokoi, A.; et al. Mean platelet volume as a potential biomarker for survival outcomes in ovarian clear cell carcinoma. Int J Clin Oncol 2023, 28, 1680–1689. [Google Scholar] [CrossRef]
- Qi, X.; Chen, J.; Wei, S.; Ni, J.; Song, L.; Jin, C.; Yang, L.; Zhang, X. Prognostic significance of platelet-to-lymphocyte ratio (PLR) in patients with breast cancer treated with neoadjuvant chemotherapy: a meta-analysis. BMJ Open 2023, 13, e074874. [Google Scholar] [CrossRef]
- Li, X.; Ren, H.; Peng, L.; Li, J. Prognostic value of pretreatment platelet count, fibrinogen and d-dimer levels in osteosarcoma patients: A meta-analysis. Medicine 2024, 103, e38463. [Google Scholar] [CrossRef]
- Shi, H.; Wang, H.; Pan, J.; Liu, Z.; Li, Z. Comparing prognostic value of preoperative platelet indexes in patients with resectable gastric cancer. Scientific Reports 2022, 12, 6480. [Google Scholar] [CrossRef]
- Yuan, Y.; Zhong, H.; Ye, L.; Li, Q.; Fang, S.; Gu, W.; Qian, Y. Prognostic value of pretreatment platelet counts in lung cancer: a systematic review and meta-analysis. BMC Pulm Med 2020, 20, 96. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Tao, L.; Qiu, J.; Xu, J.; Yang, X.; Zhang, Y.; Tian, X.; Guan, X.; Cen, X.; Zhao, Y. Tumor biomarkers for diagnosis, prognosis and targeted therapy. Signal Transduction and Targeted Therapy 2024, 9, 132. [Google Scholar] [CrossRef]
- Liu, C.J.; Li, H.Y.; Gao, Y.; Xie, G.Y.; Chi, J.H.; Li, G.L.; Zeng, S.Q.; Xiong, X.M.; Liu, J.H.; Shi, L.L.; et al. Platelet RNA signature independently predicts ovarian cancer prognosis by deep learning neural network model. Protein Cell 2023, 14, 618–622. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Giannakeas, V.; Narod, S.A.; Akbari, M.R. Emerging applications of tumour-educated platelets in the detection and prognostication of ovarian cancer. Protein Cell 2023, 14, 556–559. [Google Scholar] [CrossRef]
- Hu, H.; Song, H.; Han, B.; Zhao, H.; He, J. Tumor-educated platelet RNA and circulating free RNA: emerging liquid biopsy markers for different tumor types. Frontiers in Bioscience-Landmark 2024, 29, 80. [Google Scholar] [CrossRef]
- Hu, M.S.; Jiang, M.; Wang, Y.J.; Xu, S.F.; Jiang, F.Y.; Han, Y.T.; Liu, Z.W.; Yu, H. Platelet-related gene risk score: a predictor for pancreatic cancer microenvironmental signature, chemosensitivity and prognosis. Am J Cancer Res 2023, 13, 6113–6124. [Google Scholar]
- Li, Q.; Zhang, C.; Ren, Y.; Qiao, L.; Xu, S.; Li, K.; Liu, Y. A novel platelets-related gene signature for predicting prognosis, immune features and drug sensitivity in gastric cancer. Front Immunol 2024, 15, 1477427. [Google Scholar] [CrossRef]
- Zhao, J.; Huang, A.; Zeller, J.; Peter, K.; McFadyen, J.D. Decoding the role of platelets in tumour metastasis: enigmatic accomplices and intricate targets for anticancer treatments. Frontiers in Immunology 2023, 14. [Google Scholar] [CrossRef]
- Huber, L.T.; Kraus, J.M.; Ezić, J.; Wanli, A.; Groth, M.; Laban, S.; Hoffmann, T.K.; Wollenberg, B.; Kestler, H.A.; Brunner, C. Liquid biopsy: an examination of platelet RNA obtained from head and neck squamous cell carcinoma patients for predictive molecular tumor markers. Explor Target Antitumor Ther 2023, 4, 422–446. [Google Scholar] [CrossRef]
- Hu, Z.; Wang, N.; Zhang, Y.; Zhang, D.; Sun, S.; Yu, H.; Lin, Y.; Zhao, X.; Wang, H.; Wu, X.; et al. PD-L1 mRNA derived from tumor-educated platelets as a potential immunotherapy biomarker in non-small cell lung cancer. Translational Lung Cancer Research 2024, 13, 345–354. [Google Scholar] [CrossRef]
- Garcia-Leon, M.J.; Liboni, C.; Mittelheisser, V.; Bochler, L.; Follain, G.; Mouriaux, C.; Busnelli, I.; Larnicol, A.; Colin, F.; Peralta, M.; et al. Platelets favor the outgrowth of established metastases. Nature Communications 2024, 15, 3297. [Google Scholar] [CrossRef]
- Xu, L.; Li, X.; Li, X.; Wang, X.; Ma, Q.; She, D.; Lu, X.; Zhang, J.; Yang, Q.; Lei, S.; et al. RNA profiling of blood platelets noninvasively differentiates colorectal cancer from healthy donors and noncancerous intestinal diseases: a retrospective cohort study. Genome Med 2022, 14, 26. [Google Scholar] [CrossRef]
- Post, E.; Sol, N.; Best, M.G.; Wurdinger, T. Blood platelets as an RNA biomarker platform for neuro-oncological diseases. Neuro-Oncology Advances 2022, 4, ii61–ii65. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Song, X.; Song, X. Contents in tumor-educated platelets as the novel biosource for cancer diagnostics. Front Oncol 2023, 13, 1165600. [Google Scholar] [CrossRef] [PubMed]
- Mai, S.; Inkielewicz-Stepniak, I. Pancreatic Cancer and Platelets Crosstalk: A Potential Biomarker and Target. Front Cell Dev Biol 2021, 9, 749689. [Google Scholar] [CrossRef]
- Walraven, M.; Sabrkhany, S.; Knol, J.C.; Dekker, H.; de Reus, I.; Piersma, S.R.; Pham, T.V.; Griffioen, A.W.; Broxterman, H.J.; Oude Egbrink, M.; et al. Effects of Cancer Presence and Therapy on the Platelet Proteome. Int J Mol Sci 2021, 22. [Google Scholar] [CrossRef]
- Gerber, D.E.; Singh, H.; Larkins, E.; Ferris, A.; Forde, P.M.; Selig, W.; Basu Roy, U. A New Approach to Simplifying and Harmonizing Cancer Clinical Trials-Standardizing Eligibility Criteria. JAMA Oncol 2022, 8, 1333–1339. [Google Scholar] [CrossRef]
- Wiyarta, E.; Nugraha, D.A.; Ramadani, M.I.; Gustya, G.F.; Ammar, M.F.; Edwar, H.D.; Kheirizzad, N.; Mukhlisah, M.N.; Burhan, E.; Syahruddin, E. Clinical utility and diagnostic value of tumor-educated platelets in lung cancer: a systematic review and meta-analysis. Front Oncol 2023, 13, 1201713. [Google Scholar] [CrossRef]
- Sakai, K.; Ohara, S.; Tanaka, J.; Suda, K.; Muramatsu, T.; Uematsu, C.; Tsutani, Y.; Mitsudomi, T.; Nishio, K. Improved platelet separation performance from whole blood using an acoustic fluidics system. Cancer Sci 2024, 115, 3795–3803. [Google Scholar] [CrossRef]
- Jin, J.; Shao, Y.; Zhang, J.; Cao, J.; Tao, Z.; Hu, X. High-purity isolation platelets by gradient centrifugation plus filtration. International Journal of Laboratory Hematology 2023, 45, 187–194. [Google Scholar] [CrossRef]
- Collinson, R.J.; Boey, D.; Wilson, L.; Ng, Z.Y.; Mirzai, B.; Chuah, H.; Leahy, M.F.; Howman, R.; Linden, M.; Fuller, K.; et al. PlateletSeq: A novel method for discovery of blood-based biomarkers. Methods 2023, 219, 139–149. [Google Scholar] [CrossRef]
- Karp, J.M.; Modrek, A.S.; Ezhilarasan, R.; Zhang, Z.-Y.; Ding, Y.; Graciani, M.; Sahimi, A.; Silvestro, M.; Chen, T.; Li, S.; et al. Deconvolution of the tumor-educated platelet transcriptome reveals activated platelet and inflammatory cell transcript signatures. JCI Insight 2024, 9. [Google Scholar] [CrossRef]
- Cygert, S.; Pastuszak, K.; Górski, F.; Sieczczyński, M.; Juszczyk, P.; Rutkowski, A.; Lewalski, S.; Różański, R.; Jopek, M.A.; Jassem, J.; et al. Platelet-Based Liquid Biopsies through the Lens of Machine Learning. Cancers (Basel) 2023, 15. [Google Scholar] [CrossRef]
- Antunes-Ferreira, M.; Koppers-Lalic, D.; Würdinger, T. Circulating platelets as liquid biopsy sources for cancer detection. Mol Oncol 2021, 15, 1727–1743. [Google Scholar] [CrossRef]
- Walke, V.; Das, S.; Mittal, A.; Agrawal, A. Tumor Educated Platelets as a Biomarker for Diagnosis of Lung cancer: A Systematic Review. Asian Pac J Cancer Prev 2024, 25, 1911–1920. [Google Scholar] [CrossRef]
- Roweth, H.G.; Battinelli, E.M. Lessons to learn from tumor-educated platelets. Blood 2021, 137, 3174–3180. [Google Scholar] [CrossRef]
- Goossens, N.; Nakagawa, S.; Sun, X.; Hoshida, Y. Cancer biomarker discovery and validation. Translational Cancer Research 2015, 4, 256–269. [Google Scholar]
- Salwei, M.E.; Reale, C. Workflow analysis of breast cancer treatment decision-making: challenges and opportunities for informatics to support patient-centered cancer care. JAMIA Open 2024, 7, ooae053. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Guo, H.; Zhao, Y.; Liu, Z.; Wang, C.; Bu, J.; Sun, T.; Wei, J. Liquid biopsy in cancer: current status, challenges and future prospects. Signal Transduction and Targeted Therapy 2024, 9, 336. [Google Scholar] [CrossRef] [PubMed]
- Nicolò, E.; Gianni, C.; Pontolillo, L.; Serafini, M.S.; Munoz-Arcos, L.S.; Andreopoulou, E.; Curigliano, G.; Reduzzi, C.; Cristofanilli, M. Circulating tumor cells et al.: towards a comprehensive liquid biopsy approach in breast cancer. Transl Breast Cancer Res 2024, 5, 10. [Google Scholar] [CrossRef]
- Bravaccini, S.; Boldrin, E.; Gurioli, G.; Tedaldi, G.; Piano, M.A.; Canale, M.; Curtarello, M.; Ulivi, P.; Pilati, P. The use of platelets as a clinical tool in oncology: opportunities and challenges. Cancer Lett 2024, 607, 217044. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Hu, Y.; Hu, H. Advances and challenges in platelet counting: evolving from traditional microscopy to modern flow cytometry. Journal of Laboratory Medicine 2025, 49, 2–13. [Google Scholar] [CrossRef]
- Hindle, M.S.; Cheah, L.T.; Yates, D.M.; Naseem, K.M. Preanalytical conditions for multiparameter platelet flow cytometry. Res Pract Thromb Haemost 2023, 7, 102205. [Google Scholar] [CrossRef]
- Liao, K.; Zhang, X.; Liu, J.; Teng, F.; He, Y.; Cheng, J.; Yang, Q.; Zhang, W.; Xie, Y.; Guo, D.; et al. The role of platelets in the regulation of tumor growth and metastasis: the mechanisms and targeted therapy. MedComm (2020) 2023, 4, e350. [Google Scholar] [CrossRef]
- Wang, Y.; Dong, A.; Jin, M.; Li, S.; Duan, Y. TEP RNA: a new frontier for early diagnosis of NSCLC. J Cancer Res Clin Oncol 2024, 150, 97. [Google Scholar] [CrossRef]
- Orcutt, X.; Chen, K.; Mamtani, R.; Long, Q.; Parikh, R.B. Evaluating generalizability of oncology trial results to real-world patients using machine learning-based trial emulations. Nature Medicine 2025, 31, 457–465. [Google Scholar] [CrossRef]
- Arora, A.; Alderman, J.E.; Palmer, J.; Ganapathi, S.; Laws, E.; McCradden, M.D.; Oakden-Rayner, L.; Pfohl, S.R.; Ghassemi, M.; McKay, F.; et al. The value of standards for health datasets in artificial intelligence-based applications. Nat Med 2023, 29, 2929–2938. [Google Scholar] [CrossRef]
- Yang, S.; Wang, L.; Wu, Y.; Wu, A.; Huang, F.; Tang, X.; Kantawong, F.; Anuchapreeda, S.; Qin, D.; Mei, Q.; et al. Apoptosis in megakaryocytes: Safeguard and threat for thrombopoiesis. Front Immunol 2022, 13, 1025945. [Google Scholar] [CrossRef]
- Hufnagel, D.H.; Cozzi, G.D.; Crispens, M.A.; Beeghly-Fadiel, A. Platelets, Thrombocytosis, and Ovarian Cancer Prognosis: Surveying the Landscape of the Literature. Int J Mol Sci 2020, 21. [Google Scholar] [CrossRef]
- Isingizwe, Z.R.; Meelheim, B.A.; Benbrook, D.M. Elevated Platelet Aggregation in Patients with Ovarian Cancer: More than Just Increased Platelet Count. Cancers 2024, 16, 3583. [Google Scholar] [CrossRef]
- Li, Y.; Wang, H.; Zhao, Z.; Yang, Y.; Meng, Z.; Qin, L. Effects of the interactions between platelets with other cells in tumor growth and progression. Frontiers in Immunology 2023, 14. [Google Scholar] [CrossRef]
- Xing, S.; Zeng, T.; Xue, N.; He, Y.; Lai, Y.Z.; Li, H.L.; Huang, Q.; Chen, S.L.; Liu, W.L. Development and Validation of Tumor-educated Blood Platelets Integrin Alpha 2b (ITGA2B) RNA for Diagnosis and Prognosis of Non-small-cell Lung Cancer through RNA-seq. Int J Biol Sci 2019, 15, 1977–1992. [Google Scholar] [CrossRef]
- Dudiki, T.; Veleeparambil, M.; Zhevlakova, I.; Biswas, S.; Klein, E.A.; Ford, P.; Podrez, E.A.; Byzova, T.V. Mechanism of Tumor-Platelet Communications in Cancer. Circ Res 2023, 132, 1447–1461. [Google Scholar] [CrossRef]
- Qiu, X.; Quan, G.; Ou, W.; Wang, P.; Huang, X.; Li, X.; Shen, Y.; Yang, W.; Wang, J.; Wu, X. Unraveling TIMP1: a multifaceted biomarker in colorectal cancer. Front Genet 2023, 14, 1265137. [Google Scholar] [CrossRef]
- Wang, S.; Claret, F.-X.; Wu, W. MicroRNAs as Therapeutic Targets in Nasopharyngeal Carcinoma. Frontiers in Oncology 2019, 9. [Google Scholar] [CrossRef]
- Lin, Y.; Zhao, W.; Lv, Z.; Xie, H.; Li, Y.; Zhang, Z. The functions and mechanisms of long non-coding RNA in colorectal cancer. Front Oncol 2024, 14, 1419972. [Google Scholar] [CrossRef]
- Mondal, D.; Shinde, S.; Sinha, V.; Dixit, V.; Paul, S.; Gupta, R.K.; Thakur, S.; Vishvakarma, N.K.; Shukla, D. Prospects of liquid biopsy in the prognosis and clinical management of gastrointestinal cancers. Front Mol Biosci 2024, 11, 1385238. [Google Scholar] [CrossRef]
- Li, X.; Ma, N.; Zhang, Y.; Wei, H.; Zhang, H.; Pang, X.; Li, X.; Wu, D.; Wang, D.; Yang, Z.; et al. Circular RNA circNRIP1 promotes migration and invasion in cervical cancer by sponging miR-629-3p and regulating the PTP4A1/ERK1/2 pathway. Cell Death & Disease 2020, 11, 399. [Google Scholar] [CrossRef]
- Sabrkhany, S.; Kuijpers, M.J.E.; Knol, J.C.; Olde Damink, S.W.M.; Dingemans, A.C.; Verheul, H.M.; Piersma, S.R.; Pham, T.V.; Griffioen, A.W.; Oude Egbrink, M.G.A.; et al. Exploration of the platelet proteome in patients with early-stage cancer. J Proteomics 2018, 177, 65–74. [Google Scholar] [CrossRef]
- Shen, Y.; Yang, W.; Liu, J.; Zhang, Y. Minimally invasive approaches for the early detection of endometrial cancer. Mol Cancer 2023, 22, 53. [Google Scholar] [CrossRef]
- Chebbo, M.; Assou, S.; Pantesco, V.; Duez, C.; Alessi, M.C.; Chanez, P.; Gras, D. Platelets Purification Is a Crucial Step for Transcriptomic Analysis. Int J Mol Sci 2022, 23. [Google Scholar] [CrossRef]
- Banerjee, M.; Rowley, J.W.; Stubben, C.J.; Tolley, N.D.; Freson, K.; Nelson, B.; Nagy, B., Jr.; Fejes, Z.; Blair, A.M.; Turro, E.; et al. Prospective, international, multisite comparison of platelet isolation techniques for genome-wide transcriptomics: communication from the SSC of the ISTH. J Thromb Haemost 2024, 22, 2922–2934. [Google Scholar] [CrossRef]
- Ju, M.; Fan, J.; Zou, Y.; Yu, M.; Jiang, L.; Wei, Q.; Bi, J.; Hu, B.; Guan, Q.; Song, X.; et al. Computational Recognition of a Regulatory T-cell-specific Signature With Potential Implications in Prognosis, Immunotherapy, and Therapeutic Resistance of Prostate Cancer. Frontiers in Immunology 2022, 13. [Google Scholar] [CrossRef]
- Wen, X.; Yang, G.; Dong, Y.; Luo, L.; Cao, B.; Mengesha, B.A.; Zu, R.; Liao, Y.; Liu, C.; Li, S.; et al. Selection and Validation of Reference Genes for Pan-Cancer in Platelets Based on RNA-Sequence Data. Frontiers in Genetics 2022, 13. [Google Scholar] [CrossRef]
- Wilfinger, W.W.; Eghbalnia, H.R.; Mackey, K.; Miller, R.; Chomczynski, P. Whole blood RNA extraction efficiency contributes to variability in RNA sequencing data sets. PLoS One 2023, 18, e0291209. [Google Scholar] [CrossRef]
- Phallen, J.; Leal, A.; Woodward, B.D.; Forde, P.M.; Naidoo, J.; Marrone, K.A.; Brahmer, J.R.; Fiksel, J.; Medina, J.E.; Cristiano, S.; et al. Early Noninvasive Detection of Response to Targeted Therapy in Non-Small Cell Lung Cancer. Cancer Res 2019, 79, 1204–1213. [Google Scholar] [CrossRef]
- Park, C.K.; Lee, S.W.; Cho, H.J.; Oh, H.J.; Kim, Y.C.; Kim, Y.H.; Ahn, S.J.; Cho, J.H.; Oh, I.J. Blood-Based Biomarker Analysis for Predicting Efficacy of Chemoradiotherapy and Durvalumab in Patients with Unresectable Stage III Non-Small Cell Lung Cancer. Cancers (Basel) 2023, 15. [Google Scholar] [CrossRef]
- Mamdani, H.; Matosevic, S.; Khalid, A.B.; Durm, G.; Jalal, S.I. Immunotherapy in Lung Cancer: Current Landscape and Future Directions. Front Immunol 2022, 13, 823618. [Google Scholar] [CrossRef]
| Mechanism | Representative Evidence/Studies | Major Consequences for Platelet Function | References |
| 1. Sequestration of tumor-derived biomolecules | - Glioblastoma Example: Platelets can internalize mutant EGFRvIII mRNA in approximately 40–60% of patients with EGFRvIII-positive tumors, reflecting tumor molecular status. - Colorectal Cancer: Platelet RNA content shows a 2–3-fold increase in tumor-specific transcripts (e.g., PCA3) compared to healthy controls. - Tumor-secreted EVs (exosomes) containing integrins and oncogenic miRNAs can be absorbed by platelets, creating a “mirror” of the tumor’s genetic material. |
- Platelets become reservoirs of tumor-specific signals, enabling liquid biopsy approaches (e.g., detection of oncogenic mutations in platelets). - Potential amplification of pro-tumor pathways if sequestered molecules (e.g., growth factors) are released at metastatic sites. |
[34] |
| 2. Modulation of platelet RNA content | - RNA Splicing Events: Tumor-derived signals (e.g., thrombin, TGF-β, IL-6) induce alternative splicing in 200–300 platelet transcripts, leading to distinct cancer-associated RNA “fingerprints.” - ITGA2B & PCA3: Reports indicate 2–5-fold elevated levels of these RNAs in platelets from lung and prostate cancer patients, respectively. - Some studies showed that platelets can de novo synthesize proteins (e.g., VEGF variants) from tumor-influenced transcripts. |
- Emergence of unique “tumor-educated” RNA signatures that can distinguish cancer from non-cancer with high accuracy (~80–90% in some studies). - Platelet-driven local release of oncogenic or pro-angiogenic factors, promoting tumor cell proliferation and vascularization. |
[7] |
| 3. Megakaryocyte education and thrombopoiesis alteration | - Cytokine-Driven: Elevated IL-6 (~2–3× normal) in cancer patients stimulates hepatic TPO production, boosting megakaryocyte proliferation. - Reactive Thrombocytosis: In ovarian cancer, up to 65% of advanced cases present with high platelet counts (>400×10^9/L). - Mouse models: Blocking IL-6 reduces paraneoplastic thrombocytosis by ~50%, correlating with slower tumor growth. |
- Increased platelet production leads to higher circulating platelet counts, which can promote metastatic spread through “platelet shielding” of tumor cells. - Newly formed platelets may be “pre-loaded” with cancer-related transcripts, amplifying tumor-specific signals in circulation. |
[89,90] |
| 4. Increased platelet levels (thrombocytosis) | - Clinical Observations: A prospective cohort showed ~31% of patients with elevated platelets were later diagnosed with lung or colorectal cancer, making thrombocytosis a potential early warning sign. - Ovarian Cancer: ~50–65% incidence of thrombocytosis, correlating with advanced stage and worse prognosis. |
- Heightened platelet count is consistently linked to poor prognosis in multiple tumor types (lung, ovarian, gastric, etc.). - Elevated platelets enhance the risk of hematogenous metastasis and recurrent disease. |
[52,91] |
| 5. Tumor cell–induced platelet activation | - In Vitro Aggregation: Tumor-derived ADP, IgG, or podoplanin can cause 20–30% higher platelet aggregation than in healthy controls. - Morphological Changes: Electron cryotomography reveals disrupted microtubules and increased mitochondrial mass in up to ~65% of platelets from patients with metastatic cancer. |
- Hyperresponsive platelets form microthrombi around tumor cells, aiding immune evasion and extravasation. - Elevated levels of activation markers (soluble P-selectin, sCD40L) correlate with metastatic potential. |
[35] |
| 6. Molecular pathways of platelet–tumor interactions | - CLEC2–Podoplanin Axis: Podoplanin-expressing tumors (e.g., certain squamous cell carcinomas) bind CLEC2 on platelets, activating ITAM–Syk signaling, which can increase thrombotic risk by 2–3×. - TLR4–HMGB1: Tumor cells release HMGB1 that triggers a TLR4-mediated cascade, elevating platelet granule release. |
- Unique, tumor-specific pathways shift platelet activation from normal hemostasis to pro-metastatic activity. - Enhanced opportunity for therapeutic intervention: blocking CLEC2–podoplanin or TLR4–HMGB1 can reduce tumor-related thrombosis and metastasis in preclinical models. |
[25,92] |
| RNA Name | Cancer Type(s) | Diagnostic Accuracy / Observed Fold Change | Mechanistic or Biological Role | References |
| ITGA2B (Integrin αIIb) mRNA | Non-small cell lung cancer (NSCLC) | Elevated ~2–5× in platelets from NSCLC vs. healthy controls; AUC ~0.80–0.85 in some studies |
Encodes a platelet membrane glycoprotein important for aggregation; increased expression may reflect platelet hyper-reactivity in tumors |
[76,93] |
| PCA3 mRNA | Prostate cancer | Upregulated 3–4-fold in platelets from metastatic cases; high specificity (≥85%) when combined with other markers |
Known prostate cancer–associated non-coding RNA; possibly involved in androgen receptor signaling and disease progression |
[7,94] |
| TIMP1 mRNA | Colorectal cancer (CRC) | AUC ~0.78–0.85 for detecting CRC; often 2–3-fold higher in platelets of advanced CRC patients |
Tissue inhibitor of metalloproteinases; elevated TIMP1 in platelets may reflect increased tumor invasion and remodeling activity |
[40,95] |
| miR-34c-3p & miR-18a-5p | Nasopharyngeal carcinoma (NPC) | Combined panel AUC ~0.95; significant elevation in patient platelets, not always mirrored in plasma miRNA |
Tumor suppressor (miR-34c) and oncogenic (miR-18a) pathways; reflect complex tumor–platelet interactions unique to NPC |
[36,96] |
| LINC00534, TSPOAP-AS1, SNHG20 | Colorectal, gastric, and other GI malignancies | Each shows 2–4-fold higher expression in tumor-educated platelets; combined panel AUC ~0.78–0.82 |
Long non-coding RNAs implicated in regulating cell cycle and metastatic processes; higher levels correlate with more aggressive GI tumors |
[97,98] |
| circNRIP1 | Non-small cell lung cancer (NSCLC) | Elevated circNRIP1 in platelets correlated with ~75% sensitivity; potential synergy with other circRNAs in multi-marker panels |
Circular RNA modulating multiple signaling pathways (e.g., PI3K/AKT); may promote epithelial–mesenchymal transition in lung tumors |
[37,99] |
| Study (Year) | Cancer Type(s) | Stage of Disease | Type of Controls | Biomarker / Approach | Cohort Size | Performance Metrics | Key Findings |
| Best et al. (2015) [7] | Multi-cancer (lung, breast, colon, etc.) | Stages I–IV (mixed) | Healthy individuals (n=55) | RNA-seq of platelet mRNA (ThromboSeq) | 228 patients + 55 healthy | Sensitivity ~96%, Specificity ~90% | Platelet RNA profiles accurately distinguished cancer vs. non-cancer and identified tumor types with ~71% accuracy. |
| In ’t Veld et al. (2022) [34] | Multi-cancer (pan-cancer approach) | Stages I–IV (mixed) | Asymptomatic controls, symptomatic controls | RNA-seq of platelet mRNA (expanded ThromboSeq) | 1,096 patients + asymptomatic + symptomatic | Overall Sensitivity ~66%, Specificity ~99% (in asymptomatic) |
Large multicenter study showing robust specificity; sensitivity higher in late-stage disease than early-stage. |
| Sabrkhany et al. (2018) [100] | Lung and pancreatic | Early-stage (I–II) and various stages | Healthy donors (n=50) | Platelet proteomics (LC–MS/MS) | 131 patients + 50 healthy | AUC ~0.85–0.90 | Identified >80 proteins with significant dysregulation; protein levels normalized after tumor resection in most early-stage patients. |
| Li et al. (2023) [76,86] | Non-small cell lung cancer (NSCLC) | Stage I–III (predominantly early/mid) | Benign pulmonary nodules (n=150), healthy (n=100) | Targeted mRNA panel + machine learning | 210 NSCLC + 250 controls total | Sensitivity 80%, Specificity 73%, AUC 0.85 | A 3-gene signature (MAX, MTURN, HLA-B) was highly upregulated in NSCLC platelets; strong accuracy for early-stage lung cancer detection. |
| Liu et al. (2022) [101] | Endometrial cancer | Stages I–III | Benign endometrial lesions (n=80) | Combined TEP RNA profiling + ctDNA | 90 endometrial cancer + 80 benign | TEP AUC ~0.88 vs. ctDNA AUC ~0.75 | TEP RNA performed better than ctDNA alone for cancer detection; a combined signature further improved sensitivity in early-stage disease. |
| Type of Challenge | Description / Examples | Potential Solutions | References |
| Pre-analytical | - Platelet contamination by leukocytes or RBCs can confound RNA/protein assays - Sample handling (e.g., multiple freeze/thaw cycles) alters platelet activation status |
- Standardized platelet isolation protocols (e.g., dual-spin centrifugation, gentle pipetting) - Use of automated devices to minimize contamination and activation |
[70,102,103,104] |
| Analytical Sensitivity | - Limited RNA content per platelet requires deep sequencing or highly sensitive qPCR - Variability in RNA extraction efficiency across labs |
- Development of targeted multiplex assays focusing on robust biomarker panels - Rigorous training sets and QC standards for RNA isolation |
[72,73,74,105,106] |
| Data Analysis & Algorithmic | - Complex machine learning models may be “black boxes” for clinicians - Overfitting risk when training on small cohorts |
- Large, multi-institutional datasets to improve generalizability - Explainable AI approaches or simplified gene panels for clinical interpretation |
[73,74] |
| Specificity to Cancer | - Platelets respond to inflammation, infections, and other non-cancer conditions - Elevated platelet counts can be non-cancerous (e.g., reactive thrombocytosis) |
- Include control groups with inflammatory or autoimmune diseases - Combine TEP data with other biomarkers (ctDNA, imaging) for multi-modal specificity |
[29,75,76] |
| Inter-cancer Heterogeneity | - Not all tumors “educate” platelets to the same extent - Certain cancers (e.g., less vascularized, behind blood–brain barrier) may release fewer signals into circulation |
- Large-scale “pan-cancer” studies to capture diverse tumor profiles - Cancer-type–specific panels if broad-based signatures prove insufficient |
[11,28,53,75] |
| Dynamic Changes & Timing | - Single time-point sampling may not capture transient tumor–platelet interactions - Recent chemotherapy or surgery can alter platelet states |
- Serial sampling at multiple time points - Standardized timing of blood draw in relation to treatment (e.g., pre- vs. post-chemotherapy) |
[77,94] |
| Regulatory & Clinical Translation | - No FDA-approved TEP test to date - Need for consensus on validated biomarkers and standardized protocols |
- Large prospective clinical trials with consistent SOPs - Collaboration between academic, regulatory, and industry stakeholders for test standardization |
[78] |
| Direction / Trial Name | Focus / Rationale | Study Population / Design | Biomarker(s) Under Investigation | Primary Endpoints | Status / Expected Completion | References |
| Multi-modal Liquid Biopsy Trials | Integrating TEP RNA signatures with ctDNA and circulating proteins for comprehensive tumor profiling | Prospective cohort studies combining blood-based assays; e.g., advanced lung or ovarian cancer cohorts | Platelet RNA panels, ctDNA mutation load, tumor-specific proteins (VEGF, CEA, etc.) | Diagnostic accuracy (AUC, sensitivity, specificity), early detection of recurrence | Ongoing; projected results within next 1–2 years | [42,80] |
| Automated Platelet Isolation Devices | Development of point-of-care technologies to standardize platelet separation and reduce contamination | Pilot feasibility studies in large hospitals; testing 100–200 blood samples from cancer & non-cancer patients | Concentration and purity of platelet isolates, reproducibility of TEP RNA/protein measurements | Turnaround time for test results, reproducibility across multiple clinics | Prototypes in testing; commercial rollouts anticipated | [82,83] |
| Serial Sampling for Treatment Monitoring | Evaluating dynamic changes in TEPs over multiple time points to detect early therapeutic response or relapse | Phase II trials in lung, breast, and GI cancers, collecting pre-, mid-, and post-therapy platelet samples | Platelet RNA/protein panels (e.g., TIMP1, VEGF, IL-6 mRNA), morphological changes (MPV) | Early detection of relapse vs. imaging alone, improved patient stratification for personalized therapy | Trials expected to complete in 1–3 years | [107,108] |
| Combination of TEP with Immunotherapy Biomarkers | Improving patient selection for immune checkpoint inhibitors (ICIs) by integrating platelet activation signatures | Single-arm or randomized trials combining PD-1/PD-L1 inhibitors with TEP-based monitoring in NSCLC or UC | Platelet activation markers (sP-selectin, sCD40L), immune checkpoint protein transcripts in platelets | Objective response rate, correlation with platelet markers and tumor burden, immune-related adverse events | Early-phase clinical studies; expansions planned | [61,109] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




