Submitted:
28 February 2025
Posted:
03 March 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. PPG Signal Data Collection
2.2. Data Collection Conditions
- Resting state: The patient was in a sitting or lying position with minimal motor activity. In this condition, signals with a minimum level of artifacts were expected to be obtained, which allows them to be used as a benchmark for comparison. This mode is important for testing filters for their ability to preserve signal information without excessive smoothing of its key components, such as peaks and minimum fluctuations.
- Condition in motion: The patient performed moderate physical activity, which included arm movements, walking, and changes in body position. This condition simulated typical scenarios of using wearable devices in everyday life, where motion noise is often observed, which significantly affects signal quality. The data obtained in this condition is key to testing filters for their ability to effectively eliminate artifacts while preserving important biomedical signal features.
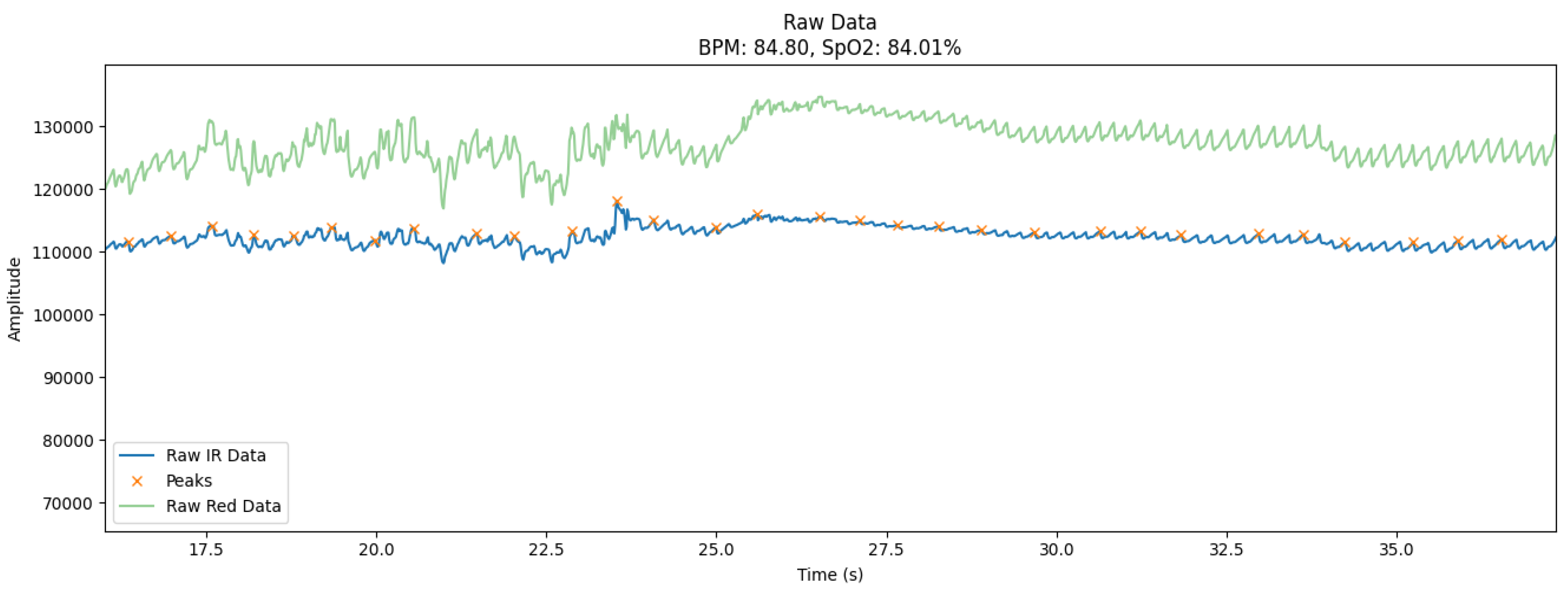
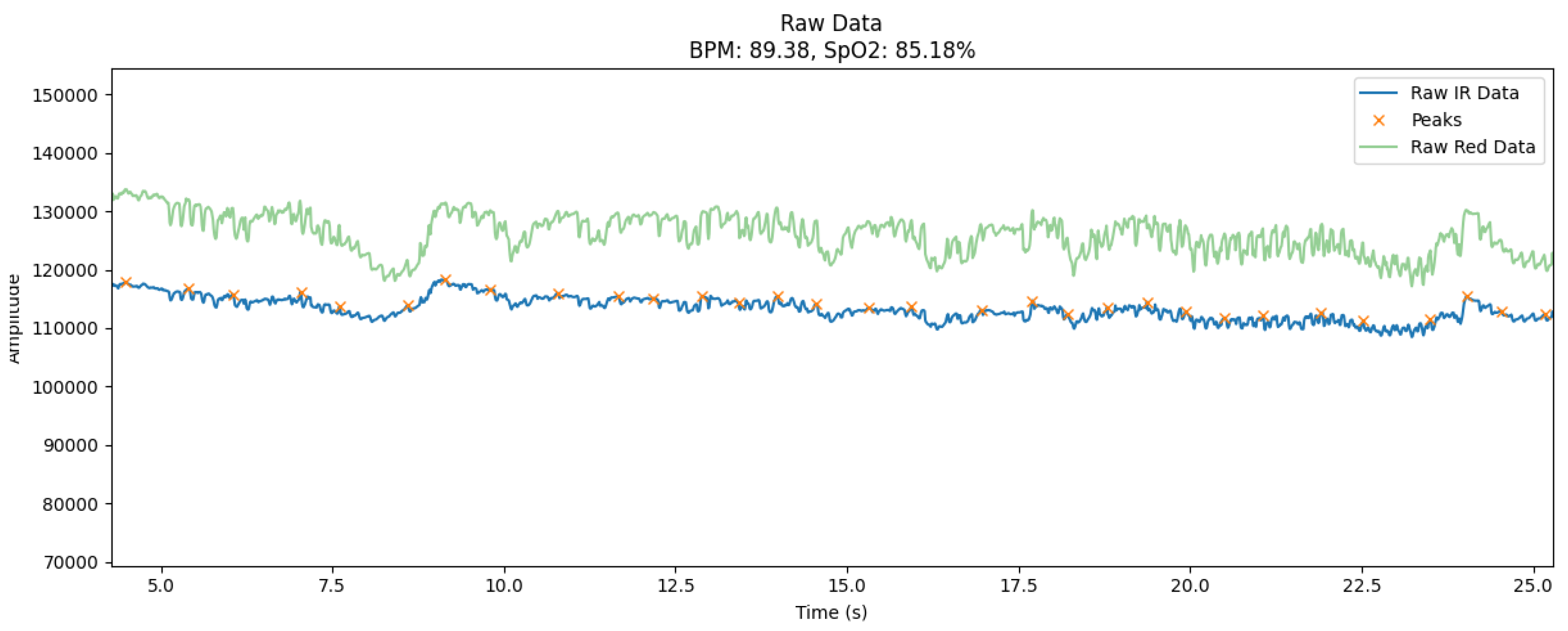
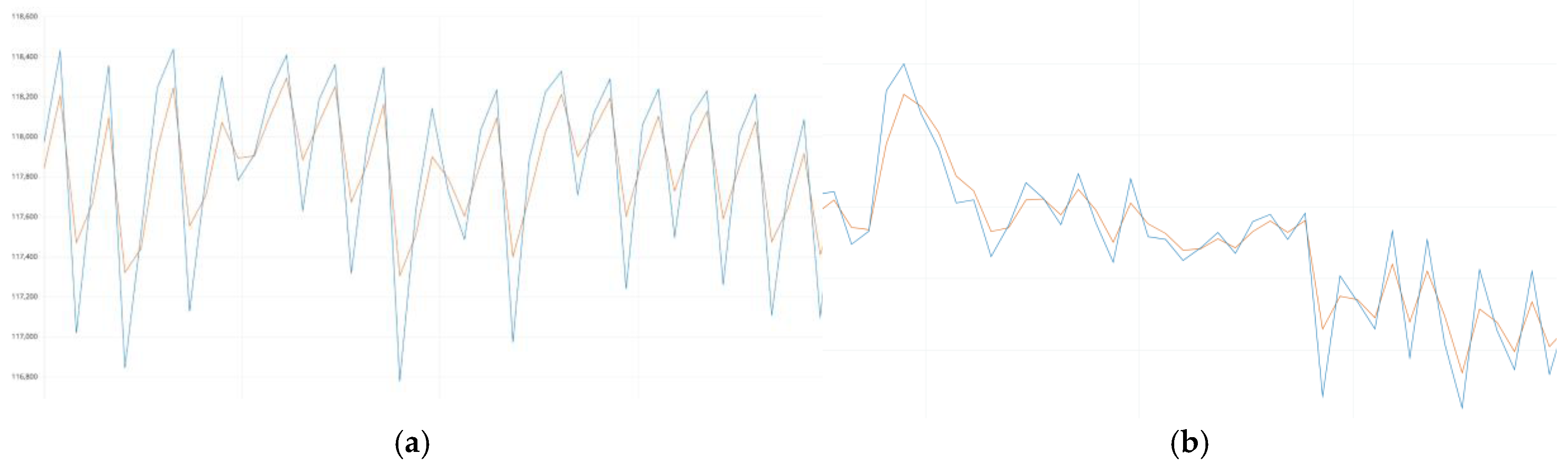
2.3. Selecting Segments for Analysis
- Blue curve (Raw IR Data): Displays the raw signal received from the infrared channel. This signal is the main one for estimating heart rate because it is sensitive to changes in blood volume in tissues.
- Green curve (Raw Red Data): Shows data from the red channel used to calculate SpO₂. This channel reflects the amplitude fluctuations caused by the oxygen saturation of the blood.
- Orange marks (Peaks): Indicates the peak values of systolic waves that correspond to the moments of maximum blood filling of the vessels. These peaks are critical for calculating BPM.
- Unstable amplitude: Both the IR and Red channels show significant fluctuations due to the movement of the sensor on the skin or slight changes in body position.
- Sudden peaks and dips: The signal occasionally contains sudden spikes in amplitude that may be due to slippage of the sensor or short-term loss of skin contact.
- Low frequency noise: Smooth changes in the baseline of the signal indicate drift caused by slow movements or uneven pressure on the skin.
2.4. Methodological Foundations and Implementation of Filtering Algorithms
2.4.1. Low Pass Filter
2.4.2. Moving Average Filter
2.4.3. Weighted Moving Average Filter
2.4.4. Kalman Filter
3. Results
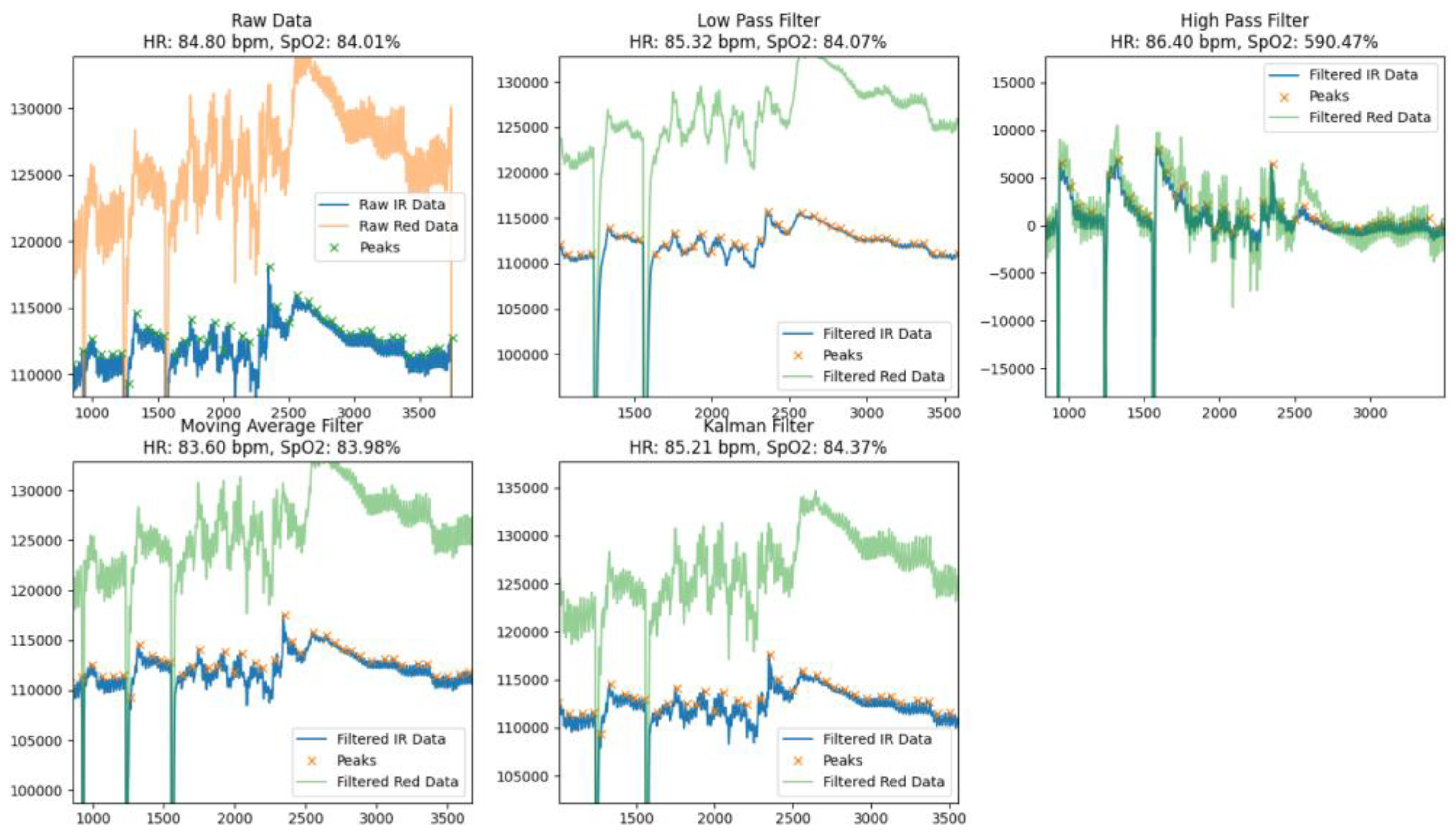
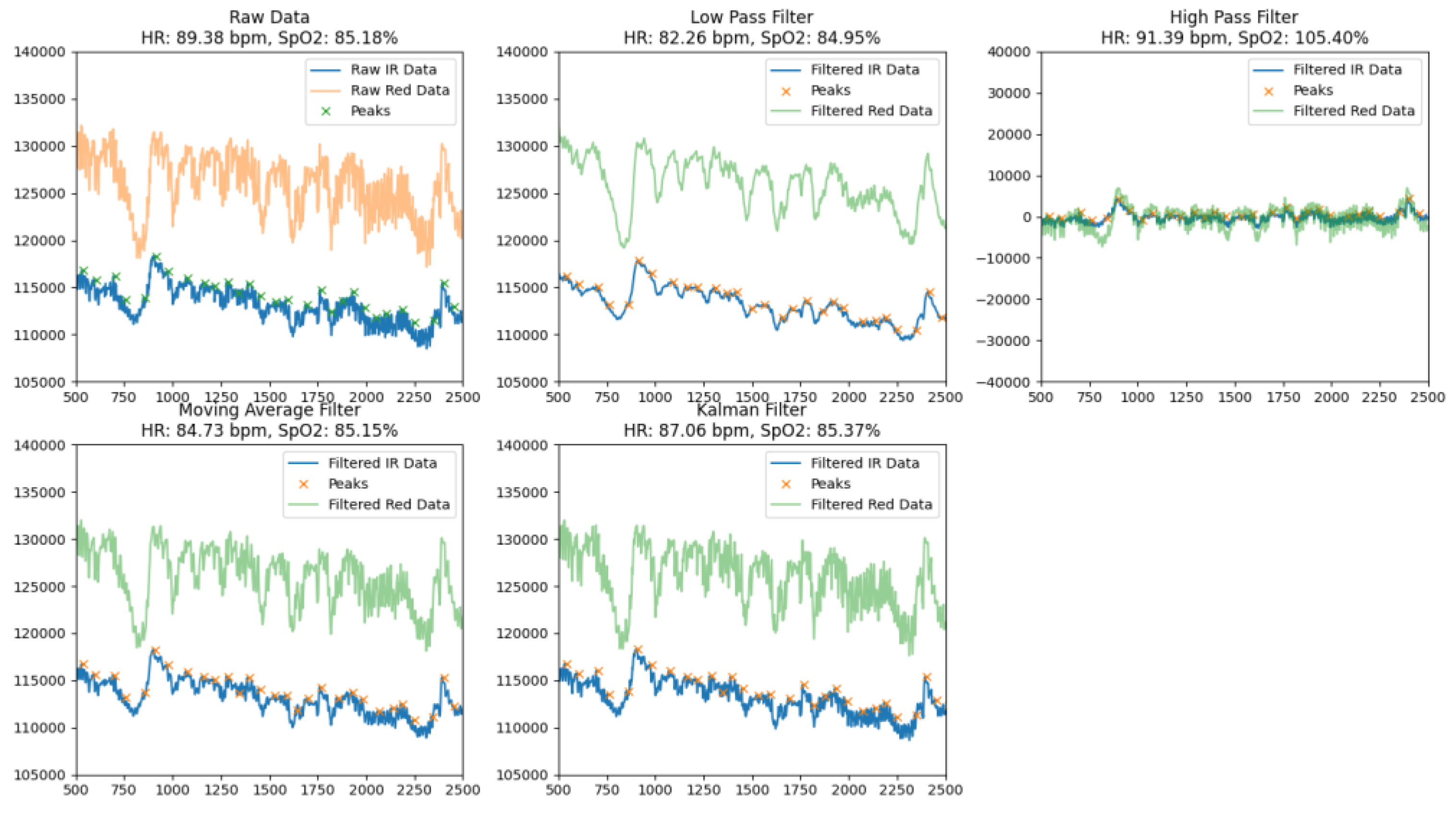
- The Low Pass Filter provided an average HR of 82.26 bpm and SpO₂ of 84.95%. Visual analysis of the processed signal indicated that it was smoothed, but there were some dips and residual noise that could lead to inaccuracies in peak detection. The filter effectively identified the main peak components of the signal, but due to smoothing, some peaks were less pronounced, which could affect the overall accuracy of the calculations.
- The High Pass Filter demonstrated average HR values of 91.39 bpm and SpO₂ of 105.40%, but the processed signal remained significantly noisy with numerous artifacts. This led to an overestimation of SpO₂ values and difficulties in correctly identifying peaks. Due to excessive noise and artifacts, the high-pass filter was less suitable for accurate calculation of physiological parameters.
- The Moving Average Filter showed results of HR of 84.73 bpm and SpO₂ of 85.15%. The signal after processing was well smoothed with well-defined peaks and minimal dips. This makes the moving average filter a reliable tool for determining basic physiological parameters. The minimal level of residual noise ensured high stability and accuracy of calculations, which allows this method to be used in real-time systems.
- The Kalman Filter showed the best results among all the methods considered, providing HR values of 87.06 bpm and SpO₂ - 85.37%. The signal after processing was almost completely cleaned of noise, with clearly defined peaks and virtually no dips. Thanks to its ability to adaptively smooth and dynamically compensate for artifacts, the Kalman filter provided high signal stability and calculation accuracy, making it ideal for use in conditions with high noise levels or motion artifacts.
4. Discussion
5. Conclusions
References
- Shovlin, A.; Ghen, M.; Simpson, P.; Mehta, K. Challenges facing medical data digitization in low-resource contexts. 2013 IEEE Global Humanitarian Technology Conference (GHTC); pp. 365–371.
- Kawala-Janik, A.; Pelc, M.; Podpora, M. Method for EEG Signals Pattern Recognition in Embedded Systems. Elektronika ir Elektrotechnika 2015, 21, 3–9. [Google Scholar] [CrossRef]
- Aziz, R.; Jawed, F.; Khan, S.A.; Sundus, H. Wearable IoT Devices in Rehabilitation: Enabling Personalized Precision Medicine. 10.4018/979-8-3693-2105-8.
- Ferdi, Y. Fractional order calculus-based filters for biomedical signal processing. 2011 1st Middle East Conference on Biomedical Engineering (MECBME); pp. 73–76.
- Lazazzera, R.; Belhaj, Y.; Carrault, G. A New Wearable Device for Blood Pressure Estimation Using Photoplethysmogram. Sensors 2019, 19, 2557. [Google Scholar] [CrossRef] [PubMed]
- ANDERSON, Brian DO; MOORE, John B. Optimal filtering. Courier Corporation, 2005.
- Smith, Steven W. The Scientist and Engineer’s Guide to Digital Signal Processing. 1st ed, California Technical Publishing, 1997.
- Gao, Y.; Tian, D.; Wang, Y. Fuzzy Self-tuning Tracking Differentiator for Motion Measurement Sensors and Application in Wide-Bandwidth High-accuracy Servo Control. Sensors 2020, 20, 948. [Google Scholar] [CrossRef] [PubMed]
- Proakis, J.G. & Manolakis, Dimitris. (1992). Digital Signal Processing.
- Chen, Y.; Li, D.; Li, Y.; Ma, X.; Wei, J. Use Moving Average Filter to Reduce Noises in Wearable PPG During Continuous Monitoring. 181. 193-203. 10.1007/978-3-319-49655-9_26.
- Lyons, Richard. (2001). Understanding Digital Signal Processing’s Frequency Domain. RF Design Magazine.
- Lee, J. Motion artifacts reduction from PPG using cyclic moving average filter. Technol. Heal. Care 2014, 22, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Eilers, P.H.C. A Perfect Smoother. Anal. Chem. 2003, 75, 3631–3636. [Google Scholar] [CrossRef] [PubMed]
- Tamura, T.; Maeda, Y.; Sekine, M.; Yoshida, M. Wearable Photoplethysmographic Sensors—Past and Present. Electronics 2014, 3, 282–302. [Google Scholar] [CrossRef]
- Kawala-Janik, A.; Pelc, M.; Podpora, M. Method for EEG Signals Pattern Recognition in Embedded Systems. Elektronika ir Elektrotechnika 2015, 21, 3–9. [Google Scholar] [CrossRef]
- Wang, Y.-J.; Chen, C.-H.; Sue, C.-Y.; Lu, W.-H.; Chiou, Y.-H. Estimation of Blood Pressure in the Radial Artery Using Strain-Based Pulse Wave and Photoplethysmography Sensors. Micromachines 2018, 9, 556. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-K.; Jo, J.; Lee, Y.; Shin, H.S.; Kwon, O.-W. Particle filter-based noise reduction of PPG signals for robust emotion recognition. In Proceedings of the 2012 IEEE International Conference on Consumer Electronics (ICCE); pp. 598–599.
- Lee, B.; Han, J.; Baek, H.J.; Shin, J.H.; Park, K.S.; Yi, W.J. Improved elimination of motion artifacts from a photoplethysmographic signal using a Kalman smoother with simultaneous accelerometry. Physiol. Meas. 2010, 31, 1585–1603. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



