Submitted:
20 February 2025
Posted:
21 February 2025
You are already at the latest version
Abstract
Keywords:
Plain Language Summary
- Identifying the causes of mental disorders will aid in the discovery of optimal treatments
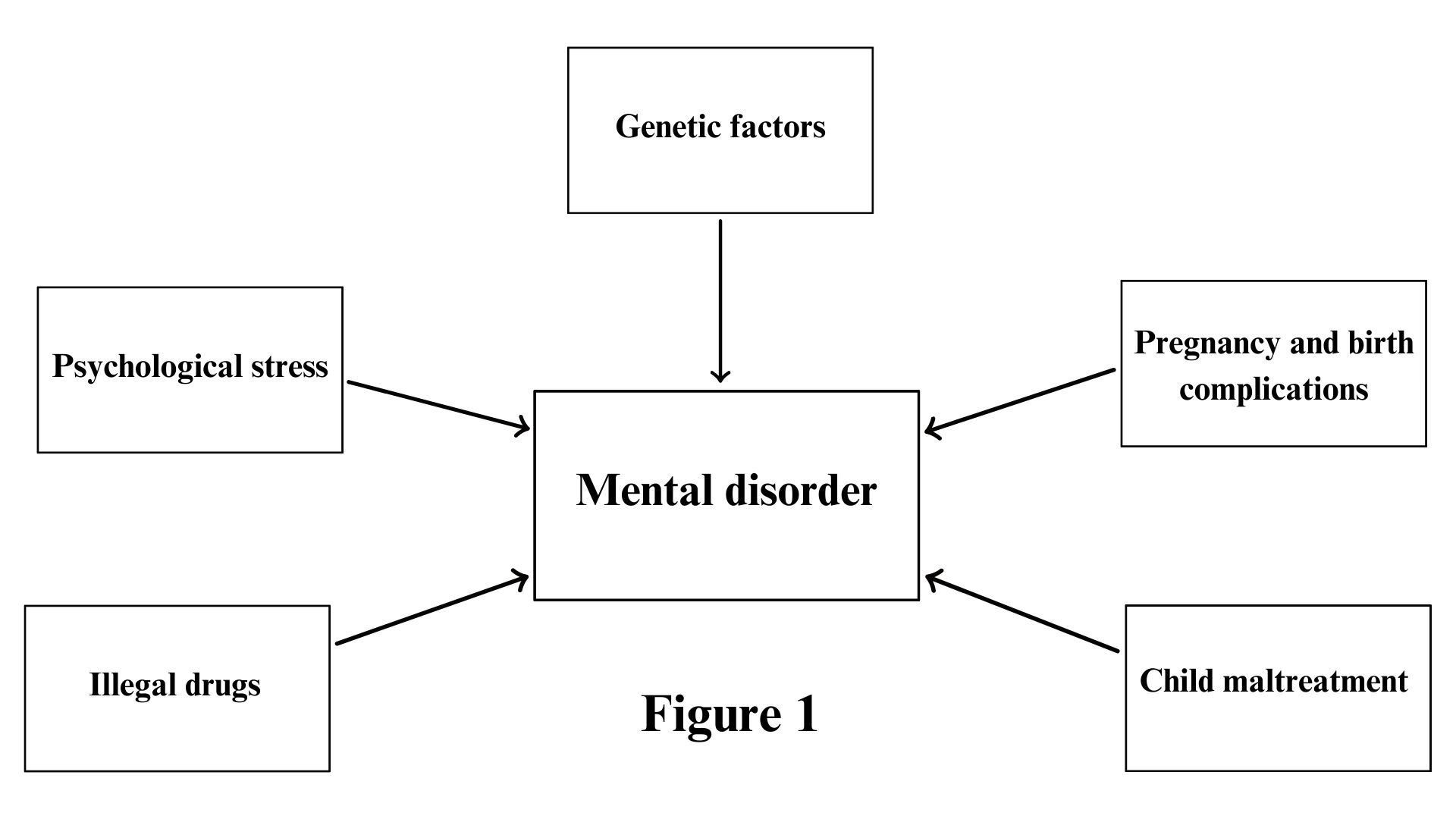
- The psychiatric diseases causes include interplays of genetic, epigenetic, and environmental factors
- Technical advances have facilitated the detection of genetic/epigenetic causes of mental diseases, decreasing even testing costs, but a valid assessment of environmental factors remains a challenge
- Stress is one of the most important environmental causative factors of mental disorders
- Assessing the history of stressful experiences in a patient´s life is a challenge because retrospective questionnaires are inaccurate and biased
- In this narrative review, we demonstrate how to assess an individual´s stress history by measuring hair and nail cortisol content, telomere length, and mitochondrial DNA copy number, as well as epigenetic markers
- We also suggest avenues for research: A quantitative prospective evaluation of environmental etiological factors including their frequency, severity, duration, and timing, and a similar assessment of protective variables are needed
1. Introduction
1.1. Polyenviromic Risk/Protective Score
1.2. Pregnancy and Birth Complications in the Etiology of Mental Disorders
1.3. Environmental Risk Factors in Mental Disorders in Adulthood
1.4. How to Assess a Stressful Experience?
- Stress Questionnaire [17]: This questionnaire detects acute perceived stress. The questions aim to discover whether individuals continue working at night once they arrive home, have low self-confidence or guilty feelings, think about problems when they are supposed to be relaxing, present mood swings or difficulty making decisions, etc. Each of 25 questions can be answered as "yes“ or "no“ resulting in a score of either 1 or 0, respectively. The total score is added and an appropriate recommendation is offered to the respondent.
- Perceived Stress Scale [18]: This scale detects stress perceived during the preceding month. The questions inquire whether the respondent is upset due to unexpected events, whether they feel nervous, stressed, irritated, and/or angry. Each of the ten questions may get a score from "0“ (never) to "4“ (very often), and the total is the sum of all answers.
- Perceived Stress Questionnaire [19]: This questionnaire detects stress experienced during the preceding month. The questions ask whether the respondent feels calm, safe, tired, and/or frustrated, whether they have trouble relaxing, are worried, lack time for themselves, feel under pressure from other people, etc. Each of the 30 questions may be scored from "1“ (almost never) to "4“ (usually). The results of the test correlate highly with anxiety traits.
1.5. Biological Markers of Perceived Acute Stress
1.6. Hair and Nail Cortisol
1.7. Telomere Length and Mitochondrial DNA Copy Number
1.8. Epigenetic Markers
2. Discussion
3. Conclusions
- Surprisingly, obtaining a valid assessment of environmental etiological factors of mental disorders seems to be more complicated than obtaining an evaluation of epigenetic/genetic variables, due to methodological drawbacks.
- Environmental factors have mostly been studied non-systematically, using different methodologies and in isolation, polyenvironmental risk or protective scores have not been widely used.
- Research into environmental protective factors has been neglected.
- Measures of external etiological factors have not been unified/standardized.
- Striving to create a valid assessment method for external etiological factors should lead to improved mental disease prevention and personalized treatment strategies.
4. Future Directions
- A quantitative prospective assessment of environmental factors is more reliable than a qualitative/dichotomic retrospective one.
- Evaluating environmental etiological factors in detail – their frequency and severity as well as the total exposure length – is crucial.
- The assessment of timing is also important, the same environmental etiological factor can be deleterious in a certain period of life, but neutral or protective in another one.
- Simultaneous or subsequent accumulations of environmental factors should be studied.
- Biological environmental etiological factors as well as biological markers of disorders under study should not be neglected.
- Protective environmental factors (like an individual´s favorable social or economic situations) should also be considered.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Hosak, L; Malekirad, M. ; Latalova, K. The Etiology of Mental Disorders. Concise, Clear and Synoptical., 1st ed.; Charles University, Karolinum Press: Prague, Czech Republic, 2022; p. 9. [Google Scholar]
- Rood, J.E.; Regev, A. The legacy of the Human Genome Project. Science. 2021, 373(6562), 1442–1443. [Google Scholar] [CrossRef] [PubMed]
- Mullin, E. The Era of Fast, Cheap Genome Sequencing Is Here. Science. /: from: https, 7 November 2023. [Google Scholar]
- Gayer-Anderson, C.; Jongsma, H.E.; Di Forti, M.; et al. The EUropean Network of National Schizophrenia Networks Studying Gene-Environment Interactions (EU-GEI): Incidence and First-Episode Case-Control Programme. Soc Psychiatry Psychiatr Epidemiol. 2020, 55(5), 645–657. [Google Scholar] [CrossRef]
- Tarricone, I.; D´Andrea, G.; Jongsma, H.E.; et al. Migration history and risk of psychosis: results from the multinational EU-GEI study. Psychol Med. 2022, 52(14), 2972–2984. [Google Scholar] [CrossRef]
- Jongsma, H.E.; Gayer-Anderson, C.; Tarricone, I.; et al. Social disadvantage, linguistic distance, ethnic minority status and first-episode psychosis: results from the EU-GEI case-control study. Psychol Med. 2021, 51(9), 1536–1548. [Google Scholar] [CrossRef] [PubMed]
- Sideli, L.; Schimmenti, A.; La Barbera, D.; et al. Choldhood Maltreatment, Educational Attainment, and IQ: Findings From a Multicentric Case-control Study of First-episode Psychosis (EU-GEI). Schizophr Bull. 2022, 48(3), 575–589. [Google Scholar] [CrossRef] [PubMed]
- Di Forti, M.; Quattrone, D.; Freeman, T.P.; et al. The contribution of cannabis use to variation in the incidence of psychotic disorder across Europe (EU-GEI): a multicentre case-control study. Lancet Psychiatry. 2019, 6(5), 427–436. [Google Scholar] [CrossRef]
- Padmanabhan, J.L.; Shah, J.L.; Tandon, N.; Keshavan, M.S. The “polyenviromic risk score”: aggregating environmental risk factors predicts conversion to psychosis in familial high-risk subjects. Schizophr Res. 2017, 181, 17–22. [Google Scholar] [CrossRef]
- International Schizophrenia Consortium. Common polygenic variation contributes to risk of schizophrenia and bipolar disorder. Nature. 2009, 460(7256), 748–752. [Google Scholar] [CrossRef]
- Hosak, L.; Hosakova, K.; Malekirad, M.; Kamaradova Koncelikova, D.; Zapletalova, J.; Latalova, K. Environmental Factors in the Etiology of Mental Disorders in the Czech Republic. Neuropsychiatr Dis Treat. 2023, 19, 349–359. [Google Scholar] [CrossRef]
- Stilo, S.A.; Murray, R.M. Non-Genetic Factors in Schizophrenia. Curr Psychiatry Rep. 2019, 21(10), 100. [Google Scholar] [CrossRef]
- Al-Haddad, B.J.S.; Oler, E.; Armistead, B.; et al. The fetal origins of mental illness. Am J Obstet Gynecol. 2019, 221(6), 549–562. [Google Scholar] [CrossRef] [PubMed]
- Vorvolakos, T.; Arseniou, S.; Samakouri, M. There is no safe threshold for lead exposure: A literature review. Psychiatriki. 2016, 27(3), 204–214. [Google Scholar] [CrossRef]
- Van den Bergh, B.R.H.; van den Heuvel, M.I.; Lahti, M.; et al. Prenatal developmental origins of behavior and mental health: The influence of maternal stress in pregnancy. Neurosci Biobehav Rev. 2020, 117, 26–64. [Google Scholar] [CrossRef] [PubMed]
- Vassos, E.; Pedersen, C.B.; Murray, R.M.; Collier, D.A.; Lewis, C.M. Meta-Analysis of the Association of Urbanicity With Schizophrenia. Schizophr Bull. 2012, 38(6), 1118–1123. [Google Scholar] [CrossRef]
- Stress Questionnaire [homepage on the Internet]. ISMA. Available from: https://isma.org.uk/sites/default/files/clients/413/Stress-Questionnaire-F2.pdf. Accessed , 2023. 15 November.
- Perceived Stress Scale [homepage on the Internet] State of New Hampshire Employee Assistance Program. Available from: https://www.das.nh.gov/wellness/docs/percieved%20stress%20scale.pdf. Accessed , 2023. 15 November.
- Shahid, A.; Wilkinson, K.; Marcu, S.; Shapiro, C.M. (Eds.) STOP, THAT and One Hundred Other Sleep Scales. 1st ed.; Springer; New York, United States of America, 2012; pp. 273-274.
- Sheeringa, M.S.; Zeanah, C.H. PTSD Semi-Structured Interview and Observation Record for Infants and Young Children., 1st ed.; Department of Psychiatry and Neurology, Tulane University Health Sciences Center: New Orleans, United States of America, 1994; pp. 1–5. [Google Scholar]
- Lightman, S.L.; Birnie, M.T.; Conway-Campbell, B.L. Dynamics of ACTH and Cortisol Secretion and Implications for Disease. Endocrine Rev. 2020, 41(3), 1–21. [Google Scholar] [CrossRef] [PubMed]
- Labad, J. The role of cortisol and prolactin in the pathogenesis and clinical expression of psychotic disorders. Psychoneuroendocrinology. 2019, 102, 24–36. [Google Scholar] [CrossRef]
- Kennis, M,.; Gerritsen, L.; van Dalen, M.; Williams, A.; Cuijpers, P.; Bockting, C. Prospective biomarkers of major depressive disorder: a systematic review and meta-analysis. Mol Psychiatry. [CrossRef]
- Juruena, M.F.; Eror, F.; Cleare, A.J.; Young, A.H. The Role of Early Life Stress in HPA Axis and Anxiety. Adv Exp Med Biol. 2020, 1191, 141–153. [Google Scholar] [CrossRef]
- Schindler-Gmelch, L.; Capito, K.; Steudte-Schmiedgen, S.; Kirschbaum, C.; Berking, M. Hair Cortisol Research in Posttraumatic Stress Disorder - 10 Years of Insights and Open Questions. A Systematic Review. Curr Neuropharmacol. [CrossRef]
- Hellhammer, D.H.; Wüst, S.; Kudielka, B.M. Salivary cortisol as a biomarker in stress research. Psychoneuroendocrinology. 2009, 34(2), 163–171. [Google Scholar] [CrossRef]
- Immanuel, S.; Teferra, M.N.; Baumert, M.; Bidargaddi, N. Heart Rate Variability for Evaluating Psychological Stress Changes in Healthy Adults: A Scoping Review. Neuropsychobiology. 2023, 82(4), 187–202. [Google Scholar] [CrossRef]
- Nicolo, A.; Massaroni, C.; Schena, E.; Sacchetti, M. The Importance of Respiratory Rate Monitoring: From Healthcare to Sport and Exercise. Sensors (Basel). 2020, 20(21), 6396. [Google Scholar] [CrossRef]
- Barki, H.; Chung, W.Y. Mental Stress Detection Using a Wearable In-Ear Plethysmography. Biosensors (Basel). 2023, 13(3), 397. [Google Scholar] [CrossRef] [PubMed]
- Klimek, A.; Mannheim, I.; Schouten, G.; Wouters, E.J.M.; Peeters, M.W.H. Wearables measuring electrodermal activity to assess perceived stress in care: a scoping review. Acta Neuropsychiatr. [CrossRef]
- Khoury, J.E.; Enlow, M.B.; Plamondon, A.; Lyons-Ruth, K. The association between adversity and hair cortisol levels in humans: A meta-analysis. Psychoneuroendocrinology. 2019, 103, 104–117. [Google Scholar] [CrossRef]
- Koumantarou Malisiova, E.; Mourikis, I.; Darviri, C.; et al. Hair cortisol concentrations in mental disorders: A systematic review. Physiol Behav. 2021, 229, 113244. [Google Scholar] [CrossRef] [PubMed]
- Bryson, H.E.; Price, A.M.; Goldfeld, S.; Mensah, F. Associations between social adversity and young children´s hair cortisol. A systematic review. Psychoneuroendocrinology. 2021, 127, 105176. [Google Scholar] [CrossRef]
- Botschek, T.; Husslein, V.; Peters, E.M.J.; Brosig, B. Hair cortisol as outcome parameter for psychological and neuropsychiatric interventions – a literature review. Front Psychiatry. 2023, 14, 1227153. [Google Scholar] [CrossRef]
- Phillips, R.; Kraeuter, A.K.; McDermott, B.; Lupien, S.; Sarnyai, Z. Human nail cortisol as a retrospective biomarker of chronic stress: A systematic review. Psychoneuroendocrinology. 2021, 123, 104903. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Liu, Y.Z.D.; Songyand, Z.; Wan, M. Telomeres-structure, function, and regulation. Exp Cell Res. 2013, 319(2), 133–141. [Google Scholar] [CrossRef]
- Aubert, G.; Lansdorp, P.M. Telomeres and aging. Physiol Rev. 2008, 88(2), 557–579. [Google Scholar] [CrossRef]
- Lin, J.; Epel, E. Stress and telomere shortening: Insights from cellular mechanisms. Ageing Res Rev. 2022, 73, 101507. [Google Scholar] [CrossRef]
- Schutte, N.S.; Malouff, J.M. The Relationship Between Perceived Stress and Telomere Length: A Meta-analysis. Stress Health. 2016, 32(4), 313–319. [Google Scholar] [CrossRef]
- Oliveira. B.S.; Zunzunegui, M.V.; Quinlan, J.; Fahmi, H.; Tu, M.T.; Guerra, R.O. Systematic review of the association between chronic social stress and telomere length: A life course perspective. Ageing Res Rev. 2016, 26, 37–52. [Google Scholar] [CrossRef] [PubMed]
- Coimbra, B.M.; Carvalho, C.M.; Moretti, P.N.; Mello, M.F.; Belangero, S.I. Stress-related telomere length in children: A systematic review. J Psychiatr Res. 2017, 92, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Darrow, S.M.; Verhoeven, J.E.; Revesz, D.; et al. The association between psychiatric disorders and telomere length: A Meta-analysis involving 14,827 persons. Psychosom Med. 2016, 78(7), 776–787. [Google Scholar] [CrossRef]
- Monroy-Jaramillo, N.; Dyukova, E.; Walss-Bass, C. Telomere length in psychiatric disorders: Is it more than an ageing marker? World J Biol Psychiatry. 2018, 19(sup2), S2–S20. [Google Scholar] [CrossRef]
- Li, X.; Wang, J.; Zhou, J.; Huang, P.; Li, J. The association between post-traumatic stress disorder and shorter telomere length: A systematic review and meta-analysis. J Affect Disord. 2017, 218, 322–326. [Google Scholar] [CrossRef] [PubMed]
- Ridout, K.K.; Khan, M.; Ridout, S.J. Adverse Childhood Experiences Run Deep: Toxic Early Life Stress, Telomeres, and Mitochondrial DNA Copy Number, the Biological Markers of Cumulative Stress. Bioessays. 2018, 40(9), e1800077. [Google Scholar] [CrossRef] [PubMed]
- Tyrka, A.R.; Carpenter, L.L.; Kao, H.T.; et al. Association of telomere length and mitochondrial DNA copy number in a community sample of healthy adults. Exp Gerontol. 2015, 66, 17–20. [Google Scholar] [CrossRef]
- Cai, N.; Chang, S.; Li, Y.; et al. Molecular signatures of major depression. Curr Biol. 2015, 25(9), 1146–1156. [Google Scholar] [CrossRef]
- Picard, M.; McEwen, B.S. Psychological Stress and Mitochondria: A Systematic Review. Psychosom Med. 2018, 80(2), 141–153. [Google Scholar] [CrossRef]
- Lutz, P.E.; Almeida, D.; Fiori, L.M.; Turecki, G. Childhood maltreatment and stress-related psychopathology: the epigenetic memory hypothesis. Curr Pharm Des. 2015, 21(11), 1413–1417. [Google Scholar] [CrossRef]
- Daskalakis, N.P.; Rijal, C.M.; King, C.; Huckins, L.M.; Ressler, K.J. Recent Genetics and Epigenetics Approaches to PTSD. Curr Psychiatry Rep. 1007. [Google Scholar]
- Fabrizio, P.; Garvis, S.; Palladino, F. Histone Methylation and Memory of Environmental Stress. Cells. 2019, 8(4), 339. [Google Scholar] [CrossRef] [PubMed]
- Poon, C.H.; Heng, B.C.; Lim, L.W. New insights on brain-derived neurotrophic factor epigenetics: from depression to memory extinction. Ann N Y Acad Sci. 2021, 1484(1), 9–31. [Google Scholar] [CrossRef] [PubMed]
- Bonnaud, E.M.; Suberbielle, E.; Malnou, C.E. Histone acetylation in neuronal (dys)function. Biomol Concepts. 2016, 7(2), 103–116. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




