Submitted:
14 February 2025
Posted:
14 February 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Pathophysiology of Dyssynchrony in Left Bundle Branch Block
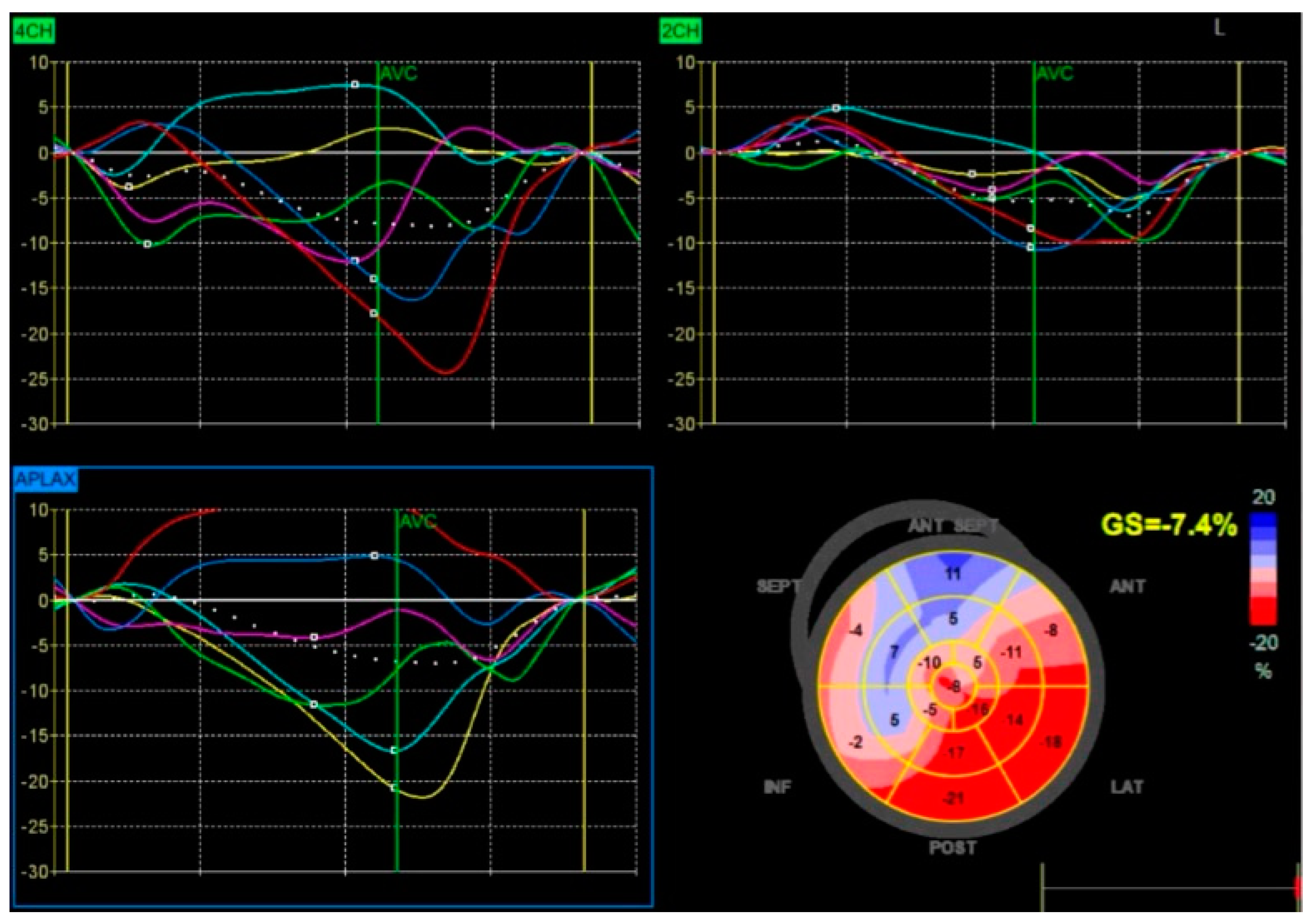
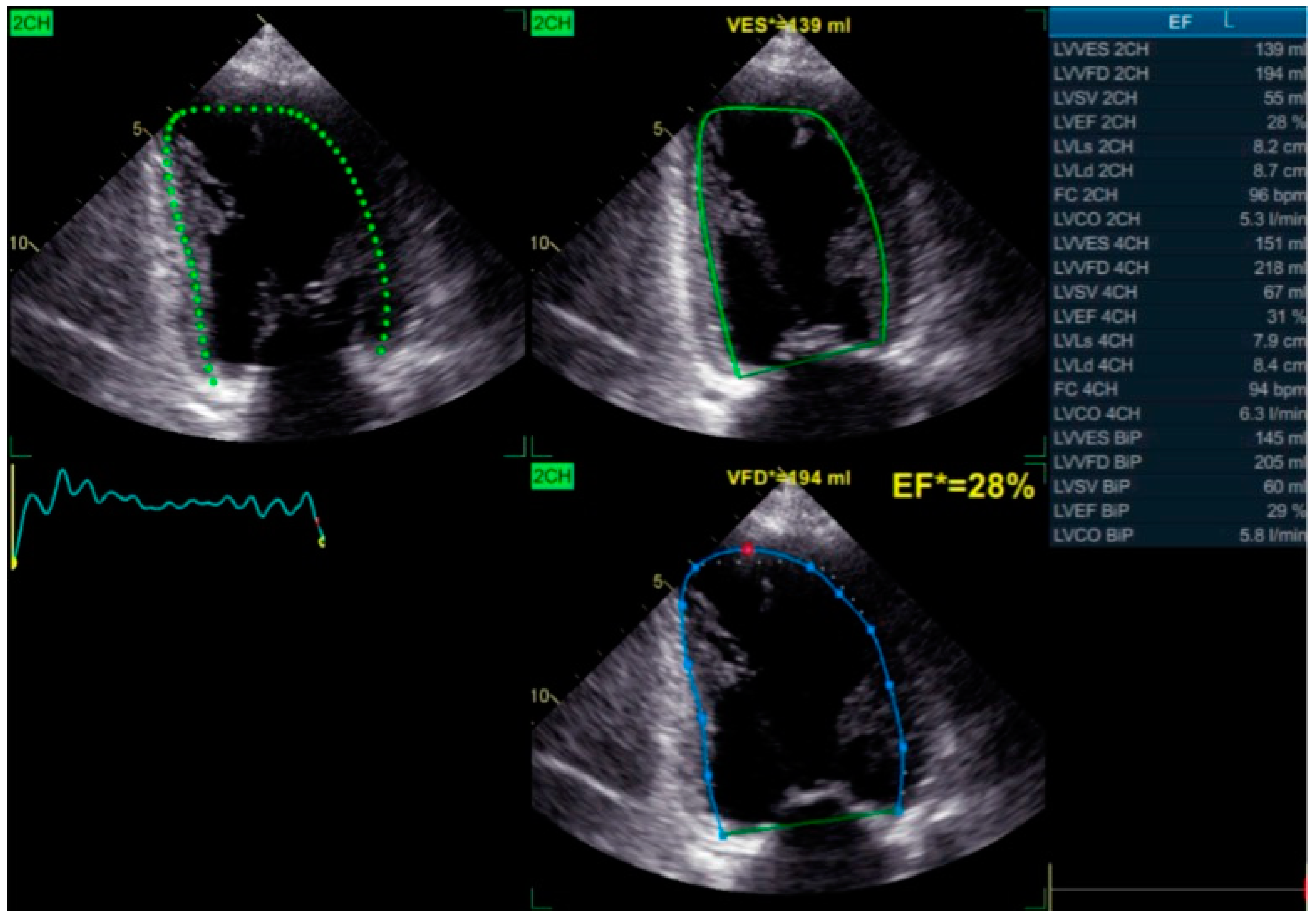
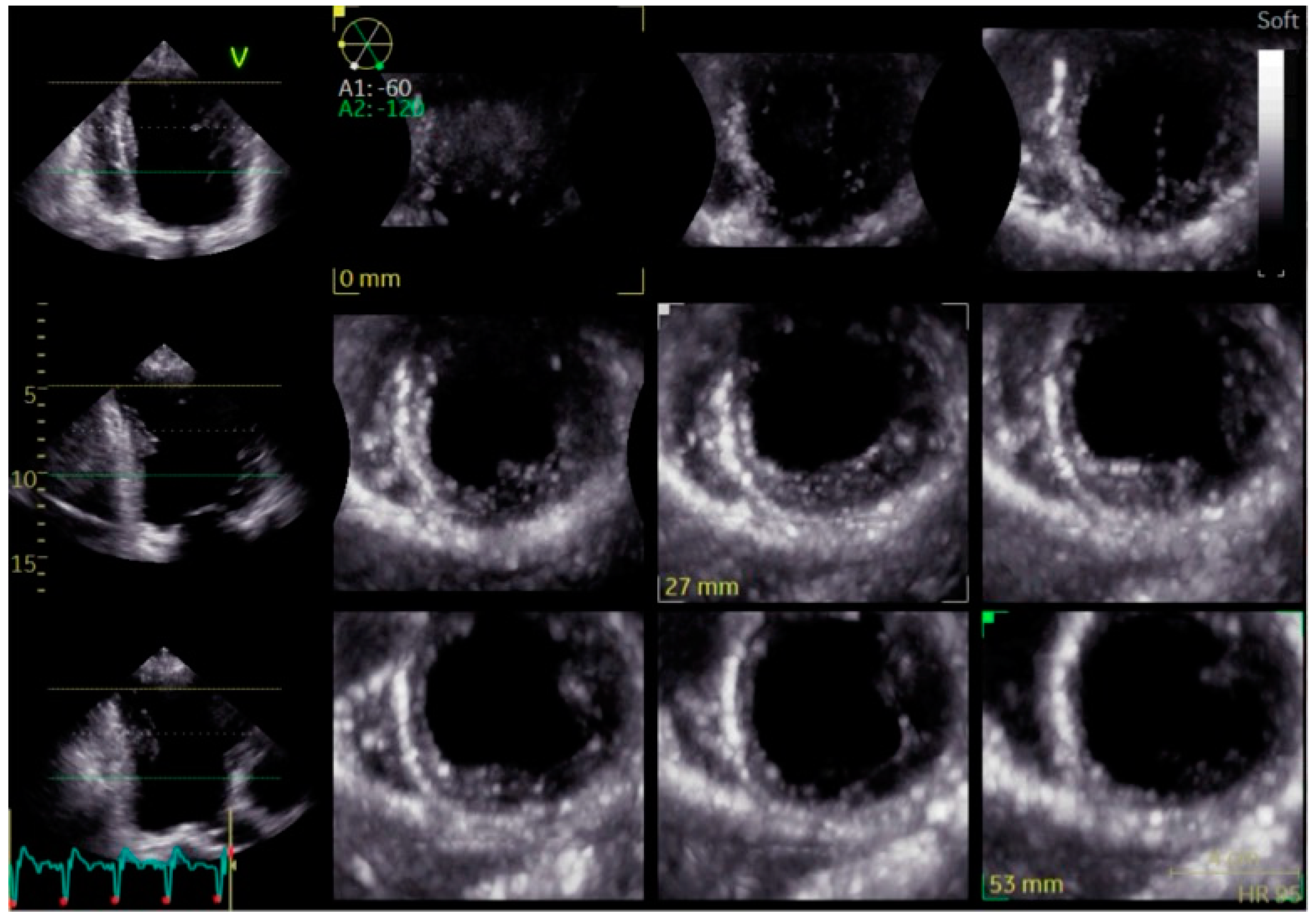
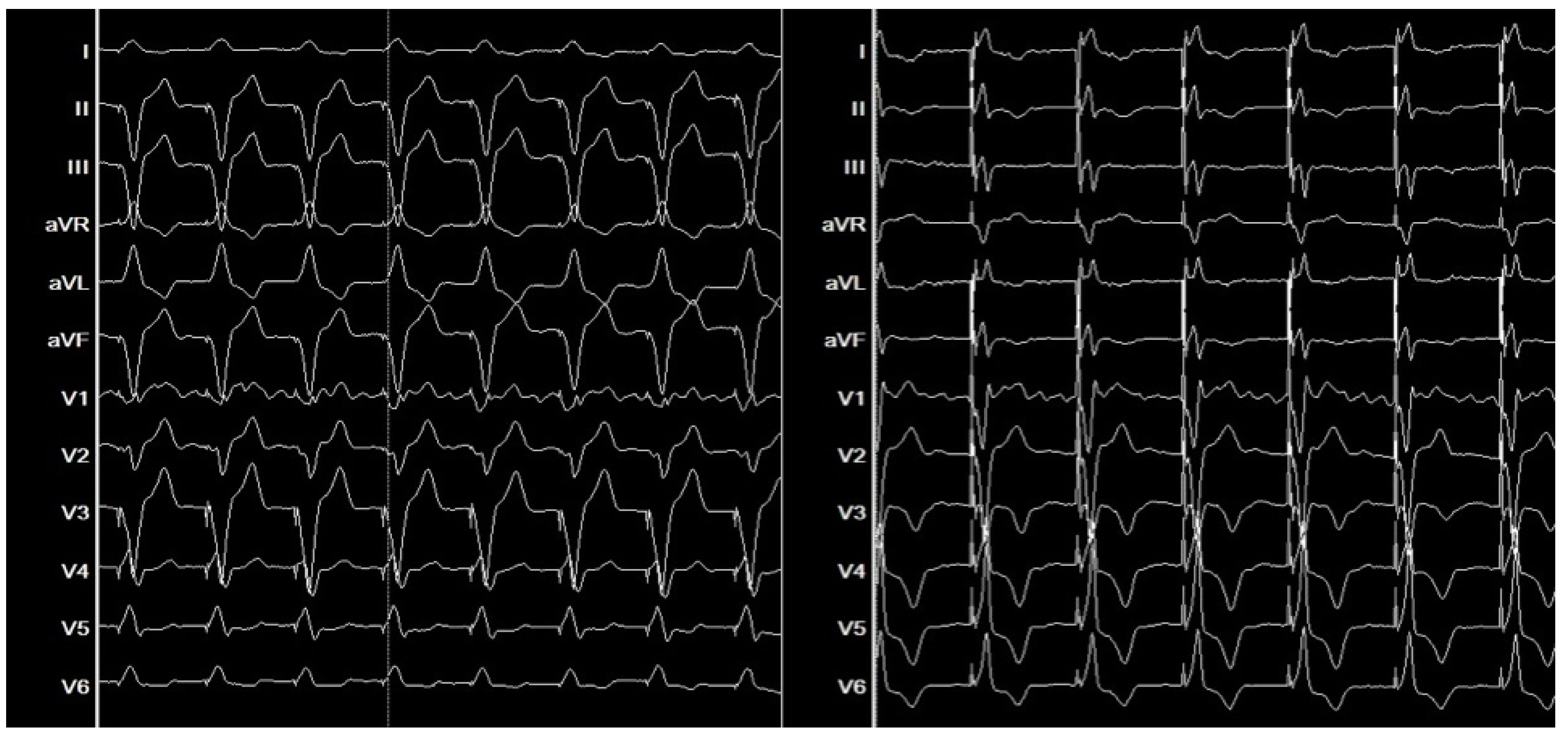
3. Imaging Modalities for Assessing Dyssynchrony
4. Clinical Outcomes of Bundle Branch Pacing
5. Echocardiographic Selection Criteria and Evidence of Reverse Remodeling Following Bundle Branch Pacing
6. Biomolecular Effects of Bundle Branch Stimulation
7. Challenges and Future Directions
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| BBP | Bundle Branch Pacing |
| HFrEF | Heart Failure with Reduced Ejection Fraction |
| TDI | Tissue Doppler Imaging |
| STE | Speckle Tracking Imaging |
| GLS | Global Longitudinal Strain |
| CRT | Cardiac Resyncronization Therapy |
| LBB | Left Bundle Branch Block |
References
- Vicent, L.; Álvarez-García, J.; Vazquez-Garcia, R.; González-Juanatey, J.R.; Rivera, M.; Segovia, J.; Pascual-Figal, D.; Bover, R.; Worner, F.; Fernández-Avilés, F.; et al. Coronary Artery Disease and Prognosis of Heart Failure with Reduced Ejection Fraction. J. Clin. Med. 2023, 12, 3028. [Google Scholar] [CrossRef] [PubMed]
- Friedman, D.J.; Emerek, K.; Kisslo, J.; Søgaard, P.; Atwater, B.D. Left bundle-branch block is associated with asimilar dyssynchronous phenotype in heart failure patients with normal and reduced ejection fractions. Am. Hear. J. 2020, 231, 45–55. [Google Scholar] [CrossRef]
- Pujol-López, M.; Tolosana, J.M.; Upadhyay, G.A.; Mont, L.; Tung, R. Left Bundle Branch Block. Cardiol. Clin. 2023, 41, 379–391. [Google Scholar] [CrossRef] [PubMed]
- Sami, A.; Iftekhar, M.F.; Khan, I.; Jan, R. Intraventricular Dyssynchrony among patients with left bundle branch block. Pak. J. Med Sci. 2018, 34, 390–392. [Google Scholar] [CrossRef] [PubMed]
- Leclercq, C.; Burri, H.; Delnoy, P.P.; A Rinaldi, C.; Sperzel, J.; Calò, L.; Concha, J.F.; Fusco, A.; Al Samadi, F.; Lee, K.; et al. Cardiac resynchronization therapy non-responder to responder conversion rate in the MORE-CRT MPP trial. Eur. 2023, 25. [Google Scholar] [CrossRef]
- Liu, P.; Wang, Q.; Sun, H.; Qin, X.; Zheng, Q. Left Bundle Branch Pacing: Current Knowledge and Future Prospects. Front. Cardiovasc. Med. 2021, 8. [Google Scholar] [CrossRef]
- Siranart, N.; Chokesuwattanaskul, R.; Prasitlumkum, N.; Huntrakul, A.; Phanthong, T.; Sowalertrat, W.; Navaravong, L.; Cheungpasitporn, W.; Jongnarangsin, K. Reverse of left ventricular remodeling in heart failure patients with left bundle branch area pacing: Systematic review and meta-analysis. Pacing Clin. Electrophysiol. 2023, 46, 459–466. [Google Scholar] [CrossRef]
- AlJaroudi, W. Left ventricular mechanical dyssynchrony in patient with CAD: The Saga continues. J. Nucl. Cardiol. 2020, 28, 3021–3024. [Google Scholar] [CrossRef]
- Bacharova, L.; de Luna, B. The Primary Alteration of Ventricular Myocardium Conduction: The Significant Determinant of Left Bundle Branch Block Pattern. Cardiol. Res. Pr. 2022, 2022, 1–8. [Google Scholar] [CrossRef]
- Sillanmäki, S.; Lipponen, J.A.; Tarvainen, M.P.; Laitinen, T.; Hedman, M.; Hedman, A.; Kivelä, A.; Hämäläinen, H.; Laitinen, T. Relationships between electrical and mechanical dyssynchrony in patients with left bundle branch block and healthy controls. J. Nucl. Cardiol. 2018, 26, 1228–1239. [Google Scholar] [CrossRef]
- Hayama, Y.; Miyazaki, A.; Ohuchi, H.; Miike, H.; Negishi, J.; Sakaguchi, H.; Kurosaki, K.; Shimizu, S.; Kawada, T.; Sugimachi, M. Septal Flash-like Motion of the Earlier Activated Ventricular Wall Represents the Pathophysiology of Mechanical Dyssynchrony in Single-Ventricle Anatomy. J. Am. Soc. Echocardiogr. 2020, 33, 612–621.e2. [Google Scholar] [CrossRef] [PubMed]
- Calle, S.; Calle, S.; Kamoen, V.; Kamoen, V.; De Buyzere, M.; De Buyzere, M.; De Pooter, J.; De Pooter, J.; Timmermans, F.; Timmermans, F. A Strain-Based Staging Classification of Left Bundle Branch Block-Induced Cardiac Remodeling. JACC: Cardiovasc. Imaging 2021, 14, 1691–1702. [Google Scholar] [CrossRef] [PubMed]
- Sze, E.; Daubert, J.P. Left bundle branch block-induced left ventricular remodeling and its potential for reverse remodeling. J. Interv. Card. Electrophysiol. 2018, 52, 343–352. [Google Scholar] [CrossRef]
- Degtiarova, G.; Claus, P.; Duchenne, J.; Schramm, G.; Nuyts, J.; Verberne, H.J.; Voigt, J.-U.; Gheysens, O. Impact of left bundle branch block on myocardial perfusion and metabolism: A positron emission tomography study. J. Nucl. Cardiol. 2019, 28, 1730–1739. [Google Scholar] [CrossRef]
- Brenyo, A.; Pietrasik, G.; Barsheshet, A.; Huang, D.T.; Polonsky, B.; McNITT, S.; Moss, A.J.; Zareba, W. QRS Fragmentation and the Risk of Sudden Cardiac Death in MADIT II. J. Cardiovasc. Electrophysiol. 2012, 23, 1343–1348. [Google Scholar] [CrossRef]
- Vezi, B.; Akinrimisi, O.P. Permanent Bi-Bundle Pacing in a Patient With Heart Failure and Left Bundle Branch Block. JACC: Case Rep. 2022, 4, 101688. [Google Scholar] [CrossRef]
- Agricola, E.; Ancona, F. Is There Any Room Left for Echocardiographic-Dyssynchrony Parameters in the Field of CRT? JACC: Cardiovasc. Imaging 2023, 16, 885–888. [Google Scholar] [CrossRef]
- Bazoukis, G.; Thomopoulos, C.; Tse, G.; Tsioufis, K.; Nihoyannopoulos, P. Global longitudinal strain predicts responders after cardiac resynchronization therapy—a systematic review and meta-analysis. Hear. Fail. Rev. 2021, 27, 827–836. [Google Scholar] [CrossRef]
- Gurgu, A.; Luca, C.-T.; Vacarescu, C.; Petrescu, L.; Goanta, E.-V.; Lazar, M.-A.; Arnăutu, D.-A.; Cozma, D. Considering Diastolic Dyssynchrony as a Predictor of Favorable Response in LV-Only Fusion Pacing Cardiac Resynchronization Therapy. Diagnostics 2023, 13, 1186. [Google Scholar] [CrossRef]
- Mandoli, G.E.; Cameli, M.; Pastore, M.C.; Benfari, G.; Malagoli, A.; D’andrea, A.; Sperlongano, S.; Bandera, F.; Esposito, R.; Santoro, C.; et al. Speckle tracking echocardiography in early disease stages: a therapy modifier? J. Cardiovasc. Med. 2023, 24, e55–e66. [Google Scholar] [CrossRef]
- Laufer-Perl, M.; Arnold, J.H.; Moshkovits, Y.; Havakuk, O.; Shmilovich, H.; Chausovsky, G.; Sivan, A.; Szekely, Y.; Arbel, Y.; Banai, S.; et al. Evaluating the role of left ventricle global longitudinal strain in myocardial perfusion defect assessment. Int. J. Cardiovasc. Imaging 2021, 38, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Pujol-López, M.; Jiménez-Arjona, R.; Garcia-Ribas, C.; Borràs, R.; Guasch, E.; Regany-Closa, M.; Graterol, F.R.; Niebla, M.; Carro, E.; Roca-Luque, I.; et al. Longitudinal comparison of dyssynchrony correction and ‘strain’ improvement by conduction system pacing: LEVEL-AT trial secondary findings. Eur. Hear. J. - Cardiovasc. Imaging 2024, 25, 1394–1404. [Google Scholar] [CrossRef] [PubMed]
- Bragança, B.; Trêpa, M.; Santos, R.; Silveira, I.; Fontes-Oliveira, M.; Sousa, M.J.; Reis, H.; Torres, S.; Santos, M. Echocardiographic Assessment of Right Ventriculo-arterial Coupling: Clinical Correlates and Prognostic Impact in Heart Failure Patients Undergoing Cardiac Resynchronization Therapy. J. Cardiovasc. Imaging 2020, 28, 109–120. [Google Scholar] [CrossRef]
- Berlot, B.; Bucciarelli-Ducci, C.; Palazzuoli, A.; Marino, P. Myocardial phenotypes and dysfunction in HFpEF and HFrEF assessed by echocardiography and cardiac magnetic resonance. Hear. Fail. Rev. 2019, 25, 75–84. [Google Scholar] [CrossRef]
- Chen, Z.; Ma, X.; Gao, Y.; Wu, S.; Xu, N.; Chen, F.; Song, Y.; Li, C.; Lu, M.; Dai, Y.; et al. Cardiac magnetic resonance–derived myocardial scar is associated with echocardiographic response and clinical prognosis of left bundle branch area pacing for cardiac resynchronization therapy. Eur. 2023, 25. [Google Scholar] [CrossRef]
- Moriña-Vázquez, P.; Moraleda-Salas, M.T.; López-Masjuan-Ríos, Á.; Esteve-Ruiz, I.; Arce-León, Á.; Lluch-Requerey, C.; Rodríguez-Albarrán, A.; Venegas-Gamero, J.; Gómez-Menchero, A.E. Improvement in electrocardiographic parameters of repolarization related to sudden death in patients with ventricular dysfunction and left bundle branch block after cardiac resynchronization through His bundle pacing. J. Interv. Card. Electrophysiol. 2023, 66, 2003–2010. [Google Scholar] [CrossRef]
- Sharma PS, Patel NR, Ravi V, et al. Clinical outcomes of left bundle branch area pacing compared to right ventricular pacing: Results from the Geisinger-Rush Conduction System Pacing Registry [published correction appears in Heart Rhythm. 2023 Jul;20(7):1100. https://doi.org/10.1016/j.hrthm.2023.05.001]. Heart Rhythm. 2022;19(1):3-11. [CrossRef]
- Zhuo, J.; Chen, C.; Lin, J.; Wang, J.; Fu, F. A long-term clinical comparative study of left bundle branch pacing versus biventricular pacing in patients with heart failure and complete left bundle branch block. Hear. Vessel. 2025, 1–11. [Google Scholar] [CrossRef]
- Felix, I.F.; Collini, M.; Fonseca, R.; Guida, C.; Armaganijan, L.; Healey, J.S.; Carvalho, G. Conduction system pacing versus biventricular pacing in heart failure with reduced ejection fraction: A systematic review and meta-analysis of randomized controlled trials. Hear. Rhythm. 2024, 21, 881–889. [Google Scholar] [CrossRef]
- Vezi, B.; Akinrimisi, O.P. Permanent Bi-Bundle Pacing in a Patient With Heart Failure and Left Bundle Branch Block. JACC: Case Rep. 2022, 4, 101688. [Google Scholar] [CrossRef]
- Jastrzębski, M.; Kiełbasa, G.; Cano, O.; Curila, K.; Heckman, L.; De Pooter, J.; Chovanec, M.; Rademakers, L.; Huybrechts, W.; Grieco, D.; et al. Left bundle branch area pacing outcomes: the multicentre European MELOS study. Eur. Hear. J. 2022, 43, 4161–4173. [Google Scholar] [CrossRef]
- Fletcher-Hall, S.D. Pacemaker-induced cardiomyopathy. J. Am. Acad. Physician Assist. 2023, 36, 1–4. [Google Scholar] [CrossRef]
- Shan, Q.-J.; Xu, H.; Zhou, X.-J.; Chang, Q.; Ji, L.; Chen, C.; Jiang, Z.-X. Effects of permanent left bundle branch area pacing on QRS duration and short-term cardiac function in pacing-indicated patients with left bundle branch block. Chin. Med J. 2021, 134, 1101–1103. [Google Scholar] [CrossRef] [PubMed]
- Vijayaraman, P.; Pokharel, P.; Subzposh, F.A.; Oren, J.W.; Storm, R.H.; Batul, S.A.; Beer, D.A.; Hughes, G.; Leri, G.; Manganiello, M.; et al. His-Purkinje Conduction System Pacing Optimized Trial of Cardiac Resynchronization Therapy vs Biventricular Pacing. JACC: Clin. Electrophysiol. 2023, 9, 2628–2638. [Google Scholar] [CrossRef] [PubMed]
- Madias, J.E. The impact of changing oedematous states on the QRS duration: implications for cardiac resynchronization therapy and implantable cardioverter/defibrillator implantation. Eur. 2005, 7, 158–164. [Google Scholar] [CrossRef]
- Cheng, Y.; Wang, Z.; Li, Y.; Qi, J.; Liu, J. Left bundle branch pacing in heart failure patients with left bundle branch block: A systematic review and meta-analysis. Pacing Clin. Electrophysiol. 2021, 45, 212–218. [Google Scholar] [CrossRef]
- Vijayaraman, P.; Sharma, P.S.; Cano, Ó.; Ponnusamy, S.S.; Herweg, B.; Zanon, F.; Jastrzebski, M.; Zou, J.; Chelu, M.G.; Vernooy, K.; et al. Comparison of Left Bundle Branch Area Pacing and Biventricular Pacing in Candidates for Resynchronization Therapy. Circ. 2023, 82, 228–241. [Google Scholar] [CrossRef]
- Auricchio, A.; Prinzen, F.W. Enhancing Response in the Cardiac Resynchronization Therapy Patient. JACC: Clin. Electrophysiol. 2017, 3, 1203–1219. [Google Scholar] [CrossRef]
- Ali, N.; Arnold, A.D.; Miyazawa, A.A.; Keene, D.; Peters, N.S.; Kanagaratnam, P.; Qureshi, N.; Ng, F.S.; Linton, N.W.F.; Lefroy, D.C.; et al. Septal scar as a barrier to left bundle branch area pacing. Pacing Clin. Electrophysiol. 2023, 46, 1077–1084. [Google Scholar] [CrossRef]
- Lisi, M.; Cameli, M.; Mandoli, G.E.; Pastore, M.C.; Righini, F.M.; D’ascenzi, F.; Focardi, M.; Rubboli, A.; Mondillo, S.; Henein, M.Y. Detection of myocardial fibrosis by speckle-tracking echocardiography: from prediction to clinical applications. Hear. Fail. Rev. 2022, 27, 1857–1867. [Google Scholar] [CrossRef]
- Ahmed I, Kayani WT. Cardiac Resynchronization Therapy. In: StatPearls. Treasure Island (FL): StatPearls Publishing; , 2023. 30 July.
- Yang, Z.; Liang, J.; Chen, R.; Pang, N.; Zhang, N.; Guo, M.; Gao, J.; Wang, R. Clinical outcomes of left bundle branch area pacing: Prognosis and specific applications. Pacing Clin. Electrophysiol. 2023, 47, 80–87. [Google Scholar] [CrossRef]
- Li, X.; Zhu, H.; Fan, X.; Wang, Q.; Wang, Z.; Li, H.; Tao, J.; Wang, H.; Liu, Z.; Yao, Y. Tricuspid regurgitation outcomes in left bundle branch area pacing and comparison with right ventricular septal pacing. Hear. Rhythm. 2022, 19, 1202–1203. [Google Scholar] [CrossRef] [PubMed]
- Cacciapuoti, F.; Marfella, R.; Paolisso, G.; Cacciapuoti, F. Is the aging heart similar to the diabetic heart? Evaluation of LV function of the aging heart with Tissue Doppler Imaging. Aging Clin. Exp. Res. 2009, 21, 22–26. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, J.; Althoff, T.; Busch, S.; Chun, K.J.; Dahme, T.; Ebert, M.; Estner, H.; Gunawardene, M.; Heeger, C.; Iden, L.; et al. „Left bundle branch (area) pacing“: Sondenpositionierung und Erfolgskriterien – Schritt für Schritt. Herzschrittmachertherapie + Elektrophysiologie 2024, 36, 1–9. [Google Scholar] [CrossRef]
- Guo, J.; Li, L.; Xiao, G.; Huang, X.; Li, Q.; Wang, Y.; Cai, B. Feasibility and stability of left bundle branch pacing in patients after prosthetic valve implantation. Clin. Cardiol. 2020, 43, 1110–1118. [Google Scholar] [CrossRef]
- Mei, Y.; Han, R.; Cheng, L.; Li, R.; He, Y.; Xie, J.; Wang, Z.; Wu, Y. Assessment of Cardiac Function and Ventricular Mechanical Synchronization in Left Bundle Branch Area Pacing by Speckle Tracking and Three-Dimensional Echocardiography. Am. J. Cardiol. 2022, 187, 1–9. [Google Scholar] [CrossRef]
- Zhu, H.; Qin, C.; Du, A.; Wang, Q.; He, C.; Zou, F.; Li, X.; Tao, J.; Wang, C.; Liu, Z.; et al. Comparisons of long-term clinical outcomes with left bundle branch pacing, left ventricular septal pacing, and biventricular pacing for cardiac resynchronization therapy. Hear. Rhythm. 2024, 21, 1342–1353. [Google Scholar] [CrossRef]
- Ponnusamy, S.S.; Arora, V.; Namboodiri, N.; Kumar, V.; Kapoor, A.; Vijayaraman, P. Left bundle branch pacing: A comprehensive review. J. Cardiovasc. Electrophysiol. 2020, 31, 2462–2473. [Google Scholar] [CrossRef]
- Cheng, S.; Li, H.; Hu, Y.; Jin, H.; Weng, S.; He, P.; Huang, H.; Liu, X.; Gu, M.; Niu, H.; et al. Left Bundle Branch Area Pacing With or Without Conduction System Capture in Heart Failure Models. JACC: Clin. Electrophysiol. 2024, 10, 2234–2246. [Google Scholar] [CrossRef]
- Chen, X.; Xu, J.; Wu, Y.; Qin, C.; Xue, S.; Hu, G.; Zou, J.; Shan, Q.; Zhou, X.; Hou, X.; et al. The association between paced left ventricular activation time and cardiac reverse remodeling in heart failure patients with left bundle branch block. J. Cardiovasc. Electrophysiol. 2024, 35, 1636–1644. [Google Scholar] [CrossRef]
- Peigh, G.; Steinberg, B.A. Mechanisms for structural remodeling with left bundle branch area pacing: more than meets the eye. J. Interv. Card. Electrophysiol. 2023, 67, 221–223. [Google Scholar] [CrossRef]
- Chen, X.; Huang, M.; Chen, Y.; Xu, H.; Wu, M. Mineralocorticoid receptor antagonists and heart failure with preserved ejection fraction: current understanding and future prospects. Hear. Fail. Rev. 2024, 30, 191–208. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Wu, Y.; Zhou, C.; Xie, J.; Zhang, Y.; Yang, X.; Xiao, J.; Wang, D.W.; Shan, C.; Zhou, X.; et al. Hyperactivation of ATF4/TGF-β1 signaling contributes to the progressive cardiac fibrosis in Arrhythmogenic cardiomyopathy caused by DSG2 Variant. BMC Med. 2024, 22, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Gong, X.; Chen, H.; Qin, S.; Zhou, N.; Su, Y.; Ge, J. Effect of Cardiac Resynchronization Therapy on Myocardial Fibrosis and Relevant Cytokines in a Canine Model With Experimental Heart Failure. J. Cardiovasc. Electrophysiol. 2017, 28, 438–445. [Google Scholar] [CrossRef] [PubMed]
- Sequeira, V.; Theisen, J.; Ermer, K.J.; Oertel, M.; Xu, A.; Weissman, D.; Ecker, K.; Dudek, J.; Fassnacht, M.; Nickel, A.; et al. Semaglutide normalizes increased cardiomyocyte calcium transients in a rat model of high fat diet-induced obesity. ESC Hear. Fail. 2024. [Google Scholar] [CrossRef]
- Uchinoumi, H.; Nakamura, Y.; Suetomi, T.; Nawata, T.; Fujinaka, M.; Kobayashi, S.; Yamamoto, T.; Yano, M.; Sano, M. Structural Instability of Ryanodine Receptor 2 Causes Endoplasmic Reticulum (ER) Dysfunction as Well as Sarcoplasmic Reticulum (SR) Dysfunction. J. Cardiol. 2025. [Google Scholar] [CrossRef]
- Bassingthwaighte, J.B. Linking Cellular Energetics to Local Flow Regulation in the Heart. Ann. New York Acad. Sci. 2008, 1123, 126–133. [Google Scholar] [CrossRef]
- Sahu, P.; Acharya, S.; Totade, M. Evolution of Pacemakers and Implantable Cardioverter Defibrillators (ICDs) in Cardiology. Cureus 2023, 15, e46389. [Google Scholar] [CrossRef]
- Nantsupawat, T.; Gumrai, P.; Apaijai, N.; Phrommintikul, A.; Prasertwitayakij, N.; Chattipakorn, S.C.; Chattipakorn, N.; Wongcharoen, W. Atrial pacing improves mitochondrial function in peripheral blood mononuclear cells in patients with cardiac implantable electronic devices. Am. J. Physiol. Circ. Physiol. 2024, 327, H1146–H1152. [Google Scholar] [CrossRef]
- Furquim, S.R.; Bocchi, E.A.; Lira, M.T.S.d.S.; Wanderley, M.R.d.B.; de Marchi, D.C.; Maciel, P.C.; Zimerman, A.; Ramires, F.J.A.; Nastari, L.; Biselli, B.; et al. Predictors of sustained reverse remodelling in patients with heart failure with reduced ejection fraction. ESC Hear. Fail. 2025. [Google Scholar] [CrossRef]
- Moraleda-Salas, M.T.; Amigo-Otero, E.; Esteve-Ruiz, I.; Arce-León, Á.; Carreño-Lineros, J.M.; Torralba, E.I.; Roldan, F.N.; Moriña-Vázquez, P. Early Improvement in Cardiac Function and Dyssynchrony After Physiological Upgrading in Pacing-Induced Cardiomyopathy. Pacing Clin. Electrophysiol. 2024. [Google Scholar] [CrossRef]
- Ladenvall, P.; Andersson, B.; Dellborg, M.; Hansson, P.-O.; Eriksson, H.; Thelle, D.; Eriksson, P. Genetic variation at the human connexin 43 locus but not at the connexin 40 locus is associated with left bundle branch block. Open Hear. 2015, 2, e000187–e000187. [Google Scholar] [CrossRef]
- Zlochiver, S. Subthreshold Parameters of Cardiac Tissue in a Bi-Layer Computer Model of Heart Failure. Cardiovasc. Eng. 2010, 10, 190–200. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



