Submitted:
13 February 2025
Posted:
17 February 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Ethical Considerations
2.3. Key Eligibility Criteria of Subjects
2.4. Randomization and Treatment Allocation
2.5. Study Procedure
2.6. Study Endpoints
2.7. Study Assessment
2.8. Statistical Analysis
3. Results
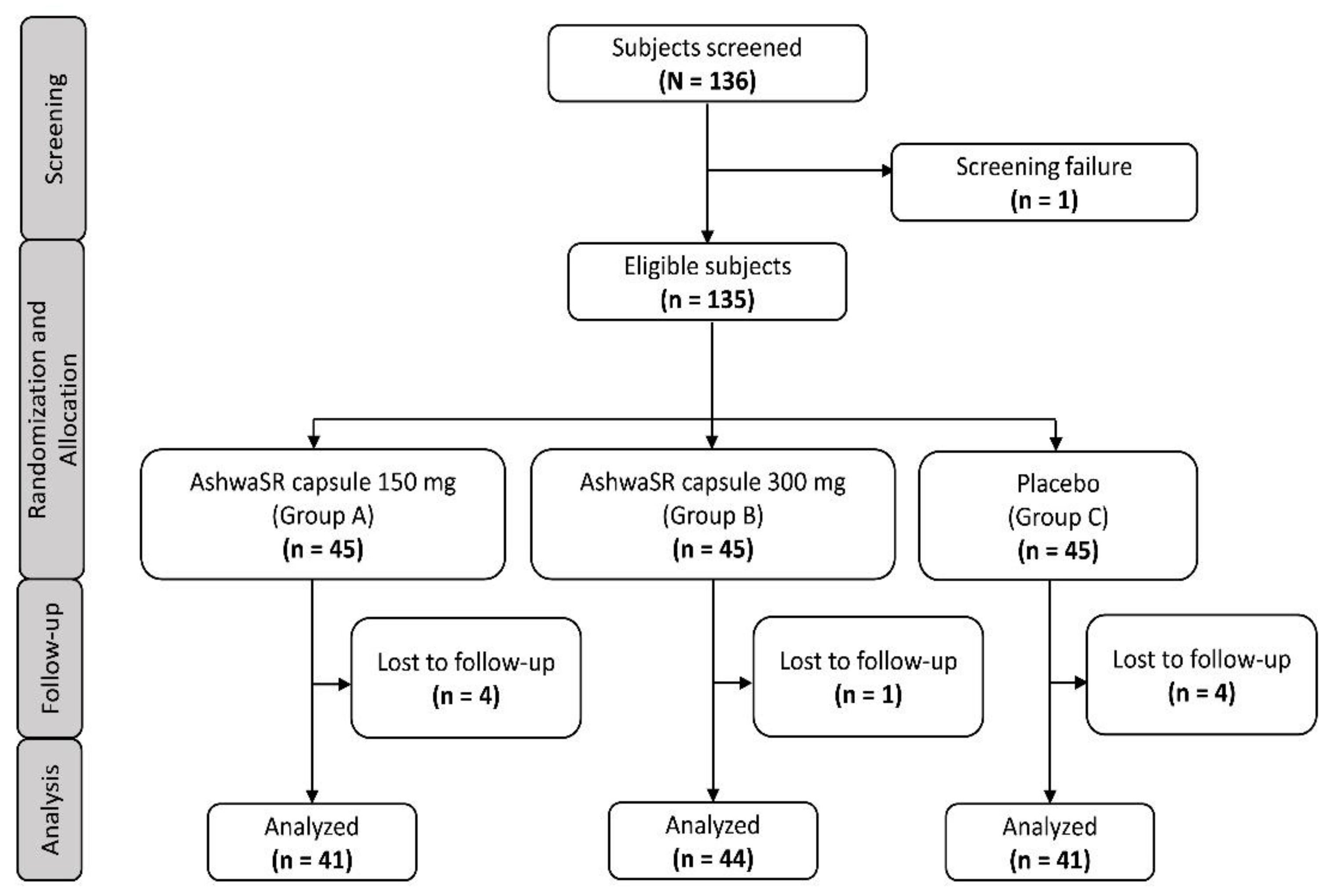
3.1. Subject Disposition and Characteristics
3.2. Primary Outcomes
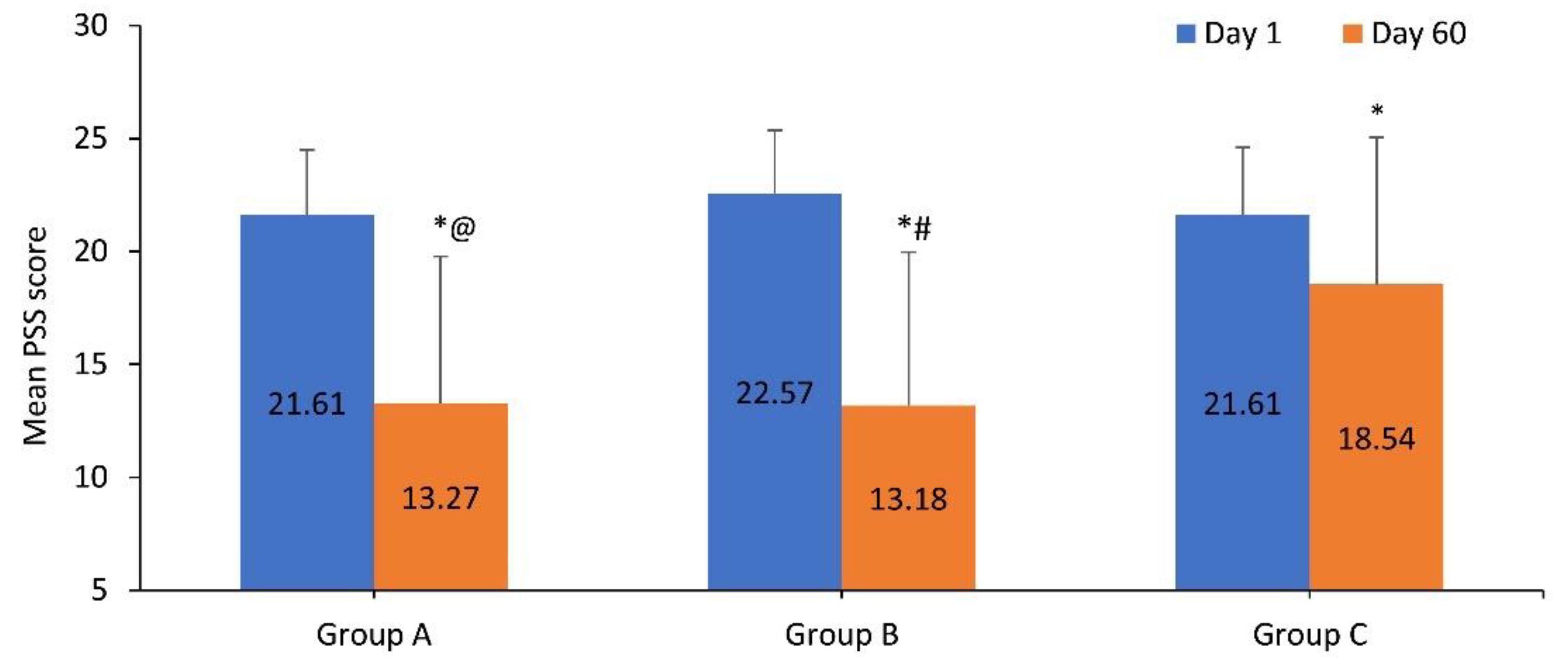
3.2.1. Change in PSS Score
3.3. Secondary Outcomes
3.3.1. Change in PSQI Global Score
3.3.2. Change in OHQ-8 Global Score
3.3.3. Change in TFEQ-R18 Global Score
3.3.4. Serum Cortisol Level
3.3.5. Subjects’ and Physicians’ Global Assessment of Therapy
3.3.6. Safety
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| SR | sustained-release |
| AshwaSR | Ashwagandha root extract SR |
| PSS | Perceived Stress Scale |
| CTRI | Clinical Trials Registry-India |
| PSQI | Pittsburgh Sleep Quality Index |
| OHQ-8 | Oxford Happiness Questionnaire-8 score |
| TFEQ-R18 | Three-Factor Eating Questionnaire- Revised 18 score |
| AEs | Adverse events |
| SD | Standard deviation |
| ANOVA | Analysis of variance |
| HSD | Honest significant difference |
References
- Chandrasekhar, K.; Kapoor, J.; Anishetty, S. A prospective, randomized double-blind, placebo-controlled study of safety and efficacy of a high-concentration full-spectrum extract of Ashwagandha root in reducing stress and anxiety in adults. Indian J. Psychol. Med. 2012, 34, 255–262. [Google Scholar] [CrossRef]
- Khan, S.I.R.; Aljammaz, G.; Alosail, L.A.; Almeshrafi, A.; Ramachandran, A.; Siddeeqh, S.; Alfadley, A. Psychological stress as a determinant of increased maximum voluntary bite force - A clinical observational study. Cureus. 2023, 15, e46106. [Google Scholar] [CrossRef]
- Ning, L.; Guan, S.; Liu, J. An investigation into psychological stress and its determinants in Xinjiang desert oil workers. Medicine 2018, 97, e0323. [Google Scholar] [CrossRef] [PubMed]
- Yau, Y.H.; Potenza, M.N. Stress and eating behaviors. Minerva Endocrinol. 2013, 38, 255–267. [Google Scholar] [PubMed]
- Mahmud, S.; Mohsin, M.; Dewan, M.N.; Muyeed, A. The global prevalence of depression, anxiety, stress, and insomnia among general population during COVID-19 pandemic: A systematic review and meta-analysis. Trends in Psychol. 2023, 31, 143–170. [Google Scholar] [CrossRef]
- Panossian, A.; Wikman, G. Effects of adaptogens on the central nervous system and the molecular mechanisms associated with their stress - Protective activity. Pharmaceuticals. 2010, 3, 188–224. [Google Scholar] [CrossRef]
- Bokelmann, J.M. Ashwagandha (Withania somnifera): Root. In J. M. Bokelmann (Ed.), Medicinal herbs in primary care. Elsevier; 2022, 195–201.
- Verma, N.; Gupta, S.K.; Tiwari, S.; Mishra, A.K. Safety of Ashwagandha root extract: A randomized, placebo-controlled, study in healthy volunteers. Complement. Ther. Med. 2021, 57, 102642. [Google Scholar] [CrossRef]
- Majeed, M.; Nagabhushanam, K.; Mundkur, L. A standardized Ashwagandha root extract alleviates stress, anxiety, and improves quality of life in healthy adults by modulating stress hormones: Results from a randomized, double-blind, placebo-controlled study. Med. 2023, 102, E35521. [Google Scholar] [CrossRef]
- Alluri, V.K.R.; Thanawala, S.; Upadhyay, V. A comparative pharmacokinetics study of Ashwagandha (Withania somnifera) Root Extract sustained-release capsules: An open-label, randomized, two treatment, two-sequence, two period, single-dose crossover clinical study. Int. J. Basic Clin. Pharmacol. 2021, 11, 26–34. [Google Scholar] [CrossRef]
- Krishnaraju, A.V.; Somepalli, V.; Thanawala, S.; Shah, R. . Efficacy and anti-inflammatory activity of Ashwagandha sustained-release formulation on depression and anxiety induced by chronic unpredictable stress: In vivo and in vitro Studies. J. Exp. Pharmacol. 2023, 15, 291–305. [Google Scholar] [CrossRef]
- Gopukumar, K.; Thanawala, S.; Somepalli, V.; Rao, T.S.S.; Thamatam, V.B.; Chauhan, S. Efficacy and safety of Ashwagandha root extract on cognitive functions in healthy, stressed adults: A randomized, double-blind, placebo-controlled study. Evidence-based Complement. Altern. Med. 2021, 2021, 8254344. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.; Kamarck, T.; Mermelstein, R. A global measure of perceived stress. J. Health. Soc. Behav. 1983, 24, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Rijal, D.; Paudel, K.; Adhikari, T.B.; Bhurtyal, A. Stress and coping strategies among higher secondary and undergraduate students during COVID-19 pandemic in Nepal. PLOS Glob. public Heal. 2023, 3, e0001533. [Google Scholar] [CrossRef] [PubMed]
- Buysse, D.J.; Reynolds, C.F. , 3rd, Monk, T.H.; Berman, S.R.; Kupfer, D.J. The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Res. 1989, 28, 193–213. [Google Scholar] [CrossRef]
- Park, B.K. The Pittsburg Sleep Quality Index (PSQI) and associated factors in middle-school Students: A Cross-sectional Study. Child Heal. Nurs. Res. 2020, 26, 55–63. [Google Scholar] [CrossRef]
- Karlsson, J.; Persson, L.O.; Sjöström, L.; Sullivan, M. Psychometric properties and factor structure of the Three-Factor Eating Questionnaire (TFEQ) in obese men and women. Results from the Swedish Obese Subjects (SOS) study. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 1715–1725. [Google Scholar] [CrossRef]
- Banna, J.C.; Panizza, C.E.; Boushey, C.J.; Delp, E.J.; Lim, E. Association between cognitive restraint, uncontrolled eating, emotional eating and BMI and the amount of food wasted in early adolescent girls. Nutrients. 2018, 10, 1279. [Google Scholar] [CrossRef]
- Hills, P.; Argyle, M. The Oxford Happiness Questionnaire: A compact scale for the measurement of psychological well-being. Pers. Individ. Dif. 2002, 33, 1071–1082. [Google Scholar] [CrossRef]
- Rao, R.; Naik, B.N.; Shekhar, S.; Nirala, S.K.; Singh, C.M.; Verma, M.; Ramalingam, A. Level of happiness among medical students in Bihar-An online survey. J. Educ. Health Promot. 2023, 12, 305. [Google Scholar] [CrossRef]
- Yaribeygi, H.; Panahi, Y.; Sahraei, H.; Johnston, T.P.; Sahebkar, A. The impact of stress on body function: A review. EXCLI J. 2017, 16, 1057–1072. [Google Scholar] [CrossRef]
- Salve, J.; Pate, S.; Debnath, K.; Langade, D. Adaptogenic and anxiolytic effects of ashwagandha root extract in healthy adults: A double-blind, randomized, placebo-controlled clinical study. Cureus. 2019, 11, e6466. [Google Scholar] [CrossRef] [PubMed]
- Eugene, A.R.; Masiak, J. The neuroprotective aspects of sleep. MEDtube. Sci. 2015, 3, 35–40. [Google Scholar] [PubMed]
- Vandekerckhove, M.; Wang, Y.L. Emotion, emotion regulation and sleep: An intimate relationship. AIMS Neurosci. 2018, 5, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Deshpande, A.; Irani, N.; Balkrishnan, R.; Benny, I.R. A randomized, double blind, placebo-controlled study to evaluate the effects of ashwagandha (Withania somnifera) extract on sleep quality in healthy adults. Sleep Med. 2020, 72, 28–36. [Google Scholar] [CrossRef]
- Hirotsu, C.; Tufik, S.; Andersen, M.L. Interactions between sleep, stress, and metabolism: From physiological to pathological conditions. Sleep Sci. 2015, 8, 143–152. [Google Scholar] [CrossRef]
- Apfeldorfer, G.; Zermati, J.P. Cognitive restraint in obesity. History of ideas, clinical description. Press medicale. 2001, 30, 1575–1580. [Google Scholar]
- Dziurkowska, E.; Wesolowski, M. Cortisol as a biomarker of mental disorder severity. J. Clin. Med. 2021, 10, 5204. [Google Scholar] [CrossRef]
- Elgar, K. Ashwagandha: A review of clinical use and efficacy. Nutr. Med. J. 2021, 1, 68–78. [Google Scholar]
- Remenapp, A.; Coyle, K.; Orange, T.; Lynch, T.; Hooper, D.; Hooper, S.; Conway, K.; Hausenblas, H.A. Efficacy of Withania somnifera supplementation on adult’s cognition and mood. J. Ayurveda Integr. Med. 2022, 13, 100510. [Google Scholar] [CrossRef]
| Parameters | Group A AshwaSR 150 mg (n = 41) |
Group B AshwaSR 300 mg (n = 44) |
Group C Placebo (n = 41) |
p-value |
|---|---|---|---|---|
| Age (years) | 34.29 ± 7.77 | 34.61 ± 8.97 | 35.49 ± 7.77 | 0.7922 |
| Body weight (kg) | 67.28 ± 10.58 | 64.65 ± 9.21 | 65.55 ± 8.57 | 0.4347 |
| Height (cm) | 164.10 ± 8.29 | 163.47 ± 7.83 | 162.18 ± 7.71 | 0.4584 |
| BMI (kg/m2) | 24.91 ± 2.85 | 24.17 ± 2.75 | 24.89 ± 2.37 | 0.3095 |
| Items | Day 1 | Day 3 | Day 7 | Day 15 | Day 30 | Day 45 | Day 60 |
|---|---|---|---|---|---|---|---|
| PSS score | |||||||
| Group A | 21.61 ± 2.89 | 20.85 ± 4.60 | 20.71 ± 4.91 | 19.63 ± 5.05* | 17.76 ± 4.32*ǂ | 16.90 ± 5.21*ǂ | 13.27 ± 6.51*ǂ |
| Group B | 22.57 ± 2.79 | 22.93 ± 3.54 | 22.11 ± 2.97 | 20.02 ± 3.59*# | 18.20 ± 3.64*# | 16.75 ± 5.34*# | 13.18 ± 6.79*# |
| Group C | 21.61 ± 2.99 | 21.76 ± 3.65 | 21.95 ± 4.22 | 21.37 ± 3.85 | 21.10 ± 3.92 | 19.78 ± 4.31* | 18.54 ± 6.51* |
| PSQI global score | |||||||
| Group A | 9.44 ± 3.35 | 9.15 ± 3.68 | 8.32 ± 3.43*ǂ | 7.56 ± 3.35*ǂ | 7.07 ± 3.58*ǂ | 6.32 ± 3.14*ǂ | 5.54 ± 3.02*ǂ |
| Group B | 9.32 ± 3.34 | 8.82 ± 3.69* | 8.07 ± 3.54*# | 7.20 ± 3.72*# | 6.25 ± 3.17*# | 5.16 ± 3.09*# | 4.52 ± 2.90*# |
| Group C | 8.17 ± 3.26 | 8.17 ± 2.92 | 8.15 ± 2.83 | 8.20 ± 2.89 | 7.98 ± 3.00 | 7.80 ± 3.27 | 7.59 ± 3.46 |
| OHQ-8 score | |||||||
| Group A | 3.14 ± 0.52ǂ | 3.26 ± 0.67 | 3.37 ± 0.62* | 3.44 ± 0.50*ǂ | 3.46 ± 0.55* | 3.63 ± 0.58*ǂ | 3.75 ± 0.60* |
| Group B | 2.93 ± 0.60# | 2.90 ± 0.60 | 3.19 ± 0.66*# | 3.32 ± 0.62*# | 3.47 ± 0.56*# | 3.72 ± 0.54*# | 4.00 ± 0.65*#$ |
| Group C | 3.47 ± 0.48 | 3.47 ± 0.36 | 3.49 ± 0.38 | 3.51 ± 0.41 | 3.55 ± 0.45 | 3.64 ± 0.50* | 3.73 ± 0.60* |
| Items | Day 1 | Day 7 | Day 30 | Day 60 |
|---|---|---|---|---|
| TFEQ-R18 - Cognitive restraint | ||||
| Group A | 13.71 ± 2.96 | 12.41 ± 2.31* | 12.02 ± 2.57*ǂ | 10.59 ± 2.77*ǂ |
| Group B | 13.93 ± 2.64 | 12.82 ± 2.69* | 11.75 ± 3.05*# | 10.93 ± 3.41*# |
| Group C | 13.00 ± 2.39 | 12.56 ± 2.52 | 12.63 ± 2.80 | 12.17 ± 3.24 |
| TFEQ-R18 - Uncontrolled eating score | ||||
| Group A | 19.20 ± 4.44 | 17.66 ± 3.95*ǂ | 16.66 ± 3.81*ǂ | 14.95 ± 3.71*ǂ |
| Group B | 18.73 ± 4.76 | 17.32 ± 4.34*# | 15.77 ± 3.55*# | 14.25 ± 3.50*# |
| Group C | 17.93 ± 3.88 | 17.95 ± 4.08 | 18.05 ± 4.40 | 17.54 ± 5.19 |
| TFEQ-R18 - Emotional eating score | ||||
| Group A | 6.37 ± 1.96 | 6.20 ± 1.74 | 5.59 ± 1.41* | 4.95 ± 1.53*ǂ |
| Group B | 6.27 ± 1.47 | 6.07 ± 1.50 | 5.43 ± 1.28* | 4.73 ± 1.34*# |
| Group C | 6.15 ± 1.73 | 6.08 ± 1.38 | 6.02 ± 1.75 | 6.02 ± 1.89 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




