Submitted:
12 March 2025
Posted:
13 March 2025
You are already at the latest version
Abstract
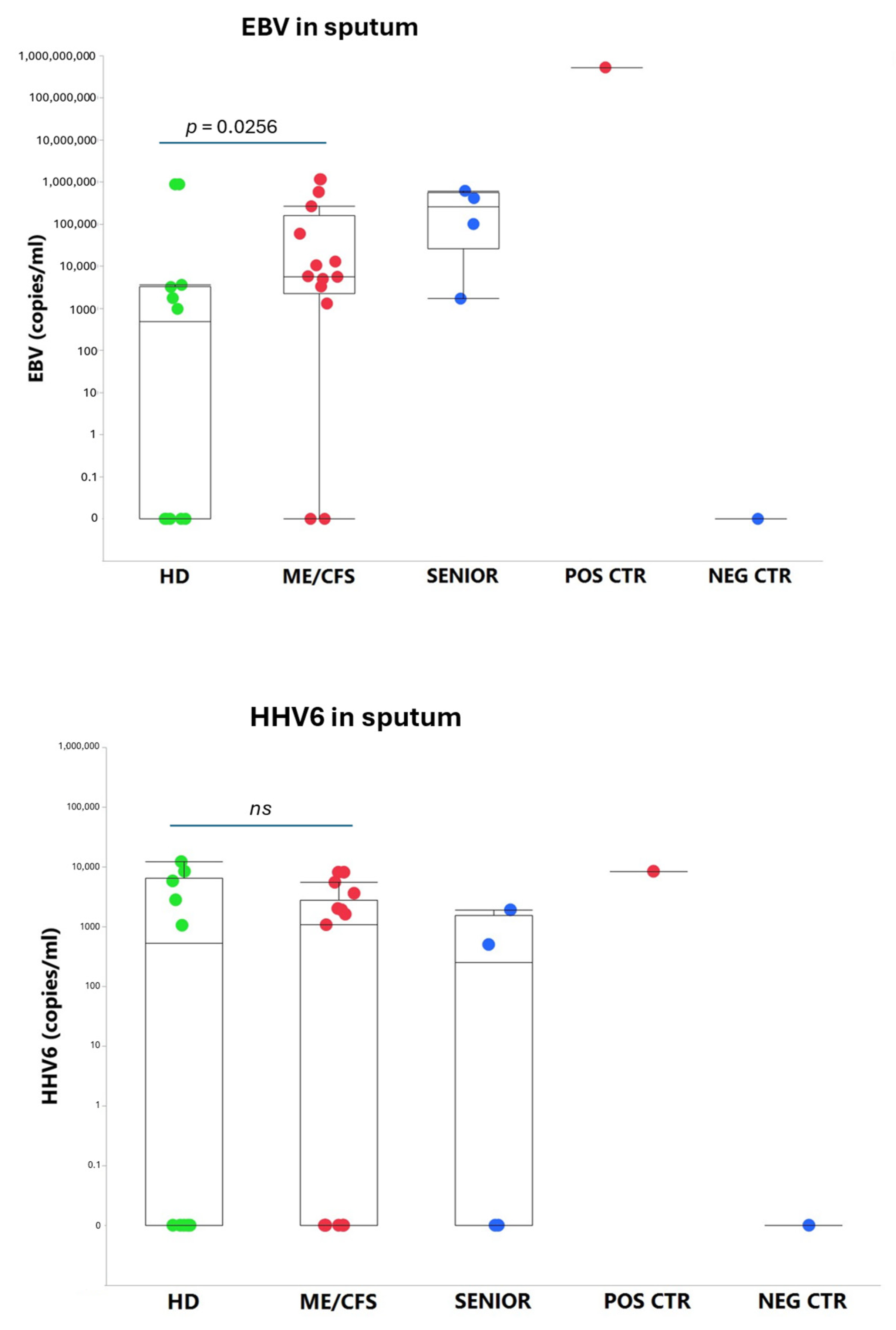
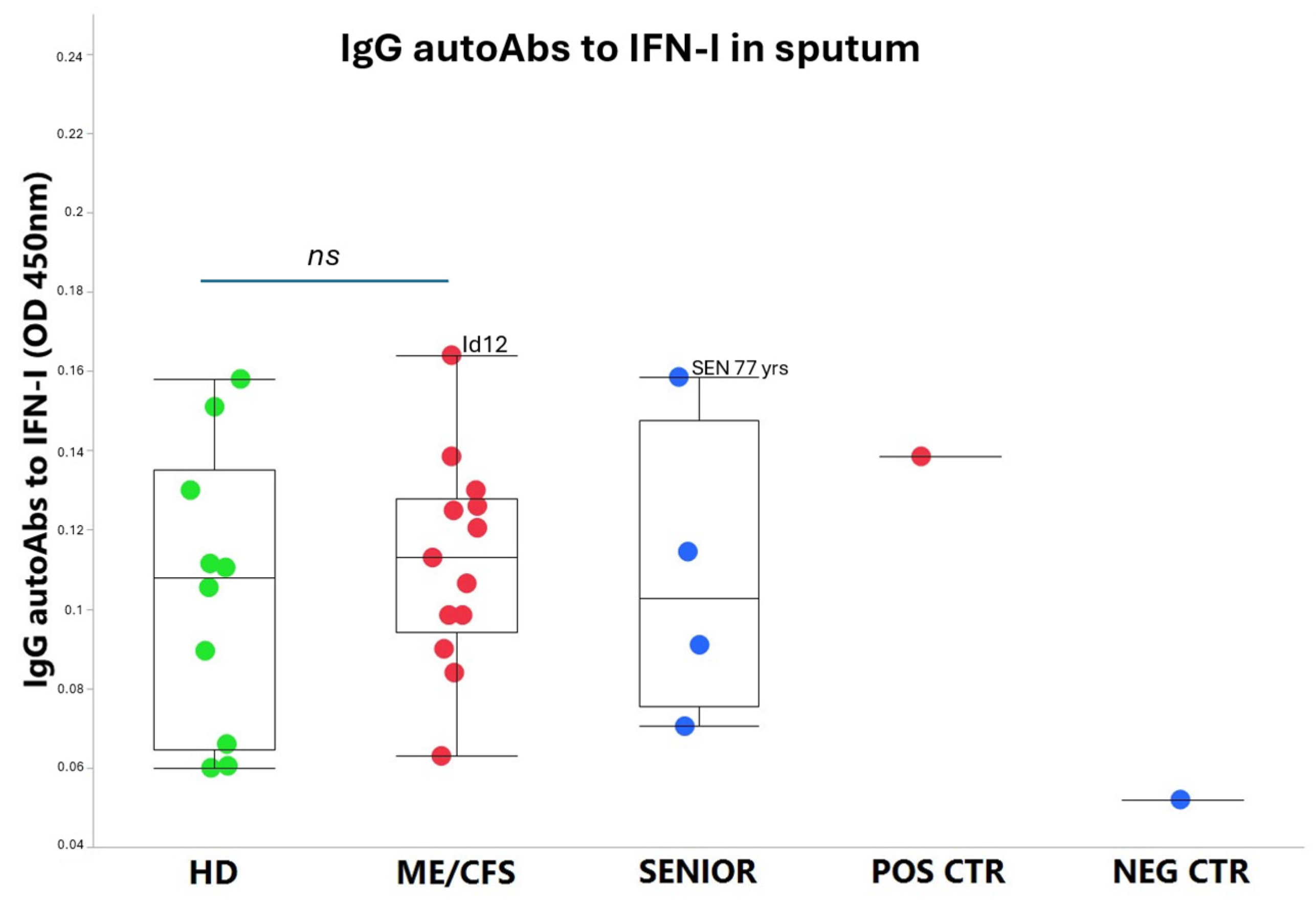
An exhausted antiviral immune response is observed in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and post-SARS-CoV-2 syndrome also termed long COVID. In this study, potential mechanisms behind this exhaustion were investigated. First, the viral load of Epstein-Barr virus (EBV), human adenovirus (HAdV), human cytomegalovirus (HCMV), human herpesvirus 6 (HHV6), and severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was determined in sputum samples (n = 29) derived from ME/CFS patients (n = 13), healthy controls (n = 10), elderly healthy controls (n = 4), and immunosuppressed controls (n = 2). Secondly, autoantibodies (autoAbs) to type I interferon (IFN-I) in sputum were analyzed to possibly explain impaired viral immunity. We found that ME/CFS patients released EBV at a significantly higher level compared to controls (p = 0.0256). HHV6 was present in ~50% of all participants at the same level. HAdV was detected in two cases with immunosuppression and severe ME/CFS, respectively. HCMV and SARS-CoV-2 were found only in immunosuppressed controls. Notably, anti-IFN-I autoAbs in ME/CFS and controls did not differ, except in a severe ME/CFS case showing an increased level. We conclude that ME/CFS patients, compared to controls, have a significantly higher load of EBV. IFN-I autoAbs cannot explain IFN-I dysfunction, with the possible exception of severe cases, also reported in severe SARS-CoV-2. We forward that additional mechanisms, such as viral evasion of IFN-I effect via degradation of IFN-receptors; may be present in ME/CFS, which demands further studies.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Ethical Permit
2.3. Sputum Collection
2.4. Analysis of EBV, HCMV, HHV6, HAdV, and SARS-CoV-2 in Sputum
2.5. ELISA Analysis of IgG autoAbs to Type-I IFN
2.6. Statistical Analyses
3. Results
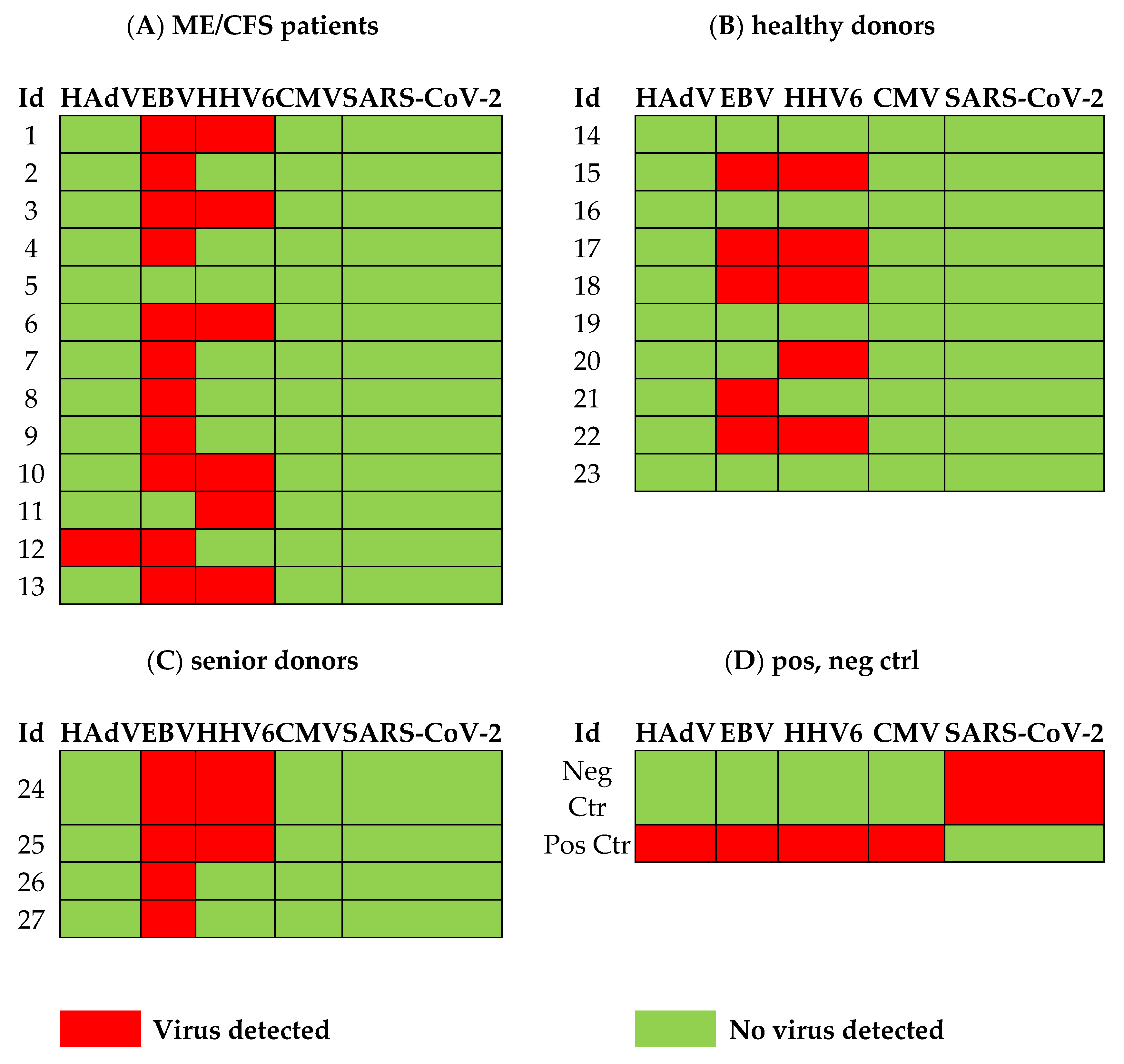
3.1. Viral Load of EBV, HCMV, HHV6, HAdV, and SARS-CoV-2 in Sputum
3.2. IgG autoAbs Against IFN-I in ME/CFS Patients
4. Discussion
4.1. ME/CFS Immune and Antiviral Dysregulation-Unknown Mechanisms
4.2. Overload of Epstein-Barr Virus in ME/CFS
4.3. Human Adenovirus and Herpesvirus Reactivation in Airways
4.4. Latent Virus—Host Immune Balance
4.5. Limitation of the Study
4.6. Future Perspectives and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vahratian, A.; Lin, J.S.; Bertolli, J.; Unger, E.R. Myalgic Encephalomyelitis/Chronic Fatigue Syndrome in Adults: United States, 2021-2022. NCHS Data Brief. 2023, 1–8. [Google Scholar]
- Valdez, A.R.; Hancock, E.E.; Adebayo, S.; Kiernicki, D.J.; Proskauer, D.; Attewell, J.R.; Bateman, L.; DeMaria, A., Jr.; Lapp, C.W.; Rowe, P.C. , et al. Estimating Prevalence, Demographics, and Costs of ME/CFS Using Large Scale Medical Claims Data and Machine Learning. Front Pediatr. 2018, 6, 412. [Google Scholar] [CrossRef] [PubMed]
- Bateman, L.; Bested, A.C.; Bonilla, H.F.; Chheda, B.V.; Chu, L.; Curtin, J.M.; Dempsey, T.T.; Dimmock, M.E.; Dowell, T.G.; Felsenstein, D. , et al. Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Essentials of Diagnosis and Management. Mayo Clin Proc. 2021, 96, 2861–2878. [Google Scholar] [CrossRef] [PubMed]
- Hanson, M.R. The viral origin of myalgic encephalomyelitis/chronic fatigue syndrome. PLoS Pathog. 2023, 19, e1011523. [Google Scholar] [CrossRef] [PubMed]
- Hickie, I.; Davenport, T.; Wakefield, D.; Vollmer-Conna, U.; Cameron, B.; Vernon, S.D.; Reeves, W.C.; Lloyd, A.; Dubbo Infection Outcomes Study, G. Post-infective and chronic fatigue syndromes precipitated by viral and non-viral pathogens: prospective cohort study. BMJ. 2006, 333, 575. [Google Scholar] [CrossRef] [PubMed]
- Naess, H.; Sundal, E.; Myhr, K.M.; Nyland, H.I. Postinfectious and chronic fatigue syndromes: clinical experience from a tertiary-referral centre in Norway. In Vivo 2010, 24, 185–188. [Google Scholar] [PubMed]
- Blomberg, J.; Gottfries, C.G.; Elfaitouri, A.; Rizwan, M.; Rosén, A. Infection Elicited Autoimmunity and Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: An Explanatory Model. Front Immunol. 2018, 9, 229. [Google Scholar] [CrossRef]
- Komaroff, A.L.; Lipkin, W.I. Insights from myalgic encephalomyelitis/chronic fatigue syndrome may help unravel the pathogenesis of postacute COVID-19 syndrome. Trends Mol Med. 2021, 27, 895–906. [Google Scholar] [CrossRef]
- Apostolou, E.; Rizwan, M.; Moustardas, P.; Sjögren, P.; Bertilson, B.C.; Bragée, B.; Polo, O.; Rosén, A. Saliva antibody-fingerprint of reactivated latent viruses after mild/asymptomatic COVID-19 is unique in patients with myalgic-encephalomyelitis/chronic fatigue syndrome. Front Immunol. 2022, 13, 949787. [Google Scholar] [CrossRef]
- Hannestad, U.; Apostolou, E.; Sjögren, P.; Bragée, B.; Polo, O.; Bertilson, B.C.; Rosén, A. Post-COVID sequelae effect in chronic fatigue syndrome: SARS-CoV-2 triggers latent adenovirus in the oral mucosa. Front Med (Lausanne). 2023, 10, 1208181. [Google Scholar] [CrossRef]
- Schindele, A.; Holm, A.; Kraft, S.; Nylander, K.; Allard, A.; Olofsson, K. Cross-evaluating Epstein-Barr virus, human papilloma virus, human cytomegalovirus and human adenovirus in nasal polyps and turbinate mucosa. Acta Otolaryngol. 2025, 145, 164–167. [Google Scholar] [PubMed]
- Wu, C.T.; Lidsky, P.V.; Xiao, Y.; Cheng, R.; Lee, I.T.; Nakayama, T.; Jiang, S.; He, W.; Demeter, J.; Knight, M.G. , et al. SARS-CoV-2 replication in airway epithelia requires motile cilia and microvillar reprogramming. Cell. 2023, 186, 112–130. [Google Scholar] [CrossRef] [PubMed]
- Vojdani, A.; Vojdani, E.; Saidara, E.; Maes, M. Persistent SARS-CoV-2 Infection, EBV, HHV-6 and Other Factors May Contribute to Inflammation and Autoimmunity in Long COVID. Viruses. 2023, 15. [Google Scholar] [CrossRef]
- Albanese, M.; Tagawa, T.; Hammerschmidt, W. Strategies of Epstein-Barr virus to evade innate antiviral immunity of its human host. Front Microbiol. 2022, 13, 955603. [Google Scholar]
- Hale, B.G. Autoantibodies targeting type I interferons: Prevalence, mechanisms of induction, and association with viral disease susceptibility. Eur J Immunol. 2023, 53, e2250164. [Google Scholar] [PubMed]
- Carruthers, B.M. Definitions and aetiology of myalgic encephalomyelitis: how the Canadian consensus clinical definition of myalgic encephalomyelitis works. J Clin Pathol. 2007, 60, 117–119. [Google Scholar] [PubMed]
- Carruthers, B.M.; van de Sande, M.I.; De Meirleir, K.L.; Klimas, N.G.; Broderick, G.; Mitchell, T.; Staines, D.; Powles, A.C.; Speight, N.; Vallings, R. , et al. Myalgic encephalomyelitis: International Consensus Criteria. J Intern Med. 2011, 270, 327–338. [Google Scholar] [PubMed]
- Hernroth, B.E.; Conden-Hansson, A.C.; Rehnstam-Holm, A.S.; Girones, R.; Allard, A.K. Environmental factors influencing human viral pathogens and their potential indicator organisms in the blue mussel, Mytilus edulis: the first Scandinavian report. Appl Environ Microbiol. 2002, 68, 4523–4533. [Google Scholar] [CrossRef] [PubMed]
- Schindele, A.; Holm, A.; Nylander, K.; Allard, A.; Olofsson, K. Mapping human papillomavirus, Epstein-Barr virus, cytomegalovirus, adenovirus, and p16 in laryngeal cancer. Discov Oncol. 2022, 13, 18. [Google Scholar]
- Corman, V.M.; Landt, O.; Kaiser, M.; Molenkamp, R.; Meijer, A.; Chu, D.K.; Bleicker, T.; Brunink, S.; Schneider, J.; Schmidt, M.L.; et al. Detection of 2019 novel coronavirus (2019-nCoV) by real-time RT-PCR. Euro Surveill 2020, 25. [Google Scholar]
- Gervais, A.; Rovida, F.; Avanzini, M.A.; Croce, S.; Marchal, A.; Lin, S.C.; Ferrari, A.; Thorball, C.W.; Constant, O.; Le Voyer, T.; et al. Autoantibodies neutralizing type I IFNs underlie West Nile virus encephalitis in approximately 40% of patients. J Exp Med 2023, 220. [Google Scholar]
- Eaton-Fitch, N.; Rudd, P.; Er, T.; Hool, L.; Herrero, L.; Marshall-Gradisnik, S. Immune exhaustion in ME/CFS and long COVID. JCI Insight. 2024, 9. [Google Scholar]
- Blomberg, J.; Rizwan, M.; Bohlin-Wiener, A.; Elfaitouri, A.; Julin, P.; Zachrisson, O.; Rosén, A.; Gottfries, C.G. Antibodies to Human Herpesviruses in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome Patients. Front Immunol. 2019, 10, 1946. [Google Scholar] [CrossRef] [PubMed]
- Apostolou, E.; Rosén, A. Epigenetic reprograming in myalgic encephalomyelitis/chronic fatigue syndrome: A narrative of latent viruses. J Intern Med. 2024, 296, 93–115. [Google Scholar] [PubMed]
- Choutka, J.; Jansari, V.; Hornig, M.; Iwasaki, A. Unexplained post-acute infection syndromes. Nat Med. 2022, 28, 911–923. [Google Scholar] [PubMed]
- Su, Y.; Yuan, D.; Chen, D.G.; Ng, R.H.; Wang, K.; Choi, J.; Li, S.; Hong, S.; Zhang, R.; Xie, J. , et al. Multiple early factors anticipate post-acute COVID-19 sequelae. Cell. 2022, 185, 881–895. [Google Scholar] [PubMed]
- Le Voyer, T.; Parent, A.V.; Liu, X.; Cederholm, A.; Gervais, A.; Rosain, J.; Nguyen, T.; Perez Lorenzo, M.; Rackaityte, E.; Rinchai, D. , et al. Autoantibodies against type I IFNs in humans with alternative NF-kappaB pathway deficiency. Nature. 2023, 623, 803–813. [Google Scholar] [PubMed]
- Bastard, P.; Vazquez, S.E.; Liu, J.; Laurie, M.T.; Wang, C.Y.; Gervais, A.; Le Voyer, T.; Bizien, L.; Zamecnik, C.; Philippot, Q. , et al. Vaccine breakthrough hypoxemic COVID-19 pneumonia in patients with auto-Abs neutralizing type I IFNs. Sci Immunol 2023, 8, eabp8966. [Google Scholar] [PubMed]
- Philippot, Q.; Fekkar, A.; Gervais, A.; Le Voyer, T.; Boers, L.S.; Conil, C.; Bizien, L.; de Brabander, J.; Duitman, J.W.; Romano, A. , et al. Autoantibodies Neutralizing Type I IFNs in the Bronchoalveolar Lavage of at Least 10% of Patients During Life-Threatening COVID-19 Pneumonia. J Clin Immunol. 2023, 43, 1093–1103. [Google Scholar] [CrossRef] [PubMed]
- Gervais, A.; Marchal, A.; Fortova, A.; Berankova, M.; Krbkova, L.; Pychova, M.; Salat, J.; Zhao, S.; Kerrouche, N.; Le Voyer, T. , et al. Autoantibodies neutralizing type I IFNs underlie severe tick-borne encephalitis in approximately 10% of patients. J Exp Med 2024, 221. [Google Scholar]
- Feng, A.; Yang, E.Y.; Moore, A.R.; Dhingra, S.; Chang, S.E.; Yin, X.; Pi, R.; Mack, E.K.; Volkel, S.; Gessner, R. , et al. Autoantibodies are highly prevalent in non-SARS-CoV-2 respiratory infections and critical illness. JCI Insight 2023, 8. [Google Scholar] [CrossRef] [PubMed]
- Walter, J.E.; Rosen, L.B.; Csomos, K.; Rosenberg, J.M.; Mathew, D.; Keszei, M.; Ujhazi, B.; Chen, K.; Lee, Y.N.; Tirosh, I. , et al. Broad-spectrum antibodies against self-antigens and cytokines in RAG deficiency. J Clin Invest. 2015, 125, 4135–4148. [Google Scholar] [PubMed]
- Ariza, M.E. Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: The Human Herpesviruses Are Back! Biomolecules. 2021, 11, 185. [Google Scholar] [CrossRef] [PubMed]
- Shikova, E.; Reshkova, V.; Kumanova capital A, C.; Raleva, S.; Alexandrova, D.; Capo, N.; Murovska, M.; On Behalf Of The European Network On Me/Cfs, E. Cytomegalovirus, Epstein-Barr virus, and human herpesvirus-6 infections in patients with myalgic small ie, Cyrillicncephalomyelitis/chronic fatigue syndrome. J Med Virol. 2020, 92, 3682–3688. [Google Scholar]
- Jeong, J.H.; Kim, K.H.; Jeong, S.H.; Park, J.W.; Lee, S.M.; Seo, Y.H. Comparison of sputum and nasopharyngeal swabs for detection of respiratory viruses. J Med Virol. 2014, 86, 2122–2127. [Google Scholar]
- Lion, T. Adenovirus persistence, reactivation, and clinical management. FEBS Lett. 2019, 593, 3571–3582. [Google Scholar] [CrossRef]
- Garnett, C.T.; Erdman, D.; Xu, W.; Gooding, L.R. Prevalence and quantitation of species C adenovirus DNA in human mucosal lymphocytes. J Virol. 2002, 76, 10608–10616. [Google Scholar] [PubMed]
- Lion, T. Adenovirus infections in immunocompetent and immunocompromised patients. Clin Microbiol Rev. 2014, 27, 441–462. [Google Scholar] [CrossRef] [PubMed]
- Kosulin, K.; Haberler, C.; Hainfellner, J.A.; Amann, G.; Lang, S.; Lion, T. Investigation of adenovirus occurrence in pediatric tumor entities. J Virol. 2007, 81, 7629–7635. [Google Scholar] [CrossRef] [PubMed]
- Virgin, H.W.; Wherry, E.J.; Ahmed, R. Redefining chronic viral infection. Cell. 2009, 138, 30–50. [Google Scholar] [CrossRef] [PubMed]
- Levin, D.; Schneider, W.M.; Hoffmann, H.H.; Yarden, G.; Busetto, A.G.; Manor, O.; Sharma, N.; Rice, C.M.; Schreiber, G. Multifaceted activities of type I interferon are revealed by a receptor antagonist. Sci Signal. 2014, 7, ra50. [Google Scholar] [CrossRef] [PubMed]
- Crow, Y.J.; Stetson, D.B. The type I interferonopathies: 10 years on. Nat Rev Immunol. 2022, 22, 471–483. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, Q.; Zheng, C.; Wang, L. MAVS: The next STING in cancers and other diseases. Crit Rev Oncol Hematol. 2024, 207, 104610. [Google Scholar] [PubMed]
- Miyagi, S.; Watanabe, T.; Hara, Y.; Arata, M.; Uddin, M.K.; Mantoku, K.; Sago, K.; Yanagi, Y.; Suzuki, T.; Masud, H. , et al. A STING inhibitor suppresses EBV-induced B cell transformation and lymphomagenesis. Cancer Sci. 2021, 112, 5088–5099. [Google Scholar] [CrossRef]
- Pasquesi, G.I.M.; Allen, H.; Ivancevic, A.; Barbachano-Guerrero, A.; Joyner, O.; Guo, K.; Simpson, D.M.; Gapin, K.; Horton, I.; Nguyen, L.L. , et al. Regulation of human interferon signaling by transposon exonization. Cell. 2024, 187, 7621–7636.e7619. [Google Scholar] [CrossRef] [PubMed]
- Peluso, M.J.; Deeks, S.G. Mechanisms of long COVID and the path toward therapeutics. Cell. 2024, 187, 5500–5529. [Google Scholar] [CrossRef] [PubMed]
- Kosulin, K.; Geiger, E.; Vecsei, A.; Huber, W.D.; Rauch, M.; Brenner, E.; Wrba, F.; Hammer, K.; Innerhofer, A.; Potschger, U. , et al. Persistence and reactivation of human adenoviruses in the gastrointestinal tract. Clin Microbiol Infect. 2016, 22, 381.e381–381.e388. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Cardona, J.J.; Whited, L.K.; Chemaly, R.F. Brincidofovir: understanding its unique profile and potential role against adenovirus and other viral infections. Future Microbiol. 2020, 15, 389–400. [Google Scholar] [CrossRef] [PubMed]
- El-Haddad, D.; El Chaer, F.; Vanichanan, J.; Shah, D.P.; Ariza-Heredia, E.J.; Mulanovich, V.E.; Gulbis, A.M.; Shpall, E.J.; Chemaly, R.F. Brincidofovir (CMX-001) for refractory and resistant CMV and HSV infections in immunocompromised cancer patients: A single-center experience. Antiviral Res. 2016, 134, 58–62. [Google Scholar] [CrossRef] [PubMed]
- Camargo, J.F.; Morris, M.I.; Abbo, L.M.; Simkins, J.; Saneeymehri, S.; Alencar, M.C.; Lekakis, L.J.; Komanduri, K.V. The use of brincidofovir for the treatment of mixed dsDNA viral infection. J Clin Virol. 2016, 83, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Hill, J.A.; Nichols, W.G.; Marty, F.M.; Papanicolaou, G.A.; Brundage, T.M.; Lanier, R.; Zerr, D.M.; Boeckh, M.J. Oral brincidofovir decreases the incidence of HHV-6B viremia after allogeneic HCT. Blood. 2020, 135, 1447–1451. [Google Scholar] [CrossRef] [PubMed]
- Grimley, M.S.; Maron, G. , editors. Preliminary Results of a Phase 2a Clinical Trial to Evaluate Safety, Tolerability and Antiviral Activity of Intravenous Brincidofovir (BCV IV) in Immunocompromised Patients with Adenovirus Infection. 65th ASH Annual Meeting; 2023; San Diego, CA: Blood.
- Kosulin, K. Intestinal HAdV Infection: Tissue Specificity, Persistence, and Implications for Antiviral Therapy. Viruses. 2019, 11. [Google Scholar] [CrossRef]
- Radke, J.R.; Cook, J.L. Human adenovirus infections: update and consideration of mechanisms of viral persistence. Curr Opin Infect Dis. 2018, 31, 251–256. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Calcedo, R.; Medina-Jaszek, A.; Keough, M.; Peng, H.; Wilson, J.M. Adenoviruses in lymphocytes of the human gastro-intestinal tract. PLoS One. 2011, 6, e24859. [Google Scholar] [CrossRef]
| ME/CFS Patients | Healthy Controls | Senior Controls | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample | Duration of | Disease | Sex | Age | Sample | Duration of | Sex | Age | Sample | Duration of | Sex | Age |
| Id | ME/CFS (yrs) | Severity | yrs | Id | ME/CFS (yrs) | yrs | Id | ME/CFS (yrs) | yrs | |||
| 1 | 8 | 1 | F | 61 | 14 | NA | F | 56 | 24 | NA | F | 65 |
| 2 | 13 | 2 | F | 60 | 15 | NA | F | 33 | 25 | NA | M | 77 |
| 3 | 10 | 2 | F | 58 | 16 | NA | F | 61 | 26 | NA | F | 72 |
| 4 | 12 | 1 | F | 56 | 17 | NA | F | 61 | 27 | NA | M | 75 |
| 5 | 28 | 1 | F | 54 | 18 | NA | F | 37 | Median | 73.5 | ||
| 6 | 12 | 1 | F | 54 | 19 | NA | F | 49 | Range | 65–77 | ||
| 7 | 10 | 1 | F | 53 | 20 | NA | F | 66 | ||||
| 8 | 14 | 1 | F | 49 | 21 | NA | M | 48 | ||||
| 9 | 14 | 2 | F | 48 | 22 | NA | F | 33 | Positive, negative control | |||
| 10 | 8 | 1 | F | 37 | 23 | NA | F | 66 | Sample | Duration of | Sex | Age |
| 11 | 17 | 2 | F | 37 | Median | 54 | Id | ME/CFS (yrs) | yrs | |||
| 12 | 12 | 3 | F | 22 | Range | 33–66 | NEG CTR | NA | F | 46 | ||
| 13 | 27 | 1 | F | 59 | POS CTR | NA | F | 54 | ||||
| Median | 12 | 52.5 | ||||||||||
| Range | 8- 28 | 22–61 | ||||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




