Submitted:
14 April 2026
Posted:
15 April 2026
You are already at the latest version
Abstract
Keywords:
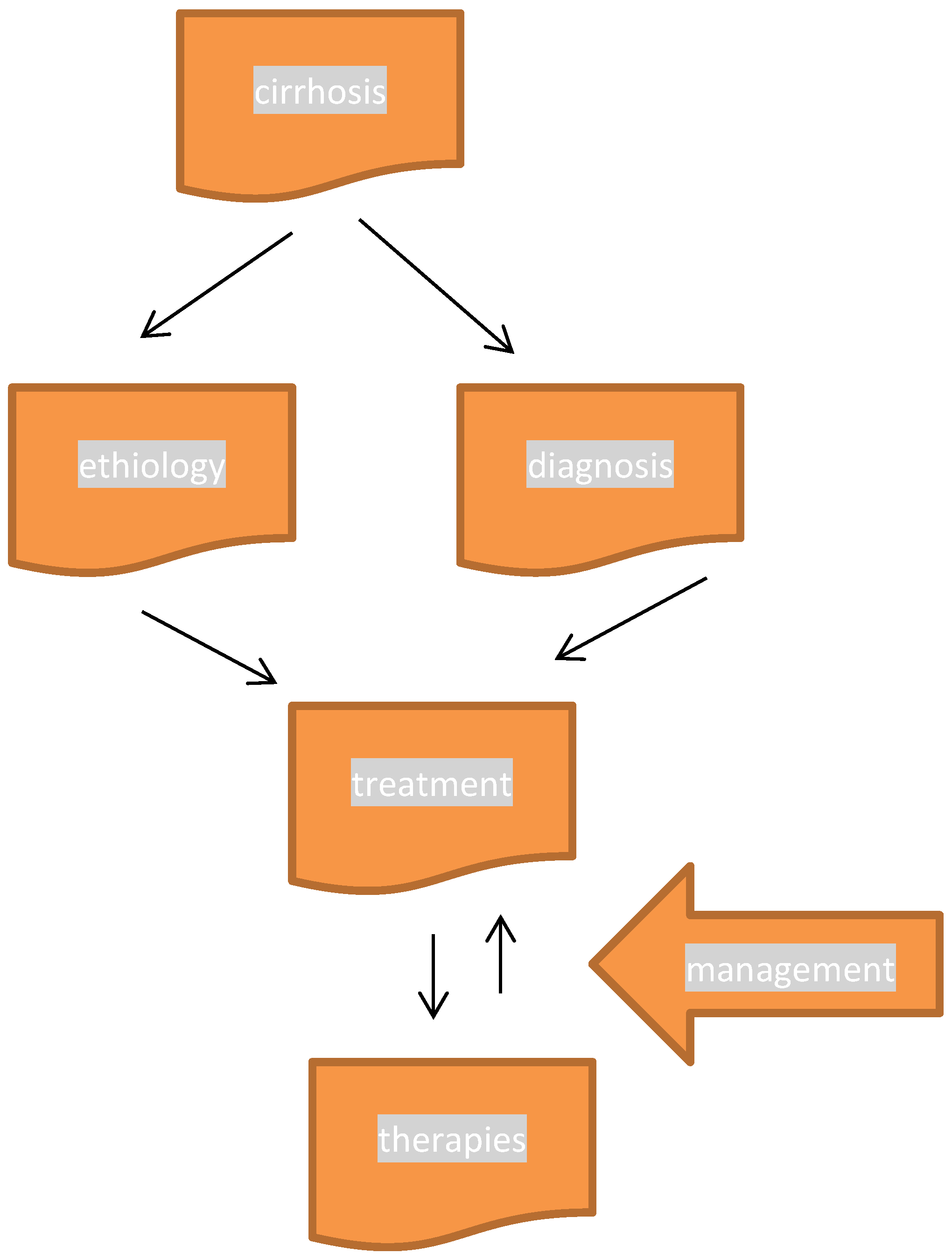
1. Introduction
2. Historical Key Points on P450
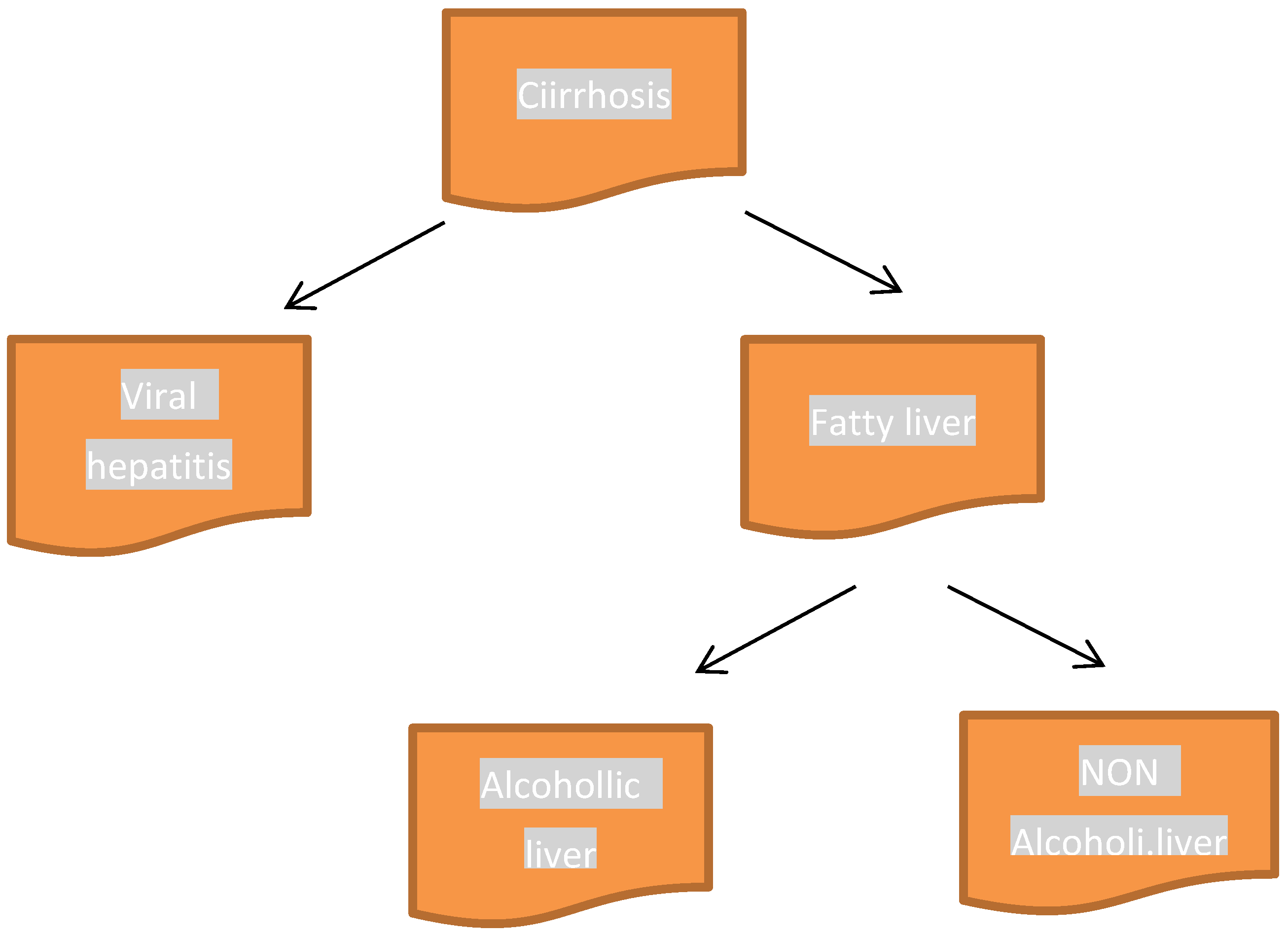
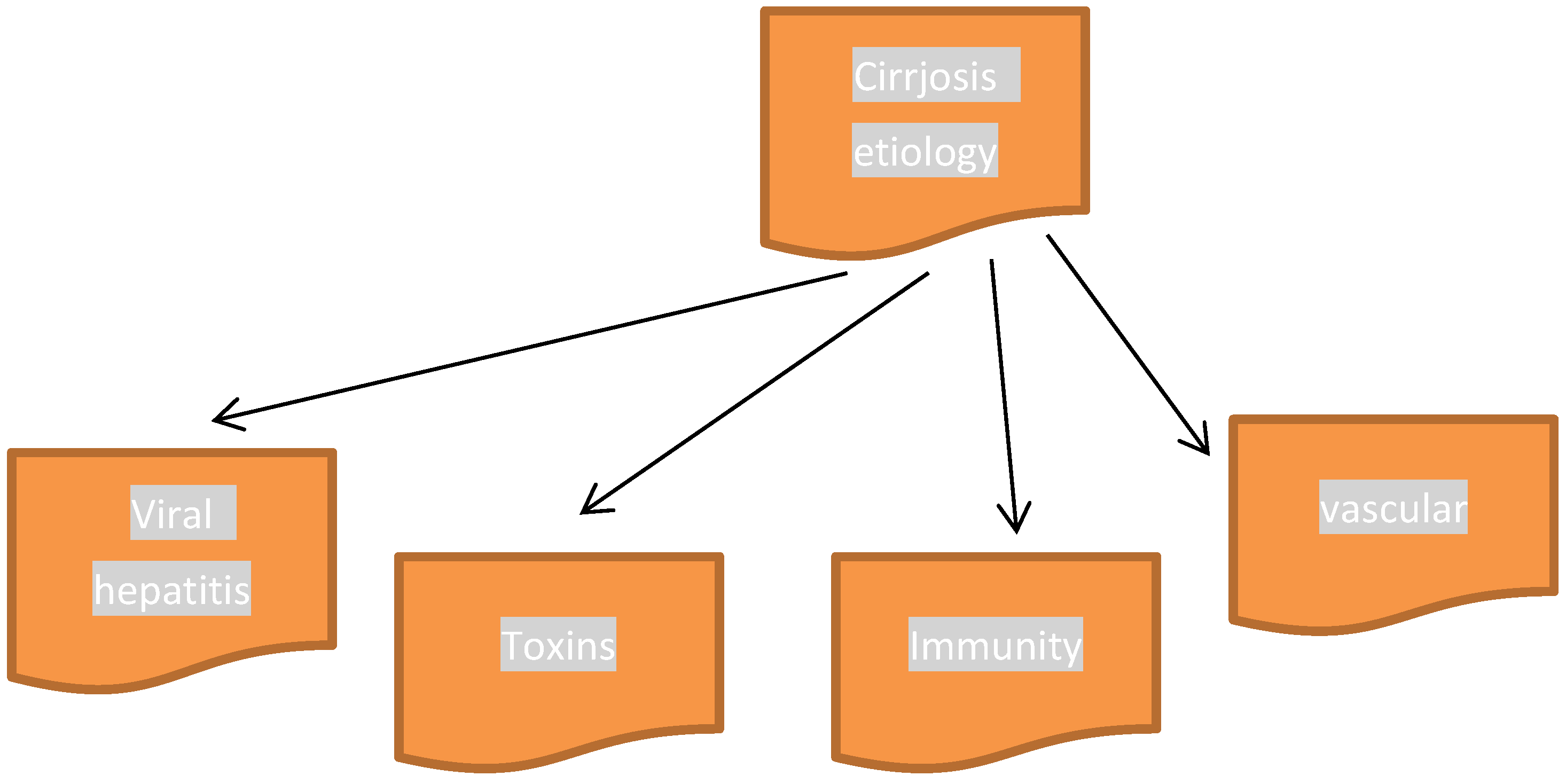
3. Cirrhosis and CYP Isoenzymes
4. Paraclinic Key Points in Cirrhosis Diagnosis
5. Complications in Cirrhosys and Trends in Management Reffering to Diagnosis
6. Conclusions
References
- Kleinbloesem, C.H.; van Harten, J.; Wilson, J.P.; Danhof, M.; van Brummelen, P.; Breimer, D.D. Nifedipine: kinetics and hemodynamic effects in patients with liver cirrhosis after intravenous and oral administration. Clin Pharmacol Ther. 1986, 40, 21–8. [Google Scholar] [CrossRef]
- Morgan, D.J.; McLean, A.J. Clinical pharmacokinetic and pharmacodynamic considerations in patients with liver disease. An update. Clin Pharmacokinet. 1995, 29, 370–91. [Google Scholar] [CrossRef]
- Lamba, J.K.; Lin, Y.S.; Schuetz, E.G.; Thummel, K.E. Genetic contribution to variable human CYP3A-mediated metabolism. Advanced Drug Delivery Reviews 2002, 54, 1271–94. [Google Scholar] [CrossRef]
- George, J.; Liddle, C.; Murray, M.; Byth, K.; Farrell, G.C. Pre-translational regulation of cytochrome P450 genes is responsible for disease-specific changes of individual P450 enzymes among patients with cirrhosis. Biochem Pharmacol. 1995, 49, 873–81. [Google Scholar] [CrossRef]
- Guengerich, F.P.; Turvy, C.G. Comparison of levels of several human microsomal cytochrome P-450 enzymes and epoxide hydrolase in normal and disease states using immunochemical analysis of surgical liver samples. J Pharmacol Exp Ther. 1991, 256, 1189–94. [Google Scholar] [CrossRef] [PubMed]
- Lown, K.; Kolars, J.; Turgeon, K.; Merion, R.; Wrighton, S.A.; Watkins, P.B. The erythromycin breath test selectively measures P450IIIA in patients with severe liver disease. Clin Pharmacol Ther. 1992, 51, 229–38. [Google Scholar] [CrossRef] [PubMed]
- Kleinbloesem, C.H.; van Brummelen, P.; Faber, H.; Danhof, M.; Vermeulen, N.P.; Breimer, D.D. Variability in nifedipine pharmacokinetics and dynamics: a new oxidation polymorphism in man. Biochem Pharmacol. 1984, 33(22), 3721–3724. [Google Scholar] [CrossRef]
- Kleinbloesem, C.H.; Van Harten, J.; Van Brummelen, P.; Breimer, D.D. Liquid chromatographic determination of nifedipine in plasma and of its main metabolite in urine. J Chromatogr. 1984, 308, 209–216. [Google Scholar] [CrossRef] [PubMed]
- van Harten, J.; van Brummelen, P.; Wilson, J.H.; Lodewijks, M.T.; Breimer, D.D. Nisoldipine: kinetics and effects on blood pressure and heart rate in patients with liver cirrhosis after intravenous and oral administration. Eur J Clin Pharmacol. 1988, 34, 387–94. [Google Scholar] [CrossRef]
- Guengerich, F. Peter, A history of the roles of cytochrome P450 enzymes in the toxicity of drugs. Toxicol Res 2020, 37(1), 1–23. [Google Scholar] [CrossRef]
- Borzelleca, J.F. Profiles in toxicology–Paracelsus: herald of modern toxicology. Toxicol Sci. 2000, 53, 2–4. [Google Scholar] [CrossRef]
- Bachmann, C.; Bickel, M.H. History of drug metabolism: the first half of the 20th century. Drug Metab Rev. 1985, 16, 185–253. [Google Scholar] [CrossRef]
- Guengerich, F.P. Introduction and historical perspective. In Biotransformation; Comprehensive toxicology (McQueen CA, Series ed) 3; Guengerich, F.P., Ed.; Elsevier: Oxford, 2018; vol 10, pp. 1–7. [Google Scholar]
- Garfinkel, D. Studies on pig liver microsomes. I. Enzymic and pigment composition of different microsomal fractions. Arch Biochem Biophys. 1958, 77, 493–509. [Google Scholar] [CrossRef]
- Klingenberg, M. Pigments of rat liver microsomes. Arch Biochem Biophys. 1958, 75, 376–386. [Google Scholar] [CrossRef]
- Testa, B.; Jenner, P. Drug metabolism: chemical and biochemical aspects; Marcel Dekker: New York, 1976. [Google Scholar]
- Oxford Textbook of Clinical Hepatology., 2nd Ed.; Bircher, J., Benhamou, J.P., McIntyre, N., Rizzetto, M., Rodes, J., Eds.; Oxford University Press, 1999. [Google Scholar]
- Sherlock, S.; Dooley, J. (Eds.) Diseases of the Liver and Biliary System., 11th Ed.; Blackwell Science: Oxford, UK; Malden, MA, 2002. [Google Scholar]
- Schiff, E.R.; Sorrell, M.F.; Maddrey, E.C. (Eds.) Schiff’s Diseases of the Liver., 9th Ed.; Lippincott, Williams & Wilkins: Philadelphia, 2003. [Google Scholar]
- Desmet, V.J.; Roskams, T. Cirrhosis reversal: a duel between dogma and myth. J Hepatol. 2004, 40, 860–7. [Google Scholar] [CrossRef]
- Wanless, I.R.; Nakashima, E.; Sherman, M. Regression of human cirrhosis. Morphologic features and the genesis of incomplete septal cirrhosis. Arch Pathol Lab Med. 2000, 124, 1599–607. [Google Scholar] [CrossRef]
- Poynard, T.; Bedossa, P.; Opolon, P. Natural history of liver fibrosis progression in patients with chronic hepatitis C. The OBSVIRC, METAVIR, CLINIVIR, and DOSVIRC groups. Lancet 1997, 349, 825–32. [Google Scholar] [CrossRef] [PubMed]
- Bellentani, S.; Pozzato, G.; Saccoccio, G.; et al. Clinical course and risk factors of hepatitis C virus related liver disease in the general population: report from the Dionysos study. Gut 1999, 44, 874–80. [Google Scholar] [CrossRef] [PubMed]
- Bellentani, S.; Saccoccio, G.; Costa, G. The Dionysos Study Group Drinking habits as cofactors of risk for alcohol induced liver damage. Gut 1997, 41, 845–50. [Google Scholar] [CrossRef] [PubMed]
- Fracanzani, A.L.; Fargion, S.; Romano, R.; et al. Portal hypertension and iron depletion in patients with genetic hemochromatosis. Hepatology 1995, 22, 1127–31. [Google Scholar] [CrossRef]
- Conn, H.; Atterbury, C. Cirrhosis. In Diseases of the Liver., 7th ed.; Schiff, L., Schiff, E., Eds.; Lippencott Company: Philadelphia; Philadelphia, 1993; pp. 875–934. [Google Scholar]
- Dienstag, J.L.; Goldin, R.D.; Heathcote, E.J.; et al. Histological outcome during long-term lamivudine therapy. Gastroenterology 2003, 124, 105–17. [Google Scholar] [CrossRef] [PubMed]
- Everson, G.T. Management of cirrhosis due to chronic hepatitis C. J Hepatol. 2005, 42 (Suppl1), S65–74. [Google Scholar] [CrossRef] [PubMed]
- Lok, A.S.; McMahon, B.J. Practice Guidelines Committee, American Association for the Study of Liver Diseases (AASLD). Chronic hepatitis B: update of recommendations. Hepatology 2004, 39, 857–61. 23. [Google Scholar] [CrossRef]
- Van Thiel, D.H.; Gavaler, J.S.; Schade, R.R. Liver disease and the hypothalamic pituitary gonadal axis. Semin Liver Dis. 1985, 5, 35–45. 24. [Google Scholar] [CrossRef]
- Tangerman, A.; Meuwese-Arends, M.T.; Jansen, J.B. Cause and composition of foetor hepaticus. Lancet 1994, 343, 483. [Google Scholar] [CrossRef]
- Di Lelio, A.; Cestari, C.; Lomazzi, A.; Beretta, L. Cirrhosis: Diagnosis with sonographic study of the liver surface. Radiology 1989, 172, 389–92. [Google Scholar] [CrossRef] [PubMed]
- Tchelepi, H.; Ralls, P.W.; Radin, R.; Grant, E. Sonography of diffuse liver disease. J Ultrasound Med. 2002, 21, 1023–32. [Google Scholar] [CrossRef] [PubMed]
- Jansen, C.; Chatterjee, D.A.; Thomsen, K.L.; Al-Kassou, B.; Sawhney, R.; Jones, H.; et al. Significant reduction in heart rate variability is a feature of acute decompensation of cirrhosis and predicts 90-day mortality. Aliment Pharmacol Ther. 2019, 50, 568–579. [Google Scholar] [CrossRef]
- Borzelleca, J.F. Profiles in toxicology–Paracelsus: herald of modern toxicology. Toxicol Sci. 2000, 53, 2–4. [Google Scholar] [CrossRef]
- Bachmann, C.; Bickel, M.H. History of drug metabolism: the first half of the 20th century. Drug Metab Rev. 1985, 16, 185–253. [Google Scholar] [CrossRef] [PubMed]
- Guengerich, F.P. Introduction and historical perspective. In Biotransformation; Comprehensive toxicology (McQueen CA, Series ed) 3; Guengerich, F.P., Ed.; Elsevier: Oxford, 2018; vol 10, pp. 1–7. [Google Scholar]
- Garfinkel, D. Studies on pig liver microsomes. I. Enzymic and pigment composition of different microsomal fractions. Arch Biochem Biophys. 1958, 77, 493–509. [Google Scholar] [CrossRef]
- Klingenberg, M. Pigments of rat liver microsomes. Arch Biochem Biophys. 1958, 75, 376–386. [Google Scholar] [CrossRef]
- Testa, B.; Jenner, P. Drug metabolism: chemical and biochemical aspects; Marcel Dekker: New York, 1976. [Google Scholar]
- Casafont Morencos, F.; de las Heras Castaño, G.; Martín Ramos, L.; López Arias, M.J.; Ledesma, F.; Pons Romero, F. Small bowel bacterial overgrowth in patients with alcoholic cirrhosis. Dig Dis Sci 1996, 41(3), 552–6. [Google Scholar] [CrossRef] [PubMed]
- Sheen, I.S.; Liaw, Y.F. The prevalence and incidence of cholecystolithiasis in patients with chronic liver diseases: a prospective study. Hepatology 1989, 9(4), 538–40. [Google Scholar] [CrossRef] [PubMed]
- John, S.; Thuluvath, P.J. Hyponatremia in cirrhosis: pathophysiology and management. World J Gastroenterol 2015, 21(11), 3197–205. [Google Scholar] [CrossRef]
- Lata, J. Hepatorenal syndrome. World J Gastroenterol 2012, 18(36), 4978–84. [Google Scholar] [CrossRef] [PubMed]
- Green, G.R. Mechanism of hypogonadism in cirrhotic males. Gut 1977, 18(10), 843–53. [Google Scholar] [CrossRef]
- Moon, A.M.; Singal, A.G.; Tapper, E.B. Contemporary epidemiology of chronic liver disease and cirrhosis. Clin Gastroenterol Hepatol. 2020, 18, 2650–2666. [Google Scholar] [CrossRef] [PubMed]
- Trebicka, J.; Fernandez, J.; Papp, M.; Caraceni, P.; Laleman, W.; Gambino, C.; et al. The PREDICT study uncovers three clinical courses of acutely decompensated cirrhosis that have distinct pathophysiology. J Hepatol. 2020, 73, 842–854. [Google Scholar] [CrossRef]
- Tapper, E.B.; Halbert, B.; Mellinger, J. Rates of and reasons for hospital readmissions in patients with cirrhosis: a multistate population-based cohort study. Clin Gastroenterol Hepatol. 2016, 14, 1181–8 e2. [Google Scholar] [CrossRef]
- Bajaj, J.S.; Reddy, K.R.; Tandon, P.; Wong, F.; Kamath, P.S.; Garcia-Tsao, G.; et al. The 3-month readmission rate remains unacceptably high in a large North American cohort of patients with cirrhosis. Hepatology. 2016, 64, 200–208. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver EASL Clinical Practice Guidelinesfor the management of patients with decompensated cirrhosis. Hepatol. 2018, 69, 406–460. [CrossRef] [PubMed]
- Macdonald, S.; Jepsen, P.; Alrubaiy, L.; Watson, H.; Vilstrup, H.; Jalan, R. Quality of life measures predict mortality in patients with cirrhosis and severe ascites. Aliment Pharmacol Ther. 2019, 49, 321–330. [Google Scholar] [CrossRef] [PubMed]
- Häussinger, D.; Dhiman, R.K.; Felipo, V.; Görg, B.; Jalan, R.; Kircheis, G.; Merli, M.; Montagnese, S.; Romero-Gomez, M.; Schnitzler, A.; Taylor-Robinson, S.D.; Vilstrup, H. Hepatic encephalopathy. Nat Rev Dis Primers 2022, 8, 43. [Google Scholar] [CrossRef] [PubMed]
- Ridola, L.; Faccioli, J.; Nardelli, S.; Gioia, S.; Riggio, O. Hepatic Encephalopathy: Diagnosis and Management. J Transl Int Med. 2020, 8, 210–219. [Google Scholar] [CrossRef]
- European Association for the Study of the Liver. EASL clinical practice guidelines on the management of ascites, spontaneous bacterial peritonitis, and hepatorenal syndrome in cirrhosis. J Hepatol. 2010, 53, 397–417. [Google Scholar] [CrossRef]
- Gravito-Soares, M.; Gravito-Soares, E.; Lopes, S.; Ribeiro, G.; Figueiredo, P. Spontaneous fungal peritonitis: a rare but severe complication of liver cirrhosis. Eur J Gastroenterol Hepatol. 2017, 29, 1010–1016. [Google Scholar] [CrossRef]
- Fiore, M.; Chiodini, P.; Pota, V.; Sansone, P.; Passavanti, M.B.; Leone, S.; Aurilio, C.; Pace, M.C. Risk of spontaneous fungal peritonitis in hospitalized cirrhotic patients with ascites: a systematic review of observational studies and meta-analysis. Minerva Anestesiol. 2017, 83, 1309–1316. [Google Scholar] [CrossRef]
- Alqahtani, S.A.; Jang, S. Pathophysiology and Management of Variceal Bleeding 2021, 81, 647–667, Baiges.
- Bachmann, C.; Bickel, M.H. History of drug metabolism: the first half of the 20th century. Drug Metab Rev. 1985, 16, 185–253. [Google Scholar] [CrossRef]
- Guengerich, F.P. Introduction and historical perspective. In Biotransformation; Comprehensive toxicology (McQueen CA, Series ed) 3; Guengerich, F.P., Ed.; Elsevier: Oxford, 2018; vol 10, pp. 1–7. [Google Scholar]
- Garfinkel, D. Studies on pig liver microsomes. I. Enzymic and pigment composition of different microsomal fractions. Arch Biochem Biophys. 1958, 77, 493–509. [Google Scholar] [CrossRef] [PubMed]
- Klingenberg, M. Pigments of rat liver microsomes. Arch Biochem Biophys. 1958, 75, 376–386. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



