Submitted:
21 December 2024
Posted:
23 December 2024
You are already at the latest version
Abstract
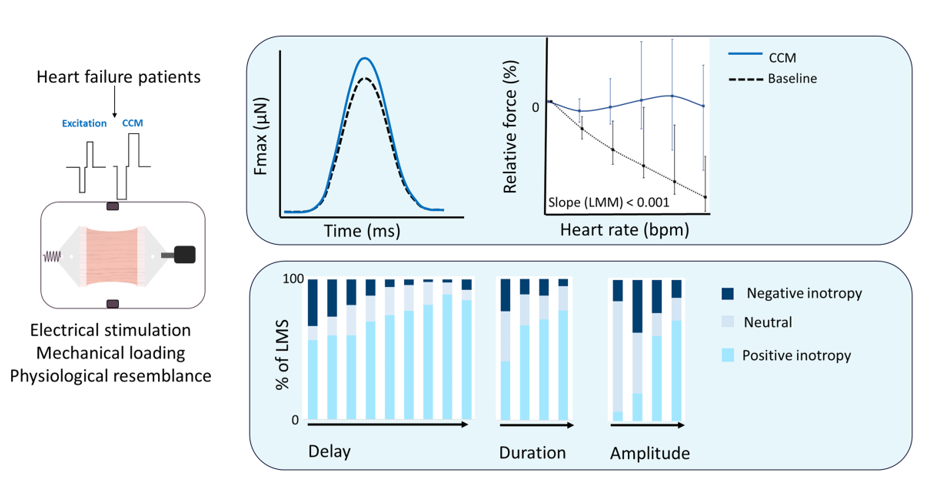
1.1 Aims: Proof-of-concept to determine the direct biomechanical effects of cardiac contractility modulation (CCM) on living myocardial slices (LMS) from patients with end-stage heart failure (HF). 1.2 Methods and results: Left ventricular LMS from patients with end-stage HF were produced and cultured in a biomimetic system with mechanical loading and electrical stimulation. CCM stimulation (80 mA, 40 ms delay, 21 ms duration) enhanced maximum contractile force (CCM: 1229 µN (587 – 2658) vs. baseline: 1066 µN (529 – 2128), p = 0.05) and area under the contractile curve (CCM: 297 (151 – 562) vs. baseline: 243 (129 – 464), p = 0.05), but did not significantly impact contractile duration, time to peak or time to relaxation. Increasing CCM stimulation delay, duration and amplitude resulted in a higher fraction of LMS with a positive inotropic response. Furthermore, CCM attenuated the negative force-frequency relationship in HF-LMS. 1.3 Conclusion: CCM stimulation enhanced contractile force in HF-LMS. The fraction of LMS exerting a positive inotropic response to CCM increased with increasing delay, duration and amplitude settings, suggesting that personalizing stimulation parameters could optimize the beneficial effects of CCM. 1.4 Translational perspective CCM is a novel device-based therapy that may improve contractile function, ejection fraction, functional outcomes and quality of life in patients with heart failure. However, continuous efforts are needed to identify true responders to CCM therapy, understand the exact mechanisms and to optimize the contractile response to CCM stimulation. The present study revealed that CCM enhanced contractile force of HF-LMS in a stimulation setting dependent manner, reaching a larger fraction of the myocardium while increasing delay, duration and amplitude. This understanding may contribute to the individualization of CCM stimulation settings.
Keywords:
1. Introduction
2. Methods
2.1. Slice Preparation
2.2. Slice Cultivation
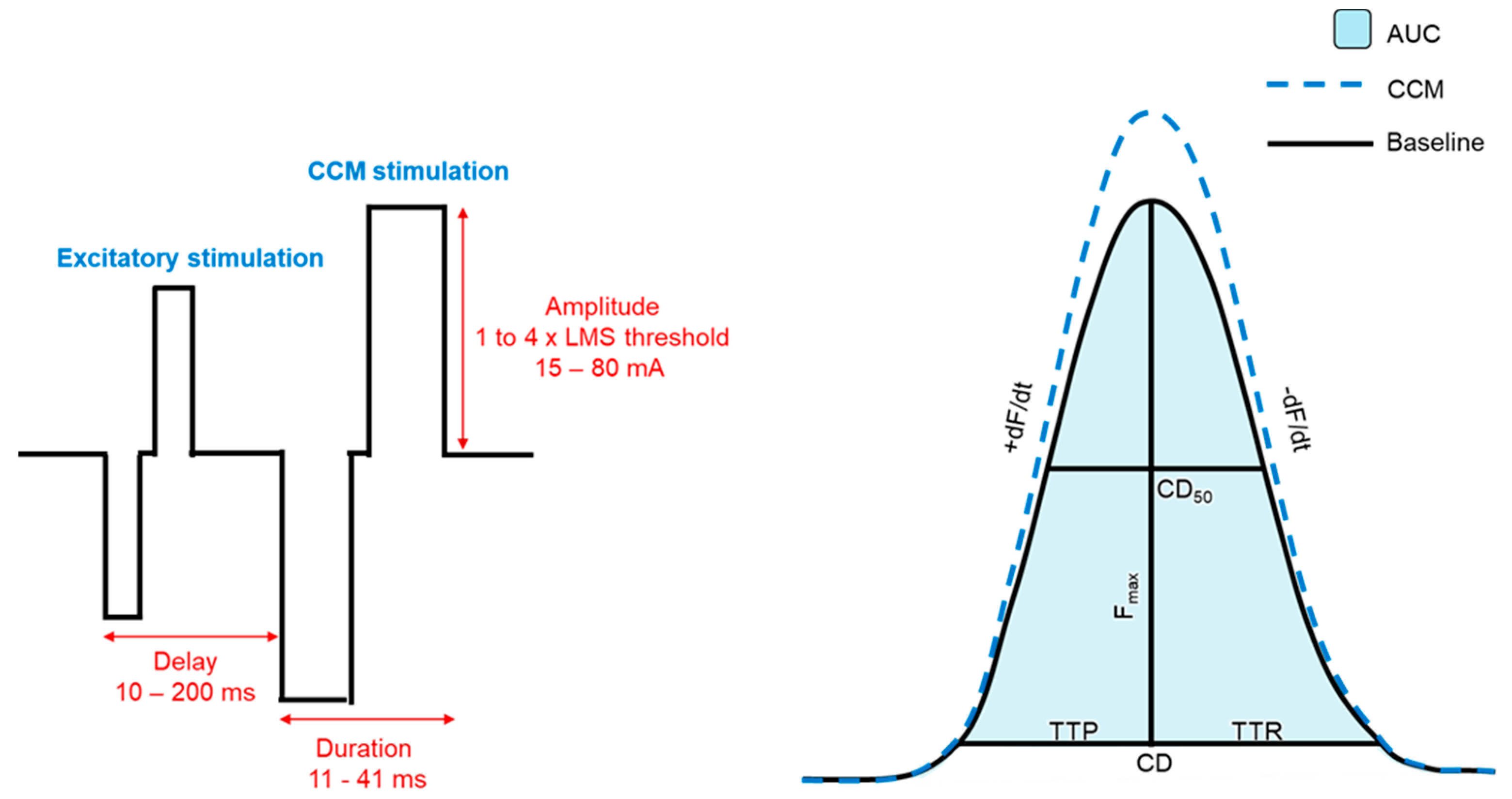
2.3. CCM Stimulation
2.4. Functional Refractory Period
2.5. Force-Frequency Relationship
2.6. Contractile Measurements
2.7. Statistical Analysis
3. Results
3.1. LMS Characteristics
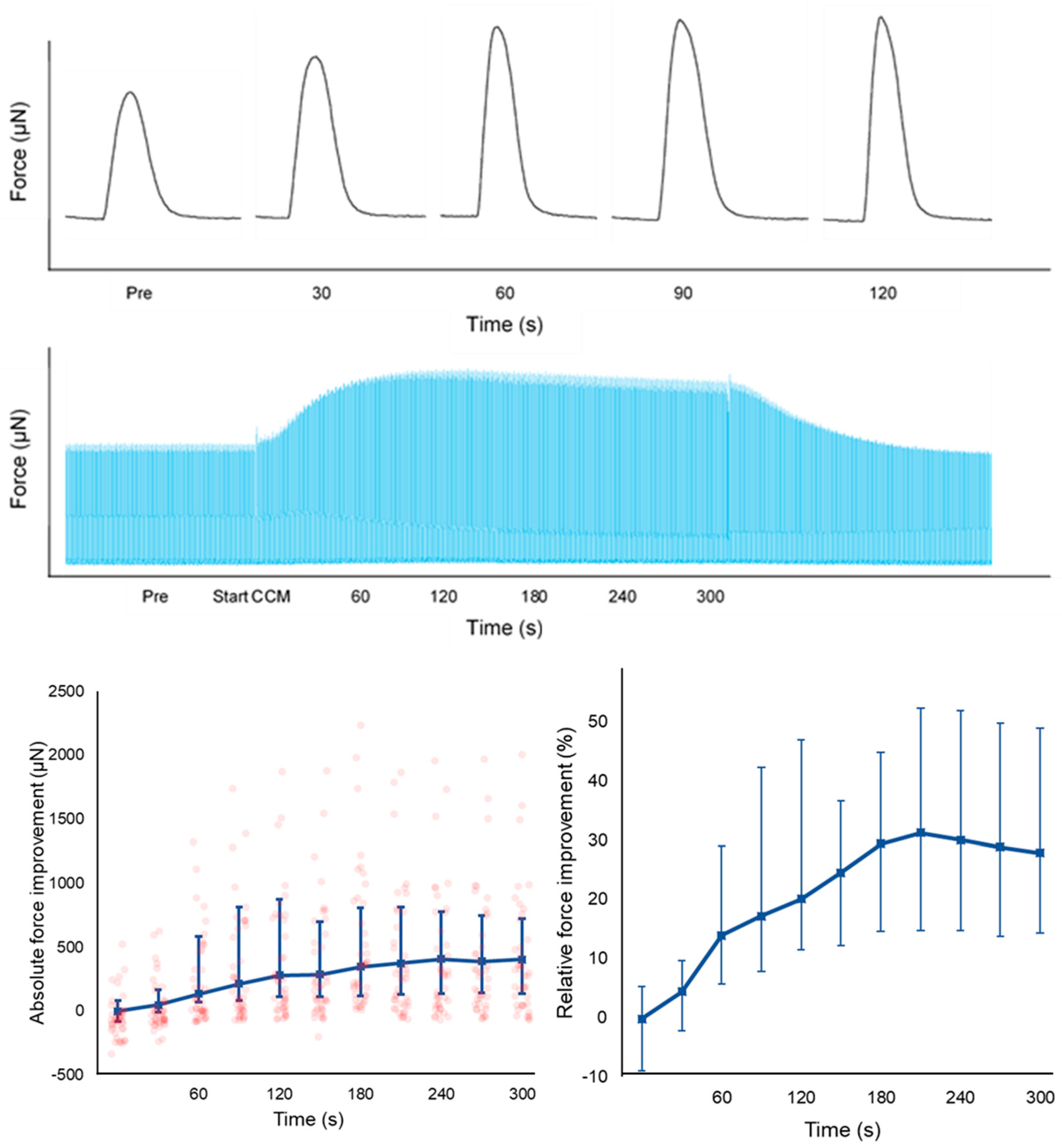
3.2. Biomechanical Effects of CCM on Contractility
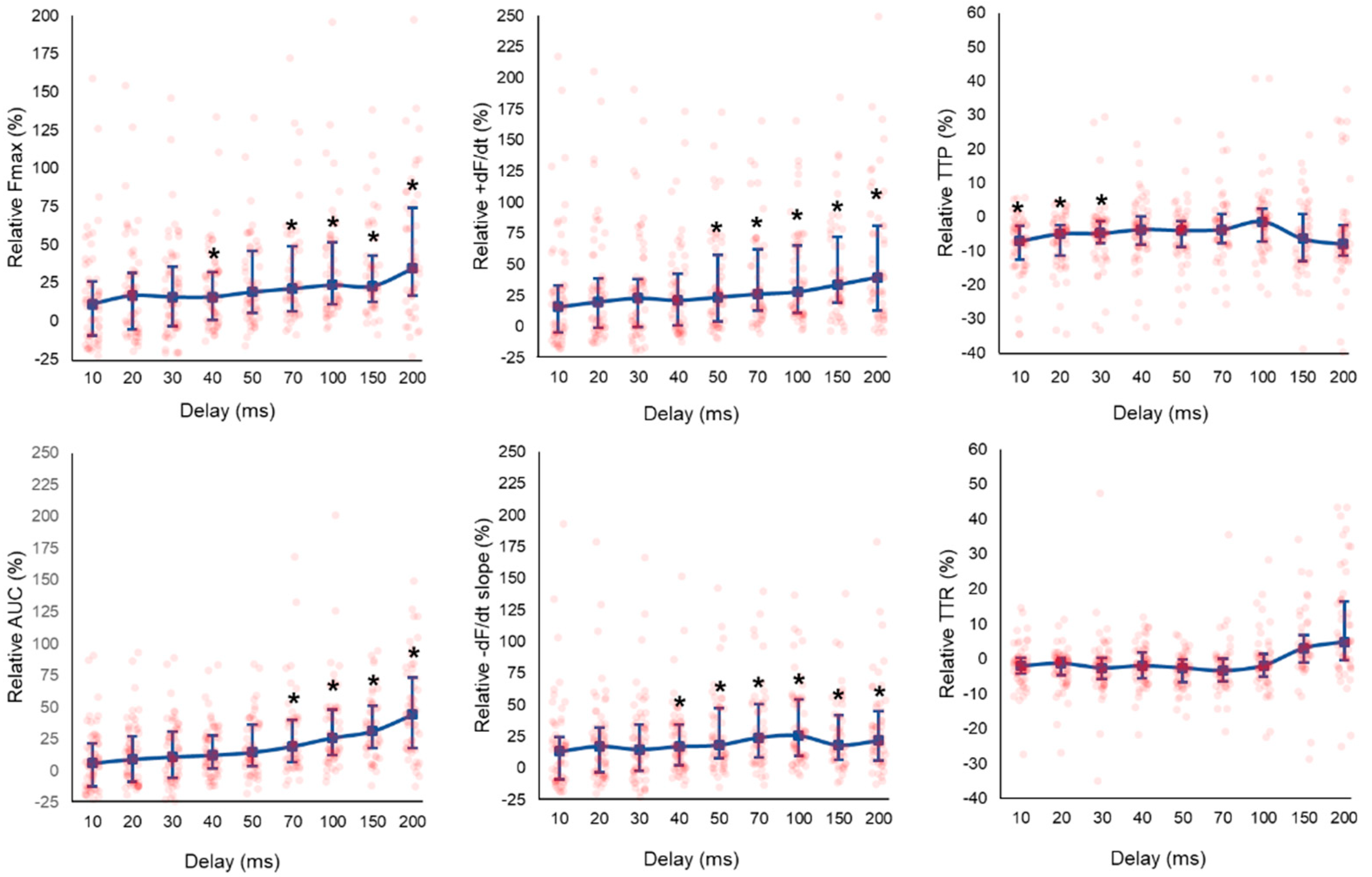
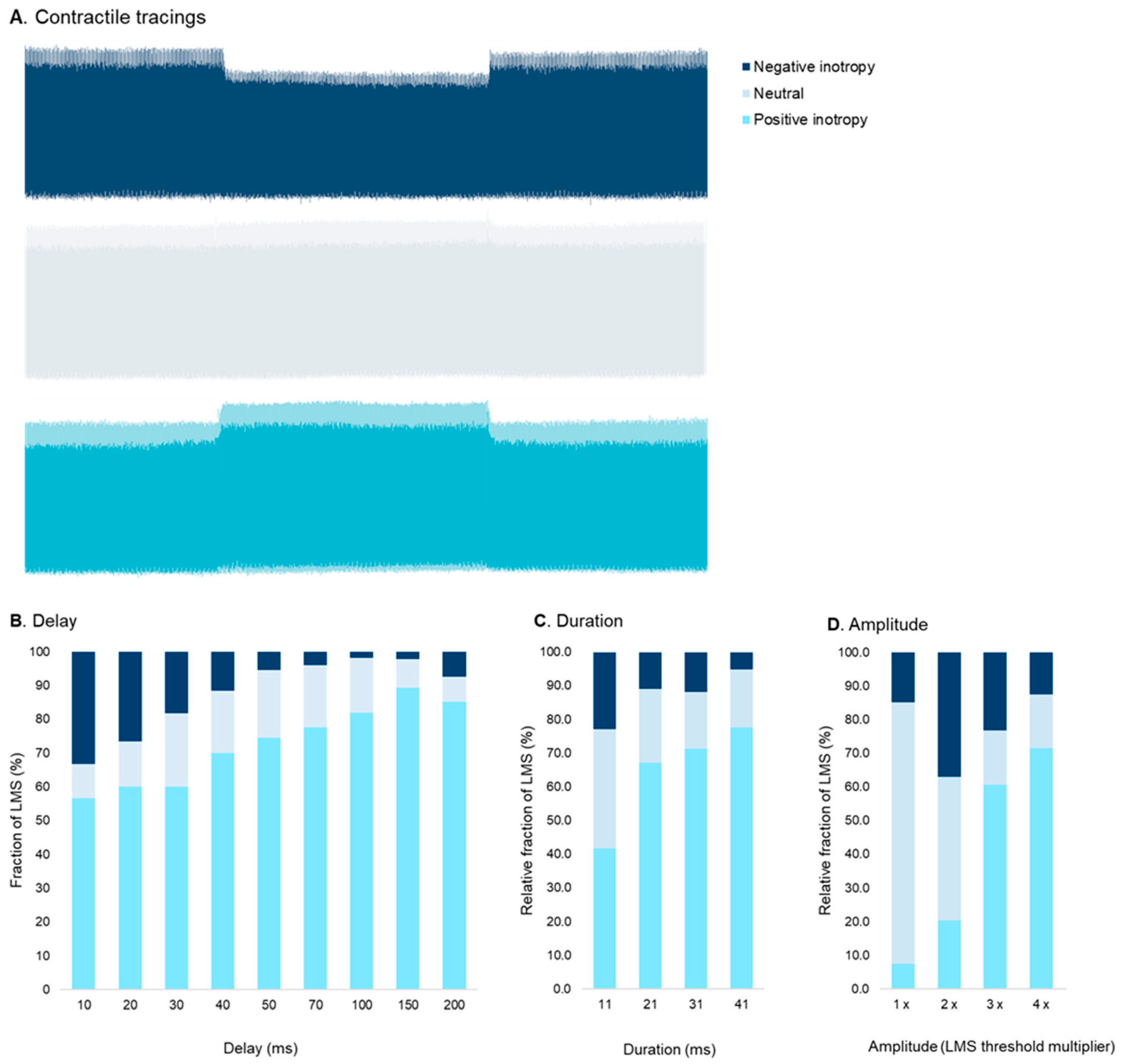
3.3. Effect of CCM Stimulation Delay
3.4. Effect of CCM Stimulation Duration
3.5. Effect of CCM Stimulation Amplitude
3.6. Force-Frequency Relationship
4. Discussion
4.1. Key Findings
4.2. Effect of CCM on the Biomechanical Profile of HF-LMS
4.3. Individual Response of LMS to CCM Stimulation
4.4. CCM and the Force Frequency Relationship in Failing Myocardium
4.5. Clinical Perspective
5. Limitations
6. Conclusion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflict of Interest
References
- Abraham WT, Kuck KH, Goldsmith RL, Lindenfeld J, Reddy VY, Carson PE, Mann DL, Saville B, Parise H, Chan R, Wiegn P, Hastings JL, Kaplan AJ, Edelmann F, Luthje L, Kahwash R, Tomassoni GF, Gutterman DD, Stagg A, Burkhoff D, Hasenfuß G (2018) A Randomized Controlled Trial to Evaluate the Safety and Efficacy of Cardiac Contractility Modulation. JACC Heart Fail 6:874-883. [CrossRef]
- Abu-Khousa M, Fiegle DJ, Sommer ST, Minabari G, Milting H, Heim C, Weyand M, Tomasi R, Dendorfer A, Volk T, Seidel T (2020) The Degree of t-System Remodeling Predicts Negative Force-Frequency Relationship and Prolonged Relaxation Time in Failing Human Myocardium. Front Physiol 11. [CrossRef]
- Amesz JH, Langmuur SJJ, Bierhuizen MFA, de Groot NMS, Manintveld OC, Taverne YJHJ (2023) Omecamtiv mecarbil in precision-cut living heart failure slices: A story of a double-edged sword. Journal of Molecular and Cellular Cardiology Plus 5:100040. [CrossRef]
- Bates D, Mächler M, Bolker B, Walker S (2015) Fitting Linear Mixed-Effects Models Using lme4. Journal of Statistical Software 67:1 - 48. [CrossRef]
- Brunckhorst CB, Shemer I, Mika Y, Ben-Haim SA, Burkhoff D (2006) Cardiac contractility modulation by non-excitatory currents: studies in isolated cardiac muscle. Eur J Heart Fail 8:7-15. [CrossRef]
- Daniel C. Pipilas AH, Jagmeet P. Singh, Theofanie Mela (2023) Cardiac Contractility Modulation for Heart Failure: Current and Future Directions. Journal of the Society for Cardiovascular Angiography & Interventions 2. [CrossRef]
- Feaster TK, Feric N, Pallotta I, Narkar A, Casciola M, Graziano MP, Aschar-Sobbi R, Blinova K (2022) Acute effects of cardiac contractility modulation stimulation in conventional 2D and 3D human induced pluripotent stem cell-derived cardiomyocyte models. Front Physiol 13:1023563. [CrossRef]
- Fischer C, Milting H, Fein E, Reiser E, Lu K, Seidel T, Schinner C, Schwarzmayr T, Schramm R, Tomasi R, Husse B, Cao-Ehlker X, Pohl U, Dendorfer A (2019) Long-term functional and structural preservation of precision-cut human myocardium under continuous electromechanical stimulation in vitro. Nat Commun 10:117. [CrossRef]
- Giallauria F, Cuomo G, Parlato A, Raval NY, Kuschyk J, Stewart Coats AJ (2020) A comprehensive individual patient data meta-analysis of the effects of cardiac contractility modulation on functional capacity and heart failure-related quality of life. ESC Heart Fail 7:2922-2932. [CrossRef]
- Hall JE (2015) Cardiac Muscle; The Heart as a Pump and Function of the Heart Valves. In: Guyton and Hall Textbook of Medical Phsyiology. Saunders.
- Hashimoto K, Perez NG, Kusuoka H, Baker DL, Periasamy M, Marbán E (2000) Frequency-dependent changes in calcium cycling and contractile activation in SERCA2a transgenic mice. Basic Res Cardiol 95:144-151. [CrossRef]
- Kuznetsova A, Brockhoff PB, Christensen RHB (2017) lmerTest Package: Tests in Linear Mixed Effects Models. Journal of Statistical Software 82:1 - 26. [CrossRef]
- McDonagh TA, Metra M, Adamo M, Gardner RS, Baumbach A, Böhm M, Burri H, Butler J, Čelutkienė J, Chioncel O, Cleland JGF, Coats AJS, Crespo-Leiro MG, Farmakis D, Gilard M, Heymans S, Hoes AW, Jaarsma T, Jankowska EA, Lainscak M, Lam CSP, Lyon AR, McMurray JJV, Mebazaa A, Mindham R, Muneretto C, Francesco Piepoli M, Price S, Rosano GMC, Ruschitzka F, Kathrine Skibelund A, Group ESCSD (2021) 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J 42:3599-3726. [CrossRef]
- Morita H, Suzuki G, Haddad W, Mika Y, Tanhehco EJ, Sharov VG, Goldstein S, Ben-Haim S, Sabbah HN (2003) Cardiac contractility modulation with nonexcitatory electric signals improves left ventricular function in dogs with chronic heart failure. J Card Fail 9:69-75. [CrossRef]
- Narkar A, Feaster TK, Casciola M, Blinova K (2022) Human in vitro neurocardiac coculture (ivNCC) assay development for evaluating cardiac contractility modulation. Physiol Rep 10:e15498. [CrossRef]
- Pappone C, Rosanio S, Burkhoff D, Mika Y, Vicedomini G, Augello G, Shemer I, Prutchi D, Haddad W, Aviv R, Snir Y, Kronzon I, Alfieri O, Ben-Haim SA (2002) Cardiac contractility modulation by electric currents applied during the refractory period in patients with heart failure secondary to ischemic or idiopathic dilated cardiomyopathy. Am J Cardiol 90:1307-1313. [CrossRef]
- Pieske B, Trost S, Schütt K, Minami K, Just H, Hasenfuss G (1998) Influence of forskolin on the force-frequency behavior in nonfailing and end-stage failing human myocardium. Basic Res Cardiol 93 Suppl 1:66-75. [CrossRef]
- Pitoulis FG, Hasan W, Papadaki M, Clavere NG, Perbellini F, Harding SE, Kirk JA, Boateng SY, de Tombe PP, Terracciano CM (2020) Intact myocardial preparations reveal intrinsic transmural heterogeneity in cardiac mechanics. J Mol Cell Cardiol 141:11-16. [CrossRef]
- Riccardi M, Sammartino AM, Adamo M, Inciardi RM, Lombardi CM, Pugliese NR, Tomasoni D, Vizzardi E, Metra M, Coats AJS, Pagnesi M (2023) Cardiac contractility modulation: an effective treatment strategy for heart failure beyond reduced left ventricular ejection fraction? Heart Fail Rev 28:1141-1149. [CrossRef]
- Tschöpe C, Kherad B, Klein O, Lipp A, Blaschke F, Gutterman D, Burkhoff D, Hamdani N, Spillmann F, Van Linthout S (2019) Cardiac contractility modulation: mechanisms of action in heart failure with reduced ejection fraction and beyond. Eur J Heart Fail 21:14-22. [CrossRef]
- van Doorn ECH, Amesz JH, Sadeghi AH, de Groot NMS, Manintveld OC, Taverne Y (2024) Preclinical Models of Cardiac Disease: A Comprehensive Overview for Clinical Scientists. Cardiovasc Eng Technol 15:232-249. [CrossRef]
- Watson SA, Terracciano CM, Perbellini F (2019) Myocardial Slices: an Intermediate Complexity Platform for Translational Cardiovascular Research. Cardiovasc Drugs Ther 33:239-244. [CrossRef]
- Wood EH, Heppner RL, Weidmann S (1969) Inotropic effects of electric currents. I. Positive and negative effects of constant electric currents or current pulses applied during cardiac action potentials. II. Hypotheses: calcium movements, excitation-contraction coupling and inotropic effects. Circ Res 24:409-445. [CrossRef]
- Writing Committee M, Members AAJC (2022) 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure. J Card Fail 28:e1-e167.
- Yu CM, Chan JY, Zhang Q, Yip GW, Lam YY, Chan A, Burkhoff D, Lee PW, Fung JW (2009) Impact of cardiac contractility modulation on left ventricular global and regional function and remodeling. JACC Cardiovasc Imaging 2:1341-1349.
- Zhang Q, Chan YS, Liang YJ, Fang F, Lam YY, Chan CP, Lee AP, Chan KC, Wu EB, Yu CM (2013) Comparison of left ventricular reverse remodeling induced by cardiac contractility modulation and cardiac resynchronization therapy in heart failure patients with different QRS durations. Int J Cardiol 167:889-893. [CrossRef]

| Patient characteristics | N = 7 |
|---|---|
| Age, years | 39 ± 18.5 |
| Male, n (%) | 3 (43) |
| Aetiology of heart failure Ischemic cardiomyopathy, n Dilated cardiomyopathy, n Chemotherapy induced, n Myocarditis, n Arrhythmogenic cardiomyopathy, n |
3 3 2 1 1 |
| Surgery LVAD implantation, n Cardiac transplantation, n LVAD in situ, n |
2 5 1 |
| Baseline | CCM | P-value | |
|---|---|---|---|
| Fmax (µN) | 1066 (529 – 2128) | 1229 (587 – 2658) | 0.050* |
| CD (ms) | 450 (396 – 485) | 429 (377 – 482) | 0.297 |
| CD50 (ms) | 223 (210 – 256) | 217 (199 – 246) | 0.056 |
| –dF/dt (µN/s) | -6461 (-13355 - -3020) | -6968 (-16660 - -3692) | 0.043* |
| +dF/dt (µN/s) | 8148 (4109 – 16488) | 10145 (5086 – 22260) | 0.050* |
| AUC (µN.s) | 243 (129 - 464) | 297 (151 - 562) | 0.053 |
| TTP (ms) | 166 (152 - 187) | 160 (142 - 185) | 0.357 |
| TTR (ms) | 270 (230 – 305) | 268 (221 - 303) | 0.388 |
| Baseline | CCM | P-value | |
|---|---|---|---|
| Fmax (µN) | 1066 (626 - 2113) | 1324 (801 - 2738) | 0.030* |
| CD (ms) | 441 (393 - 485) | 411 (370 - 474) | 0.163 |
| CD50 (ms) | 224 (216 - 252) | 217 (202 - 242) | 0.043* |
| –dF/dt (µN/s) | -6461 (-13183 - -3828) | -7914 (-16915 - -4505) | 0.028* |
| +dF/dt (µN/s) | 8880 (5419 – 16282) | 11275 (6629 – 24770) | 0.027* |
| AUC (µN.s) | 243 (156 - 461) | 306 (192 - 582) | 0.040* |
| TTP (ms) | 166 (154 - 186) | 159 (142 - 173) | 0.209 |
| TTR (ms) | 267 (228 - 301) | 260 (220 - 281) | 0.272 |
| Patient etiology | Baseline (µN) | CCM (µN) | % | LMS (n) |
|---|---|---|---|---|
| Ischemic cardiomyopathy | 1411 (1042 – 2113) | 2376 (1239 – 2918) | 68.4 | 20 |
| Ischemic cardiomyopathy | 582 (405 – 863) | 786 (532 – 1113) | 35.2 | 5 |
| Myocarditis-induced dilated cardiomyopathy | 1103 (204 – 1485) | 1164 (317 – 1928) | 12.3 | 5 |
| Arrhythmogenic cardiomyopathy | 2668 (1247 – 4071) | 2927 (1267 – 4202) | 9.7 | 15 |
| Ischemic cardiomyopathy | 1103 (624 – 2167) | 1164 (629 – 2002) | 5.5 | 8 |
| Chemo-induced dilated cardiomyopathy | 267 (161 – 384) | 266 (143 – 454) | -0.3 | 11 |
| Chemo-induced dilated cardiomyopathy | 2267 (1652 – 2880) | 2172 (1671 – 2671) | -4.2 | 2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




