Submitted:
10 December 2024
Posted:
11 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Data Collection
2.2. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Post-authorisation measures: questions and answers. European Medicines Agency post-authorisation procedural advice for users of the centralised procedure. Available online: https://www.ema.europa.eu/en/pharmacovigilance-post-authorisation/post-authorisation-measures-questions-and-answers (accessed on 23 September 2024).
- Zeitoun, J.D.; Ross, J.S.; Atal, I.; Vivot, A.; Downing, N. S.; Baron, G.; Ravaud, P. Postmarketing studies for novel drugs approved by both the FDA and EMA between 2005 and 2010: a cross-sectional study. BMJ Open 2017, 7(12), e018587. [Google Scholar] [CrossRef] [PubMed]
- Guideline on good pharmacovigilance practices (GVP). Available online: https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-good-pharmacovigilance-practices-gvp-module-viii-post-authorisation-safety-studies-rev-3_en.pdf. (accessed on 23 September 2024).
- Carroll, R.; Ramagopalan, S.V.; Cid-Ruzafa, J.; Lambrelli, D.; McDonald, L. An analysis of characteristics of post-authorisation studies registered on the ENCePP EU PAS Register. F1000Res. 2017, 6, 1447. [Google Scholar] [CrossRef]
- ICH E11(R1) guideline on clinical investigation of medicinal products in the pediatric population - Scientific guideline. Available online: https://www.ema.europa.eu/en/ich-e11r1-guideline-clinical-investigation-medicinal-products-pediatric-population-scientific-guideline (accessed on 8 November 2024).
- Ethical considerations for clinical trials on medicinal products conducted with minors. Recommendations of the expert group on clinical trials for the implementation of Regulation (EU) No 536/2014 on clinical trials on medicinal products for human use. Revision 1. Available online: https://health.ec.europa.eu/system/files/2018-02/2017_09_18_ethical_consid_ct_with_minors_0.pdf. (accessed on 8 November 2024).
- Sultana, J.; Crisafulli, S.; Almas, M.; Ippazio, C.A.; Esme, B.; et al. Overview of the European post-authorisation study register post-authorization studies performed in Europe from September 2010 to December 2018. Pharmacoepidemiol Drug Saf 2022, 31(6), 689–705. [Google Scholar] [CrossRef] [PubMed]
- Maier, W.C.; Christensen, R.A.; Anderson, P. Post-approval Studies for Rare Disease Treatments and Orphan Drugs. In Rare Diseases Epidemiology: Update and Overview, 2nd ed.; Posada De La Paz, M., Taruscio, D., Groft, S.C., Eds.; Springer International Publishing: Cham, Switzerland, 2017; Volume 1031, pp. 197–205. [Google Scholar] [CrossRef]
- Catalogue of RWD studies. Available online: https://catalogues.ema.europa.eu/catalogue-rwd-studies (accessed on 23 September 2024).
- REGULATION (EC) No 1901/2006 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 12 December 2006 on medicinal products for pediatric use and amending Regulation (EEC) No 1768/92, Directive 2001/20/EC, Directive 2001/83/EC and Regulation (EC) No 726/2004. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A32006R1901 (accessed on 23 September 2024).
- FAQs on the RWD Catalogues, the old EU PAS Register & ENCePP Resource Database. Question 47. Can I register a study that is not conducted in the EU? Available online: https://catalogues.ema.europa.eu/support (accessed on 6 November 2024).
- Subramanian, D.; Cruz, C.V.; Garcia-Bournissen, F. Systematic Review of Early Phase Pediatric Clinical Pharmacology Trials. J Pediatr Pharmacol Ther 2022, 27(7), 609–617. [Google Scholar] [CrossRef] [PubMed]
- Toma, M.; Felisi, M.; Bonifazi, D.; Bonifazi, F.; Giannuzzi, V.; et al. Pediatric Medicines in Europe: The Pediatric Regulation—Is It Time for Reform? Front Med 2021, 8, 593281. [Google Scholar] [CrossRef] [PubMed]
- ENVI POSITION FOR THE NEW PHARMACEUTICAL LEGISLATION: WHAT’S NEW FOR PEDIATRICS? Accessed May 20, 2024 Available online: https://eptri.eu/news/envi-position-for-the-new-pharmaceutical-legislation-whats-new-for-pediatrics (accessed on 23 September 2024). Parliament adopts its position on EU pharmaceutical reform. European Parliament. Available online: https://www.europarl.europa.eu/news/en/press-room/20240408IPR20308/parliament-adopts-its-position-on-eu-pharmaceutical-reform (accessed on 23 September 2024).
| Studies on pediatric populations registered in the EU PAS Register N= 165 (%) |
|
|---|---|
| Study type | |
| Observational study | 142 (86.1) |
| Survey | 9 (5.5) |
| Review or meta-analysis | 7 (4.2) |
| Clinical trial | 4 (2.4) |
| Other | 3 (1.8) |
| Study design | |
| Descriptive study | 18 (10.9) |
| Cohort studies | 103 (62.4) |
| Cross sectional studies | 8 (4.8) |
| Case control studies | 7 (4.2) |
| Other | 14 (8.5) |
| More than 1 | 13 (8.0) |
| Unknown | 2 (1.2) |
| Status of the Study | |
| Finalized | 95 (57.6) |
| Ongoing | 49 (29.7) |
| Planned | 21 (12.7) |
| Funding Details | |
| Private | 109 (66.1) |
| Public | 45 (27.3) |
| Mixed | 9 (5.5) |
| Unknown | 1 (0.6) |
| Scope of the study* | |
| Risk Assessment | 82 (49.7) |
| Drug Utilization | 39 (23.6) |
| Effectiveness Evaluation | 35 (21.2) |
| Disease Epidemiology | 27 (16.4) |
| Other Scope | 22 (13.3) |
| ENCePP Seal | |
| Yes | 7 (4.2) |
| No | 158 (95.8) |
| Requested by a regulator | |
| Yes | 83 (50.3) |
| No | 80 (48.5) |
| Unknown | 2 (1.2) |
| Country | |
| EU | 120 (72.7) |
| Non-EU | 45 (27.3) |
| RMP status | |
| EU RMP 1 | 12 (7.4) |
| EU RMP 2 | 4 (2.4) |
| EU RMP 3 | 44 (26.6) |
| Non-EU RMP only | 10 (6.1) |
| Unknown | 13 (7.8) |
| Not applicable | 82 (49.7) |
| Data collection** | |
| Primary data | 48 (33.8) |
| Secondary data | 76 (53.2) |
| Mixed | 18 (12.7) |
| Use of reference drug for formal comparison | |
| Yes | 36 (21.8) |
| No | 129 (78.2) |
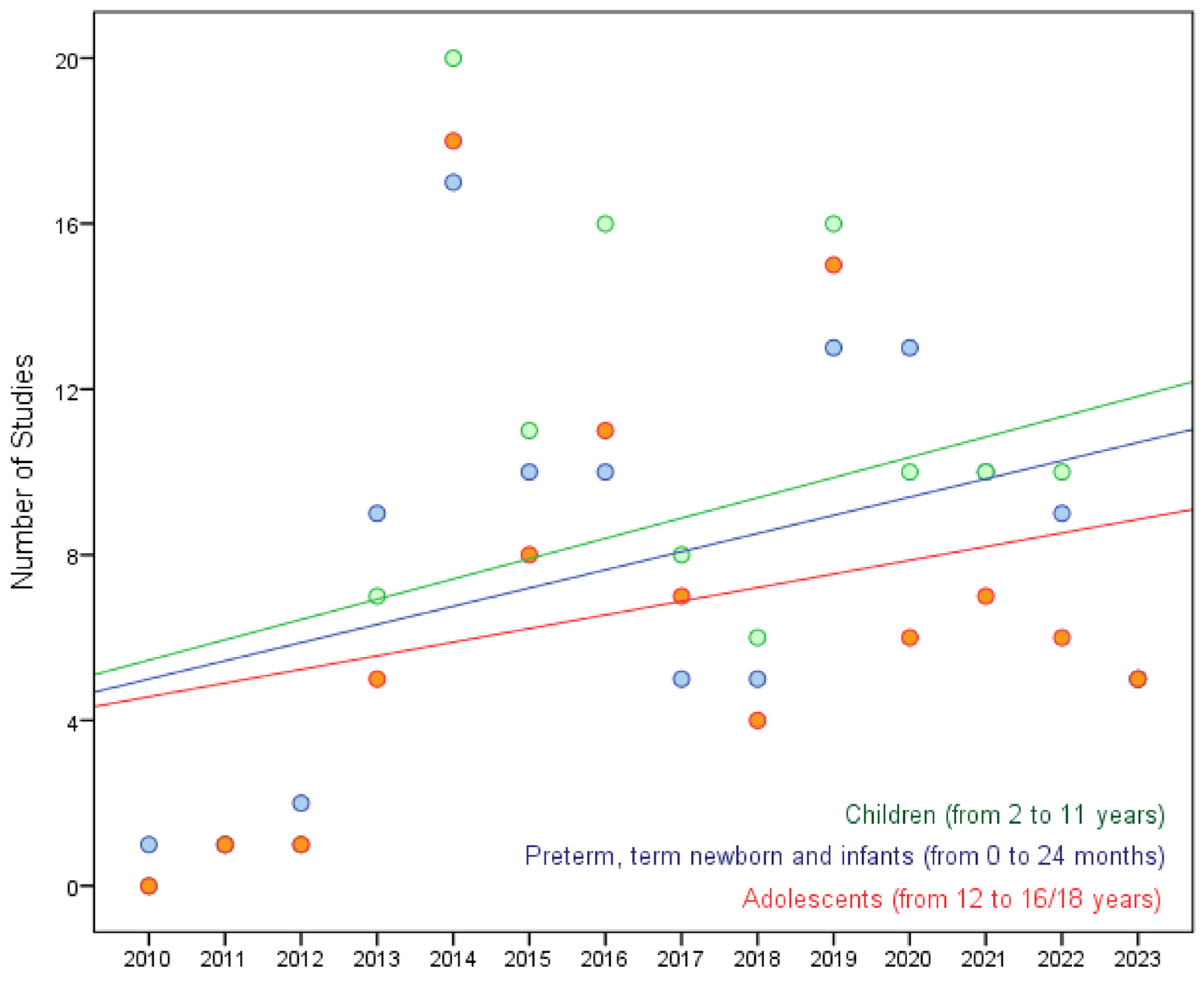
| Subjects age range*** | |
| Preterm newborn infants | 43 (26.1) |
| Term newborn infants (0 to 27 days) | 71 (43.0) |
| Infants and toddlers (28 days to 23 months) | 102 (61.8) |
| Children (2 to 11 years) | 121 (73.3) |
| Adolescents 12 to 16/18 ₸ | 94 (57.0) |
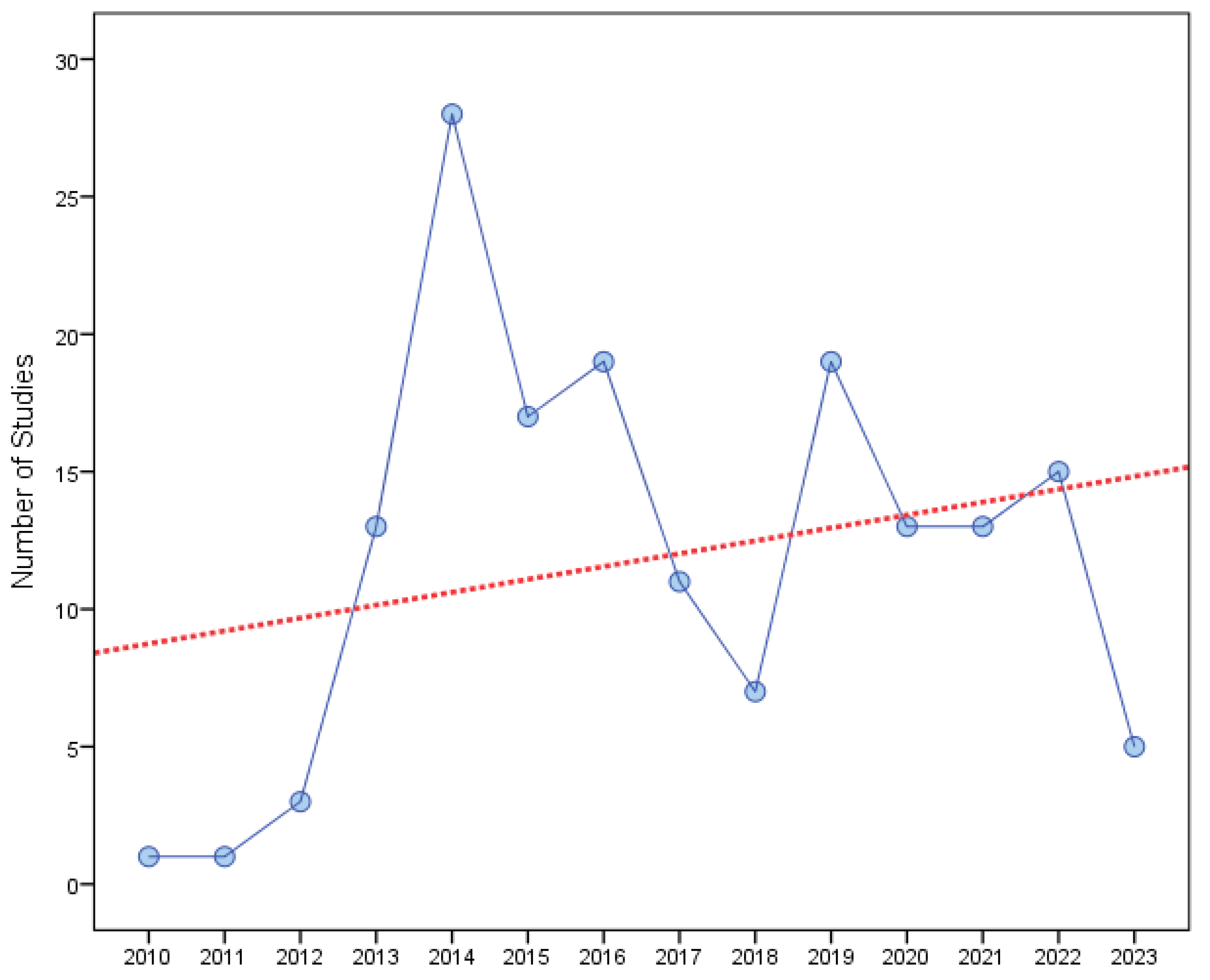
| Year of study registration | Frequency | Percent | |
|---|---|---|---|
| 2010 | 1 | .6 | |
| 2011 | 1 | .6 | |
| 2012 | 3 | 1.8 | |
| 2013 | 13 | 7.9 | |
| 2014 | 28 | 17.0 | |
| 2015 | 17 | 10.3 | |
| 2016 | 19 | 11.5 | |
| 2017 | 11 | 6.7 | |
| 2018 | 7 | 4.2 | |
| 2019 | 19 | 11.5 | |
| 2020 | 13 | 7.9 | |
| 2021 | 13 | 7.9 | |
| 2022 | 15 | 9.1 | |
| 2023 | 5 | 3.0 | |
| Total | 165 | 100.0 | |
| Funding Source | Frequency | Percent | |
|---|---|---|---|
| Pharmaceutical companies | 103 | 62.4 | |
| Government body | 25 | 15.2 | |
| EU funding scheme | 5 | 3.0 | |
| Research councils | 1 | .6 | |
| Other* | 16 | 9.7 | |
| More than one | 14 | 8.5 | |
| Unknown | 1 | .6 | |
| Total | 165 | 100.0 | |
| Year of study registration | Pharmaceutical Companies |
Government Body |
||
|---|---|---|---|---|
| Count | % | Count | % | |
| 2011 | 0 | 0.0 | 0 | 0.0 |
| 2012 | 0 | 0.0 | 0 | 0.0 |
| 2013 | 3 | 27.3 | 3 | 27.3 |
| 2014 | 24 | 85.7 | 2 | 7.1 |
| 2015 | 9 | 52.9 | 4 | 23.5 |
| 2016 | 12 | 66.7 | 3 | 16.7 |
| 2017 | 7 | 63.6 | 3 | 27.3 |
| 2018 | 4 | 66.7 | 0 | 0.0 |
| 2019 | 12 | 80.0 | 3 | 20.0 |
| 2020 | 9 | 81.8 | 1 | 9.1 |
| 2021 | 9 | 69.2 | 1 | 7.7 |
| 2022 | 9 | 64.3 | 3 | 21.4 |
| 2023 | 5 | 100.0 | 0 | 0.0 |
| Total | 103 | 68.2 | 23 | 15.2 |
| Therapeutic area | Frequency | Percent | |
|---|---|---|---|
| A: Alimentary tract and metabolism | 6 | 3.6 | |
| B: Blood and blood forming organs | 8 | 4.8 | |
| C: Cardiovascular system | 3 | 1.8 | |
| D: Dermatologicals | 1 | .6 | |
| H: Systemic hormonal preparation, excluding sex hormones and insulin | 9 | 5.5 | |
| J: Anti-infective for systemic uses | 41 | 24.8 | |
| L: Antineoplastic and immunomodulating agents | 24 | 14.5 | |
| M: Muscolo-skeletal system | 2 | 1.2 | |
| N: Nervous system | 29 | 17.6 | |
| P: Antiparasitic products, insecticides and repellents | 1 | .6 | |
| R: Respiratory system | 9 | 5.5 | |
| S: Sensory organs | 1 | .6 | |
| V: Various | 6 | 3.6 | |
| Others* | 25 | 15.2 | |
| Total | 165 | 100.0 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




