Submitted:
06 December 2024
Posted:
09 December 2024
You are already at the latest version
Abstract

Keywords:
1. Introduction
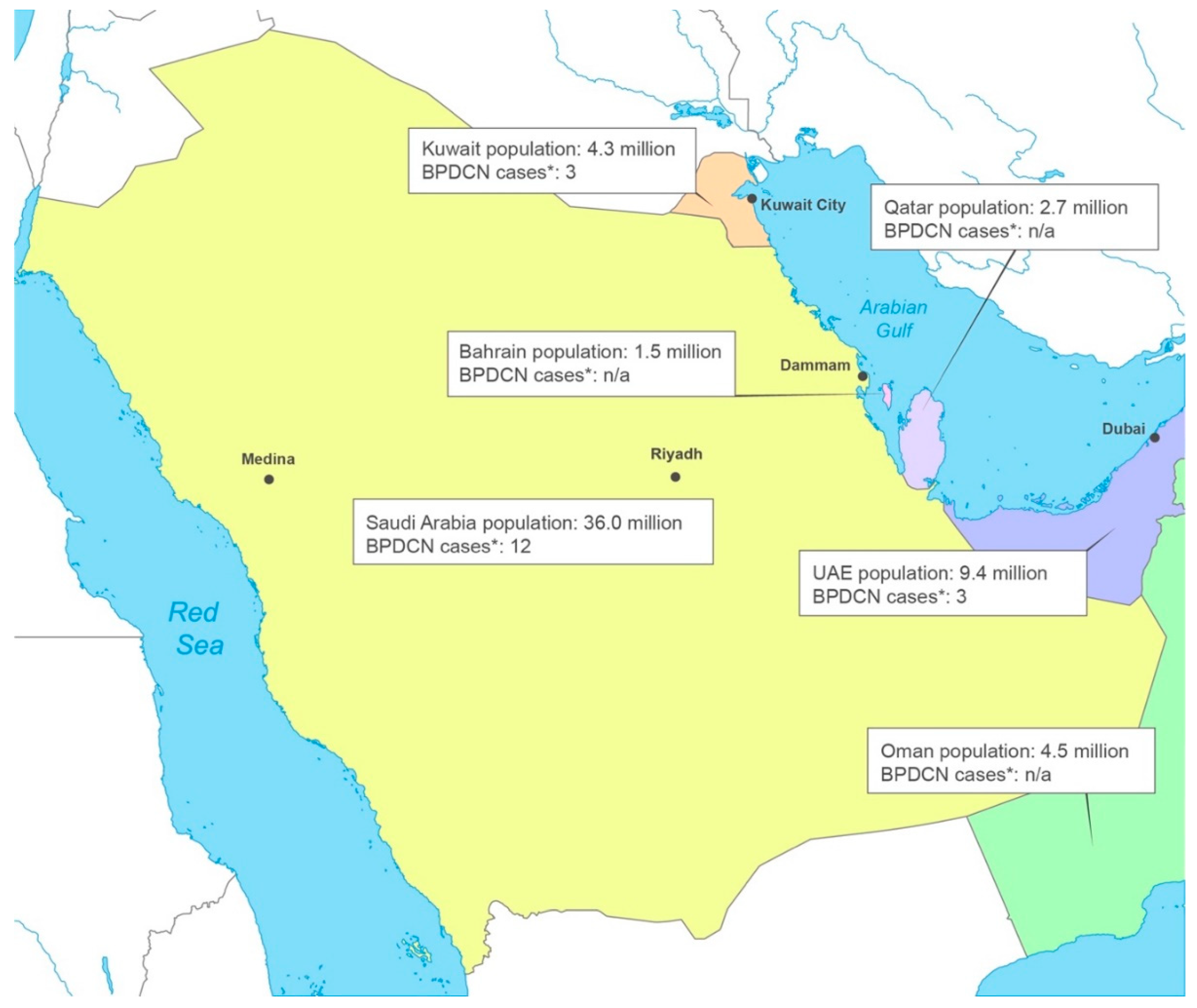
BPDCN in the countries of the GCC
2. Presentation and prognosis of BPDCN
3. Diagnosis of BPDCN
4. Proposed diagnostic pathways
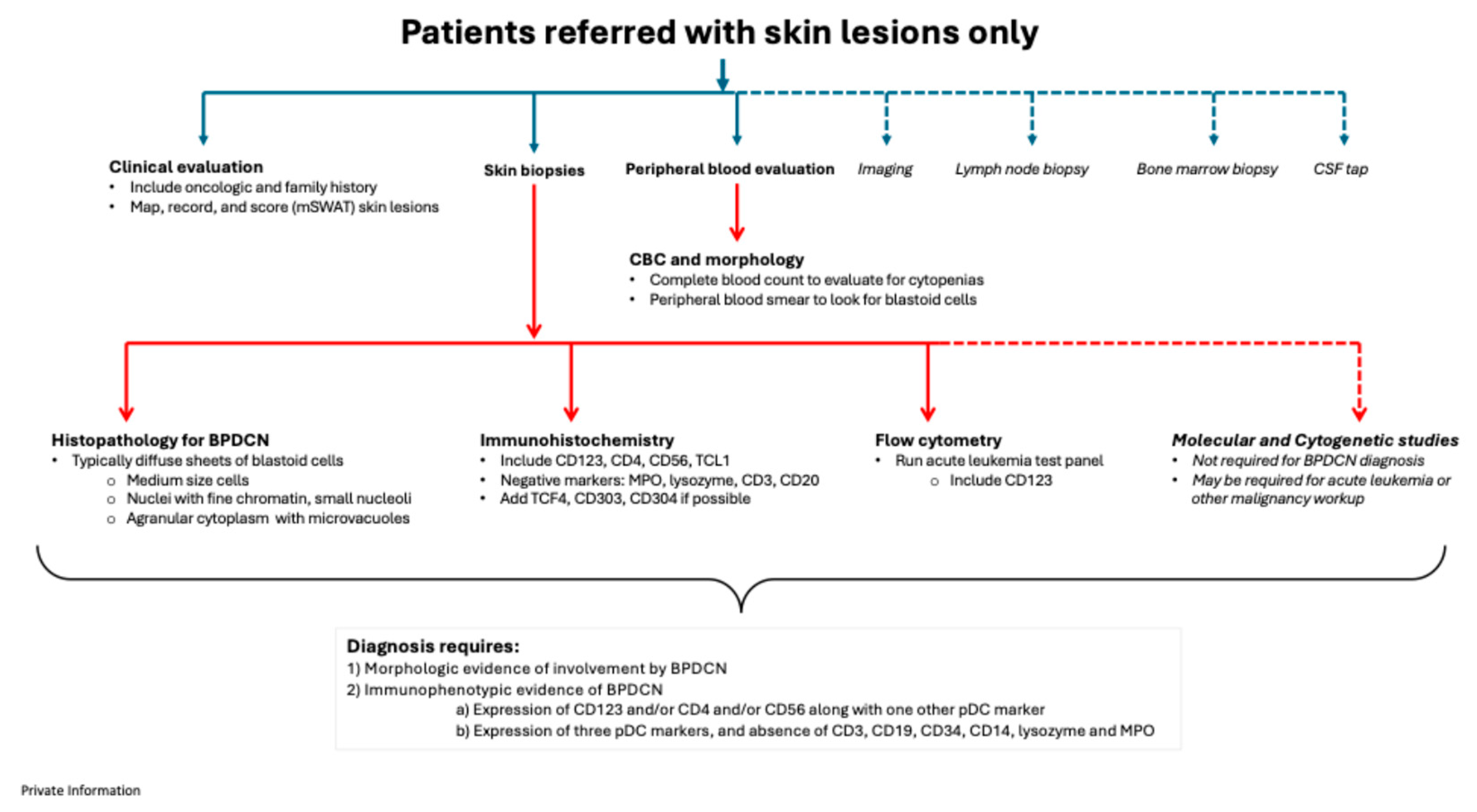
4.1. Patients with skin lesions
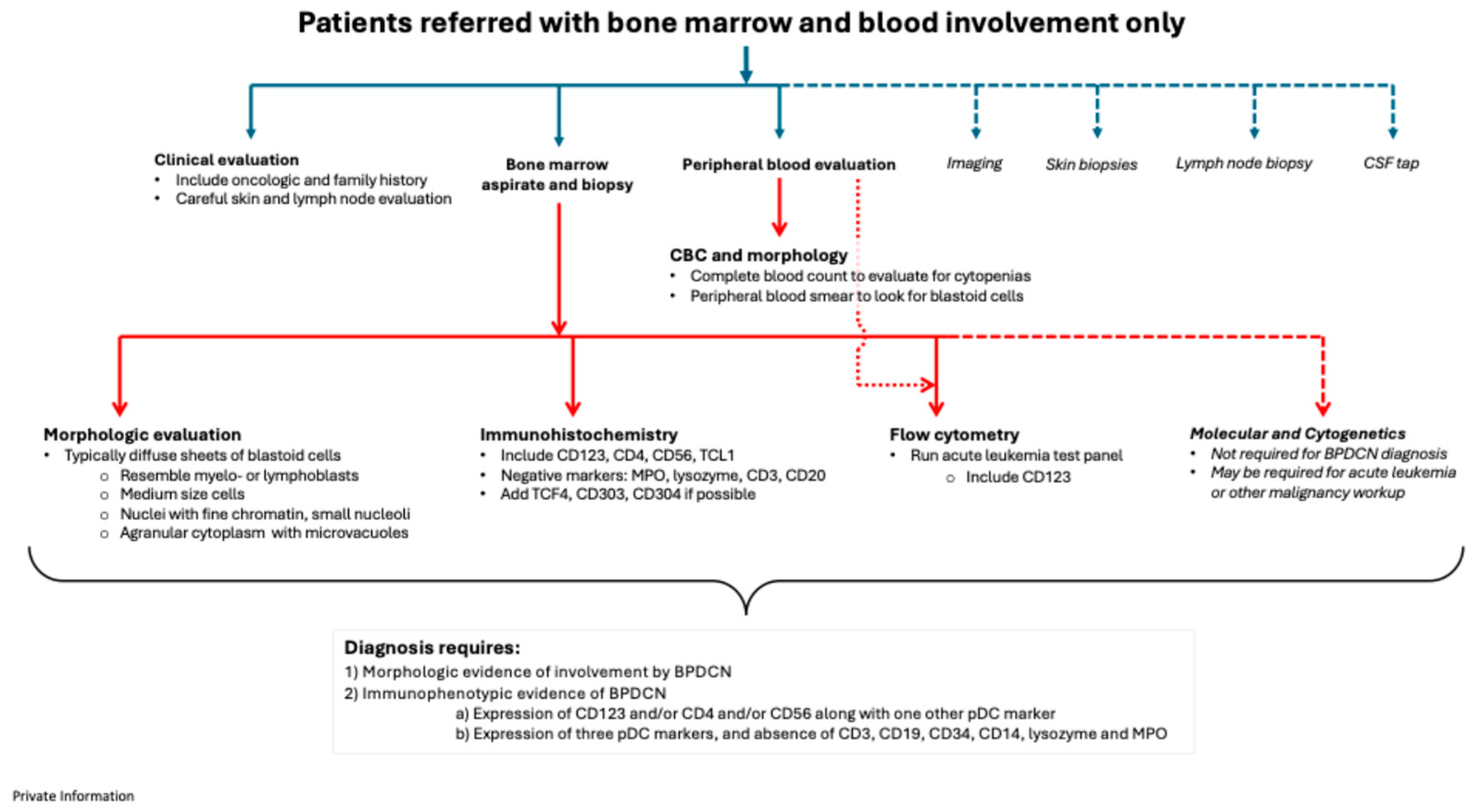
4.2. Patients with bone marrow and blood involvement
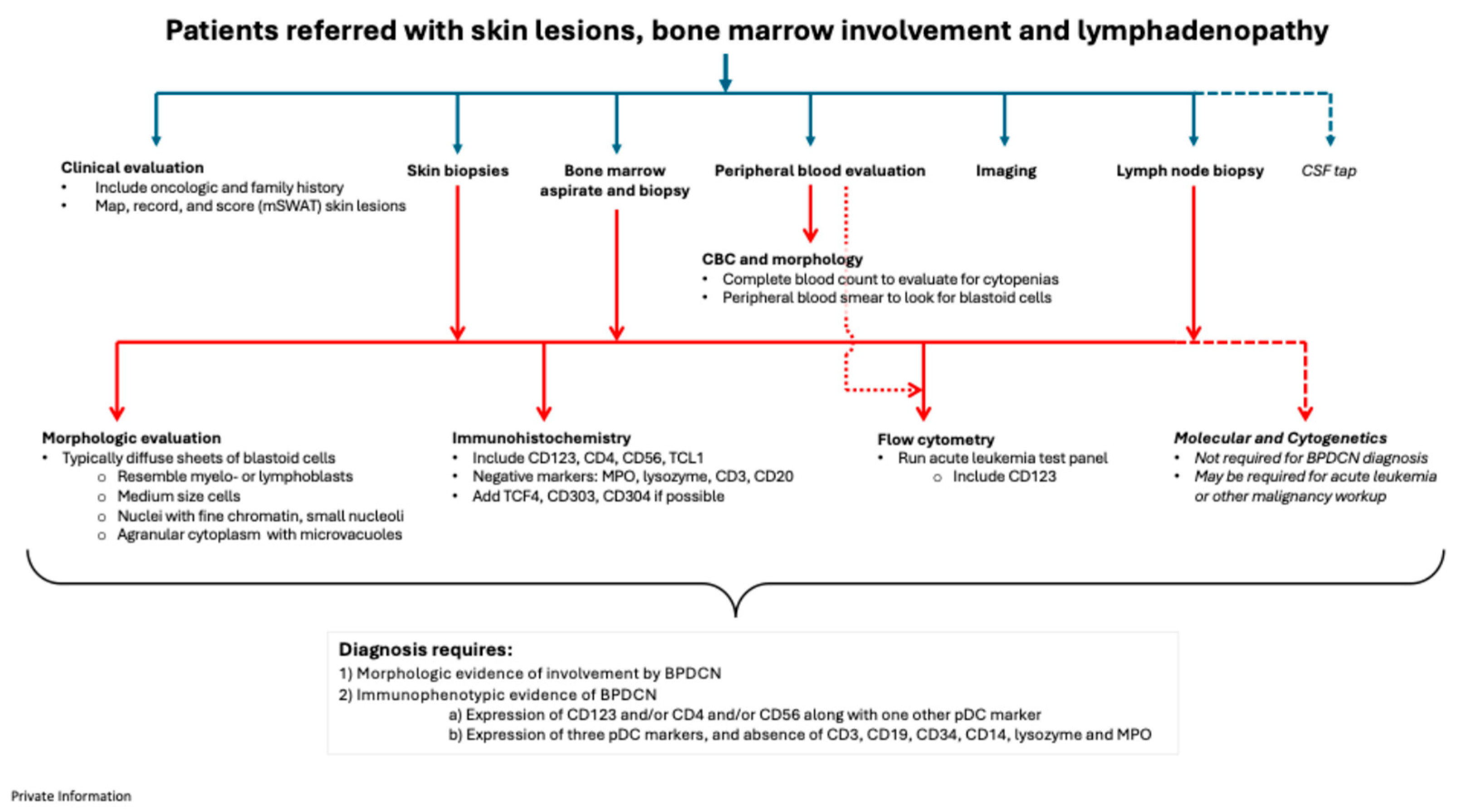
4.3. Patients with skin, bone marrow, and lymph node involvement
5. Practical recommendations for diagnosis of BPDCN in the GCC
5.1. Sample collection
5.2. Patient examinations
5.3. Differential diagnosis
5.4. Differential diagnosis
6. Treatment and monitoring
7. Conclusions and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Khoury J.D., Solary E., Abla O., et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Leukemia 2022, 36, 1703-1719. [CrossRef]
- Ohgami R.S., Aung P.P., Gru A.A., et al. An Analysis of the Pathologic Features of Blastic Plasmacytoid Dendritic Cell Neoplasm Based on a Comprehensive Literature Database of Cases. Arch Pathol Lab Med 2023, 147, 837-846. [CrossRef]
- Sullivan J.M., Rizzieri D.A. Treatment of blastic plasmacytoid dendritic cell neoplasm. Hematology Am Soc Hematol Educ Program 2016, 2016, 16-23. [CrossRef]
- Garnache-Ottou F., Vidal C., Biichle S., et al. How should we diagnose and treat blastic plasmacytoid dendritic cell neoplasm patients? Blood Adv 2019, 3, 4238-4251. [CrossRef]
- Huang L., Wang F. Primary blastic plasmacytoid dendritic cell neoplasm: a US population-based study. Front Oncol 2023, 13, 1178147. [CrossRef]
- Adachi M., Maeda K., Takekawa M., et al. High expression of CD56 (N-CAM) in a patient with cutaneous CD4-positive lymphoma. Am J Hematol 1994, 47, 278-82. [CrossRef]
- Assaf C., Gellrich S., Whittaker S., et al. CD56-positive haematological neoplasms of the skin: a multicentre study of the Cutaneous Lymphoma Project Group of the European Organisation for Research and Treatment of Cancer. J Clin Pathol 2007, 60, 981-9. [CrossRef]
- Brody J.P., Allen S., Schulman P., et al. Acute agranular CD4-positive natural killer cell leukemia. Comprehensive clinicopathologic studies including virologic and in vitro culture with inducing agents. Cancer 1995, 75, 2474-83. [CrossRef]
- Vardiman J.W., Thiele J., Arber D.A., et al. The 2008 revision of the World Health Organization (WHO) classification of myeloid neoplasms and acute leukemia: rationale and important changes. Blood 2009, 114, 937-51. [CrossRef]
- Arber D.A., Orazi A., Hasserjian R.P., et al. International Consensus Classification of Myeloid Neoplasms and Acute Leukemias: integrating morphologic, clinical, and genomic data. Blood 2022, 140, 1200-1228. [CrossRef]
- Arber D.A., Orazi A., Hasserjian R., et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood 2016, 127, 2391-405. [CrossRef]
- Huang Y., Wang Y., Chang Y., et al. Myeloid Neoplasms with Elevated Plasmacytoid Dendritic Cell Differentiation Reflect the Maturation Process of Dendritic Cells. Cytometry A 2020, 97, 61-69. [CrossRef]
- Zalmai L., Viailly P.J., Biichle S., et al. Plasmacytoid dendritic cells proliferation associated with acute myeloid leukemia: phenotype profile and mutation landscape. Haematologica 2021, 106, 3056-3066. [CrossRef]
- Beird H.C., Khan M., Wang F., et al. Features of non-activation dendritic state and immune deficiency in blastic plasmacytoid dendritic cell neoplasm (BPDCN). Blood Cancer J 2019, 9, 99. [CrossRef]
- Jain A., Sweet K. Blastic Plasmacytoid Dendritic Cell Neoplasm. J Natl Compr Canc Netw 2023, 21, 515-521. [CrossRef]
- Lee Y.J., Kim Y., Park S.H., Jo J.C. Plasmacytoid dendritic cell neoplasms. Blood Res 2023, 58, 90-95. [CrossRef]
- Sapienza M.R., Benvenuto G., Ferracin M., et al. Newly-Discovered Neural Features Expand the Pathobiological Knowledge of Blastic Plasmacytoid Dendritic Cell Neoplasm. Cancers (Basel) 2021, 13, . [CrossRef]
- Sapienza M.R., Pileri A., Derenzini E., et al. Blastic Plasmacytoid Dendritic Cell Neoplasm: State of the Art and Prospects. Cancers (Basel) 2019, 11, . [CrossRef]
- Shi Y., Wang E. Blastic plasmacytoid dendritic cell neoplasm: a clinicopathologic review. Arch Pathol Lab Med 2014, 138, 564-9. [CrossRef]
- Wang W., Khoury J.D., Miranda R.N., et al. Immunophenotypic characterization of reactive and neoplastic plasmacytoid dendritic cells permits establishment of a 10-color flow cytometric panel for initial workup and residual disease evaluation of blastic plasmacytoid dendritic cell neoplasm. Haematologica 2021, 106, 1047-1055. [CrossRef]
- Wang Y., Xiao L., Yin L., Zhou L., Deng Y., Deng H. Diagnosis, treatment, and genetic characteristics of blastic plasmacytoid dendritic cell neoplasm: A review. Medicine (Baltimore) 2023, 102, e32904. [CrossRef]
- Lin X., Wang L., Hu Q., et al. Incidence, prognostic factors, and survival outcomes in patients with blastic plasmacytoid dendritic cell neoplasm: A retrospective study in the Surveillance, Epidemiology, and End Results database. Eur J Haematol 2023, 110, 743-753. [CrossRef]
- Basic population data for 2021, all countries. Available online: https://databank.worldbank.org/embed/2021-Population-Data/id/67b1e32b (accessed on May 1, 2024).
- Alam H., Saeed N., Rashid A. Indispensable role of immunophenotyping in diagnosing leukemic phase of blastic plasmacytoid dendritic cell neoplasm without cutaneous manifestation. Leuk Res Rep 2022, 17, 100317. [CrossRef]
- Dalle S., Beylot-Barry M., Bagot M., et al. Blastic plasmacytoid dendritic cell neoplasm: is transplantation the treatment of choice? Br J Dermatol 2010, 162, 74-9. [CrossRef]
- Martin-Martin L., Lopez A., Vidriales B., et al. Classification and clinical behavior of blastic plasmacytoid dendritic cell neoplasms according to their maturation-associated immunophenotypic profile. Oncotarget 2015, 6, 19204-16. [CrossRef]
- Pagano L., Valentini C.G., Pulsoni A., et al. Blastic plasmacytoid dendritic cell neoplasm with leukemic presentation: an Italian multicenter study. Haematologica 2013, 98, 239-46. [CrossRef]
- Hayat A., Khan A., Baloch G., Shaikh N. Pancytopenia; study for clinical features and etiological pattern at tertiary care settings in Abbottabad. Professional Med J 2014, 21, 060-065.
- El Achi H., Dupont E., Paul S., Khoury J.D. CD123 as a Biomarker in Hematolymphoid Malignancies: Principles of Detection and Targeted Therapies. Cancers (Basel) 2020, 12, . [CrossRef]
- Wang W., Xu J., Khoury J.D., et al. Immunophenotypic and Molecular Features of Acute Myeloid Leukemia with Plasmacytoid Dendritic Cell Differentiation Are Distinct from Blastic Plasmacytoid Dendritic Cell Neoplasm. Cancers (Basel) 2022, 14, . [CrossRef]
- Eisenwort G., Sadovnik I., Keller A., et al. Phenotypic characterization of leukemia-initiating stem cells in chronic myelomonocytic leukemia. Leukemia 2021, 35, 3176-3187. [CrossRef]
- Tse E., Kwong Y.L. The diagnosis and management of NK/T-cell lymphomas. J Hematol Oncol 2017, 10, 85. [CrossRef]
- Cho-Vega J.H., Medeiros L.J., Prieto V.G., Vega F. Leukemia cutis. Am J Clin Pathol 2008, 129, 130-42. [CrossRef]
- Li L., Wang Y., Lian C.G., Hu N., Jin H., Liu Y. Clinical and pathological features of myeloid leukemia cutis. An Bras Dermatol 2018, 93, 216-221. [CrossRef]
- Alsomali D.Y., Bakshi N., Kharfan-Dabaja M., El Fakih R., Aljurf M. Diagnosis and Treatment of Subcutaneous Panniculitis-like T-cell Lymphoma: A Systematic Literature Review. Hematol Oncol Stem Cell Ther 2023, 16, 110-116. [CrossRef]
- Laribi K., Baugier de Materre A., Sobh M., et al. Blastic plasmacytoid dendritic cell neoplasms: results of an international survey on 398 adult patients. Blood Adv 2020, 4, 4838-4848. [CrossRef]
- Pemmaraju N., Sweet K.L., Stein A.S., et al. Long-Term Benefits of Tagraxofusp for Patients With Blastic Plasmacytoid Dendritic Cell Neoplasm. J Clin Oncol 2022, 40, 3032-3036. [CrossRef]
- Syed Y.Y. Tagraxofusp: First Global Approval. Drugs 2019, 79, 579-583. [CrossRef]
- Valentini C.G., Pagano L. Tagraxofusp for blastic plasmacytoid dendritic cell neoplasm: a 2-speed cure in the United States and European Union. Blood Adv 2023, 7, 7084-7086. [CrossRef]
- Angelucci E., Deconinck E., Manteigas D., Zuurman M., Herling M. Durable Outcomes with Manageable Safety Leading to Prolonged Survival with Tagraxofusp for Treatment-Naive Patients with Blastic Plasmacytoid Dendritic Cell Neoplasm: Updated Results from a European Named Patient Program. Blood 2023, 142 (Supplement 1), 547. [CrossRef]
- Herling M., Angelucci E., Manteigas D., Zuurman M., Deconinck E. Real world (RW) study of patients (pts) with relapsed or refractory (RR) blastic plasmacytoid dendritic cell neoplasm (BPDCN) treated with tagraxofusp (TAG). J Clin Oncol 2024, 42, e18533. [CrossRef]
- Pemmaraju N., Wilson N.R., Garcia-Manero G., et al. Characteristics and outcomes of patients with blastic plasmacytoid dendritic cell neoplasm treated with frontline HCVAD. Blood Adv 2022, 6, 3027-3035. [CrossRef]
- Aung P.P., Pemmaraju N., Torres-Cabala C.A., et al. Apparent partial loss of CD123 expression in blastic plasmacytoid dendritic cell neoplasm after treatment with CD123-targeted therapy: A novel finding and possible diagnostic pitfall. J Dermatol 2020, 47, e354-e355. [CrossRef]
- Cai T., Gouble A., Black K.L., et al. Targeting CD123 in blastic plasmacytoid dendritic cell neoplasm using allogeneic anti-CD123 CAR T cells. Nat Commun 2022, 13, 2228. [CrossRef]
- Hiraga J., Tomita A., Sugimoto T., et al. Down-regulation of CD20 expression in B-cell lymphoma cells after treatment with rituximab-containing combination chemotherapies: its prevalence and clinical significance. Blood 2009, 113, 4885-93. [CrossRef]
| Marker1 | BPDCN | AML | T-ALL | T-cell lymphoma |
| BCL-2 | + | +/- | +/- | + |
| CD3 (cytoplasmic) | - | - | + | ++ |
| CD4 | + | +/- | +/- | +/- |
| CD8 | - | +/- | +/- | +/- |
| CD14 | - | +/- | - | - |
| CD19 | - | +/- | - | - |
| CD20 | - | - | - | - |
| CD34 | - | +/- | +/- | - |
| CD56 | + | +/- | +/- | +/- |
| CD123 | ++ | +/- | +/- | - |
| CD303 | + | - | +/- | - |
| CD304 | + | - | +/- | - |
| TCF4 | ++ | - | - | - |
| TCL1 | + | +/- | +/- | - |
| TdT | +/- | +/- | +/- | - |
| Lysozyme | - | +/- | - | - |
| Myeloperoxidase | - | +/- | - | - |
| Differential Diagnosis | Notes |
| Acute myeloid leukemia (AML) with monocytic differentiation (AML-M5) | Majority of blast cells from monocytic lineage. One-third of cases lack CD34. Cases are usually positive for lysozyme, often lack myeloperoxidase and (weakly) co-express CD4 and/or CD123, and/or CD56[29] |
| Plasmacytoid dendritic cell-AML (pDC-AML) | Rare subtype of AML with ≥2% pDC expansion and interstitial distribution of pDCs intermixed with leukemic blasts. pDC may also be present in loose clusters. Positive for CD13, CD34, CD36, CD38, CD123, CD303, HLA-DR, TCL1. Negative for CD56 and CD13. Also frequently associated with RUNX1 mutation[30] |
| Mature plasmacytoid dendritic cell proliferation associated with myeloid neoplasm (MPDCP) | Mature, fully differentiated blast cells. Positive for CD123, CD4, HLA-DR. Negative for CD56, CD3, CD19, CD14, MPO[12] |
| Chronic myelomonocytic leukemia (CMML) | Characterized by dysplastic bone marrow cells and persistent peripheral blood monocytosis. Two variants: dysplastic and proliferative. Splenomegaly is common and risk of transformation to AML is high. Cells are positive for CD34, CD33, CD117, CD123, CD133. Negative for CD25, CD26, CD38[31] |
| Extranodal NK/T-cell lymphoma | Rare, disfiguring disease that can affect the upper respiratory and digestive tract, skin or (rarely) bone marrow. Strongly associated with EBV infection of lymphoma cells. Cytolytic cells with cytotoxic granules including granzyme B, perforin and TIA-1 or positivity for CD56 must be present. May also be positive for CD2, cytoplasmic CD3, and EBER[32]. Negative for CD123, CD4, TCF4 |
| T-cell prolymphocytic leukemia/lymphoma | Cells co-express CD3, CD4 and TCL1. A subset of cases may co-express CD56 |
| Leukemia cutis | Usually associated with AML. Characterized by single or multiple firm papules, nodules, and plaques that may be skin-colored, red, brown or purple. Expression of myeloperoxidase, CD15, CD43, CD45, CD34 (variable), CD68 (variable), and CD13 (variable)[33,34] |
| Subcutaneous panniculitis-like T-cell lymphoma | Rare cytotoxic T-cell lymphoma that exhibits leukemic infiltration of subcutaneous adipose tissue accompanied by large number of macrophages. Cells are positive for CD2, CD3, CD7, CD8, TIA-1; Negative for CD4, CD30, CD56[35] |
| Summary of Recommendations |
|
|
|
|
|
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




