Submitted:
03 December 2024
Posted:
04 December 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
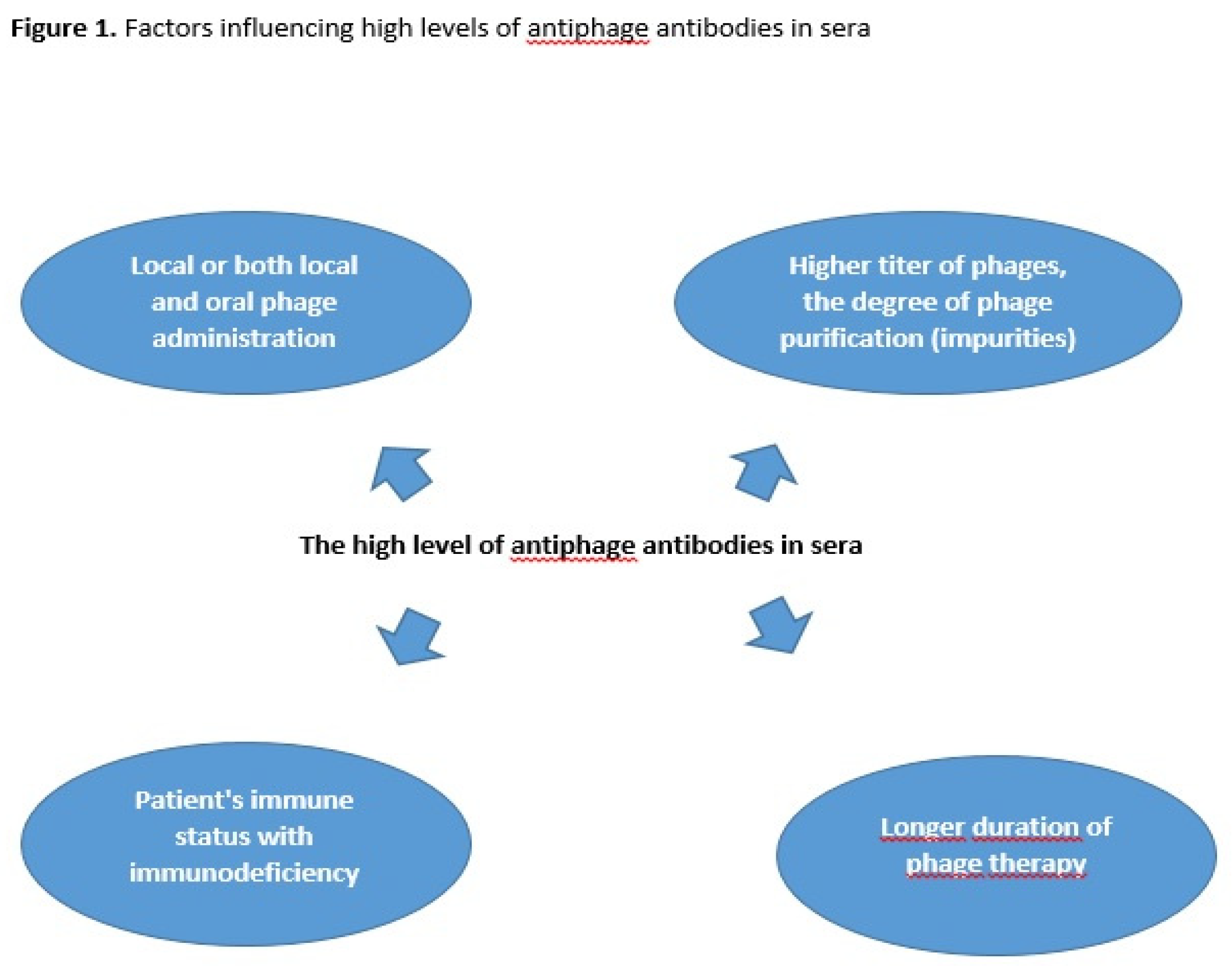
2. Materials and Methods
2.1. Patients
2.2. Bacteriophages
2.3. Plate Phage Neutralization Test
2.4. Categories of the Results of PT
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Górski, A.; Międzybrodzki R.; Węgrzyn, G.; Jończyk-Matysiak, E.; Borysowski, J.; Weber-Dąbrowska, B. Phage therapy: Current status and perspectives. Med. Res. Rev. 2020, 40(1), 459-463. [CrossRef]
- Liu, K.; Wang, C.; Zhou, X.; Guo, X.; Yang, Y.; Liu, W.; Zhao, R.; Song H. Bacteriophage therapy for drug-resistant Staphylococcus aureus infections. Front. Cell Infect. Microbiol. 2024, 14, 1336821. [CrossRef]
- Olawade, D.B.; Fapohunda, O.; Egbon, E.; Ebiesuwa, O.A.; Usman, S.O.; Faronbi, A.O.; Fidelis, S.C. Phage therapy: A targeted approach to overcoming antibiotic resistance. Microb. Pathog. 2024, 197, 107088. Epub ahead of print. [CrossRef]
- Dąbrowska, K.; Miernikiewicz, P.; Piotrowicz, A.; Hodyra, K.; Owczarek, B.; Lecion, D.; Kaźmierczak, Z.; Letarov, A.; Górski A. Immunogenicity studies of proteins forming the T4 phage head surface. J. Virol. 2014, 88(21), 12551-7. [CrossRef]
- Gembara, K.; Dąbrowska, K. Interaction of bacteriophages with the immune system: induction of bacteriophage-specific antibodies. Methods Mol. Biol. 2024, 2734, 183-196. [CrossRef]
- Majewska, J.; Kaźmierczak, Z.; Lahutta, K.; Lecion, D.; Szymczak, A.; Miernikiewicz, P.; Drapała, J.; Harhala, M.; Marek-Bukowiec, K.; Jędruchniewicz, N.; Owczarek, B.; Górski, A.; Dąbrowska, K. Induction of phage-specific antibodies by two therapeutic staphylococcal bacteriophages administered per os. Front. Immunol. 2019, 10, 2607. [CrossRef]
- Łusiak-Szelachowska, M.; Żaczek, M.; Weber-Dąbrowska, B.; Międzybrodzki, R.; Kłak, M.; Fortuna, W.; Letkiewicz S., Rogóż P., Szufnarowski K., Jończyk-Matysiak E., Owczarek, B.; Górski, A. Phage neutralization by sera of patients receiving phage therapy. Viral Immunol. 2014, 27, 295-304. [CrossRef]
- Łusiak-Szelachowska, M.; Żaczek, M.; Weber-Dąbrowska, B.; Międzybrodzki, R.; Letkiewicz, S.; Fortuna, W.; Rogóż, P.; Szufnarowski, K.; Jończyk-Matysiak, E.; Olchawa, E.; Walaszek, K. M.; Górski, A. Antiphage activity of sera during phage therapy in relation to its outcome. Future Microbiol. 2017, 12, 109-117. [CrossRef]
- Żaczek, M.; Łusiak-Szelachowska, M.; Jończyk-Matysiak, E.; Weber-Dąbrowska, B.; Międzybrodzki, R.; Owczarek, B.; Kopciuch, A.; Fortuna, W.; Rogóż, P.; Górski A. Antibody production in response to staphylococcal MS-1 phage cocktail in patients undergoing phage therapy. Front. Microbiol. 2016, 7, 1681. [CrossRef]
- Łusiak-Szelachowska, M.; Żaczek, M.; Weber-Dąbrowska, B.; Kłak, M.; Międzybrodzki, R.; Fortuna, W.; Rogóż, P.; Szufnarowski, K.; Jończyk-Matysiak, E.; Górski A. Antiphage activity of sera from patients receiving staphylococcal phage preparations. In: Microbes in the spotlight: recent progress in the understanding of beneficial and harmful microorganisms; Méndez-Vilas A., Eds; BrownWalker Press, 2016; pp. 245-249.
- Nick, JA.; Dedrick, R.M.; Gray, A.L.; Vladar, E.K.; Smith, B.E.; Freeman, K.G.; Malcolm, K.C.; Epperson, L.E.; Hasan, N.A.; Hendrix, J.; Callahan, K.; Walton, K.; Vestal, B.; Wheeler, E.; Rysavy, N.M.; Poch, K.; Caceres, S.; Lovell, V.K.; Hisert, K.B.; de Moura, V.C.; Chatterjee, D.; De, P.; Weakly, N.; Martiniano, S.L.; Lynch, D.A.; Daley, C.L.; Strong, M.; Jia, F.; Hatfull, G.F.; Davidson, R.M. Host and pathogen response to bacteriophage engineered against Mycobacterium abscessus lung infection. Cell. 2022, 185(11), 1860-1874.e12. [CrossRef]
- Le, T.; Nang, S.C.; Zhao, J.; Yu, H. H.; Li, J.; Gill, J. J.; Liu, M.; Aslam, S. Therapeutic potential of intravenous phage as standalone therapy for recurrent drug-resistant urinary tract infections. Antimicrob Agents Chemother. 2023, 67(4), e0003723. [CrossRef]
- Międzybrodzki, R.; Borysowski, J.; Weber-Dąbrowska, B.; Fortuna, W.; Letkiewicz, S.; Szufnarowski, K.; Pawełczyk, Z.; Rogóż, P.; Kłak, M.; Wojtasik, E.; Górski, A. Clinical aspects of phage therapy. Adv. Virus Res. 2012, 83, 73-121. [CrossRef]
- Łobocka, M.; Hejnowicz, M.S.; Dąbrowski, K.; Gozdek, A.; Kosakowski, J.; Witkowska, M.; Ulatowska, M.I.; Weber-Dąbrowska, B.; Kwiatek, M.; Parasion, S.; Gawor, J.; Kosowska, H.; Głowacka, A. Genomics of staphylococcal Twort-like phages--potential therapeutics of the post-antibiotic era. Adv. Virus Res. 2012, 83, 143-216. [CrossRef]
- Pescovitz, M.D.; Torgerson, T.R.; Ochs, H.D.; Ocheltree. E.; McGee, P.; Krause-Steinrauf, H.; Lachin, J. M.; Canniff, J.; Greenbaum, C.; Herold, K. C.; Skyler, J. S.; Weinberg, A.; Type 1 Diabetes TrialNet Study Group. Effect of rituximab on human in vivo antibody immune responses. J. Allergy Clin. Immunol. 2011, 128(6), 1295-1302. [CrossRef]
- Adams, M.H. Methods of study of bacterial viruses. In Bacteriophages; Adams, M.H., Eds.; Interscience: New York, NY, USA, 1959; pp. 443–522.
- Dan, J.M.; Lehman, S.M.; Al-Kolla, R.; Penziner, S.; Afshar, K.; Yung, G.; Golts, E.; Law, N.; Logan, C.; Kovach, Z.; Mearns, G.; Schooley, R.T.; Aslam, S.; Crotty, S. Development of host immune response to bacteriophage in a lung transplant recipient on adjunctive phage therapy for a multidrug-resistant pneumonia. J. Infect. Dis. 2023, 227(3), 311-316. [CrossRef]
- Dedrick, R.M.; Freeman, K.G.; Nguyen, J.A.; Bahadirli-Talbott, A.; Smith, B.E.; Wu, A.E.; Ong, A.S.; Lin, C.T.; Ruppel, L.C.; Parrish, N.M.; Hatfull, G.F.; Cohen, K.A. Potent antibody-mediated neutralization limits bacteriophage treatment of a pulmonary Mycobacterium abscessus infection. Nat. Med. 2021, 27, 1357–1361. [CrossRef]
- Dedrick, R.M.; Freeman, K.G.; Nguyen, J.A.; Bahadirli-Talbott, A.; Cardin, M.E.; Cristinziano, M.; Smith, B.E.; Jeong, S.; Ignatius, E.H.; Lin, C.T.; Cohen, K.A.; Hatfull, G.F. Nebulized bacteriophage in a patient with refractory Mycobacterium abscessus lung disease. Open Forum Infect. Dis. 2022, 9(7), ofac194. [CrossRef]
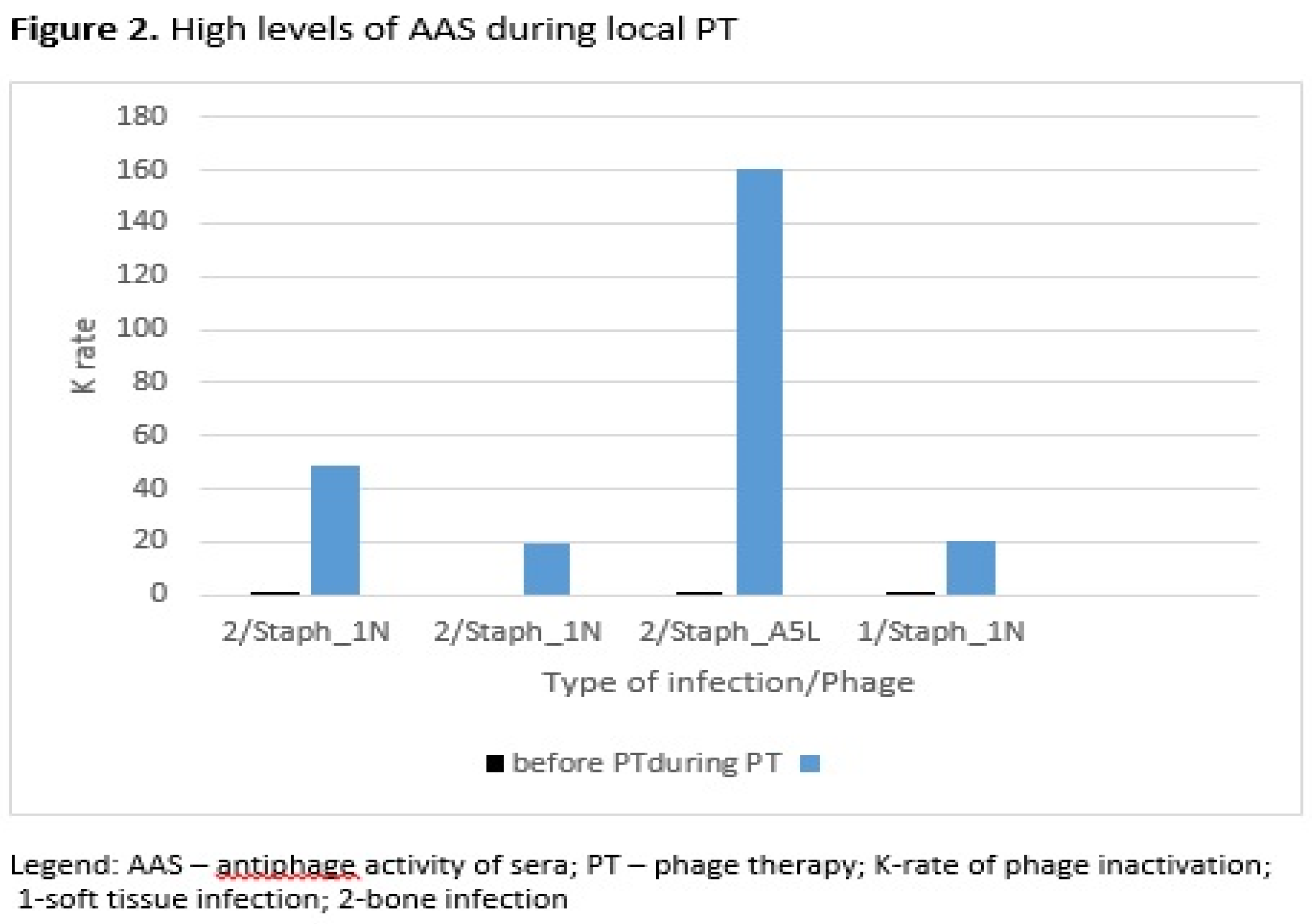
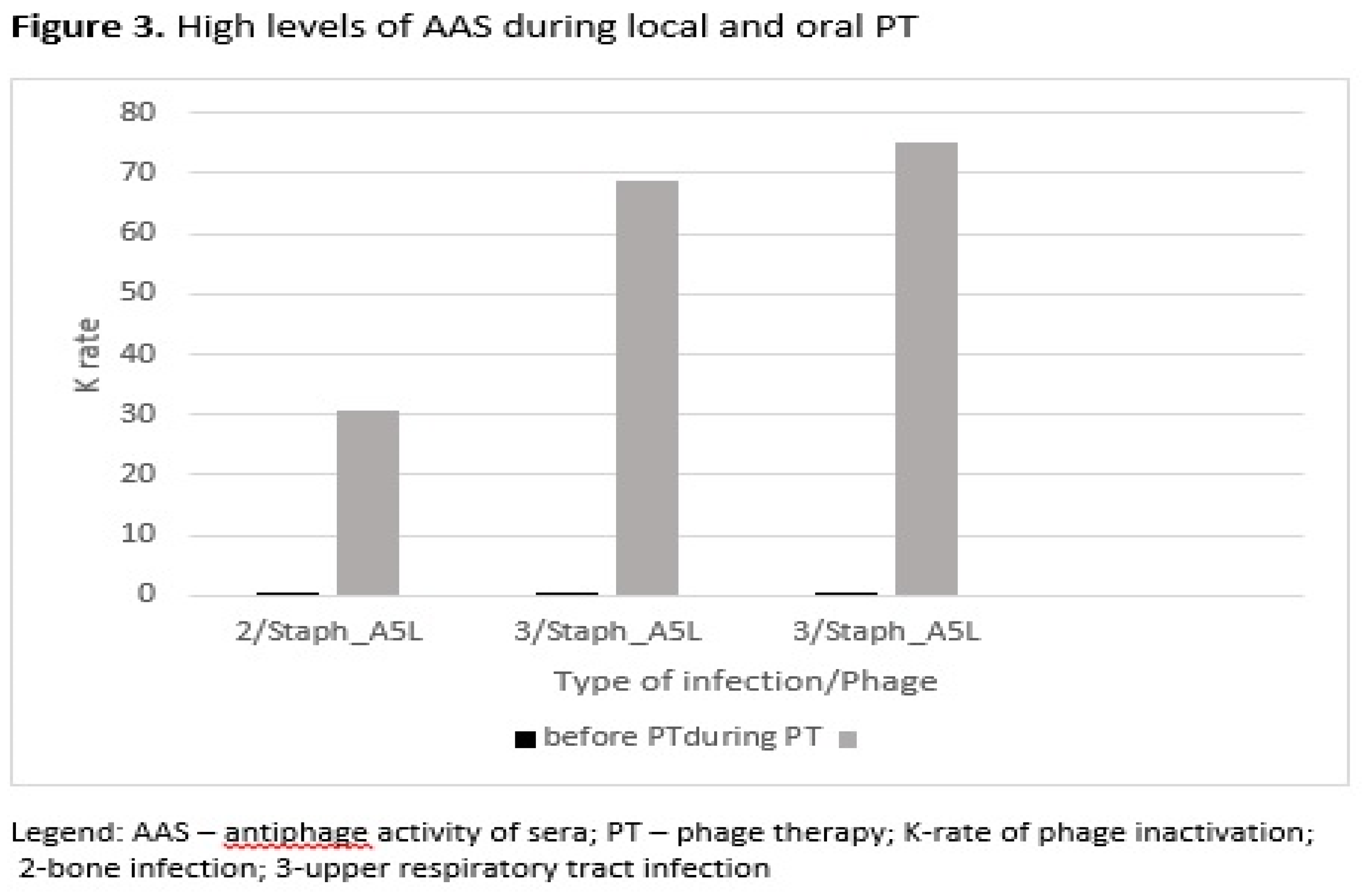
| Patient No. | Type of infection a |
Phages used in PT |
K before PT | K during PT |
Days of PT |
Clinical outcome of PT b |
|---|---|---|---|---|---|---|
| 1 | 1 | Staph_1N | 0.27 | 0.36 | 14 | D |
| 2 | 2 | Staph_1N | 0.15 | 0.29 | 18 | F |
| 3 | 3 | Staph_1N | 0.19 | 0.67 | 18 | F |
| 4 | 2 | Staph_A5L | 0.04 | 0.38 | 21 | F |
| 5 | 2 | Staph_1N | 0.05 | 48.86 | 21 | E |
| 6 | 2 | Staph_1N | 0.00 | 19.51 | 28 | D |
| 7 | 2 | Staph_1N | 0.02 | 0.02 | 28 | C |
| 8 | 2 | Staph_A5L | 0.25 | 160.08 | 35 | D |
| 9 | 1 | Staph_A5L | 0.05 | 0.25 | 56 | D |
| 10 | 1 | Staph_1N | 0.36 | 20.15 | 56 | C |
| 11 | 2 | Staph_1N | 0.02 | 143.12 | 2.5 month after PT | C |
| Patient No. | Type of infection a |
Phages used in PT | K before PT |
K during PT |
Days of PT |
Clinical outcome of PT b |
|---|---|---|---|---|---|---|
| 1 | 1 | Staph_1N | 0.06 | 0.32 | 12 | E |
| 2 | 1 | Staph_A5L | 0.14 | 0.15 | 14 | E |
| 3 | 1 | Staph_A5L | 0.47 | 2.59 | 14 | B |
| 4 | 1 | Staph_1N | 0.08 | 0.10 | 14 | B |
| 5 | 3 | Staph_A5L | 0.03 | 0.72 | 14 | C |
| 6 | 1 | Staph_1N | 0.18 | 0.12 | 17 | B |
| 7 | 3 | Staph_1N | 0.02 | 1.42 | 17 | E |
| 8 | 1 | Staph_1N | 0.02 | 0.27 | 18 | A |
| 9 | 2 | Staph_A5L | 0.52 | 30.56 | 21 | C |
| 10 | 2 | Staph_A5L | 0.03 | 0.34 | 24 | D |
| 11 | 1 | Staph_A5L | 0.26 | 2.62 | 28 | C |
| 12 | 3 | Staph_A5L | 0.02 | 68.94 | 30 | E |
| 13 | 3 | Staph_1N | 0.34 | 0.53 | 47 | F |
| 14 | 1 | Staph_1N | 0.46 | 0.58 | 55 | F |
| 15 | 3 | Staph_A5L | 0.11 | 75.12 | 56 | E |
| 16 | 2 | Staph_1N | 0.14 | 71.79 | 1 month after PT | D |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




