Submitted:
27 November 2024
Posted:
27 November 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
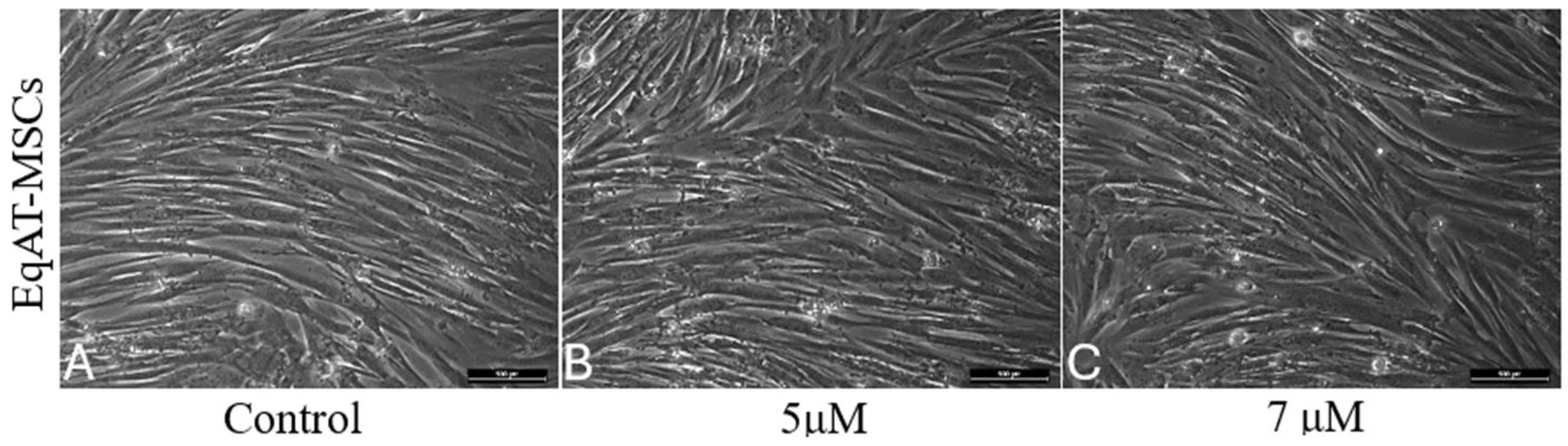
2.1. Morphological changes
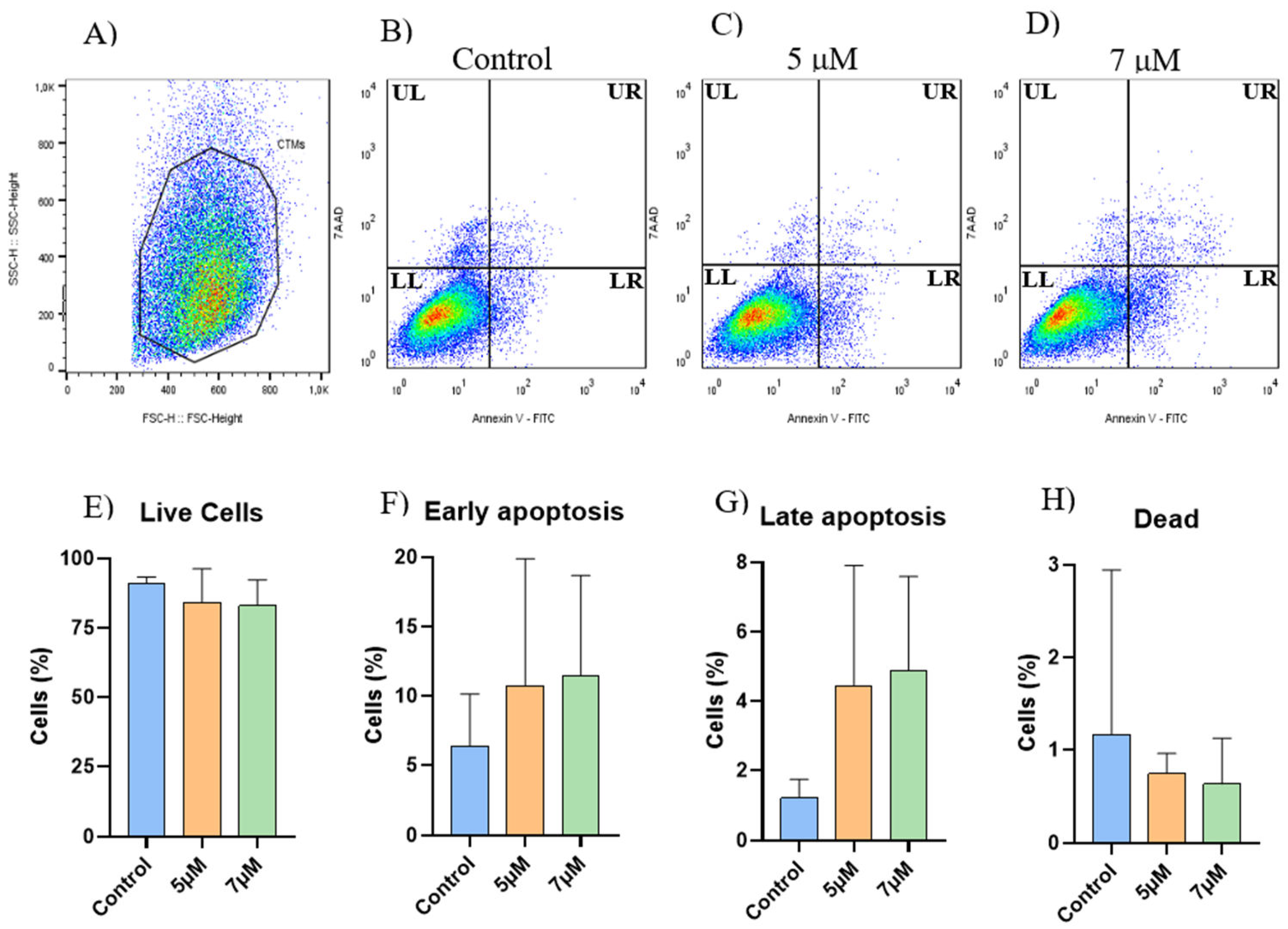
2.2. Apoptosis Assay
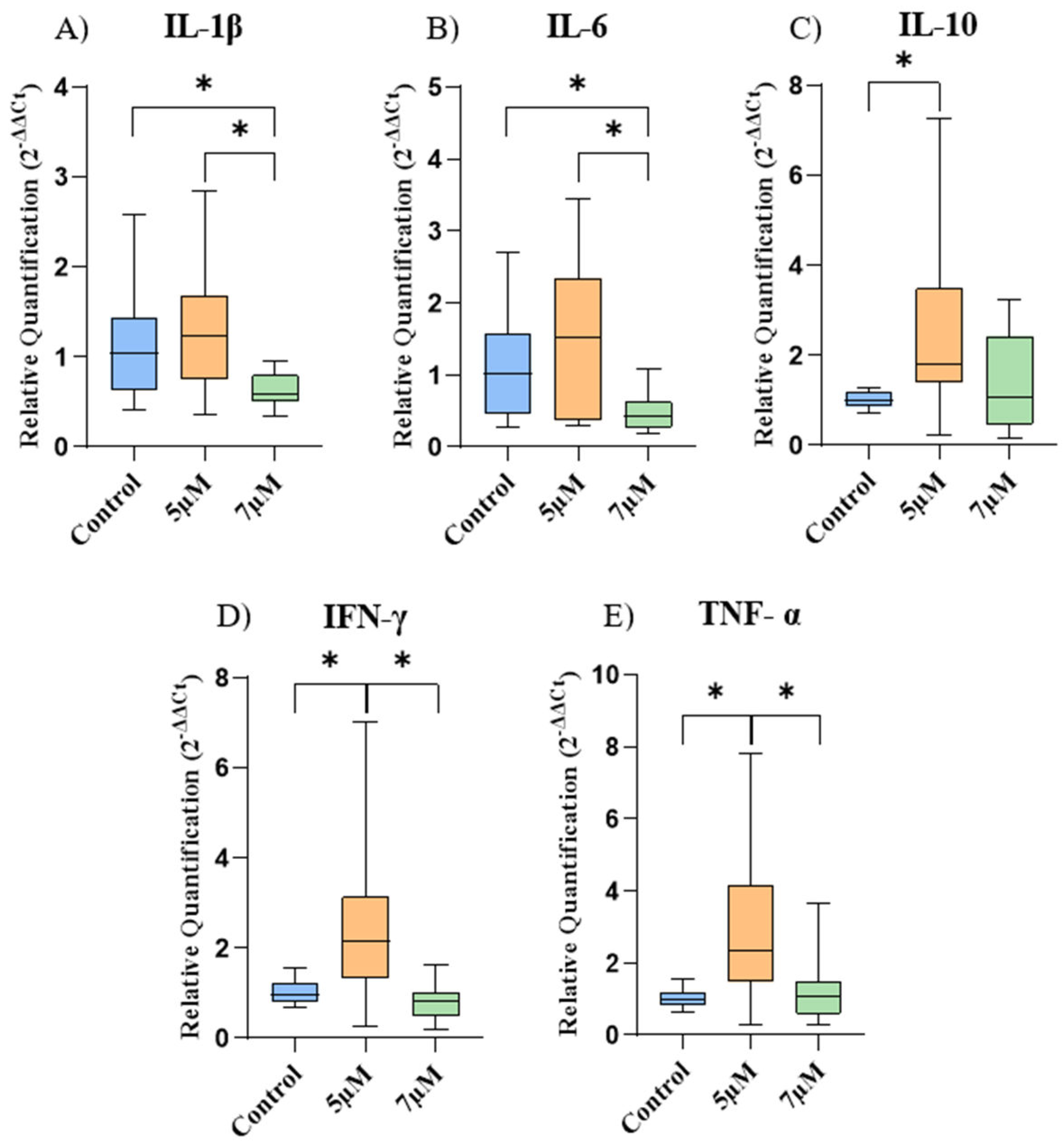
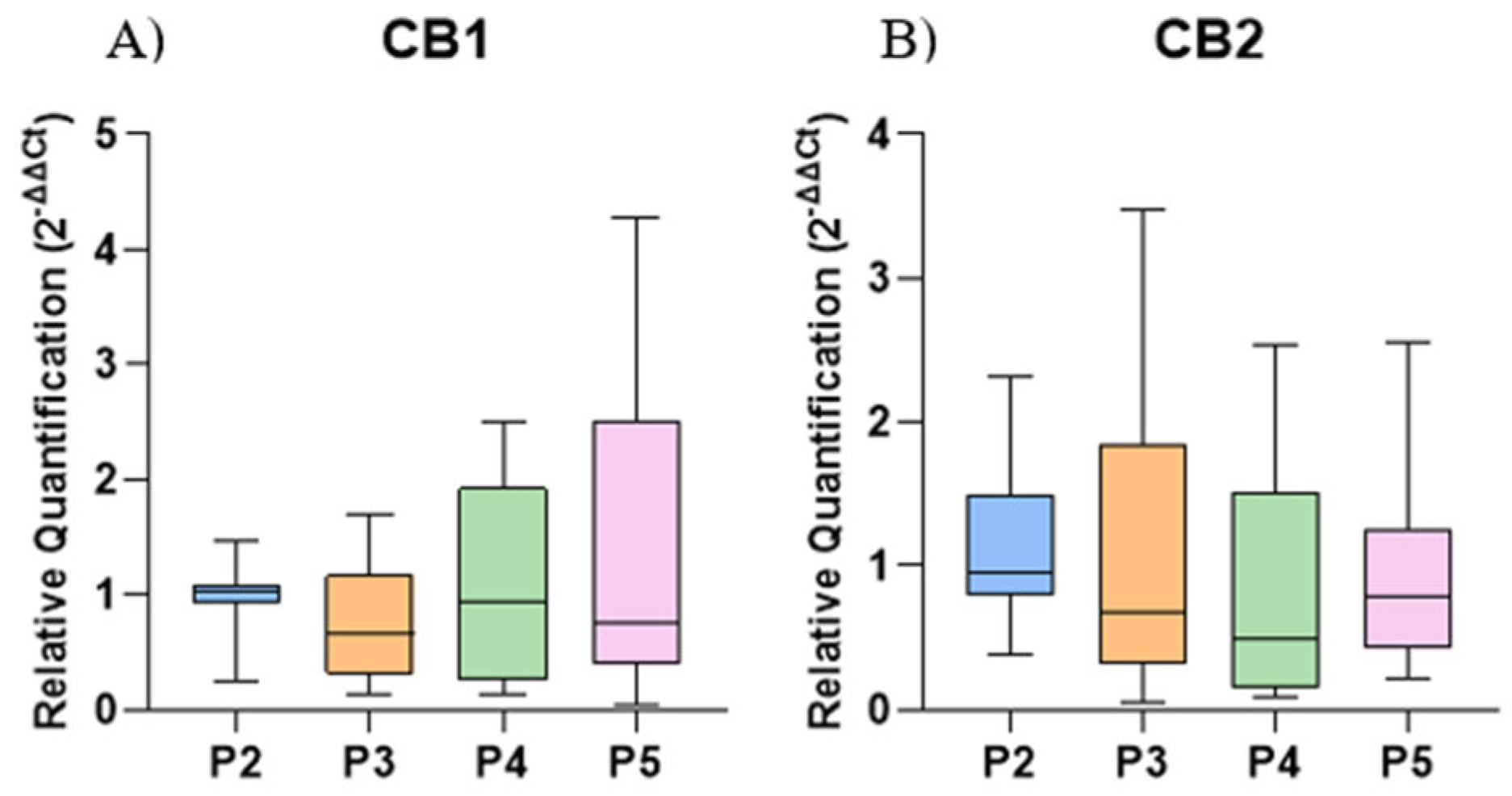
2.3. Gene Expression
3. Discussion
4. Materials and Methods
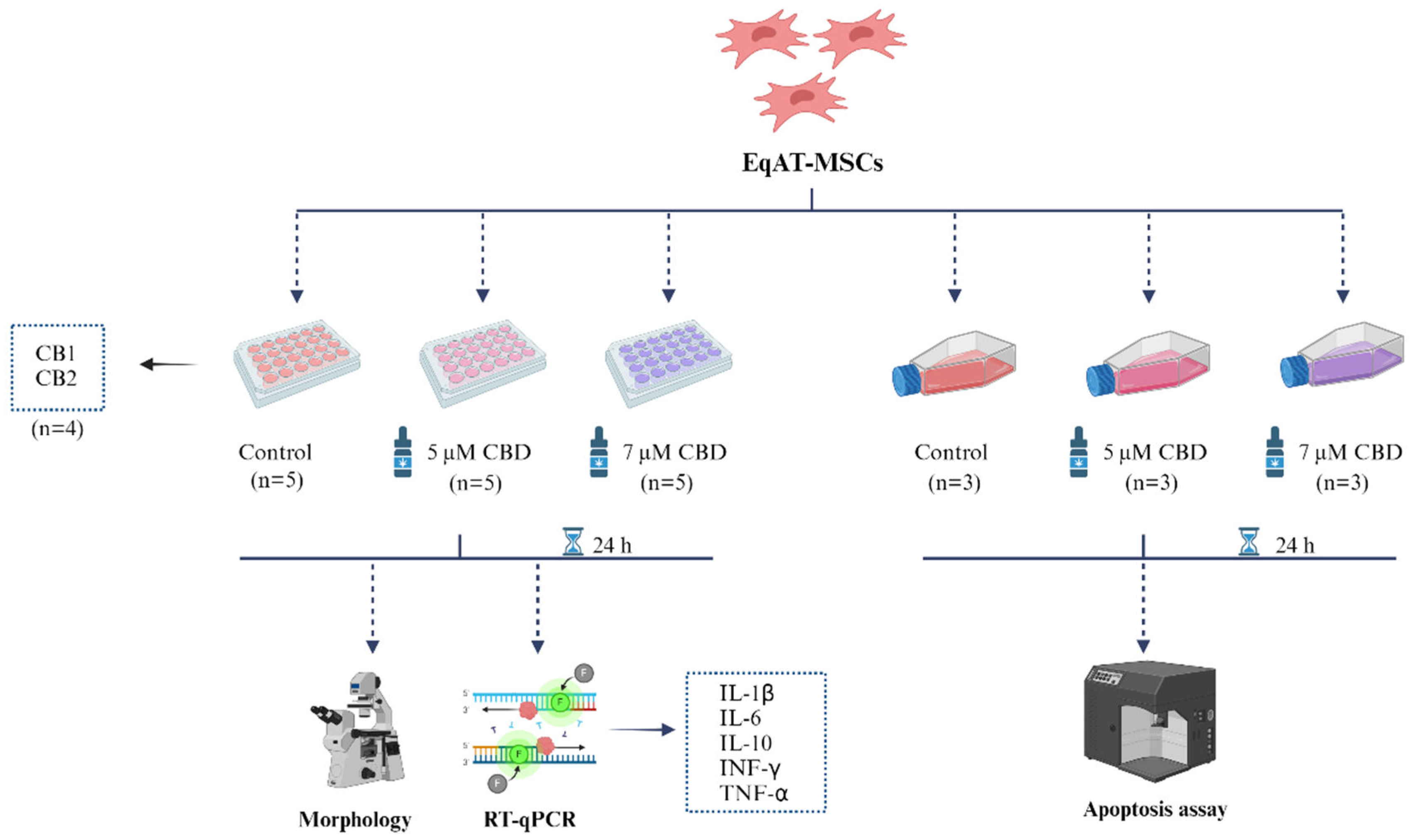
4.1. Experimental design
4.2. EqAT-MSCs
4.3. CBD-Rich Cannabis Extract
4.4. EqAT-MSCs primed with CBD-rich cannabis extract
4.5. Morphological Evaluation
4.6. Apoptosis Assay
4.7. Gene Expression
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Data Availability Statement
Conflicts of Interest
References
- Andrzejewska, A.; Dabrowska, S.; Lukomska, B.; Janowski, M. Mesenchymal stem cells for neurological disorders. Adv. Sci. 2021, 8, 2002944. [CrossRef]
- Fan, X.L.; Zhang, Y.; Li, X.; Fu, Q.L. Mechanisms underlying the protective effects of mesenchymal stem cell-based therapy. Cell. Mol. Life Sci. 2020, 77, 2771-2794. [CrossRef]
- Alvites, R.; Branquinho, M.; Sousa, A.C.; Lopes, B.; Sousa, P.; Maurício, A.C. Mesenchymal stem/stromal cells and their paracrine activity—immunomodulation mechanisms and how to influence the therapeutic potential. Pharmaceutics 2022, 14, 381. [CrossRef]
- Chen, P.M.; Liu, K.J.; Hsu, P.J.; Wei, C.F.; Bai, C.H.; Ho, L.J.; Sytwu, H.K.; Yen, B.L. Induction of immunomodulatory monocytes by human mesenchymal stem cell-derived hepatocyte growth factor through ERK1/2. J. Leukoc. Biol. 2014, 96, 295-303. [CrossRef]
- Maličev, E.; Jazbec, K. An overview of mesenchymal stem cell heterogeneity and concentration. Pharmaceuticals 2024, 17, 350. [CrossRef]
- Smith, R.K.; Garvican, E.R.; Fortier, L.A. The current ‘state of play’of regenerative medicine in horses: what the horse can tell the human. Regen. Med. 2014, 9, 673-685. [CrossRef]
- Ribitsch, I.; Baptista, P.M.; Consiglio, A.L.; Melotti, L.; Patruno, M.; Jenner, F.; Feichter, E.S.; Dutton, L.C.; Connolly, D.J.; Steenbeek, F.G.V.; Dudhia, J.; Penning, L.C. Large animal models in regenerative medicine and tissue engineering: to do or not to do. Front. Bioeng. Biotechnol. 2020, 8, 972. [CrossRef]
- Haider, K.H. Priming mesenchymal stem cells to develop “super stem cells”. World J. Stem Cells 2024, 16, 623. [CrossRef]
- Noronha, N.C.; Mizukami, A.; Oliveira, C.C.; Cominal, J.G.; Rocha, J.L.M.; Covas, D.T.; Swiech, K.; Malmegrim, K.C.R. Priming approaches to improve the efficacy of mesenchymal stromal cell-based therapies. Stem cell Res. Ther. 2019, 10, 131. [CrossRef]
- Al-Mrahleh, M.; Matar, S.; Jafar, H.; Wehaibi, S.; Aslam, N.; Awidi, A. Human Wharton’s jelly-derived mesenchymal stromal cells primed by tumor necrosis factor-α and interferon-γ modulate the innate and adaptive immune cells of type 1 diabetic patients. Front. Immunol. 2021, 12, 732549. [CrossRef]
- Amorim, R.M.; Clark, K.C.; Walker, N.J.; Kumar, P.; Herout, K.; Borjesson, D.L.; Wang, A. Placenta-derived multipotent mesenchymal stromal cells: a promising potential cell-based therapy for canine inflammatory brain disease. Stem Cell Res. Ther. 2020, 11, 304. [CrossRef]
- Barrachina, L.; Remacha, A.R.; Romero, A.; Vázquez, F.J.; Albareda, J.; Prades, M.; Gosálvez, J.; Roy, R.; Zaragoza, P.; Martín-Burriel, I.; Rodellar, C. Priming equine bone marrow-derived mesenchymal stem cells with proinflammatory cytokines: implications in immunomodulation–immunogenicity balance, cell viability, and differentiation potential. Stem Cells Dev. 2017, 26, 15-24. [CrossRef]
- Mohammadpour, H.; Pourfathollah, A.A.; Zarif, M.N.; Hashemi, S.M. Increasing proliferation of murine adipose tissue-derived mesenchymal stem cells by TNF-α plus IFN-γ. Immunopharmacol. Immunotoxicol. 2016, 38, 68-76. [CrossRef]
- Seo, Y.; Shin, T.H.; Kim, H.S. Current strategies to enhance adipose stem cell function: an update. Int. J. Mol. Sci. 2019, 20, 3827. [CrossRef]
- Ferreira, L.V.O.; Kamura, B.C.; Oliveira, J.P.M.; Chimenes, N.D.; Carvalho, M.; Santos, L.A.; Dias-Melicio, L.A.; Amorim, R.L.; Amorim, R.M. In vitro transdifferentiation potential of equine mesenchymal stem cells into Schwann-like cells. Stem Cells Dev. 2023, 32, 422-432. [CrossRef]
- Yang, Y.; Lee, E.H.; Yang, Z. Hypoxia-conditioned mesenchymal stem cells in tissue regeneration application. Tissue Eng. Part B Rev. 2022, 28, 966-977. [CrossRef]
- Iffland, K.; Grotenhermen, F. An update on safety and side effects of cannabidiol: a review of clinical data and relevant animal studies. Cannabis Cannabinoid Res. 2017, 2, 139-154. [CrossRef]
- Martínez, V.; De-Hond, A.I.; Borrelli, F.; Capasso, R.; Castillo, M.D.; Abalo, R. Cannabidiol and other non-psychoactive cannabinoids for prevention and treatment of gastrointestinal disorders: useful nutraceuticals? Int. J. Mol. Sci. 2020, 21, 3067. [CrossRef]
- Martini, S.; Gemma, A.; Ferrari, M.; Cosentino, M.; Marino, F. Effects of cannabidiol on innate immunity: experimental evidence and clinical relevance. Int. J. Mol. Sci. 2023, 24, 3125. [CrossRef]
- Fellous, T.; Maio, F.; Kalkan, H.; Carannante, B.; Boccella, S.; Petrosino, S.; Maione, S.; Marzo, V.; Iannotti, F.A. Phytocannabinoids promote viability and functional adipogenesis of bone marrow-derived mesenchymal stem cells through different molecular targets. Biochem. Pharmacol. 2020, 175, 113859. [CrossRef]
- Li, L.; Feng, J.; Sun, L.; Xuan, Y.W.; Wen, L.; Li, Y.X.; Yang, S.; Zhu, B.; Tian, X.Y.; Li, S.; Zhao, L.S.; Dang, R.J.; Jiao, T.; Zhang, H.S.; Wen, N. Cannabidiol promotes osteogenic differentiation of bone marrow mesenchymal stem cells in the inflammatory microenvironment via the CB2-dependent p38 MAPK signaling pathway. Int. J. Stem Cells 2022, 15, 405-414. [CrossRef]
- Libro, R.; Scionti, D.; Diomede, F.; Marchisio, M.; Grassi, G.; Pollastro, F.; Piattelli, A.; Bramanti, P.; Mazzon, E.; Trubiani, O. Cannabidiol modulates the immunophenotype and inhibits the activation of the inflammasome in human gingival mesenchymal stem cells. Front. Physiol. 2016, 7, 559. [CrossRef]
- Henson, J.D.; Vitetta, L.; Quezada, M.; Hall, S. Enhancing endocannabinoid control of stress with cannabidiol. J. Clin. Med. 2021, 10, 5852. [CrossRef]
- Stasiulewicz, A.; Znajdek, K.; Grudzién, M.; Pawinski, T.; Sulkowska, J.I. A guide to targeting the endocannabinoid system in drug design. Int. J. Mol. Sci. 2020, 21, 2778. [CrossRef]
- Lee, B.C.; Kang, K.S. Functional enhancement strategies for immunomodulation of mesenchymal stem cells and their therapeutic application. Stem Cell Res. Ther. 2020, 11, 397. [CrossRef]
- Li, M.; Jiang, Y.; Hou, Q.; Zhao, Y.; Zhong, L.; Fu, X. Potential pre-activation strategies for improving therapeutic efficacy of mesenchymal stem cells: current status and future prospects. Stem Cell Res. Ther. 2022, 13, 146. [CrossRef]
- Kowalczuk, A.; Marycz, K.; Garbowska, K.K.; Kornicka, J.; Zadrozny, M.B.; Groborz, S. Cannabidiol (CBD) protects adipose-derived mesenchymal stem cells (ASCs) against endoplasmic reticulum stress development and its complications. Int. J. Environ. Res. Public Health 2022, 19, 10864. [CrossRef]
- Naya, N.M.; Kelly, J.; Corna, G.; Golino, M.; Abbate, A.; Toldo, S. Molecular and cellular mechanisms of action of cannabidiol. Molecules 2023, 28, 5980. [CrossRef]
- Rajan, T.S.; Giacoppo, S.; Scionti, D.; Diomede, F.; Grassi, G.; Pollastro, F.; Piattelli, A.; Bramanti, P.; Mazzon, E.; Trubiani. Cannabidiol activates neuronal precursor genes in human gingival mesenchymal stromal cells. J. Cell. Biochem. 2017, 118, 1531-1546. [CrossRef]
- Schmuhl, E.; Ramer, R.; Salamon, A.; Peters, K.; Hinz, B. Increase of mesenchymal stem cell migration by cannabidiol via activation of p42/44 MAPK. Biochem. Pharmacol. 2014, 87, 489-501. [CrossRef]
- Lu, H.C.; Mackie, K. Review of the endocannabinoid system. Biol. Psychiatry Cogn. Neurosci. Neuroimaging 2021, 6, 607-615. [CrossRef]
- Bari, M.; Tedesco, M.; Battista, N.; Pasquariello, N.; Pucci, M.; Gasperi, V.; Scaldaferri, M.L.; Farini, D.; Felici, M.; Maccarrone. Characterization of the endocannabinoid system in mouse embryonic stem cells. Stem Cells Dev. 2011, 20, 139-147. [CrossRef]
- Ruhl, T.; Karthaus, N.; Kim, B.S.; Beier, J.P. The endocannabinoid receptors CB1 and CB2 affect the regenerative potential of adipose tissue MSCs. Exp. Cell Res. 2020, 389, 111881. [CrossRef]
- Mechoulam, R.; Parker, L.A. The endocannabinoid system and the brain. Annu. Rev. Psychol. 2013, 64, 21-47. [CrossRef]
- Libro, R.; Diomede, F.; Scionti, D.; Piattelli, A.; Grassi, G.; Pollastro, F.; Bramanti, P.; Mazzon, E.; Trubiani, O. Cannabidiol modulates the expression of Alzheimer’s disease-related genes in mesenchymal stem cells. Int. J. Mol. Sci. 2016, 18, 26. [CrossRef]
- Kozela, E.; Pietr, M.; Juknat, A.; Rimmerman, N.; Levy, R.; Vogel, Z. Cannabinoids Δ9-tetrahydrocannabinol and cannabidiol differentially inhibit the lipopolysaccharide-activated NF-κB and interferon-β/STAT proinflammatory pathways in BV-2 microglial cells. J. Biol. Chem. 2010, 285, 1616-1626. [CrossRef]
- Ma, H.; Xu, F.; Liu, C.; Seeram, N.P. A network pharmacology approach to identify potential molecular targets for cannabidiol's anti-inflammatory activity. Cannabis and Cannabinoid Res. 2021, 6, 288-299. [CrossRef]
- Liu, C.; Ma, H.; Slitt, A.L.; Seeram, N.P. Inhibitory effect of cannabidiol on the activation of NLRP3 inflammasome is associated with its modulation of the P2X7 receptor in human monocytes. J. Nat. Prod. 2020, 83, 2025-2029. [CrossRef]
- Minshawi, F.; Lanvermann, S.; McKenzie, E.; Jeffery, R.; Couper, K.; Papoutsopoulou, S.; Roers, A.; Muller, W. The generation of an engineered interleukin-10 protein with improved stability and biological function. Front. Immunol. 2020, 11, 1794. [CrossRef]
- Mittal, S.K.; Roche, P.A. Suppression of antigen presentation by IL-10. Curr. Opin. Immunol. 2015, 34, 22-27. [CrossRef]
- Neumann, C.; Scheffold, A.; Rutz, S. Functions and regulation of T cell-derived interleukin-10. Semin. Immunol. 2019, 44, 101344. [CrossRef]
- Turner, S.; Barker, V.D.; Adams, A.A. Effects of cannabidiol on the in vitro lymphocyte pro-inflammatory cytokine production of senior horses. J. Equine Vet. Sci. 2021, 103, 103668. [CrossRef]
- Turner, S.; Knych, H.K.; Adams, A.A. The effects of cannabidiol on immune function and health parameters in senior horses. Vet. Immunol. Immunopathol. 2023, 257, 110549. [CrossRef]
- Li, X.; Köner, H.; Liu, X. Susceptibility to intracellular infections: contributions of TNF to immune defense. Front. Microbiol. 2020, 11,1643. [CrossRef]
- Kak, G.; Raza, M.; Tiwari, B.K. Interferon-gamma (IFN-γ): Exploring its implications in infectious diseases. Biomol. Concepts 2018, 9, 64-79. [CrossRef]
- Barberini, D.J.; Freitas, N.P.P.; Magnoni, M.S.; Maia, L.; Listoni, A.J.; Heckler, M.C.; Sudano, M.J.; Golim, M.A.; Landim-Alvarenga, F.C.; Amorim, R.M. Equine mesenchymal stem cells from bone marrow, adipose tissue and umbilical cord: immunophenotypic characterization and differentiation potential. Stem Cell Res. Ther. 2014, 5, 25. [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods 2001, 25, 402-408. [CrossRef]
| Gene | Forward | Reverse |
| CB1 | GAGCAAGGACCTGAGACATGCT | TGCGCAGTGCCTTCACAT |
| CB2 | AAAGAAGAGGCCCCGAAGTC | CCACTGGGTGATTTTCACATCA |
| IL-1β | GCAGCCATGGCAGCAGTA | ATTGCCGCTGCAGTAAGTCA |
| IL-6 | AACAACTCACCTCATCCTTCGAA | CGAACAGCTCTCAGGCTGAAC |
| IL-10 | CGGCCCAGACATCAAGGA | TCGGAGGGTCTTCAGCTTTTC |
| INF-γ | CTGTCGCCCAAAGCTAACCT | GGCCTCGAAATGGATTCTGA |
| TNF-α | TTGGATGGGCTGTACCTCATC | GGGCAGCCTTGGCCTTT |
| ACTB | CGGCGGCTCCATTCTG | CTGCTTGCTGATCCACATCTG |
| B2M | CACCCAGCAGAGAATGGAAAG | CGGATGGAACCCAGAGACA |
| GAPDH | GGCAAGTTCCATGGCACAGT | GGGCTTTCCGTTGATGACAA |
| HPRT | GCTCGAGATGTGATGAAGGAGAT | CCCCCTTGAGCACACAGAGT |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




