Submitted:
25 November 2024
Posted:
26 November 2024
You are already at the latest version
Abstract
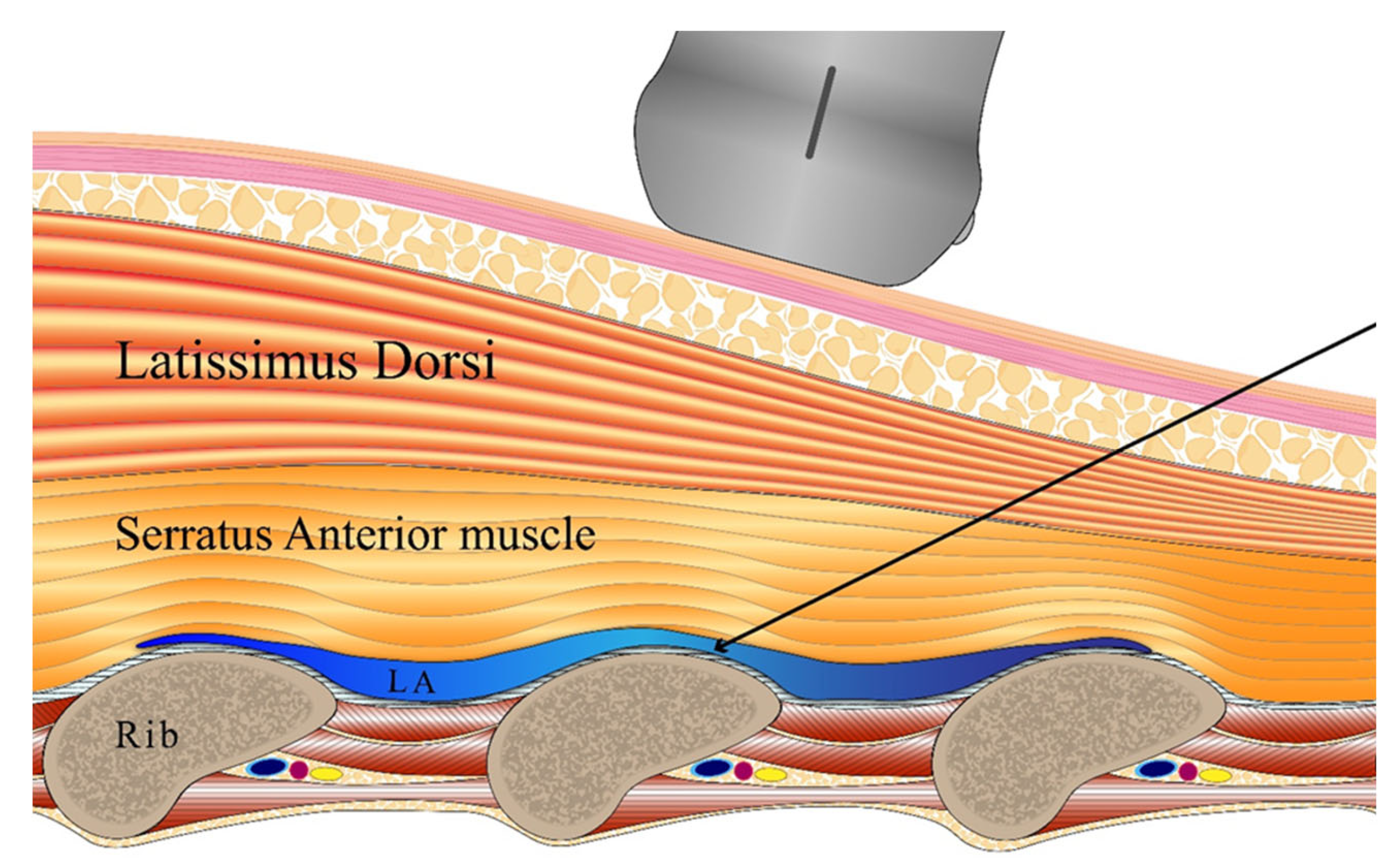
Background: Video-assisted thoracoscopic surgery (VATS) is associated with less postoperative pain than traditional open thoracotomy. However, trocar and chest tube placement may damage the intercostal nerves, causing significant discomfort. An ultrasound-guided serratus anterior plane block (SAPB) is a promising mode of pain management; this reduces the need for opioids and the associated side-effects. This study evaluated whether SAPB, compared to intravenous analgesia alone, reduces opioid consumption after thoracoscopic, lung wedge resection. Methods: In total, 22 patients undergoing VATS lung wedge resections were randomized into two groups (SAPB and control): both received intravenous patient-controlled analgesia (PCA) and one group received additional SAPB. The primary outcome was cumulative intravenous fentanyl consumption at 8 hours postoperatively. The visual analog scale (VAS) pain scores, and the incidence of postoperative complications were assessed over 48 h post-surgery. Results: Fentanyl consumption by 8 h post-surgery was significantly lower in the SAPB group than in the control group (183 ± 107 μg vs. 347 ± 202 μg, p = 0.035). Although VAS scores decreased with time in both groups, the differences were not statistically significant. The SAPB group required less opioids by 48 h. No significant between-group differences were observed in postoperative complications, including nausea and vomiting. Conclusions: SAPB effectively reduced opioid consumption after VATS lung wedge resection. SABP may serve as a valuable component of multimodal pain management.
Keywords:
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Aberle, D.R.; Adams, A.M.; Berg, C.D.; Black, W.C.; Clapp, J.D.; Fagerstrom, R.M.; Gareen, I.F.; Gatsonis, C.; Marcus, P.M.; Sicks, J.D. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med 2011, 365, 395–409. [Google Scholar] [CrossRef] [PubMed]
- de Koning, H.J.; van der Aalst, C.M.; de Jong, P.A.; Scholten, E.T.; Nackaerts, K.; Heuvelmans, M.A.; Lammers, J.J.; Weenink, C.; Yousaf-Khan, U.; Horeweg, N.; et al. Reduced Lung-Cancer Mortality with Volume CT Screening in a Randomized Trial. N Engl J Med 2020, 382, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.W.Y.; Leung, C.S.; Tsz, C.H.; Lee, B.T.Y.; Chan, H.K.; Sihoe, A.D.L. Does low-dose computed tomography screening improve lung cancer-related outcomes?—a systematic review. Video-Assisted Thoracic Surgery 2020, 5. [Google Scholar] [CrossRef]
- Nagahiro, I.; Andou, A.; Aoe, M.; Sano, Y.; Date, H.; Shimizu, N. Pulmonary function, postoperative pain, and serum cytokine level after lobectomy: a comparison of VATS and conventional procedure. Ann Thorac Surg 2001, 72, 362–365. [Google Scholar] [CrossRef]
- Neustein, S.M.; McCormick, P.J. Postoperative analgesia after minimally invasive thoracoscopy: what should we do? Can J Anaesth 2011, 58, 423-425, 425-427. [Google Scholar] [CrossRef]
- Bayman, E.O.; Parekh, K.R.; Keech, J.; Selte, A.; Brennan, T.J. A Prospective Study of Chronic Pain after Thoracic Surgery. Anesthesiology 2017, 126, 938–951. [Google Scholar] [CrossRef]
- Yie, J.C.; Yang, J.T.; Wu, C.Y.; Sun, W.Z.; Cheng, Y.J. Patient-controlled analgesia (PCA) following video-assisted thoracoscopic lobectomy: comparison of epidural PCA and intravenous PCA. Acta Anaesthesiol Taiwan 2012, 50, 92–95. [Google Scholar] [CrossRef]
- Michael A. Gropper, L.I.E., Lee A. Fleisher, Jeanine P. Wiener-Kronish. Miller's Anesthesia, 9th Edition ed.; Michael A. Gropper, L.I.E., Lee A. Fleisher, Jeanine P. Wiener-Kronish, Neal H. Cohen, Kate Leslie, Ed.; Elsevier: 2019; Volume 2, pp. 680-741.
- Steinthorsdottir, K.J.; Wildgaard, L.; Hansen, H.J.; Petersen, R.H.; Wildgaard, K. Regional analgesia for video-assisted thoracic surgery: a systematic review. Eur J Cardiothorac Surg 2014, 45, 959–966. [Google Scholar] [CrossRef]
- Gottschalk, A.; Cohen, S.P.; Yang, S.; Ochroch, E.A. Preventing and treating pain after thoracic surgery. Anesthesiology 2006, 104, 594–600. [Google Scholar] [CrossRef]
- Khalil, A.E.; Abdallah, N.M.; Bashandy, G.M.; Kaddah, T.A. Ultrasound-Guided Serratus Anterior Plane Block Versus Thoracic Epidural Analgesia for Thoracotomy Pain. J Cardiothorac Vasc Anesth 2017, 31, 152–158. [Google Scholar] [CrossRef]
- Hotta, K.; Endo, T.; Taira, K.; Sata, N.; Inoue, S.; Takeuchi, M.; Seo, N.; Endo, S. Comparison of the analgesic effects of continuous extrapleural block and continuous epidural block after video-assisted thoracoscopic surgery. J Cardiothorac Vasc Anesth 2011, 25, 1009–1013. [Google Scholar] [CrossRef] [PubMed]
- Blanco, R.; Parras, T.; McDonnell, J.G.; Prats-Galino, A. Serratus plane block: a novel ultrasound-guided thoracic wall nerve block. Anaesthesia 2013, 68, 1107–1113. [Google Scholar] [CrossRef] [PubMed]
- Mayes, J.; Davison, E.; Panahi, P.; Patten, D.; Eljelani, F.; Womack, J.; Varma, M. An anatomical evaluation of the serratus anterior plane block. Anaesthesia 2016, 71, 1064–1069. [Google Scholar] [CrossRef] [PubMed]
- Madabushi, R.; Tewari, S.; Gautam, S.K.; Agarwal, A.; Agarwal, A. Serratus anterior plane block: a new analgesic technique for post-thoracotomy pain. Pain Physician 2015, 18, E421–424. [Google Scholar]
- Hards, M.; Harada, A.; Neville, I.; Harwell, S.; Babar, M.; Ravalia, A.; Davies, G. The effect of serratus plane block performed under direct vision on postoperative pain in breast surgery. J Clin Anesth 2016, 34, 427–431. [Google Scholar] [CrossRef]
- Kazior, M.R.; King, A.B.; Lopez, M.G.; Billings, F.T.t.; Costello, W.T. Serratus anterior plane block for minimally invasive valve surgery thoracotomy pain. J Clin Anesth 2019, 56, 48–49. [Google Scholar] [CrossRef]
- Ökmen, K.; Ökmen, B.M. The efficacy of serratus anterior plane block in analgesia for thoracotomy: a retrospective study. J Anesth 2017, 31, 579–585. [Google Scholar] [CrossRef]
- Baamer, R.M.; Iqbal, A.; Lobo, D.N.; Knaggs, R.D.; Levy, N.A.; Toh, L.S. Utility of unidimensional and functional pain assessment tools in adult postoperative patients: a systematic review. Br J Anaesth 2022, 128, 874–888. [Google Scholar] [CrossRef]
- Myles, P.S.; Troedel, S.; Boquest, M.; Reeves, M. The pain visual analog scale: is it linear or nonlinear? Anesth Analg 1999, 89, 1517–1520. [Google Scholar] [CrossRef]
- Myles, P.S.; Urquhart, N. The linearity of the visual analogue scale in patients with severe acute pain. Anaesth Intensive Care 2005, 33, 54–58. [Google Scholar] [CrossRef]
- Kim, D.H.; Oh, Y.J.; Lee, J.G.; Ha, D.; Chang, Y.J.; Kwak, H.J. Efficacy of Ultrasound-Guided Serratus Plane Block on Postoperative Quality of Recovery and Analgesia After Video-Assisted Thoracic Surgery: A Randomized, Triple-Blind, Placebo-Controlled Study. Anesth Analg 2018, 126, 1353–1361. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Li, Y.; Zhang, Y.; Fang, X. Effects of serratus anterior plane block for postoperative analgesia after thoracoscopic surgery compared with local anesthetic infiltration: a randomized clinical trial. J Pain Res 2019, 12, 2411–2417. [Google Scholar] [CrossRef] [PubMed]
- Semyonov, M.; Fedorina, E.; Grinshpun, J.; Dubilet, M.; Refaely, Y.; Ruderman, L.; Koyfman, L.; Friger, M.; Zlotnik, A.; Klein, M.; et al. Ultrasound-guided serratus anterior plane block for analgesia after thoracic surgery. J Pain Res 2019, 12, 953–960. [Google Scholar] [CrossRef] [PubMed]
- Piracha, M.M.; Thorp, S.L.; Puttanniah, V.; Gulati, A. "A Tale of Two Planes": Deep Versus Superficial Serratus Plane Block for Postmastectomy Pain Syndrome. Reg Anesth Pain Med 2017, 42, 259–262. [Google Scholar] [CrossRef]
- Abdallah, F.W.; Cil, T.; MacLean, D.; Madjdpour, C.; Escallon, J.; Semple, J.; Brull, R. Too Deep or Not Too Deep?: A Propensity-Matched Comparison of the Analgesic Effects of a Superficial Versus Deep Serratus Fascial Plane Block for Ambulatory Breast Cancer Surgery. Reg Anesth Pain Med 2018, 43, 480–487. [Google Scholar] [CrossRef]
- Johnston, D.F.; Black, N.D.; O'Halloran, R.; Turbitt, L.R.; Taylor, S.J. Cadaveric findings of the effect of rib fractures on spread of serratus plane injections. Can J Anaesth 2019, 66, 738–739. [Google Scholar] [CrossRef]
- Almeida, C.R. Serratus anterior plane block for posterior rib fractures: why and when may it work? Reg Anesth Pain Med 2021, 46, 835–836. [Google Scholar] [CrossRef]
- Ökmen, K.; Metin Ökmen, B. Evaluation of the effect of serratus anterior plane block for pain treatment after video-assisted thoracoscopic surgery. Anaesth Crit Care Pain Med 2018, 37, 349–353. [Google Scholar] [CrossRef]
- Park, M.H.; Kim, J.A.; Ahn, H.J.; Yang, M.K.; Son, H.J.; Seong, B.G. A randomised trial of serratus anterior plane block for analgesia after thoracoscopic surgery. Anaesthesia 2018, 73, 1260–1264. [Google Scholar] [CrossRef]
- Viti, A.; Bertoglio, P.; Zamperini, M.; Tubaro, A.; Menestrina, N.; Bonadiman, S.; Avesani, R.; Guerriero, M.; Terzi, A. Serratus plane block for video-assisted thoracoscopic surgery major lung resection: a randomized controlled trial. Interact Cardiovasc Thorac Surg 2020, 30, 366–372. [Google Scholar] [CrossRef]
- Rosenberger, D.C.; Pogatzki-Zahn, E.M. Chronic post-surgical pain - update on incidence, risk factors and preventive treatment options. BJA Educ 2022, 22, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Kaplowitz, J.; Papadakos, P.J. Acute pain management for video-assisted thoracoscopic surgery: an update. J Cardiothorac Vasc Anesth 2012, 26, 312–321. [Google Scholar] [CrossRef] [PubMed]
- Katz, J.; Jackson, M.; Kavanagh, B.P.; Sandler, A.N. Acute pain after thoracic surgery predicts long-term post-thoracotomy pain. Clin J Pain 1996, 12, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Feray, S.; Lubach, J.; Joshi, G.P.; Bonnet, F.; Van de Velde, M. PROSPECT guidelines for video-assisted thoracoscopic surgery: a systematic review and procedure-specific postoperative pain management recommendations. Anaesthesia 2022, 77, 311–325. [Google Scholar] [CrossRef]
- Finnerty, D.T.; McMahon, A.; McNamara, J.R.; Hartigan, S.D.; Griffin, M.; Buggy, D.J. Comparing erector spinae plane block with serratus anterior plane block for minimally invasive thoracic surgery: a randomised clinical trial. Br J Anaesth 2020, 125, 802–810. [Google Scholar] [CrossRef]
| Control Group (11) | SAPB Group (10) | p-Value | |
|---|---|---|---|
| Age (years) | 52.3 ± 19.8 | 46.1 ± 15.7 | 0.442 |
| Sex (male, %) | 8 (73%) | 5 (50%) | 0.387 |
| Height (cm) | 166.9 ± 8.2 | 166.2 ± 10.8 | 0.867 |
| Weight (kg) | 63.1 ± 9.1 | 66.5 ± 11.9 | 0.458 |
| BMI (kg/m2) | 22.6 ± 2.9 | 24.2 ± 4.3 | 0.347 |
| Anesthesia time | 90.5 ± 31.2 | 72.7 ± 20.7 | 0.145 |
| Operation time | 58.6 ± 30.6 | 38.2 ± 15.4 | 0.720 |
| Number of ports | 2.8 ± 0.4 | 2.8 ± 0.4 | 0.920 |
| Bleeding volume | 40 ± 37.9 | 21 ± 24.2 | 0.185 |
| Intraoperative fluid, mL | 353.6 ± 189.4 | 350.5 ± 262.9 | 0.976 |
| ASA status score | |||
| 1 | 5 (45%) | 4 (40%) | 0.632 |
| 2 | 6 (55%) | 5 (50%) | |
| 3 | 1 (10%) | ||
| Operator | |||
| L | 6 | 4 | 0.515 |
| S | 5 | 6 |
| Control Group | SAPB Group | p-Value | |
|---|---|---|---|
| Total fentanyl consumption, μg | |||
| 4 h | 264.0 ± 168.9 | 132.6 ± 91.2 | 0.042 |
| 8 h | 346.9 ± 202.6 | 183.3 ± 107.1 | 0.035 |
| 12 h | 410.5 ± 226.8 | 208.8 ± 141.6 | 0.260 |
| 24 h | 579.5 ± 294 | 339.9 ± 206 | 0.045 |
| 48 h | 679.6 ± 319 | 383.7 ± 231.9 | 0.013 |
| Visual analog pain scores Resting |
|||
| PACU | 3.3 ± 0.9 | 2.6 ± 1.3 | 0.174 |
| 4 h | 3.1 ± 1.4 | 2.5 ± 1.2 | 0.306 |
| 8 h | 3.5 ± 1.2 | 2.9 ± 1 | 0.269 |
| 12 h | 3.4 ± 1.1 | 2.5 ± 1.4 | 0.139 |
| 24 h | 3 ± 1.2 | 2.2 ± 1 | 0.117 |
| 48 h | 2.9 ± 1.7 | 1.6 ± 0.7 | 0.036 |
| During coughing | |||
| PACU | 4.5 ± 0.9 | 3.5 ± 1.6 | 0.077 |
| 4 h | 4.1 ± 1.5 | 3.6 ± 1.5 | 0.466 |
| 8 h | 4 ± 1.4 | 3.1 ± 1.2 | 0.131 |
| 12 h | 3.9 ± 1.6 | 2.7 ± 0.9 | 0.049 |
| 24 h | 3.7 ± 2 | 2.3 ± 0.8 | 0.046 |
| 48 h | 3.5 ± 1.8 | 2 ± 1.2 | 0.029 |
| Tramadol administrations | |||
| 0-24 h | 5 | 6 | 0.258 |
| 24-48 h | 2 | 2 | 0.916 |
| Control Group | SAPB Group | p-Value | |
|---|---|---|---|
| Number of ramosetron administrations | |||
| 0–24 | 0 | 0 | N/A |
| 24–48 | 0 | 0 | N/A |
| Postoperative mean pressure [mmHg] | |||
| PACU | 90.1 ± 7.8 | 97.4 ± 17.9 | 0.237 |
| 8 h | 81.8 ± 12 | 87.8 ± 12.3 | 0.268 |
| 24 h | 84 ± 7.7 | 84.5 ± 12.1 | 0.913 |
| SPO2 at 8 h post-surgery | 98.5 ± 0.4 | 98.8 ± 0.7 | 0.389 |
| Continuous use of an oxygen mask | 1 | 1 | N/A |
| Side-effects of local anesthetics | 0 | 0 | N/A |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




