Submitted:
06 November 2024
Posted:
07 November 2024
You are already at the latest version
Abstract
Background/Objectives: Pentacyclic Triterpenoids are increasingly studied as anti-cancer agents with many advantages comparative to synthetic chemotherapeutics. The aim of this study was to prepare liposomal and nanostructured lipid formulations including a standardized extract of silver birch (Betula pendula) outer bark (TTs) and to evaluate their potential as anti-cancer agents in vitro, using Melanoma B16-F10 and Walker carcinoma cells. Methods: Appropriate solvents were selected for an efficient TTs extraction, and original recipes were used to obtain Pegylated liposomes and nanolipid complexes with entrapped TTs, comparative to pure standards (betulinic acid and doxorubicin) in similar conditions. The composition, morphology and sizes of all nanoformulations were checked by High Performance Liquid Chromatography/Mass spectrometry, Transmission Electronic Microscopy and Diffraction Light Scattering. The entrapment efficiency and their impact on cell viability, cell cycle arrest and apoptosis by Flow Cytometry was also measured on both cancer cell lines. Conclusions: The standardized TTs showed good stability, synergistic effects of main components and superior comparative to pure betulinic acid. According to experimental data, TTs showed good entrapment in liposomal and NLC nanoformulations, both delivery systems including natural, biodegradable ingredients and enhanced bioavailability. The apoptosis and necrosis effects were more pronounced for TTs liposomal formulations in both types of cancer cells, with lower cytotoxicity comparative to Doxorubicin, and can be correlated with their increased bioavailability.
Keywords:
1. Introduction
2. Results
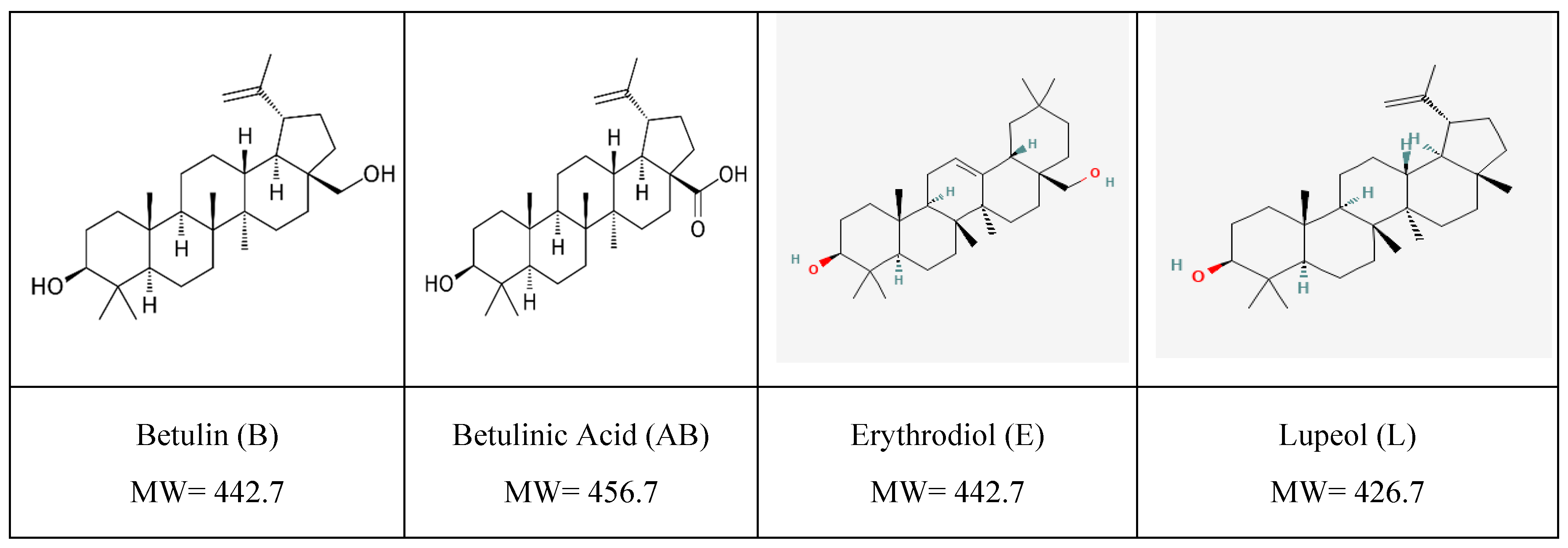
2.1. Preparation and Characterization of the TTs Extract
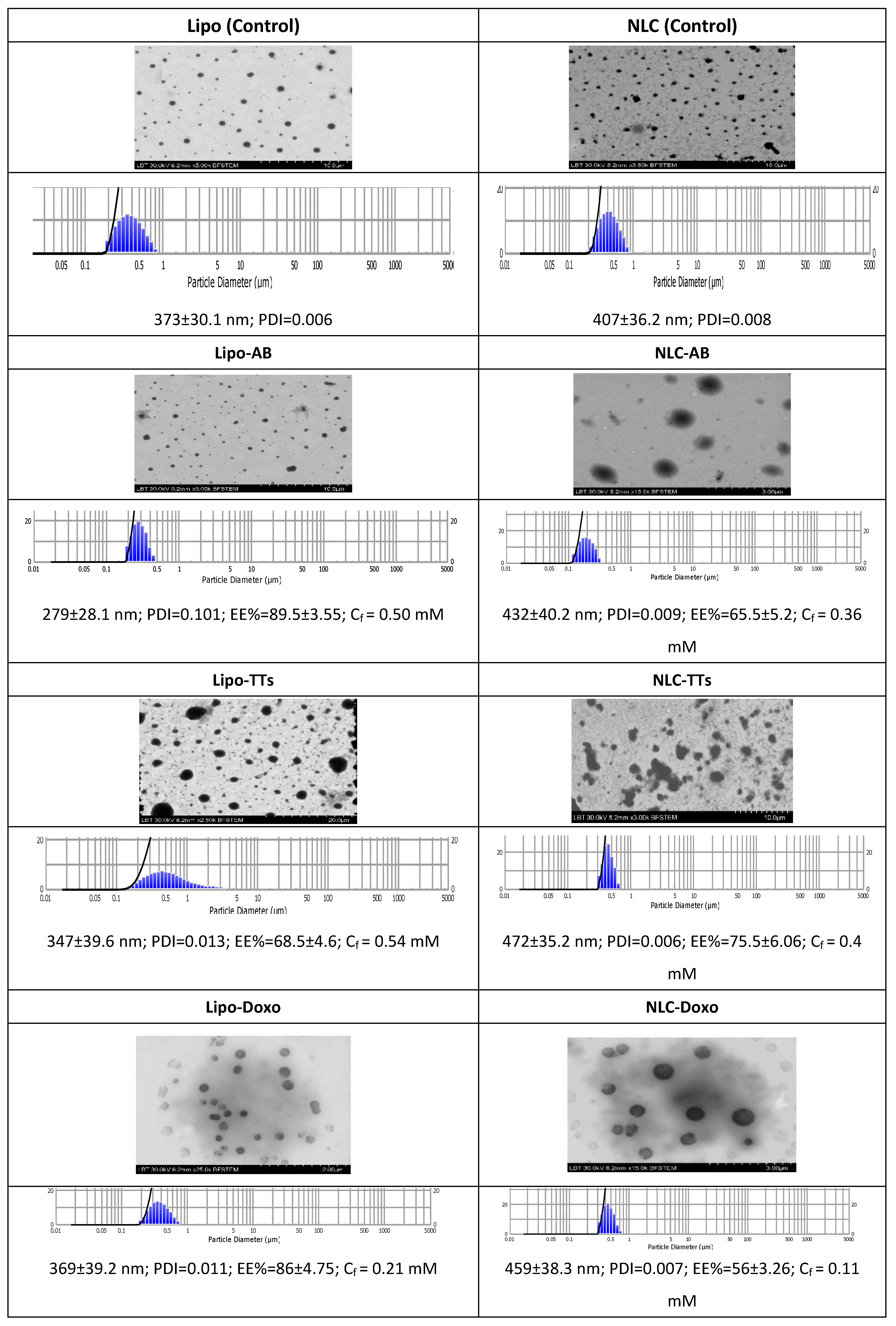
2.2. Preparation and Characterization of the Nanoformulations with Entrapped TTs, AB and Doxo
2.3. Cell Viability and Cytotoxicity
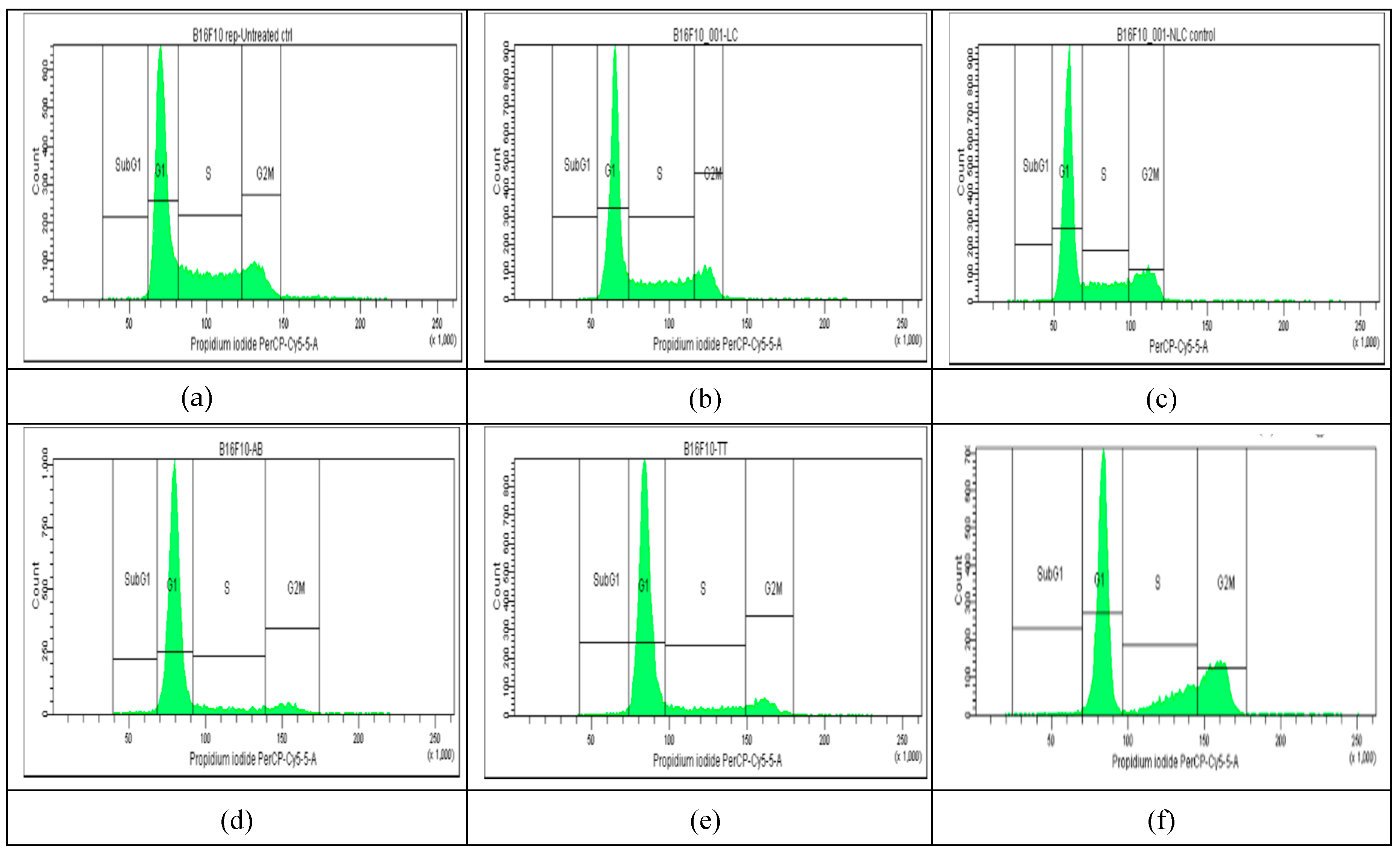
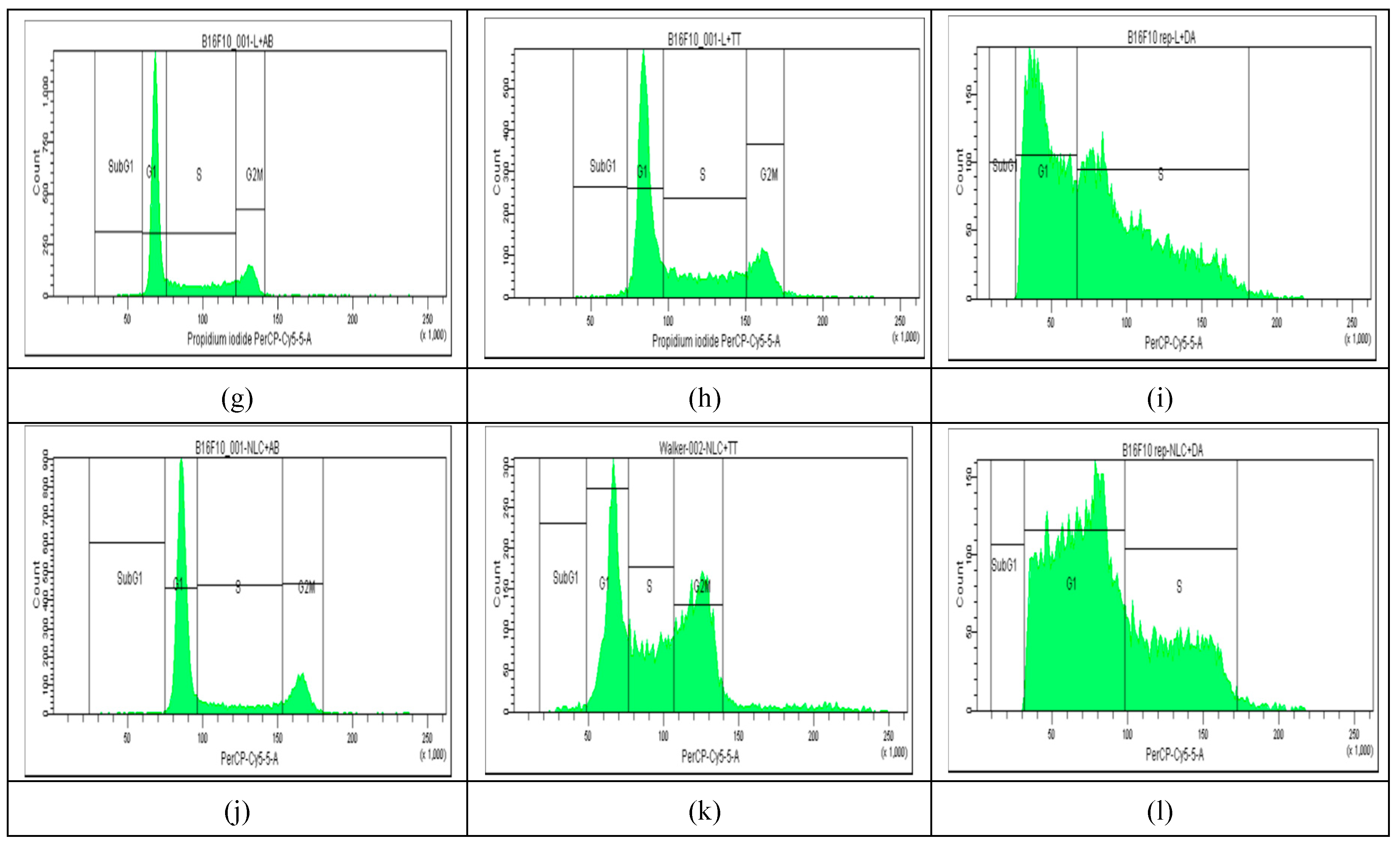
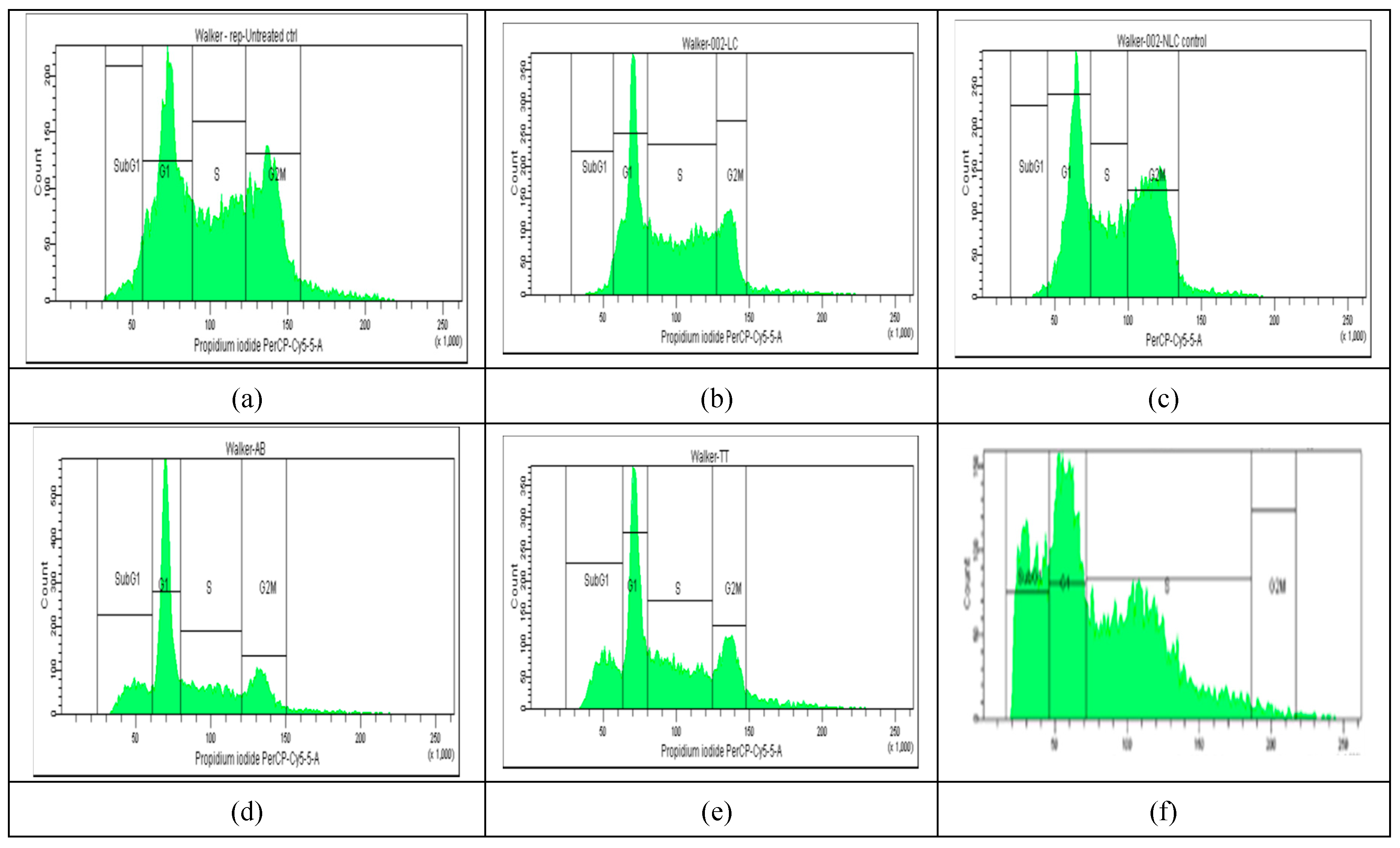
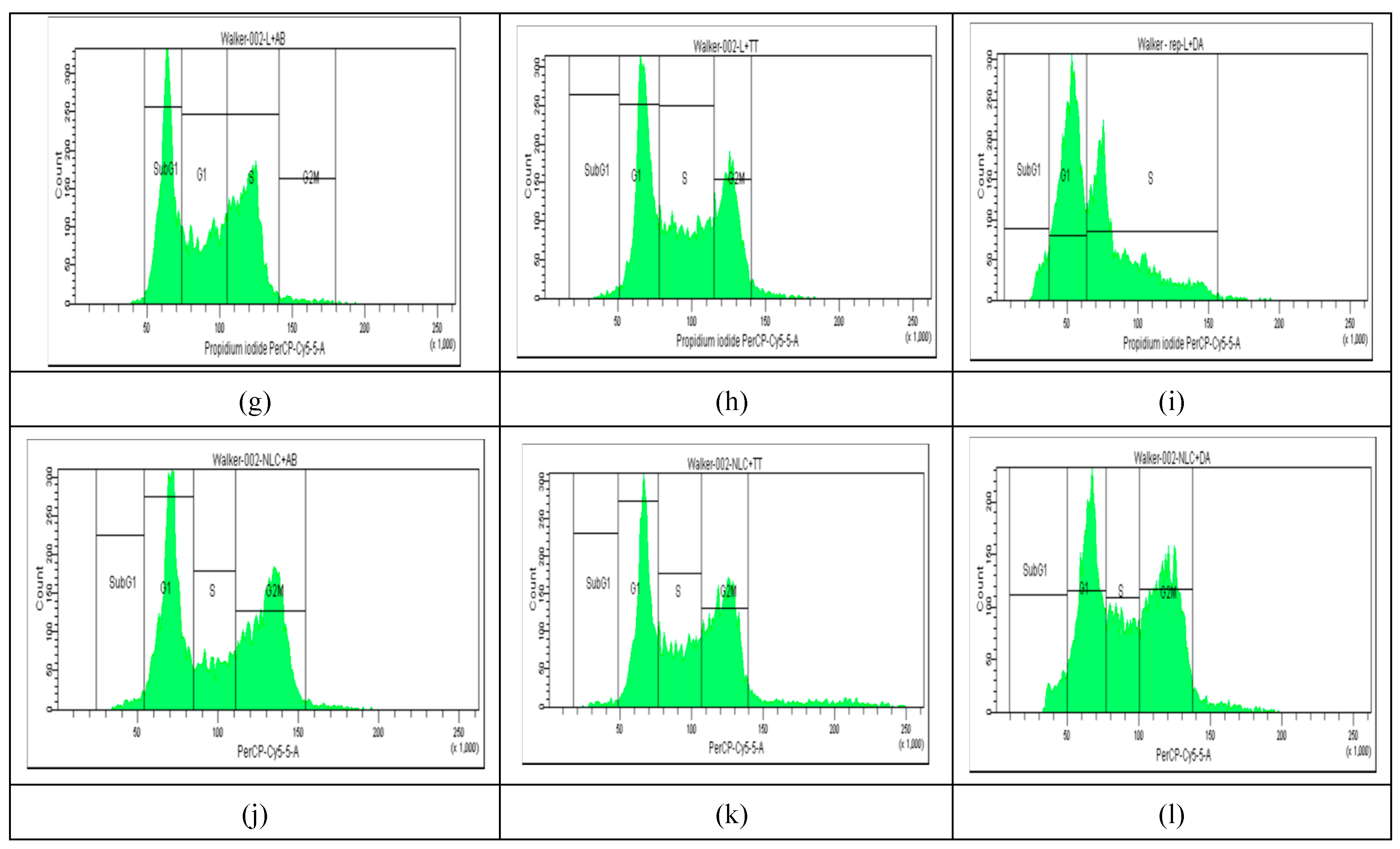
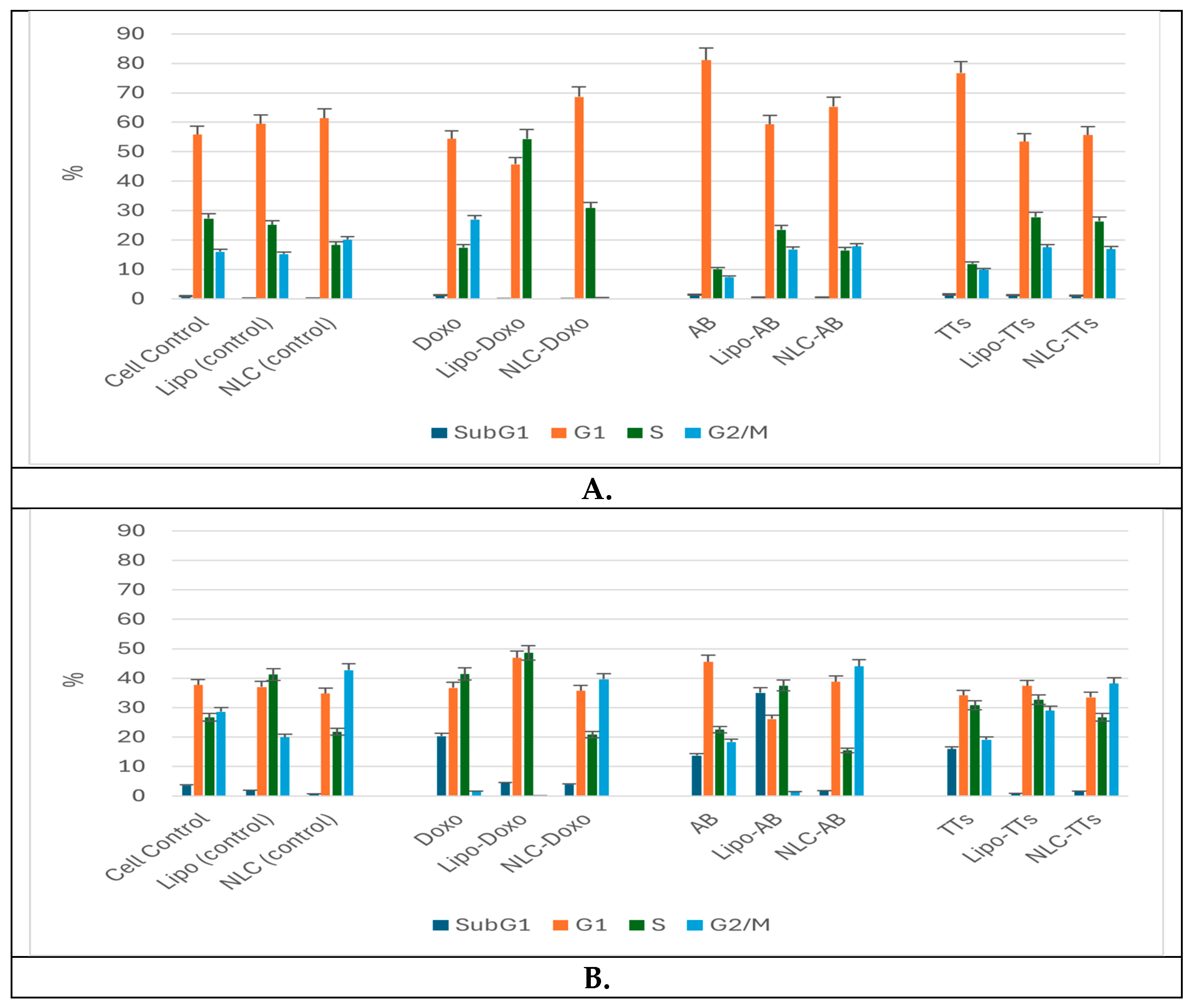
2.4. Cell Cycle Distribution
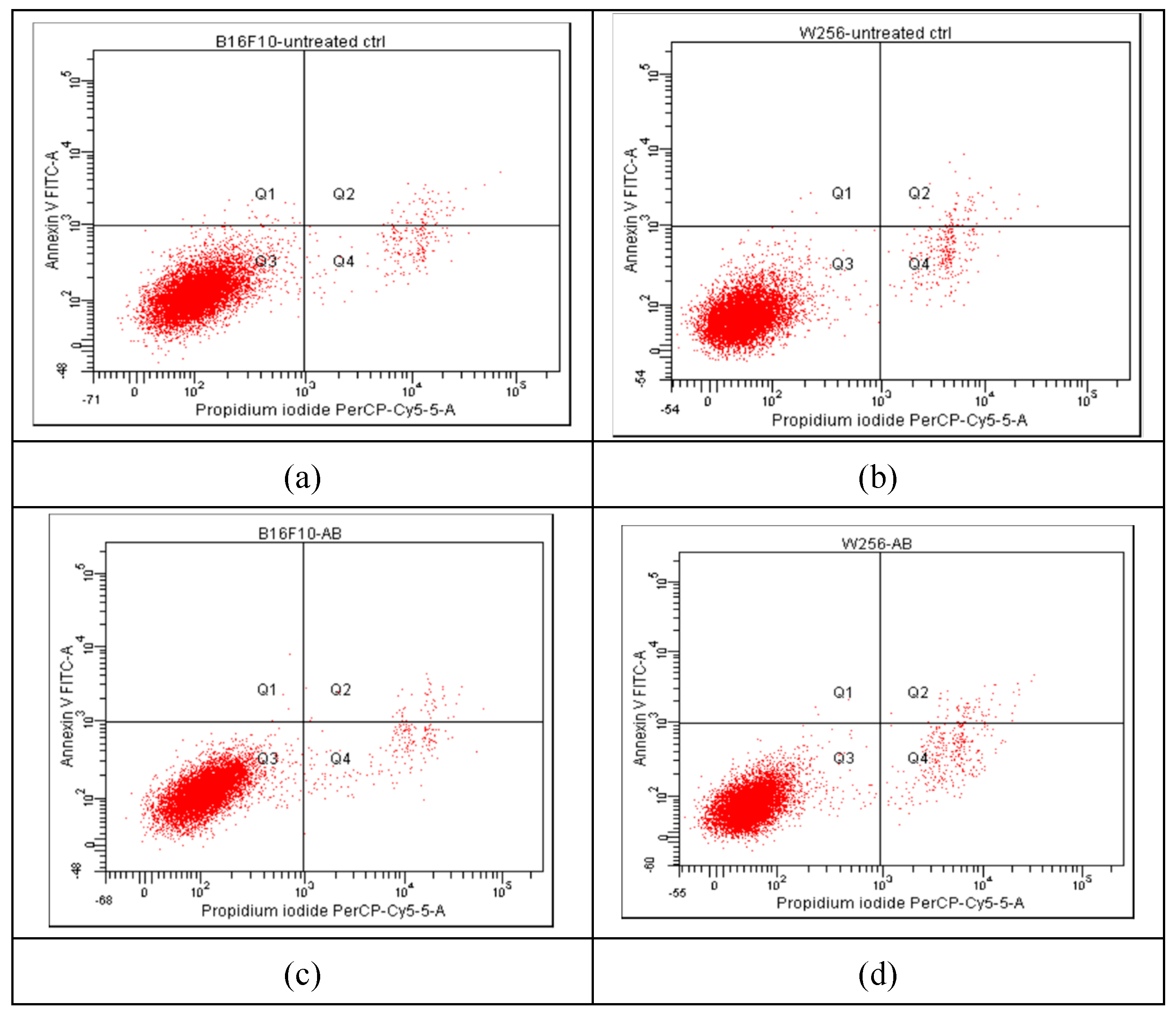
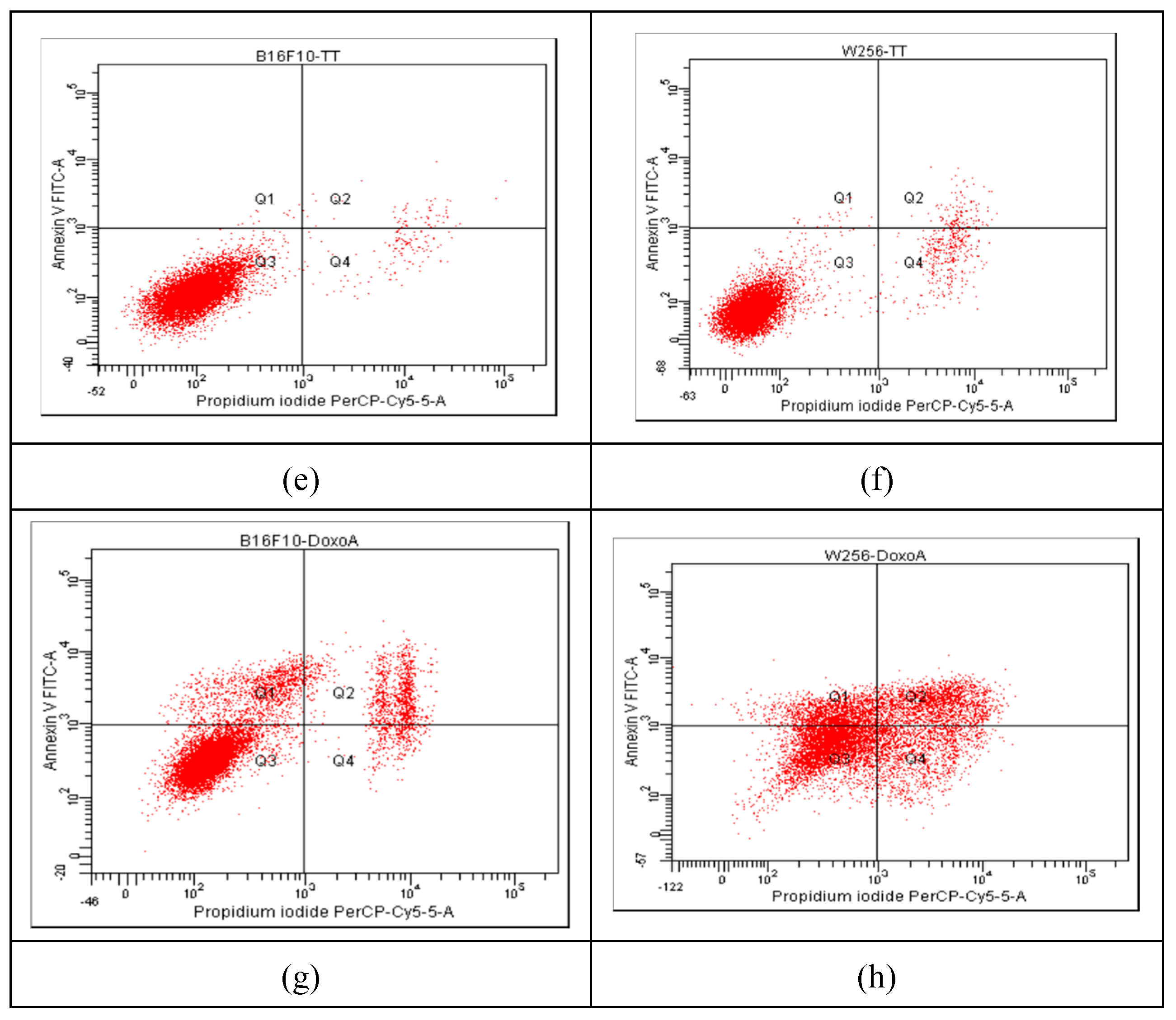
2.5. Apoptosis
3. Discussion
4. Materials and Methods
4.1. Extraction of TTs from Birch Bark
4.2. Preparation of PEGylated Liposomes Using the Ethanol Injection Method
4.3. Preparation of NLC Formulations
4.4. Entrapment Efficiency, Size Determination and Morphology of Liposomes vs NLCs
4.5. Cell Cultures
4.6. Viability Assays
4.7. Cell Cycle Analysis by Flow Cytometry
4.8. Apoptosis and Necrosis Assays by Flow Cytometry
4.9. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Kovač-Bešović EE, Durić K, Kalođera Z, Sofić E: Identification and isolation of pharmacologically active triterpenes in Betulae Cortex, Betula pendula Roth., Betulaceae. Bosn J Basic Med Sci 2009, 9, 31–38.
- Holonec, L.; Ranga, F.; Crainic, D.; Truţa, A.; Socaciu, C*. Evaluation of Betulin and Betulinic Acid Content in Birch Bark from Different Forestry Areas of Western Carpathians. Not Bot Horti Agrobo 2012, 40, 99-105.
- Šiman, P.; Filipová, A.; Tichá, A.; Niang, M.; Bezrouk, A.; Havelek, R. Effective Method of Purification of Betulin from Birch Bark: The Importance of Its Purity for Scientific and Medicinal Use. Plos One 2016, 11, e0154933.
- Demets, O.V.; Takibayeva, A.T.; Kassenov, R.Z.; Aliyeva, M.R. Methods of Betulin Extraction from Birch Bark. Molecules 2022, 27, 3621.
- Liu, S.; Liu, H.; Zhang, L.; Ma, C.; Abd El-Aty, A. M. Edible pentacyclic triterpenes: A review of their sources, bioactivities, bioavailability, self-assembly behavior, and emerging applications as functional delivery vehicles. Crit Rev Food Sci Nutr 2022, 1-17.
- Bag, B. G.; Majumdar, R. Self-assembly of Renewable Nano-sized Triterpenoids. Chem Record 2017, 17, 841–873.
- Pisha, E.; Chai, H.; Lee, I.S.; Chagwedera, T.E.; Farnsworth, N.R.; Cordell, A.C.; Beecher, C.W.W.; Fong, H.H.S.; Kinghorn, A.D.; Brown, D.M.; Wani, M.C.; Wall, M.E.; Hieken, T.J.; Das Gupta, T.K.; Pezzuto, J.M. Discovery of betulinic acid as a selective inhibitor of human Melanoma that functions by induction of apoptosis. Nat Med 1995, 1, 1046–1051.
- Cichewicz, R.H.; Kouzi, S.A. Chemistry, biological activity, and chemotherapeutic potential of betulinic acid for the prevention and treatment of cancer and HIV infection. Med Res Rev 2004, 24, 90–114.
- Furtado, N.A.J.C.; Pirson, L.; Edelberg, H.; Miranda, L.M.; Loira-Pastoriza, C.; Preat V.; Larondelle, Y.; André, C.M. Pentacyclic Triterpene Bioavailability: An Overview of In Vitro and In Vivo Studies. Molecules 2017, 22, 400.
- Sohag, A.A.; Hossain, T.; Rahaman, A.; Rahman, P.; Hasan, M.S.; Das, R.C.; Khan, K., Sikder, M.H.; Alam, M.; Uddin, J.; Rahman, H.; Islam, T.; Moon, I.S.; Hannan, A. Molecular pharmacology and therapeutic advances of the pentacyclic triterpene lupeol. Phytomedicine: Int J Phytother Phytopharmacol 2022, 99, 154012.
- Ghante MH, Jamkhande P.G, Role of Pentacyclic Triterpenoids in Chemoprevention and Anticancer Treatment: An Overview on Targets and Underling Mechanisms. J Pharmacopuncture 2019, 22, 55–67.
- Patlolla,J.M.R.; Rao,C.V.Triterpenoids for cancer prevention and treatment: current status and future prospects. Curr Pharmaceut Biotechnol 2012, 13, 147–155.
- Hordyjewska, A.; Ostapiuk, A.; Horecka, A. Betulin and betulinic acid: triterpenoids derivatives with a powerful biological potential. Phytochem Rev 2019, 18, 929-951.
- Hordyjewska, A.; Prendecka-Wróbel, M.; Kurach, Ł.; Horecka, A.; Olszewska, A.; Pigo ´n-Zaj ˛ac, D.; Małecka-Massalska, T.; Kurzepa, J. Antiproliferative Properties of Triterpenoids by ECIS Method—A New Promising Approach in Anticancer Studies? Molecules 2022, 27, 3150.
- Król, S.K.; Kiełbus, M.; Rivero-Muller, A.; Stepulak, A. Comprehensive review on betulin as a potent anticancer agent. BioMed Res Intl 2015, Article ID 584189.
- Nistor, M.; Rugina, D.; Diaconeasa, Z.; Socaciu, C.; Socaciu, M.A. Pentacyclic Triterpenoid Phytochemicals with Anticancer Activity: Updated Studies on Mechanisms and Targeted Delivery. Int J Mol Sci 2023, 24, 12923.
- Moghaddam, M.G.; Ahmad, F.B.H.; Kermani, A.S. Biological activity of betulinic acid: A review. Pharmacol Pharm 2012, 3, 119–123.
- Zuco, V.; Supino, R.; Righetti, SC.; Cleris, K.; Marchesi, E.; Gambacorti-Passerini, C.; Formelli, F. Selective cytotoxicity of betulinic acid on tumor cell lines, but not normal cells. Cancer Lett 2002, 175, 17–25.
- Ali-Seyed, M.; Jantan, I.; Vijayaraghavan, K.; Bukhari, S. N. A. Betulinic Acid: Recent Advances in Chemical Modifications, Effective Delivery, and Molecular Mechanisms of a Promising Anticancer Therapy. Chemical Biol & Drug Design 2015, 87, 517–536.
- Schmidt, M.L.; Kuzmanoff, K.L.; Ling-Indeck, L.; Pezzuto, J.M. Betulinic acid induces apoptosis in human neuroblastoma cell lines. Eur J Cancer 1997, 33, 2007–2010.
- Kwon, H.J.; Shim, J.S.; Kim, J.H.; Cho, H.Y.; Yum, Y.N.; Kim, S.H.; Yu, J. Betulinic acid inhibits growth factor-induced in vitro angiogenesis via the modulation of mitochondrial function in endothelial cells. Jpn J Cancer Res 2002, 93, 417–425.
- Fulda, S.; Scaffidi, C.; Susin, SA.; Krammer, PH.; Kroemer, G.; Peter, ME.; Debatin, K-M. Activation of mitochondria and release of mitochondrial apoptogenic factors by betulinic acid. J Biol Chem 1998, 273, 33942–33948.
- Dehelean, C.A.; Feflea, S.; Molnár, J.; Zupko, I.; Soica, C. Betulin as an antitumor agent tested in vitro on A431, HeLa and MCF7, and as an angiogenic inhibitor in vivo in the CAM assay. Nat Prod Commun 2012, 7, 981–985.
- Pinzaru, I.; Sarau, C.; Coricovac, D.; Marcovici, I.; Utescu, C.; Tofan, S.; Popovici, RA.; Manea, HC.; Pavel, IE.; Soica, C.; Dehelean, C. Silver Nanocolloids Loaded with Betulinic Acid with Enhanced Antitumor Potential: Physicochemical Characterization and In Vitro Evaluation. Nanomaterials (Basel), 2021, 11, 152.
- Gómez-Favela Mario Armando, David Ulises Santos-Ballardo, Magdalena Elizabeth Bergés-Tiznado, Dulce Libna Ambriz-Pérez, Chapter 6 - Nanoformulations applied to the delivery of terpenes, Editor(s): J. Basilio Heredia, Erick P. Gutiérrez-Grijalva, Angel Licea-Claverie, Janet Alejandra Gutierrez-Uribe, Jayanta Kumar Patra, In: Nanotechnology in Biomedicine, Phytochemical Nanodelivery Systems as Potential Biopharmaceuticals, Elsevier, 2023, 221-256, ISBN 9780323903905.
- Mierina, I.; Vilskersts, R.; Turks M. Delivery Systems for Birch-bark Triterpenoids and their Derivatives in Anticancer Research. Curr Med Chem 2020, 27, 1308 – 1336.
- Kaps, A.; Gwiazdo´n, P.; Chodurek, E. Nanoformulations for Delivery of Pentacyclic Triterpenoids in Anticancer Therapies. Molecules 2021, 26, 1764.
- Kuznetsova, S.A.; Shakhtshneider, T.; Mikhailenko, M.A.; Malyar, Y. Preparation and antitumor activity of betulin dipropionate and its composites. Biointerface Res Appl Chem 2021,12, 6873-6894.
- Chen, X.; Lu, S.; Gong, F.; Sui, X.; Liu,T.; Wang,T. Research on the synthesis of nanoparticles of betulinic acid and their targeting antitumor activity. J Biomed Mater Res 2022,110, 1789-1795.
- Saneja, A.; Arora, D.; Kumar, R.; Dubey, R.D.; Panda, A.K.; Gupta, P.N. Therapeutic applications of betulinic acid nanoformulations. Ann NY Acad Sci 2018, 1421, 5–18.
- Zhang, D.-M.; Xu, H.-G.; Wang, L.; Li, Y.-J.; Sun, P.-H.; Wu, X.-M.; Wang, G.J.; Chen, W.M.; Ye, W.-C. Betulinic Acid and its Derivatives as Potential Antitumor Agents. Med Res Rev 2015, 35, 1127–1155.
- Sharma, N.; Deepak.; C., Sandeep, K. Nanoparticles: Fundamental and Prospectives. Res J Pharmaceut Biol Chem Sci 2018, 9, 152–164.
- Li, Q.; Cai, T.G.; Huang, Y.H.; Xia, X.; Cole, S.P.C.; Cai, Y. A Review of the Structure, Preparation, and Application of NLCs, PNPs, and PLNs. Nanomaterials 2017, 7, 122–135.
- Khan S, Sharma A, Jain V. An Overview of Nanostructured Lipid Carriers and its Application in Drug Delivery through Different Routes. Adv Pharm Bull 2023, 13, 446-460.
- Mahmoud, K.; Swidan, S.; El-Nabarawi, M.; Teaima, M. Lipid based nanoparticles as a novel treatment modality for hepatocellular carcinoma: A comprehensive review on targeting and recent advances. J Nanobiotechnol 2022, 20, 109.
- Tran, M.A.; Watts, R.J.; Robertson, G.P. Use of liposomes as drug delivery vehicles for treatment of melanoma. Pigment Cell Melanoma Res 2009, 22, 388-399.
- Taléns-Visconti, R.; Díez-Sales, O.; de Julián-Ortiz, J.V.; Nácher, A. Nanoliposomes in Cancer Therapy: Marketed Products and Current Clinical Trials. Int J Mol Sci 2022, 23, 23084249.
- Liu, W.-K.; Ho, J.C.K.; Cheung, F.W.K.; Liu, B.P.L.; Ye, W.-C.; Che, C.-T. Apoptotic activity of betulinic acid derivatives on murine melanoma B16 cell line. Eur J Pharmacol 2004, 498, 71–78.
- Abraham, S.A.; Waterhouse, D.N.; Mayer, L.D.; Cullis, P.R.; Madden, T.D.; Bally, M.B. The liposomal formulation of Doxorubicin. Methods Enzymol 2005, 391, 71-97.
- Kciuk, M.; Gielecińska, A.; Mujwar, S.; Kołat, D.; Kałuzińska-Kołat, Ż.; Celik, I.; Kontek, R. Doxorubicin-An Agent with Multiple Mechanisms of Anticancer Activity. Cells 2023, 12, 659.
- Arshad, R.; Arshad, M. S.; Rahdar, A.; Hassan, D.; Behzadmehr, R.; Ghotekar, S.; Medina, D.I.; Sadanand Pandey, S. Nanomaterials as an advanced nano-tool for the Doxorubicin delivery/ Co-Delivery-A Comprehensive Review. J Drug Deliv Technol 2023, 83,104432.
- Sesarman, A.; Tefas, L.; Sylvester, B.; Licarete, E.; Rauca, V.; Luput, L.; Patras, L.; Banciu, M.; Porfire, A. Anti-angiogenic and anti-inflammatory effects of long-circulating liposomes coencapsulating curcumin and Doxorubicin on C26 murine colon cancer cells. Pharm Rep 2010, 70, 331–339.
- Bisht, A., Avinash, D., Sahu, K. K., Patel, P., Das Gupta, G., & Kurmi, B. D. A comprehensive review on doxorubicin: mechanisms, toxicity, clinical trials, combination therapies and nanoformulations in breast cancer. Drug Deliv Transl Res, 2024, 1-32.
- Dhiman N.; Awasthi R.; Sharma B.; Kharkwal H.; Kulkarni G.T. Lipid Nanoparticles as Carriers for Bioactive Delivery. Frontiers in Chemistry 2021, 9, nr. 580118.
- Mishra, V.; Bansal, K.K.; Verma, A.; Yadav, N.; Thakur, S.; Sudhakar, K.; Rosenholm, J.M. Solid Lipid Nanoparticles: Emerging Colloidal Nano Drug Delivery Systems. Pharmaceutics 2018, 10, 191.
- Müller, R.H.; Alexiev, U.; Sinambela, P.; Keck, C.M. Nanostructured Lipid Carriers (NLC): The Second Generation of Solid Lipid Nanoparticles. In: Percutaneous Penetration Enhancers Chemical Methods in Penetration Enhancement; Springer: Berlin/Heidelberg, Germany, 2016; pp. 161–185.
- Khosa, A.; Reddi, S.; Saha, R.N. Nanostructured lipid carriers for site-specific drug delivery, Biomed & Pharmacother 2018, 103, 598-613.
- Muntean, P.; Socaciu, C.; Socaciu, M. Lipid nanostructured particles as emerging carriers for targeted delivery of bioactive molecules: Applications in food and biomedical sciences (an overview). Bull UASVM Food Sci Technol 2020, 77, 37–46.
- Biswasroy, P.; Pradhan, D.; Pradhan, DK.; Ghosh, G.; Rath, G. Development of Betulin-Loaded Nanostructured Lipid Carriers for the Management of Imiquimod-Induced Psoriasis. AAPS Pharm SciTech 2024, 25, 57.
- Socaciu, M.; Diaconeasa, Z.; Socaciu, C. Simple and fast procedure to incorporate Doxorubicine in small unilamellar liposomes: Effects on liposome size and zeta potential. Studia UBB Chemia LXIV 2019, 3, 181–192.
- Rugină, D.; Pop R.; Nistor, M.; Cenariu, M.; Tăbăran F.; Socaciu,C.; Socaciu, M.A. Distinct in vitro effects of liposomal and nanostructured lipid nanoformulations with entrapped acidic and neutral doxorubicin on B16-F10 melanoma and Walker 256 carcinoma cells. Discover Oncology 2024 (submitted, under revision).
- Fan, M. ; Xu, S. ; Xia, S. Zhang, X. Preparation of salidroside nano-liposomes by ethanol injection method and in vitro release study. Eur Food Res Technol 2008, 227, 167–174.
- Liu Y.; Gao, D.; Zhang, X.; Liu, Z.; Dai, K.; Ji,B.; Wang, Q.; Luo, L. Antitumor drug effect of betulinic acid mediated by polyethylene glycol modified liposomes. Materials Sci Engineering C 2016, 64, 124–132.
- Ibrahim, M.; Abuwatfa, W.H.; Awad, N.S.; Sabouni, R.; Husseini, G.A. Encapsulation, Release, and Cytotoxicity of Doxorubicin Loaded in Liposomes, Micelles, and Metal-Organic Frameworks: A Review. Pharmaceutics 2022, 14, 254.
- Rus, I.; Tertiș, M.; Paşcalău, V.; Pavel C.; Melean B.; Suciu M.; Moldovan C.; Topală T.; Popa C.; Săndulescu R.; Cristea C. Simple And Fast Analytical Method For The Evaluation Of The Encapsulation and Release Profile Of Doxorubicin From Drug Delivery Systems, Farmacia 2021, 69, 670-681.
- Zhu, L.; Lin, M. The Synthesis of Nano-Doxorubicin and its Anticancer Effect. Anticancer Agents Med Chem 2021, 21, 2466-2477.
- Mahmoudi, M.; Azadmanesh, K.; Shokrgozar, M.A.; Journeay, W.S.; Laurent S. Effect of Nanoparticles on the Cell Life Cycle. Chem Rev 2011, 111, 3407-3432. [CrossRef]

| A. | Melanoma B16-F10 | Walker 256 | t (M vs W256) | significance |
| Doxo | 0.18±0.5 | 0.87±0.14 | -2.555 | NS |
| Lipo-Doxo | 1.03±0.11 | 1.66±0.18 | -10.203 | ** |
| NLC-Doxo | 2.14±0.12 | 1.35±0.12 | 11.401 | ** |
| AB | 10.22±4.03 | 13.53±1.65 | -3.087 | * |
| Lipo-AB | 15.52±1.1 | 14.42±1.13 | 0.62 | NS |
| NLC-AB | 22.58±0.03 | 27.33±2.8 | -279.883 | *** |
| TTs | 19.25±2.33 | 28.55±3.2 | -8.074 | * |
| Lipo-TTs | 23.58±0.03 | 27.25±1.8 | -215.883 | *** |
| NLC-TTs | 20.22±0.9 | 19.48±1.55 | -0.111 | NS |
| B. | Melanoma B16-F10 | Walker 256 | ||
| t | significance | t | significance | |
| NLC-Doxo/Doxo | 28.083 | ** | 6.86 | * |
| Lipo-Doxo/Doxo | 13.023 | ** | 7.540556 | * |
| NLC-Doxo/Lipo Doxo | 16.078 | ** | -4.70083 | NS |
| NLC-AB/AB | 716.97 | *** | 6.975 | * |
| Lipo-AB/AB | 4.402 | * | -0.27168 | NS |
| NLC-AB/Lipo-AB | 410.733 | *** | 6.93875 | * |
| NLC-TTs/TTs | -0.444 | NS | -11.8774 | ** |
| Lipo-TTs/TTs | 250.253 | *** | -2.67361 | NS |
| NLC-TTs/Lipo-TTs | -6.563 | * | -10.5726 | ** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




