Submitted:
01 November 2024
Posted:
05 November 2024
You are already at the latest version
Abstract
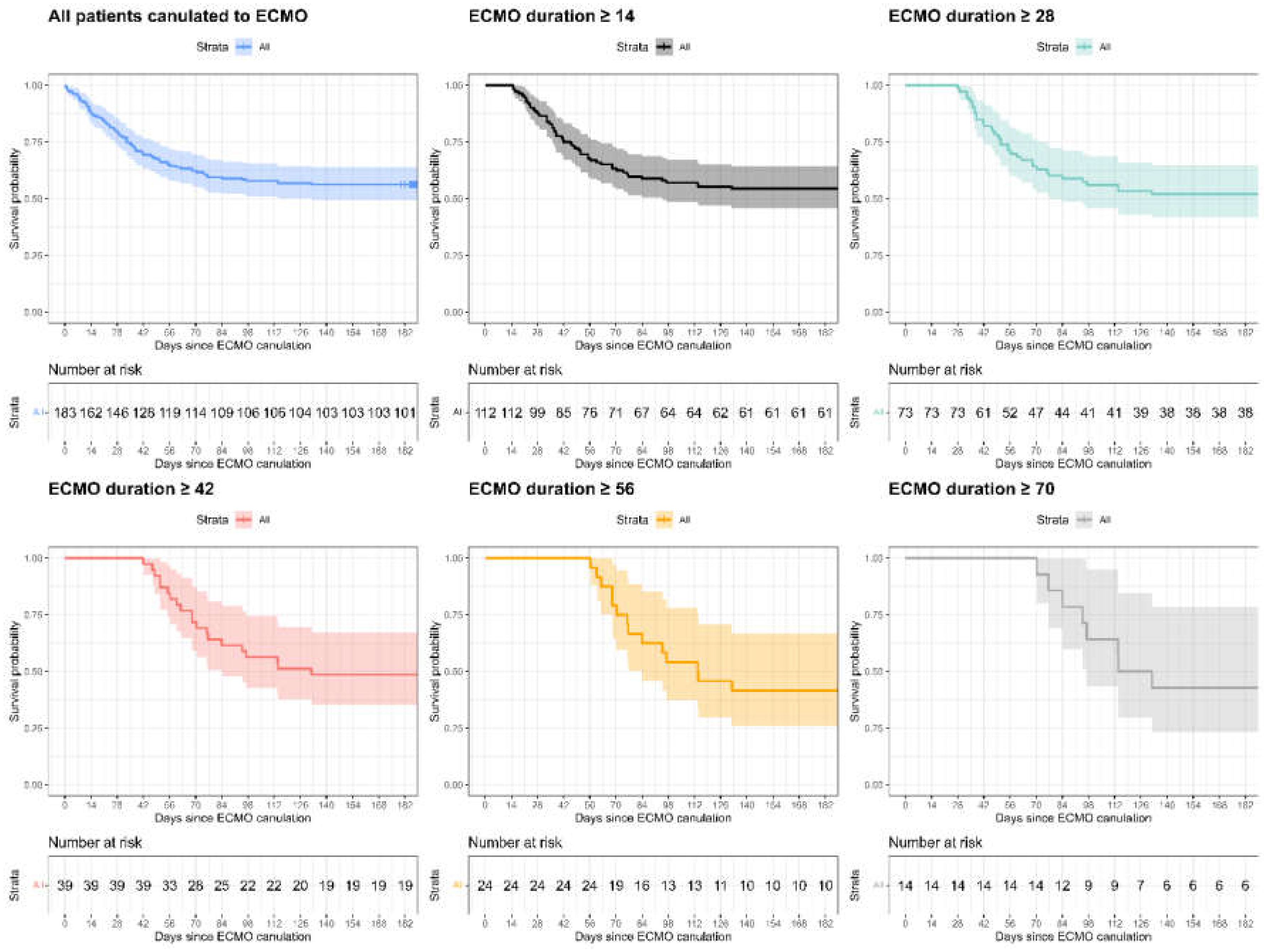
Background: Predicting whether extracorporeal membrane oxygenation (ECMO) treatment duration affects prognosis is important both medically and economically. We conducted a retrospective, multicenter study to better understand the outcomes of patients treated with VV ECMO for a prolonged duration, analyzing data from the Israel ECMO registry. The study included all adult patients treated with VV-ECMO due to COVID-19-induced respiratory failure. The primary outcomes were survival rates up to 180 days from cannulation. Results: One hundred and eighty-eight patients were included in the study. The median age was 50 years (IQR 42, 50) and 69% were male. Patients were mechanically ventilated for a median of 2.5 days before cannulation (IQR 0.5, 5). The mean ECMO support duration was 29.9 days, with a maximal duration of 189.9 days. Survival rate for 180 days was 56%. We found no change in survival for patients on ECMO for 14, 28, or 56 days. Every day of mechanical ventilation before cannulation correlated with an 11% greater risk for prolonged ECMO treatment (p=0.01). Conclusions: For COVID-19-induced ARDS patients treated with VV-ECMO, ECMO treatment duration did not affect mortality. The longer the duration of mechanical ventilation before ECMO cannulation, the higher the risk for prolonged ECMO treatment.
Keywords:
1. Background
2. Methods
2.1. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Posluszny J, Engoren M, Napolitano LM, Rycus PT, Bartlett RH, centers Em. Predicting Survival of Adult Respiratory Failure Patients Receiving Prolonged (>/=14 Days) Extracorporeal Membrane Oxygenation. ASAIO J. 2020;66:825-33.
- Paden ML, Conrad SA, Rycus PT, Thiagarajan RR, Registry E. Extracorporeal Life Support Organization Registry Report 2012. ASAIO J. 2013;59:202-10.
- Thiagarajan, R.R.; Barbaro, R.P.; Rycus, P.T.; Mcmullan, D.M.; Conrad, S.A.; Fortenberry, J.D.; Paden, M.L. Extracorporeal Life Support Organization Registry International Report 2016. ASAIO J. 2016, 63, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Peek, G.J.; Mugford, M.; Tiruvoipati, R.; Wilson, A.; Allen, E.; Thalanany, M.M.; Hibbert, C.L.; Truesdale, A.; Clemens, F.; Cooper, N.; et al. Efficacy and economic assessment of conventional ventilatory support versus extracorporeal membrane oxygenation for severe adult respiratory failure (CESAR): a multicentre randomised controlled trial. Lancet 2009, 374, 1351–1363. [Google Scholar] [CrossRef]
- Pham T, Combes A, Roze H, Chevret S, Mercat A, Roch A, et al. Extracorporeal membrane oxygenation for pandemic influenza A(H1N1)-induced acute respiratory distress syndrome: a cohort study and propensity-matched analysis. Am J Respir Crit Care Med. 2013;187:276-85.
- ELSO Registry [Internet]. https://www.elso.org/Registry/ELSOLiveRegistryDashboard. Accessed 18 Sept 2024.
- Schmidt, M.; Bailey, M.; Sheldrake, J.; Hodgson, C.; Aubron, C.; Rycus, P.T.; Scheinkestel, C.; Cooper, D.J.; Brodie, D.; Pellegrino, V.; et al. Predicting Survival after Extracorporeal Membrane Oxygenation for Severe Acute Respiratory Failure. The Respiratory Extracorporeal Membrane Oxygenation Survival Prediction (RESP) Score. Am. J. Respir. Crit. Care Med. 2014, 189, 1374–1382. [Google Scholar] [CrossRef]
- Dreier, E.; Malfertheiner, M.V.; Dienemann, T.; Fisser, C.; Foltan, M.; Geismann, F.; Graf, B.; Lunz, D.; Maier, L.S.; Müller, T.; et al. ECMO in COVID-19—prolonged therapy needed? A retrospective analysis of outcome and prognostic factors. [CrossRef]
- Li X, Guo Z, Li B, Zhang X, Tian R, Wu W, et al. Extracorporeal Membrane Oxygenation for Coronavirus Disease 2019 in Shanghai, China. ASAIO J. 2020;66:475-81.
- Yang, Y.; Rali, A.S.; Inchaustegui, C.; Alakbarli, J.; Chatterjee, S.; Herlihy, J.P.; George, J.; Shafii, A.; Nair, A.; Simpson, L. Extracorporeal Membrane Oxygenation in Coronavirus Disease 2019-associated Acute Respiratory Distress Syndrome: An initial US Experience at a High-volume Centre. Card. Fail. Rev. 2020, 6, e17. [Google Scholar] [CrossRef] [PubMed]
- Kon, Z.N.; Dahi, S.; Evans, C.F.; Byrnes, K.A.; Bittle, G.J.; Wehman, B.; Rector, R.P.; McCormick, B.M.; Herr, D.L.; Sanchez, P.G.; et al. Long-Term Venovenous Extracorporeal Membrane Oxygenation Support for Acute Respiratory Distress Syndrome. Ann. Thorac. Surg. 2015, 100, 2059–2063. [Google Scholar] [CrossRef] [PubMed]
- Na, S.J.; Jung, J.-S.; Hong, S.-B.; Cho, W.H.; Lee, S.-M.; Cho, Y.-J.; Park, S.; Koo, S.-M.; Park, S.Y.; Chang, Y.; et al. Clinical outcomes of patients receiving prolonged extracorporeal membrane oxygenation for respiratory support. Ther. Adv. Respir. Dis. 2019, 13. [Google Scholar] [CrossRef] [PubMed]
- Levy, L.; Deri, O.; Huszti, E.; Nachum, E.; Ledot, S.; Shimoni, N.; Saute, M.; Sternik, L.; Kremer, R.; Kassif, Y.; et al. Timing of Lung Transplant Referral in Patients with Severe COVID-19 Lung Injury Supported by ECMO. J. Clin. Med. 2023, 12, 4041. [Google Scholar] [CrossRef] [PubMed]
- Flinspach, A.N.; Raimann, F.J.; Bauer, F.; Zacharowski, K.; Ippolito, A.; Booke, H. Therapy and Outcome of Prolonged Veno-Venous ECMO Therapy of Critically Ill ARDS Patients. J. Clin. Med. 2023, 12, 2499. [Google Scholar] [CrossRef] [PubMed]
- Stern, D.R.; Michalak, L.A.; Beckett, A.R.; Tabachnick, D.R.; Tatooles, A.J. Outcomes of patients with COVID-19 supported by venovenous extracorporeal membrane oxygenation for greater than 90 days. JTCVS Open 2023, 16, 450–459. [Google Scholar] [CrossRef] [PubMed]
- Chandel, A.; Fabyan, K.D.; Mendelsohn, S.; Puri, N.; Damuth, E.; Rackley, C.R.; Conrad, S.A.; King, C.S.; Green, A.M. Prevalence and Survival of Prolonged Venovenous Extracorporeal Membrane Oxygenation for Acute Respiratory Distress Syndrome: An Analysis of the Extracorporeal Life Support Organization Registry. Crit. Care Med. 2024, 52, 869–877. [Google Scholar] [CrossRef] [PubMed]
- Tran, A.; Fernando, S.M.; Rochwerg, B.; Barbaro, R.P.; Hodgson, C.L.; Munshi, L.; MacLaren, G.; Ramanathan, K.; Hough, C.L.; Brochard, L.J.; et al. Prognostic factors associated with mortality among patients receiving venovenous extracorporeal membrane oxygenation for COVID-19: a systematic review and meta-analysis. Lancet Respir. Med. 2022, 11, 235–244. [Google Scholar] [CrossRef]
- Tan, Z.; Su, L.; Chen, X.; He, H.; Long, Y. Relationship between the Pre-ECMO and ECMO Time and Survival of Severe COVID-19 Patients: A Systematic Review and Meta-Analysis. J. Clin. Med. 2024, 13, 868. [Google Scholar] [CrossRef] [PubMed]
- Makhoul, M.; Keizman, E.; Carmi, U.; Galante, O.; Ilgiyaev, E.; Matan, M.; Słomka, A.; Sviri, S.; Eden, A.; Soroksky, A.; et al. Outcomes of Extracorporeal Membrane Oxygenation (ECMO) for COVID-19 Patients: A Multi-Institutional Analysis. Vaccines 2023, 11, 108. [Google Scholar] [CrossRef] [PubMed]
- Barbaro, R.P.; MacLaren, G.; Boonstra, P.S.; Combes, A.; Agerstrand, C.; Annich, G.; Diaz, R.; Fan, E.; Hryniewicz, K.; Lorusso, R.; et al. Extracorporeal membrane oxygenation for COVID-19: evolving outcomes from the international Extracorporeal Life Support Organization Registry. Lancet 2021, 398, 1230–1238. [Google Scholar] [CrossRef] [PubMed]
| Demographics | N = 188 |
|---|---|
| Age on admission in years, median (IQR) | 50 (42, 58) |
| Gender (Male), N (%) | 129 (69%) |
| BMI, kg/m2 median (IQR) | 31 (27, 35) |
| Medical history | |
| Smoking | 11 (5.9%) |
| COPD | 4 (2.1%) |
| Diabetes | 42 (22%) |
| Chronic Renal disease | 5 (2.7%) |
| Cardiovascular disease | 16 (8.5%) |
| SOFA score on admission, median (IQR) | 8.00 (6.00, 11.00) |
| PRE-ECMO conventional mechanical ventilation | 162 (86.1%) |
| Duration of pre-ECMO invasive mechanical ventilation (days), median (IQR) | 2.5 (0.5, 5.0) |
| PRE-ECMO prone positioning | 63 (34%) |
| PRE-ECMO Inotropic or vasoactive drugs | 33 (18%) |
| PRE-ECMO Nitric oxide | 100 (53%) |
| All patients N=183 |
Patients still on ECMO on day 14 N=112 |
P-value* | Patients still on ECMO on day 28 N=73 |
P-value* | Patients still on ECMO on day 42 N=39 |
P-value* | Patients still on ECMO on day 56 N=24 |
P-value* | |
|---|---|---|---|---|---|---|---|---|---|
| 60 days survival Rate (95% CI) |
0.63 (0.57-0.71) |
0.66 (0.58-0.75) |
0.636 | 0.68 (0.58-0.70) |
0.440 |
0.79 (0.68-0.93) |
0.036 | --- | --- |
| 90 days survival Rate (95% CI) |
0.58 (0.52-0.66) |
0.59 (0.50-0.69) |
0.916 | 0.59 (0.48-0.71) |
0.988 | 0.62 (0.48-0.79) |
0.732 | 0.62 (0.45-0.85) |
0.756 |
| 180 days survival Rate (95% CI) |
0.56 (0.49-0.63) |
0.54 (0.46-0.65) |
0.840 | 0.52 (0.41-0.64) |
0.496 | 0.49 (0.35-0.67) |
0.468 | 0.41 (0.25-0.66) |
0.132 |
| ECMO duration > 56 days | |||
|---|---|---|---|
| Factor | IRR | 95% CI | p-value |
| PRE ECMO-mechanical ventilation (days) | 1.11 | 0.98, 1.09 | 0.01 |
| SOFA score | 0.90 | 0.78, 1.03 | 0.13 |
| PRE ECMO-prone positioning | 1.53 | 0.69, 3.31 | 0.29 |
| PRE ECMO-nitric oxide | 0.74 | 0.29, 1.88 | 0.52 |
| PRE ECMO-inotropic vasoactive drugs | 1.24 | 0.31, 3.64 | 0.73 |
| Characteristic | All patients cannulated to ECMO, N = 183 | ||
|---|---|---|---|
| HR | 95% CI | p-value | |
| Gender (male) | 1.61 | 0.96, 2.70 | 0.069 |
| Age on admission (years) | 1.03 | 1.01, 1.05 | 0.001 |
| BMI (kg/m2) | 1.00 | 0.96, 1.03 | 0.816 |
| SOFA score on the day of cannulation | 1.12 | 1.03, 1.22 | 0.006 |
| COPD | 0.45 | 0.06, 3.22 | 0.425 |
| Diabetes | 0.90 | 0.53, 1.55 | 0.714 |
| Cardiovascular disease | 1.46 | 0.73, 2.93 | 0.283 |
| Smoking | 1.45 | 0.63, 3.34 | 0.379 |
| Days between Symptoms and intubation | 1.00 | 1.00, 1.01 | 0.364 |
| Days on invasive mechanical ventilation before ECMO cannulation | 1.01 | 0.97, 1.05 | 0.602 |
| PRE-ECMO prone positioning | 0.96 | 0.60, 1.52 | 0.851 |
| PRE-ECMO conventional mechanical ventilation | 1.03 | 0.63, 1.66 | 0.920 |
| PRE-ECMO inotropic vasoactive drugs | 0.76 | 0.41, 1.41 | 0.383 |
| PRE-ECMO nitric oxide | 1.16 | 0.75, 1.81 | 0.506 |
| Complications | Number of patients (N = 188) |
Mortality rates | |
|---|---|---|---|
| Renal | Need for renal replacement therapy | 26 (13%) | 61% |
| CNS | Convulsions | 5 (2.7%) | 60% |
| Brain infarct | 4 (2.1%) | 25% | |
| Brain edema diffuse ischemia | 1 (0.5%) | 100% | |
| Infections | Bacteremia sepsis | 61 (32%) | 38.3% |
| Ventilation-associated pneumonia | 50 (27%) | 24.5% | |
| Mechanical | Cannula repositioning and reinsertion | 17 (9%) | 25% |
| Entire circuit replacement | 29 (15%) | 25% | |
| Tubing or oxygenator clotting requiring replacement | 24 (13%) | 30.4% | |
| Pump failure or breakage | 3 (1.6%) | 66.6% | |
| Bleeding | Number of blood products (mean (range)) | 9 (0-269) | N/A |
| GI bleeding | 14 (7.4%) | 53.8% | |
| Peripheral cannulation and or indwelling lines site bleeding | 25 (13%) | 37.5% | |
| Mucosal bleeding mouth or nose | 25 (13%) | 32% | |
| Thrombocytopenia HIT | 16 (8.5%) | 25% | |
| Hemothorax on ECMO | 6 (3.2%) | 66.7% | |
| Pulmonary | Pneumothorax on ECMO | 15 (8%) | 40% |
| Characteristic | N | OR | 95% CI | p-value |
|---|---|---|---|---|
| Convulsions | 188 | 5.04 | 0.73, 99.6 | 0.2 |
| Bacteremia | 188 | 1.04 | 0.56, 1.92 | 0.9 |
| Ventilator-associated pneumonia | 188 | 0.47 | 0.23, 0.92 | 0.030 |
| Urinary tract infection | 188 | 0.33 | 0.05, 1.41 | 0.2 |
| Cannula dislodgment requiring repositioning and reinsertion | 188 | 0.64 | 0.21, 1.75 | 0.4 |
| Entire circuit replacement | 188 | 0.59 | 0.25, 1.32 | 0.2 |
| Tubing or oxygenator clotting requiring replacement | 188 | 0.56 | 0.22, 1.36 | 0.2 |
| Pump failure or breakage | 188 | 2.46 | 0.23, 53.4 | 0.5 |
| Number of Blood products | 188 | 1.00 | 1.00, 1.01 | 0.4 |
| GI bleeding | 188 | 1.68 | 0.56, 5.30 | 0.4 |
| Peripheral cannulation and or indwelling lines site bleeding | 188 | 0.94 | 0.40, 2.20 | 0.9 |
| Mucosal bleeding mouth nose | 188 | 0.94 | 0.40, 2.20 | 0.9 |
| Heparin Induced Thrombocytopenia HIT | 188 | 0.71 | 0.23, 1.99 | 0.5 |
| Hemothorax on ECMO | 188 | 6.37 | 1.00, 123 | 0.094 |
| Pneumothorax on ECMO | 188 | 0.79 | 0.26, 2.30 | 0.7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




