Submitted:
30 October 2024
Posted:
31 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Participants
2.3. Randomization and Masking
2.4. Procedures
2.5. Efficacy Outcomes
2.6. Safety Outcomes
2.7. Statistical Analysis
3. Results
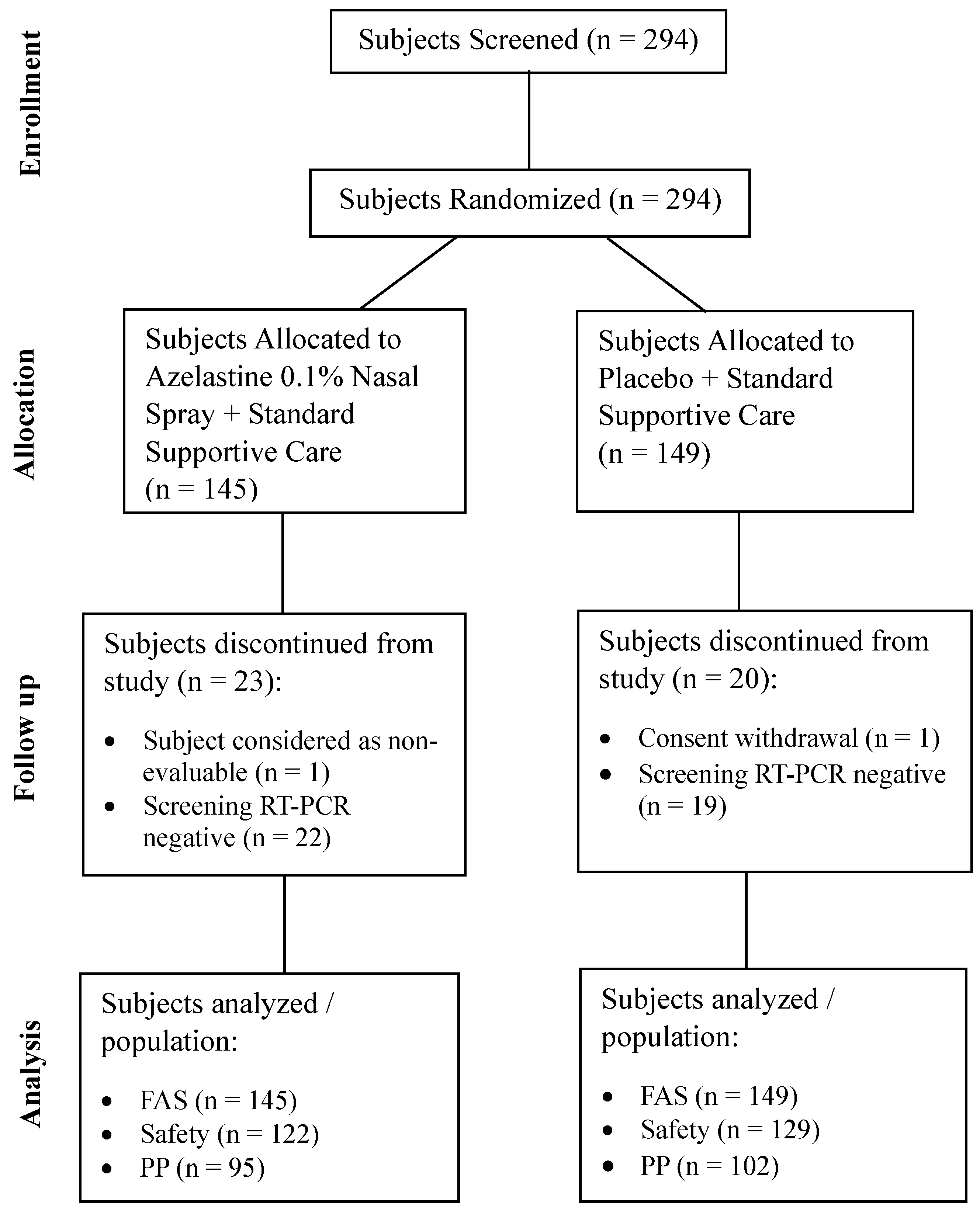
3.1. Subject Disposition
3.2. Subjects’ Baseline Characteristics
3.3. Efficacy
3.3.1. Rate of COVID-19 Related Hospitalization
3.3.2. Viral SARS-CoV-2 RNA Load Reduction
3.4. Exploratory Analysis for Robustness of the Primary Endpoint
3.4.1. Subjects Demonstrating a 10-Fold Decrease in SARS-CoV-2 Virus Load
3.4.2. Subjects with Negative Conversion of SARS-CoV-2 RT-PCR
3.4.3. Change in Symptom Severity (Based on MoH FW Checklist)
3.4.4. Change in Subject Status (Based on WHO’s 11-Category Ordinal Score)
3.5. Safety
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Panahi, Yunes, Armita Mahdavi Gorabi, Sona Talaei, Fatemeh Beiraghdar, Abolfazl Akbarzadeh, Vahideh Tarhriz, and Hassan Mellatyar. "An Overview on the Treatments and Prevention against Covid-19." Virology Journal 20, no. 1 (2023): 23. [CrossRef]
- Cascella M, Rajnik M, and et al. Aleem A. Features, Evaluation, and Treatment of Coronavirus (Covid-19) [Updated 2023 Aug 18]. Treasure Island (FL): StatPearls Publishing.
- update, World Health Organization: COVID-19 epidemiological. "World Health Organization: Covid-19 Epidemiological Update " https://www.who.int/publications/m/item/covid-19-epidemiological-update---22-december-2023.
- COVID-19, Ministry of Health and Family Welfare. "Ministry of Health and Family Welfare. Covid-19." https://www.mohfw.gov.in/.
- Balasubramani, Karuppusamy, Venkatesh Ravichandran, Kumar Arun Prasad, Mu Ramkumar, Sulochana Shekhar, Meenu Mariya James, Naveen Kumar Kodali, Sujit Kumar Behera, Natarajan Gopalan, Rakesh Kumar Sharma, Devojit Kumar Sarma, M. Santosh, Aditya Prasad Dash, and Praveen Balabaskaran Nina. "Spatio-Temporal Epidemiology and Associated Indicators of Covid-19 (Wave-I and Ii) in India." Sci Rep 14, no. 1 (2024): 220. [CrossRef]
- Parasher, Anant. "Covid-19: Current Understanding of Its Pathophysiology, Clinical Presentation and Treatment." Postgraduate Medical Journal 97, no. 1147 (2020): 312-20. [CrossRef]
- Killingley, Ben, Alex J. Mann, Mariya Kalinova, Alison Boyers, Niluka Goonawardane, Jie Zhou, Kate Lindsell, Samanjit S. Hare, Jonathan Brown, Rebecca Frise, Emma Smith, Claire Hopkins, Nicolas Noulin, Brandon Löndt, Tom Wilkinson, Stephen Harden, Helen McShane, Mark Baillet, Anthony Gilbert, Michael Jacobs, Christine Charman, Priya Mande, Jonathan S. Nguyen-Van-Tam, Malcolm G. Semple, Robert C. Read, Neil M. Ferguson, Peter J. Openshaw, Garth Rapeport, Wendy S. Barclay, Andrew P. Catchpole, and Christopher Chiu. "Safety, Tolerability and Viral Kinetics During Sars-Cov-2 Human Challenge in Young Adults." Nature Medicine 28, no. 5 (2022): 1031-41. [CrossRef]
- Yang, Li, Rong-juan Pei, Heng Li, Xin-na Ma, Yu Zhou, Feng-hua Zhu, Pei-lan He, Wei Tang, Ye-cheng Zhang, Jin Xiong, Shu-qi Xiao, Xian-kun Tong, Bo Zhang, and Jian-ping Zuo. "Identification of Sars-Cov-2 Entry Inhibitors among Already Approved Drugs." Acta Pharmacol Sin 42, no. 8 (2021): 1347-53. [CrossRef]
- Ghahremanpour, M. M., J. Tirado-Rives, M. Deshmukh, J. A. Ippolito, C. H. Zhang, I. Cabeza de Vaca, M. E. Liosi, K. S. Anderson, and W. L. Jorgensen. "Identification of 14 Known Drugs as Inhibitors of the Main Protease of Sars-Cov-2." ACS Med Chem Lett 11, no. 12 (2020): 2526-33. [CrossRef]
- Jain, R., and S. Mujwar. "Repurposing Metocurine as Main Protease Inhibitor to Develop Novel Antiviral Therapy for Covid-19." Struct Chem (2020): 1-13. [CrossRef]
- Odhar, H. A., S. W. Ahjel, Aama Albeer, A. F. Hashim, A. M. Rayshan, and S. S. Humadi. "Molecular Docking and Dynamics Simulation of Fda Approved Drugs with the Main Protease from 2019 Novel Coronavirus." Bioinformation 16, no. 3 (2020): 236-44. [CrossRef]
- Reznikov, L. R., M. H. Norris, R. Vashisht, A. P. Bluhm, D. Li, Y. J. Liao, A. Brown, A. J. Butte, and D. A. Ostrov. "Identification of Antiviral Antihistamines for Covid-19 Repurposing." Biochem Biophys Res Commun 538 (2021): 173-79. [CrossRef]
- Konrat, R., H. Papp, J. Kimpel, A. Rössler, V. Szijártó, G. Nagy, M. Madai, S. Zeghbib, A. Kuczmog, Z. Lanszki, T. Gesell, Z. Helyes, G. Kemenesi, F. Jakab, and E. Nagy. "The Anti-Histamine Azelastine, Identified by Computational Drug Repurposing, Inhibits Infection by Major Variants of Sars-Cov-2 in Cell Cultures and Reconstituted Human Nasal Tissue." Front Pharmacol 13 (2022): 861295. [CrossRef]
- Fischhuber, K., Z. Bánki, J. Kimpel, N. Kragl, A. Rössler, A. Bolze, B. Muellauer, J. Angerer, G. Nagy, E. Nagy, and V. Szijarto. "Antiviral Potential of Azelastine against Major Respiratory Viruses." Viruses 15, no. 12 (2023). [CrossRef]
- Klussmann, Jens Peter, Maria Grosheva, Peter Meiser, Clara Lehmann, Eszter Nagy, Valéria Szijártó, Gábor Nagy, Robert Konrat, Michael Flegel, Frank Holzer, Dorothea Groß, Charlotte Steinmetz, Barbara Scherer, Henning Gruell, Maike Schlotz, Florian Klein, Paula Aguiar de Aragão, Henning Morr, Helal Al Saleh, Andreas Bilstein, Belisa Russo, Susanne Müller-Scholtz, Cengizhan Acikel, Hacer Sahin, Nina Werkhäuser, Silke Allekotte, and Ralph Mösges. "Early Intervention with Azelastine Nasal Spray May Reduce Viral Load in Sars-Cov-2 Infected Patients." Sci Rep 13, no. 1 (2023): 6839. [CrossRef]
- (Dte.GHS), AIIMS/ ICMR-COVID-19 National Task Force/Joint Monitoring Group. "Aiims/ Icmr-Covid-19 National Task Force/Joint Monitoring Group (Dte.Ghs) Ministry of Health & Family Welfare, Government of India. Clinical Guidance for the Syndromic Management of Suspected Covid–19 Cases." https://dghs.gov.in/WriteReadData/News/202104290258250563281SyndromicapproachforCOVID-19.pdf.
- Marc, Aurélien, Marion Kerioui, François Blanquart, Julie Bertrand, Oriol Mitjà, Marc Corbacho-Monné, Michael Marks, and Jeremie Guedj. "Quantifying the Relationship between Sars-Cov-2 Viral Load and Infectiousness." eLife 10 (2021): e69302. [CrossRef]
- Winchester, Stephen, Sarah John, Kashif Jabbar, and Isaac John. "Clinical Efficacy of Nitric Oxide Nasal Spray (Nons) for the Treatment of Mild Covid-19 Infection." Journal of Infection 83, no. 2 (2021): 237-79. [CrossRef]
- Shmuel, Klang, Megiddo Dalia, Lapidot Tair, and Naparstek Yaakov. "Low Ph Hypromellose (Taffix) Nasal Powder Spray Could Reduce Sars-Cov-2 Infection Rate Post Mass-Gathering Event at a Highly Endemic Community: An Observational Prospective Open Label User Survey." Expert Review of Anti-infective Therapy 19, no. 10 (2021): 1325-30. [CrossRef]
- Sinha, S., K. N, V. K. Suram, S. S. Chary, S. Naik, V. B. Singh, M. K. Jain, C. P. Suthar, S. Borthakur, V. Sawardekar, N. Sk, N. Reddy, L. Talluri, P. Thakur, M. Reddy, M. Panapakam, and R. Vattipalli. "Efficacy and Safety of Molnupiravir in Mild Covid-19 Patients in India." Cureus 14, no. 11 (2022): e31508. [CrossRef]
- Bernal, Angélica Jayk, Monica M. Gomes da Silva, Dany B. Musungaie, Evgeniy Kovalchuk, Antonio Gonzalez, Virginia Delos Reyes, Alejandro Martín-Quirós, Yoseph Caraco, Angela Williams-Diaz, Michelle L. Brown, Jiejun Du, Alison Pedley, Christopher Assaid, Julie Strizki, Jay A. Grobler, Hala H. Shamsuddin, Robert Tipping, Hong Wan, Amanda Paschke, Joan R. Butterton, Matthew G. Johnson, and Carisa De Anda. "Molnupiravir for Oral Treatment of Covid-19 in Nonhospitalized Patients." New England Journal of Medicine 386, no. 6 (2022): 509-20. [CrossRef]
- Tandon, Monika, Wen Wu, Keith Moore, Stephen Winchester, Yuan-Po Tu, Christopher Miller, Rahul Kodgule, Amol Pendse, Shabbir Rangwala, and Shashank Joshi. "Sars-Cov-2 Accelerated Clearance Using a Novel Nitric Oxide Nasal Spray (Nons) Treatment: A Randomized Trial." The Lancet Regional Health - Southeast Asia 3 (2022).
- Dings, C., P. Meiser, F. Holzer, M. Flegel, D. Selzer, E. Nagy, R. Mösges, J. P. Klussmann, and T. Lehr. "Pharmacometric Modeling of the Impact of Azelastine Nasal Spray on Sars-Cov-2 Viral Load and Related Symptoms in Covid-19 Patients." Pharmaceutics 14, no. 10 (2022). [CrossRef]
| Parameter | Statistics | Azelastine 0.1% nasal spray + standard supportive care (N=122) |
Placebo + standard supportive care (N=129) |
Total (N=251) |
|---|---|---|---|---|
| Age (Year) | N | 122 | 129 | 251 |
| Mean (SD) | 39.3 (14.24) | 40.4 (15.93) | 39.9 (15.11) | |
| Median | 37.0 | 38.0 | 37.0 | |
| Min, Max | 19.0, 87.0 | 19.0, 88.0 | 19.0, 88.0 | |
| Height (cm) | n | 122 | 129 | 251 |
| Mean (SD) | 161.0 (6.99) | 160.0 (7.23) | 160.5 (7.12) | |
| Median | 159.0 | 157.0 | 158.0 | |
| Min, Max | 148.0, 177.8 | 147.3, 179.0 | 147.3, 179.0 | |
| Weight (kg) | n | 122 | 129 | 251 |
| Mean (SD) | 67.2 (8.26) | 66.2 (9.54) | 66.7 (8.94) | |
| Median | 67.6 | 65.9 | 67.0 | |
| Min, Max | 48.0, 84.6 | 42.7, 85.0 | 42.7, 85.0 | |
| BMI (kg/m2) | n | 122 | 129 | 251 |
| Mean (SD) | 26.0 (3.31) | 25.8 (3.30) | 25.9 (3.30) | |
| Median | 26.0 | 25.7 | 25.8 | |
| Min, Max | 17.9, 36.4 | 16.9, 33.7 | 16.9,36.4 | |
| Gender | ||||
| Male | n (%) | 81 (66.4) | 72 (55.8) | 153 (61.0) |
| Female | n (%) | 41 (33.6) | 57 (44.2) | 98 (39.0) |
| Race | ||||
| Asian | n (%) | 122 (100.0) | 129 (100.0) | 251 (100.0) |
| Ethnicity | ||||
| Not Hispanic or Latino | n (%) | 122 (100.0) | 129 (100.0) | 251 (100.0) |
| Smoking Status | ||||
| Ex smoker | n (%) | 1 (0.8) | 5 (3.9) | 6 (2.4) |
| Never | n (%) | 121 (99.2) | 124 (96.1) | 245 (97.6) |
| Ct value at baseline | Mean (SD) | 23.39 (3.875) | 23.68 (3.634) | 23.54 (3.749) |
| Median | 23.08 | 23.50 | 23.42 | |
| Min, Max | 14.2, 34.9 | 14.4, 35.3 | 14.2, 35.3 | |
| Vaccination Status | ||||
| Unvaccinated subjects | n (%) | 24 (19.7) | 30 (23.3) | 54 (21.5) |
| Vaccinated subjects | n (%) | 98 (80.3) | 99 (76.7) | 197 (78.5) |
| Dose 1 | 98 (80.3) | 99 (76.7) | 197 (78.5) | |
| Dose 2 | 47 (38.5) | 44 (34.1) | 91 (36.3) | |
| Dose 3 (Booster dose) | 4 (3.3) | 4 (3.1) | 8 (3.2) | |
| Visit | Statistics | Azelastine 0.1% Nasal Spray +Standard Supportive Care (N=122) |
Placebo + Standard Supportive Care (N=129) |
*p-value Between Treatment |
Total (N=251) |
|---|---|---|---|---|---|
| Baseline (day 1) | Mean (SD) | 0 (0) | 0 (0) | - | 0 (0) |
| Median | 0 | 0 | 0 | ||
| Min, Max | 0, 0 | 0, 0 | 0, 0 | ||
| Mean CFB through day 3 | Mean (SD) | -2.80 (0.274) | -2.62(0.261) | <0.0001 | -2.71 (0.282) |
| Median | -2.81 | -2.62 | -2.71 | ||
| Min, Max | -3.5, -2.1 | -3.2, -1.8 | -3.5, -1.8 | ||
| #p-value of difference | <0.0001 | <0.0001 | <0.0001 | ||
| Mean CFB through day 6 | Mean (SD) | -5.05 (0.293) | -4.94 (0.246) | 0.0013 | -4.99 (0.275) |
| Median | -5.04 | -4.95 | -4.98 | ||
| Min, Max | -6.0, -4.4 | -5.6, -4.2 | -6.0, -4.2 | ||
| #p-value of difference | <0.0001 | <0.0001 | <0.0001 | ||
| Mean CFB through day 11 | Mean (SD) | -5.93 (0.221) | -5.85 (0.205) | 0.0041 | -5.89 (0.216) |
| Median | -5.93 | -5.87 | -5.90 | ||
| Min, Max | -6.4, -5.3 | -6.3, -5.3 | -6.4, -5.3 | ||
| #p-value of difference | <0.0001 | <0.0001 | <0.0001 |
| Statistics | Azelastine 0.1% Nasal Spray +Standard Supportive Care (N=122) | Placebo + Standard Supportive Care (N=129) | p-value # between treatment | Total (N=251) |
|---|---|---|---|---|
| Mean (SD) | 1014.960 (35.2527) | 982.977 (31.6776) | <0.0001 | 998.523 (37.0381) |
| Median | 1015.56 | 984.46 | 998.58 | |
| Min, Max | 907.29, 1110.64 | 900.16, 1058.96 | 900.16, 1110.64 |
| System Organ Class / Preferred Term |
Statistics | Azelastine 0.1% nasal spray + standard supportive care (N=122) |
Placebo + standard supportive care (N=129) |
Total (N=251) |
|---|---|---|---|---|
| Gastrointestinal disorders | n (%) | 14 (11.5) | 13 (10.1) | 27 (10.8) |
| Nausea | n (%) | 13 (10.7) | 8 (6.2) | 21 (8.4) |
| Vomiting | n (%) | 1 (0.8) | 5 (3.9) | 6 (2.4) |
| General disorders and administration site conditions | n (%) | 16 (13.1) | 13 (10.1) | 29 (11.6) |
| Asthenia | n (%) | 15 (12.3) | 13 (10.1) | 28 (11.2) |
| Malaise | n (%) | 1 (0.8) | 0 | 1 (0.4) |
| Metabolism and nutrition disorders | n (%) | 1 (0.8) | 0 (0) | 1 (0.4) |
| Decreased appetite | n (%) | 1 (0.8) | 0 (0) | 1 (0.4) |
| Musculoskeletal and connective tissue disorders | n (%) | 6 (4.9) | 12 (9.3) | 18 (7.2) |
| Myalgia | n (%) | 6 (4.9) | 12 (9.3) | 18 (7.2) |
| Nervous system disorders | n (%) | 8 (6.6) | 10 (7.8) | 18 (7.2) |
| Headache | n (%) | 6 (4.9) | 10 (7.8) | 16 (6.4) |
| Ageusia | n (%) | 2 (1.6) | 0 (0) | 2 (0.8) |
| Respiratory, thoracic and mediastinal disorders | n (%) | 2 (1.6) | 3 (2.3) | 5 (2.0) |
| Cough | n (%) | 2 (1.6) | 1 (0.8) | 3 (1.2) |
| Oropharyngeal pain | n (%) | 0 (0) | 1 (0.8) | 1 (0.4) |
| Throat pain | n (%) | 0 (0) | 1 (0.8) | 1 (0.4) |
| Skin and subcutaneous tissue disorders | n (%) | 1 (0.8) | 0 (0) | 1 (0.4) |
| Pruritus | n (%) | 1 (0.8) | 0 (0) | 1 (0.4) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




