Submitted:
30 October 2024
Posted:
31 October 2024
You are already at the latest version
Abstract
Keywords:
Introduction
Perspectives on the Use of AI in Drug Development, Manufacturing, and Clinical Trials
A Review of the FDA and CTTI Joint Workshop 2024: Keynote Speaker
- Ethical Considerations: It emphasizes fairness, accountability, and transparency in AI systems to prevent discrimination and ensure responsible use.
- Research and Development: The Order calls for increased investment in AI research and development, encouraging collaboration among government, industry, and academia to advance technology while addressing safety and ethical concerns.
- Regulatory Framework for health care and drug development: This encompasses a wide range of activities, from clinical research to post-market surveillance, all subject to established regulations and FDA guidance. Relevant agencies are tasked with developing regulations and guidelines for AI development, deployment, and monitoring to ensure safety and ethical standards compliance.
- Quality Assurance: The Department of Health and Human Services (HHS) is responsible for establishing a strategy to maintain quality in AI-enabled healthcare technology through premarket assessment and post-market oversight. The testing and validation standards required to ensure data quality, reliability, reproducibility, and accuracy across drug development are also crucial. Incorporating open-source and real-world data into AI model development and appropriate documentation related to data source selection, inclusion, and exclusion is essential for effective AI implementation. Understanding how quality standards will impact overall product development requirements, both within and outside the U.S., is vital. Furthermore, determining the necessary transparency and reporting requirements to address trends and propose changes in light of postmarket safety issues or other real-world data is crucial.
- International Collaboration: The Order promotes international cooperation to establish global standards and best practices for AI.
- Public Engagement: It supports engaging the public and stakeholders in discussions about AI technologies, promoting transparency, and involving various communities in decision-making.
- Monitoring and Evaluation: Ongoing monitoring and evaluation of AI systems are outlined to assess their impact and effectiveness, with policies and regulations adapting based on emerging technologies and societal implications.
The Broader Role and Applications of AI in Drug and Biologics Development: Lessons from the FDA Workshop and Industry
Optimizing Model Design Through Multidisciplinary Expertise
Using the Data We Have, Creating the Data We Need: Clinical Development, Clinical Data Management, and Analysis
Balancing Model Performance, Explainability, and Transparency
Identifying Gaps, Addressing Challenges, and Charting the Path Forward
Data Integrity and Quality Challenges in AI-Driven Drug Development Governance Considerations: Practical Guidelines for AI Implementation
Access, Fairness, and Accountability: Lessons from Economic, Law, Ethics, and Politics:
Ethical and Compliance Challenges on AI’s Expanding Role in Clinical Trials
Ethical and Legal Considerations of Privacy and Nondiscrimination
Regulatory and Compliance Framework for AI in Drug Development
A Global Regulatory Landscape: EU vs. US and Industry Initiatives
Comparative Perspective on AI in Clinical Manufacturing and Commercialization
WHO Guidelines and Perspectives on AI
Integration and Regulation of AL/ML in Pharmaceutical Manufacturing
5.3.1. Digital Twins and Predictive Modeling
5.3.2. Emerging AI-Focused Standards for Advanced Manufacturing Technologies
5.3.3. AI-Enhanced Manufacturing Processes Monitoring
- Implementation of manufacturing data exchange standards
- Cybersecurity monitoring to safeguard network and information integrity
- Digital twin or digital surrogate simulations for process testing and control
- Reliability, prognostics, and health management of manufacturing equipment
- Product quality monitoring to ensure compliance with regulatory standards
- System-level evaluations to assess overall process efficiency and effectiveness
- Human interactivity and feedback mechanisms through natural language processing
- Trust and trustworthiness requirements for AI systems
Conclusions and Path Forward
- Personalization: Leveraging AI to advance personalized medicine, ensuring treatments are tailored to individual patient profiles.
- Regulatory Frameworks: Developing and refining robust AI validation and monitoring frameworks to ensure compliance and safety.
- Ethical Considerations: Addressing data privacy, security, and decision-making issues to maintain public trust and uphold patient rights.
References
- FLETCHER, K.H. Matter with a mind; a neurological research robot. Research 1951, 4, 305–307. [Google Scholar] [PubMed]
- Duan, Y.; Edwards, J.S.; Dwivedi, Y.K. Artificial intelligence for decision making in the era of Big Data – evolution, challenges and research agenda. International Journal of Information Management 2019, 48, 63–71. [Google Scholar] [CrossRef]
- Mijwil, M.M.; Abttan, R.A. Artificial Intelligence: A Survey on Evolution and Future Trends. Asian Journal of Applied Sciences 2021, 9. [Google Scholar] [CrossRef]
- Tufail, S.; Riggs, H.; Tariq, M.; Sarwat, A.I. Advancements and Challenges in Machine Learning: A Comprehensive Review of Models, Libraries, Applications, and Algorithms. Electronics 2023, 12, 1789. [Google Scholar] [CrossRef]
- Mariam, Z.; Niazi, S.K.; Magoola, M. Unlocking the Future of Drug Development: Generative AI, Digital Twins, and Beyond. BioMedInformatics 2024, 4, 1441–1456. [Google Scholar] [CrossRef]
- Zhang, X.-Y.; Liu, C.-L.; Suen, C.Y. Towards Robust Pattern Recognition: A Review. Proceedings of the IEEE 2020, 108, 894–922. [Google Scholar] [CrossRef]
- Statista. Global total corporate artificial intelligence (AI) investment from 2015 to 2022(in billion U.S. dollars) https://www.statista.com/statistics/941137/ai-investment-and-funding-worldwide/2024 [.
- TURINGAMI—COMPUTINGMACHINERYANDINTELLIGENCE Mind. 1950;LIX(236):433-60.
- Gardner, H. The Mind’s new science: Cognitive revolution in the computer age: Baisc Books, Inc.; 1985.
- Molenaar, I. The concept of hybrid human-AI regulation: Exemplifying how to support young learners’ self-regulated learning. Computers and Education: Artificial Intelligence 2022, 3, 100070. [Google Scholar] [CrossRef]
- McCorduck, P. Machines who think: A personal inquiry into the history and prospects of artificial intelligence. 1979.
- Newell, A. ; JC Shaw, and Herbert Simon “Report on a General Problem-Solving Program”. Carnegie Mellon University Archives; 1959. Report No.: 1982-0001.
- OECD Gender Equality https://www.oecd.org/en/topics/policy-issues/gender-equality.html2021 [.
- The EU AI Act: European Commission; 2024 [Available from: https://digital-strategy.ec.europa.eu/en/policies/regulatory-framework-ai.
- Policy Guidance on AI for Childern: Unicef- Innocenti – Global Office of Research and Foresight; 2021 [Available from: https://www.unicef.org/innocenti/reports/policy-guidance-ai-children.
- Muehlematter, U.J.; Bluethgen, C.; Vokinger, K.N. FDA-cleared artificial intelligence and machine learning-based medical devices and their 510(k) predicate networks. The Lancet Digital Health 2023, 5, e618–e26. [Google Scholar] [CrossRef]
- FDA Artificial Intelligence in Drug Manufacturing https://www.fda.gov/science-research/artificial-intelligence-and-medical-products/artificial-intelligence-fda-publications: FDA; 2023 [https://www.fda.gov/science-research/artificial-intelligence-and-medical-products/artificial-intelligence-fda-publications]. Available from: http://wcms-internet.fda.gov/media/165743/download?attachment.
- FDA Using Artificial Intelligence and Machine Learning in the development of Drug and Biological Products https://www.fda.gov/science-research/artificial-intelligence-and-medical-products/artificial-intelligence-fda-publications: FDA; 2023 [https://www.fda.gov/medical-devices/software-medical-device-samd/good-machine-learning-practice-medical-device-development-guiding-principles]. Available from: http://wcms-internet.fda.gov/media/167973/download?attachment.
- EMA Concept Paper on the revision of Annex 11 of the guidelines on Good Manufacturing Practice for medicinal products – Computerised Systems.
- EMA Concept Paper on the revision of Annex 11 of the guidelines on Good Manufacturing Practice for medicinal products – Computerised Systems https://www.gmp-compliance.org/gmp-news/revision-of-the-eu-gmp-guide-annex-11-computerised-systems-presentation-of-concept-paper: EMA; 2023 [https://www.gmp-compliance.org/gmp-news/revision-of-the-eu-gmp-guide-annex-11-computerised-systems-presentation-of-concept-paper]. Available from: https://www.ema.europa.eu/documents/regulatory-procedural-guideline/concept-paper-revision-annex-11-guidelines-good-manufacturing-practice-medicinal-products_en.pdf.
- Simeone, O. Machine Learning for Engineers.: Cambridge University press; 2022.
- Niazi, S.K.M.Z. Computer-Aided Drug Design and Drug Discovery: A Prospective Analysis. Pharmaceuticals (Basel) 2023, 17, 22. [Google Scholar] [CrossRef]
- Niazi, S.K.; Mariam, Z. Recent Advances in Machine-Learning-Based Chemoinformatics: A Comprehensive Review. International Journal of Molecular Sciences 2023, 24, 11488. [Google Scholar] [CrossRef]
- Pfizer. Pfizer Is Using AI to Discover Breakthrough Medicines https://insights.pfizer.com/pfizer-is-using-ai-to-discover-breakthrough-medicines/#:~:text=Breakthrough%20therapies%20require%20breakthrough%20technologies,tablets)%2C%20to%20patients%20faster.: Pfizer; 2022 [.
- AstraZeneca- Data Science and AI: Ben Sidders JJ, and Sajan Khosla. Unlocking new science insights https://www.astrazeneca.com/what-science-can-do/topics/data-science-ai/unlocking-potential-data-ai-driven-drug-discovery-development.html: AstraZeneca; 2023 [.
- EMA Reflection paper on the use of artificial intelligence in the lifecycle of medicines https://www.ema.europa.eu/en/news/reflection-paper-use-artificial-intelligence-lifecycle-medicines: EMA; 2023 [Available from: https://www.ema.europa.eu/use-artificial-intelligence-ai-medicinal-product-lifecycle.
- FDA Using Artificial Intelligence and Machine Learning in the development of Drug and Biological Products. 2023.
- Workshop To Consider Artificial Intelligence in Drug and Biological Product Development; Public Workshop. FDA website; 2024.
- FDA’s Diversity Action Plans to Improve Enrollment of Participants from Underrepresented Populations in Clinical Studies. 2024.
- FDA Public Workshop To Consider Artificial Intelligence in Drug and Biological Product Development https://www.federalregister.gov/documents/2024/07/10/2024-15125/workshop-to-consider-artificial-intelligence-in-drug-and-biological-product-development-public: FDA; 2024 [.
- Yadav, N.; Pandey, S.; Gupta, A.; Dudani, P.; Gupta, S.; Rangarajan, K. Data Privacy in Healthcare: In the Era of Artificial Intelligence. Indian Dermatol Online J 2023, 14, 788–792. [Google Scholar] [CrossRef] [PubMed]
- Noorbakhsh-Sabet, N.; Zand, R.; Zhang, Y.; Abedi, V. Artificial Intelligence Transforms the Future of Health Care. Am J Med 2019, 132, 795–801. [Google Scholar] [CrossRef] [PubMed]
- Reflection paper on the use of Artificial Intelligence (AI) in 5 the medicinal product lifecycle 2023.
- Qi Liu, R.H.; Hsieh, J.; Zhu, H.; Tiwari, M.; Liu, G.; Jean, D.; ElZarrad, M.K.; Fakhouri, T.; Berman, S.; Dunn, B.; Diamond, M.C.; Huang, S.-M. Landscape Analysis of the Application of Artificial Intelligence and Machine Learning in Regulatory Submissions for Drug Development From 2016 to 2021. 2022.
- House, T.W. Executive Order on Safe, Secure, and Trustworthy Development and Use of Artificial Intelligence https://www.whitehouse.gov/briefing-room/presidential-actions/2023/10/30/executive-order-on-the-safe-secure-and-trustworthy-development-and-use-of-artificial-intelligence/: The White House; 2023 [.
- House, T.W. Executive Order on AI (13960- Section 5): Promoting the Use of Trustworthy Artificial Intelligence in the Federal Government https://www.whitehouse.gov/briefing-room/presidential-actions/2023/10/30/executive-order-on-the-safe-secure-and-trustworthy-development-and-use-of-artificial-intelligence/: The White House; 2023 [.
- Freedman, D.H. Hunting for New Drugs with AI. Nature 2019, 576, S49–s53. [Google Scholar] [CrossRef]
- Tyzack, J.D.K. ; J Computational methods and tools to predict cytochrome P450 metabolism for drug discovery. Chem Biol Drug Des 2019, 93, 377–386. [Google Scholar] [CrossRef]
- Cyclica Drives Drug Discovery Through AI-Augmented Integrated Network of Cloud-Based Technologies together with Bayer. https://www.cyclicarx.com/press-releases/cyclica-drives-drug-discovery-through-ai-augmented-integrated-network-of-cloud-based-technologies-together-with-bayer: Business Wire; 2024 [.
- Exscientia reaches first major milestone with GSK https://www.research-tree.com/newsfeed/article/exscientia-reaches-first-major-milestone-with-gsk-876775: RNS-R; 2019 [.
- Exscientia Announces First AI-Designed Immuno-Oncology Drug to Enter Clinical Trials https://investors.exscientia.ai/press-releases/press-release-details/2021/Exscientia-Announces-First-AI-Designed-Immuno-Oncology-Drug-to-Enter-Clinical-Trials/default.aspx: Exscientia; 2021 [.
- The startup fighting cancer with AI https://www.wired.com/story/ai-cancer-drugs-berg-pharma-startup/: Wired; 2016 [.
- Top 22 Rising Startups Applying Artificial Intelligence in Cancer https://www.omdena.com/blog/top-startups-ai-in-cancer: Omdena; 2022 [.
- Baricitinib, First Identified By BenevolentAI As A COVID-19 Treatment, Is Granted Emergency Use In India In Response To Its Escalating Crisis https://www.benevolent.com/news-and-media/press-releases-and-in-media/baricitinib-first-identified-benevolentai-covid-19-treatment-granted-emergency-use-india-response-its-escalating-crisis/: Benevolent; 2021 [.
- Insilico, Taisho form AI partnership to tackle age-related diseases and mortal cells https://www.fiercebiotech.com/medtech/insilico-taisho-form-ai-partnership-to-tackle-age-related-diseases-and-mortal-cells: Fierce Biotech; 2020 [.
- FDA Discussion Paper- Artificial Intelligence and Machine Learning in Software as a Medical Device. 2021.
- Muehlematter, U.J.D.P.; Vokinger, K.N. Approval of artificial intelligence and machine learning-based medical devices in the USA and Europe (2015-20): A comparative analysis.: Lancet Digit Health; 2021.
- FDA Guidance on Data Integrity and Compliance with Drug cGMP. 2018.
- FDA. FDA Guidance on Data Integrity and Compliance with Drug cGMP https://www.fda.gov/regulatory-information/search-fda-guidance-documents/data-integrity-and-compliance-drug-cgmp-questions-and-answers: FDA; 2018 [Available from: https://www.google.com/url?sa=t&source=web&rct=j&opi=89978449&url=https://www.fda.gov/media/119267/download&ved=2ahUKEwjoouqW5vCIAxUXLFkFHYtONWYQFnoECBcQAQ&usg=AOvVaw3vi6io30HRwjEGMoYen3jB.
- FDA Good Clinical Practice https://www.fda.gov/science-research/clinical-trials-and-human-subject-protection/regulations-good-clinical-practice-and-clinical-trials2024 [.
- Kanate, A.S.; Kumar, A.; Dreger, P.; Dreyling, M.; Le Gouill, S.; Corradini, P.; et al. Maintenance Therapies for Hodgkin and Non-Hodgkin Lymphomas After Autologous Transplantation: A Consensus Project of ASBMT, CIBMTR, and the Lymphoma Working Party of EBMT. JAMA Oncol 2019, 5, 715–722. [Google Scholar] [CrossRef] [PubMed]
- HealthCare, E.D.f.t.Q.o.M. Biotherapeutics https://www.edqm.eu/en//biotherapeutics#P4%C2%A0Bio_Procedure2024 [.
- Bakker, E.; Starokozhko, V.; Kraaijvanger, J.W.M.; Heerspink, H.J.L.; Mol, P.G.M. Precision medicine in regulatory decision making: Biomarkers used for patient selection in European Public Assessment Reports from 2018 to 2020. Clin Transl Sci 2023, 16, 2394–2412. [Google Scholar] [CrossRef]
- FDA Good Machine Learning Practice for Medical Device Development: Guiding Principles. 2021.
- Creemers, J.H.A.; Ankan, A.; Roes, K.C.B.; Schröder, G.; Mehra, N.; Figdor, C.G.; et al. In silico cancer immunotherapy trials uncover the consequences of therapy-specific response patterns for clinical trial design and outcome. Nat Commun 2023, 14, 2348. [Google Scholar] [CrossRef]
- Vasey, B.; Nagendran, M.; Campbell, B.; Clifton, D.A.; Collins, G.S.; Denaxas, S.; et al. Reporting guideline for the early-stage clinical evaluation of decision support systems driven by artificial intelligence:, D.E.C.I.D.E.-A.I. Nat Med 2022, 28, 924–933. [Google Scholar] [CrossRef]
- Shi, M.; Jiang, R.; Hu, X.; Shang, J. A privacy protection method for health care big data management based on risk access control. Health Care Manag Sci 2020, 23, 427–442. [Google Scholar] [CrossRef]
- Wang, X.T.; Schuette, P.; Kam, M. FDA experiences with a centralized statistical monitoring tool. J Biopharm Stat. 2024, 1–7. [Google Scholar] [CrossRef]
- Koneswarakantha, B.; Ménard, T.; Rolo, D.; Barmaz, Y.; Bowling, R. Harnessing the Power of Quality Assurance Data: Can We Use Statistical Modeling for Quality Risk Assessment of Clinical Trials? Ther Innov Regul Sci 2020, 54, 1227–1235. [Google Scholar] [CrossRef]
- Khin, N.A.; Grandinetti, C.; Dixey, H.; Yu, B.; Skeete, R.; Ayalew, K.; et al. Tackling Challenging Data Integrity Topics in 2020: Update on Good Clinical Practice Perspectives from the, U. S.F.D.A.; MHRAUK Clin Pharmacol Ther 2022, 112, 31–43. [Google Scholar] [CrossRef] [PubMed]
- Wallach, J.D.; Deng, Y.; Polley, E.C.; Dhruva, S.S.; Herrin, J.; Quinto, K.; et al. Assessing the use of observational methods and real-world data to emulate ongoing randomized controlled trials. Clin Trials 2023, 20, 689–698. [Google Scholar] [CrossRef] [PubMed]
- Niazi, S.K.; Al-Shaqha, W.M.; Mirza, Z. Proposal of International Council for Harmonization (ICH) Guideline for the Approval of Biosimilars. J Mark Access Health Policy 2023, 11, 2147286. [Google Scholar] [CrossRef] [PubMed]
- FDA Advanced Manufacturing Technologies Designation Program https://www.fda.gov/regulatory-information/search-fda-guidance-documents/advanced-manufacturing-technologies-designation-program: FDA; 2024 [https://www.fda.gov/regulatory-information/search-fda-guidance-documents/advanced-manufacturing-technologies-designation-program].
- Niazi, S.K. Biosimilars Adoption: Recognizing and Removing the RoadBlocks. Clinicoecon Outcomes Res 2023, 15, 281–294. [Google Scholar] [CrossRef]
- Nicholas, S.W.G. Data, Rivalry and Government Power: Machine Learning Is Changing Everything. GLOB ASIA 2019, 23. [Google Scholar]
- Oren Bracha & Frank Pasquale, 93 CORNELL L. REV. 1149, 1180 (2008). . Federal Search Commission? Access, Fairness, and Accountability in the Law of Search. CORNELL L REV 2008, 93, 1149–1180.
- NOUSSAIRAJWVDGCN The Nexus Between Artificial Intelligence and Economic. Springer Briefs in Economics 2013, 97, 1–110.
- Michael Froomkin, I.K.J.P. When AIs Outperform Doctors: Confronting the Challenges of a Tort-Induced Over-Reliance on Machine Learning. ARIZ L REV 2019, 61, 88. [Google Scholar]
- Mazer, B.L.; Paulson, N.; Sinard, J.H. Protecting the Pathology Commons in the Digital Era. Arch Pathol Lab Med 2020, 144, 1037–1040. [Google Scholar] [CrossRef]
- Nomikos, G.G.; Feltner, D.E. Translational medicine in CNS drug development. London, United Kingdom ; San Diego, CA: Academic Press; 2019. xvi, 442 pages p.
- Ducci, F. NATURAL MONOPOLIES IN DIGITAL PLATFORM MARKETS. 5, editor: Cambridge University Press; 2020 June. 47-75 p.
- David Lehr & Paul Ohm, 51 653, 655 (2017). Playing with the Data: What Legal Scholars Should Learn About Machine Learning. UC DAVIS L REV 2017, 51, 653. [Google Scholar]
- Maurice, E. Stucke, 2 . 275, 285–286, 302 (2018). Should We Be Concerned About Data-opolies? 2 GEO L TECH REV 2018, 275, 285–286. [Google Scholar]
- Jon Schmid TS, Anton Shenk. Evaluating Natural Monopoly Conditions in the AI Foundation Model Market https://www.rand.org/pubs/research_reports/RRA3415-1.html: RAND; 2024 [.
- Calo, M.R. The Boundaries of Privacy Harm. 86 Indiana Law Journal 2011, 86, 1151–1152. [Google Scholar]
- Amanda Levendowski, 93 579 (2018). How Copyright Law Can Fix Artificial Intelligence’s Implicit Bias Problem. WASH L REV 2018, 93, 579. [Google Scholar]
- Thierer, A.O.S.A. Counterpoint: Regulators Should Allow the Greatest Space for AI Innovation https://cacm.acm.org/opinion/counterpoint-regulators-should-allow-the-greatest-space-for-ai-innovation/2018 [33].
- Solow-Niederman, A. Administering Artificial Intelligence. S CAL L REV 2020, 93. [Google Scholar] [CrossRef]
- EU AI Act. 2024.
- van Kolfschooten, H.B. A health-conformant reading of the GDPR’s right not to be subject to automated decision-making. Med Law Rev 2024, 32, 373–391. [Google Scholar] [CrossRef]
- Rezaeikhonakdar, D. AI Chatbots and Challenges of HIPAA Compliance for AI Developers and Vendors. J Law Med Ethics 2023, 51, 988–995. [Google Scholar] [CrossRef]
- Gottardelli, B.; Gatta, R.; Nucciarelli, L.; Tudor, A.M.; Tavazzi, E.; Vallati, M.; et al. GEN-RWD Sandbox: bridging the gap between hospital data privacy and external research insights with distributed analytics. BMC Med Inform Decis Mak 2024, 24, 170. [Google Scholar] [CrossRef]
- FDADP Using Artificial Intelligence and Machine Learning in the development of Drug and Biological Products. 2023.
- FDA Discussion Paper- Artificial Intelligence in Drug Manufacturing. 2024.
- FDA Diversity Action Plans to Improve Enrollment of Participants from Underrepresented Populations in Clinical Studies https://www.fda.gov/regulatory-information/search-fda-guidance-documents/diversity-action-plans-improve-enrollment-participants-underrepresented-populations-clinical-studies: FDA; 2024 [.
- Hutson, M. Cutting to the chase.: Nature, Health Sciences; 2024.
- Olaoye, E.F.G. Privacy and data protection in AI-enabled healthcare systems 2024.
- DG The FDA just opened the door to let AI make medical decisions on its own.: Quartz 2018; 2021.
- FDA permits marketing of artificial intelligence-based device to detect certain diabetes-related eye problems. . 2021.
- Ethically Aligned Design: A Vision for Prioritizing Human Well-being with Autonomous and Intelligent Systems. The IEEE Global Initiative on Ethics of Autonomous and Intelligent Systems.. IEEE; 2020.
- obin, A.; Ienca, M.; Vayena, E. The global landscape of AI ethics guidelines.: Nature Machine Intelligence; 2019; pp. 389–99.
- WHO (2021) Report, Ethics and governance of arti fi cial intelligence.: WHO; 2021.
- WHO (2023) Regulatory considerations on arti fi cial intelligence for health. World Health Organization.. WHO; 2023.
- Liu, C.; Ayyar, V.S.; Zheng, X.; Chen, W.; Zheng, S.; Mody, H.; et al. Model-Based Cellular Kinetic Analysis of Chimeric Antigen Receptor-T Cells in Humans. Clin Pharmacol Ther 2021, 109, 716–727. [Google Scholar] [CrossRef]
- Kirouac, D.C.; Zmurchok, C.; Morris, D. Making drugs from T cells: The quantitative pharmacology of engineered T cell therapeutics. npj Systems Biology and Applications. npj Systems Biology and Applications; 2024.
- Tanna, J.; McCann, C.D.; Smith, R.; Pitino, A.; Asgedom, A.; Kong, S.L.; et al. Environmental monitoring of current good manufacturing practices cleanroom facilities for manufacturing of cellular therapy products in an academic hospital setting. Cytotherapy 2024. [Google Scholar] [CrossRef]
- Niazi, S.K. Gene Editing: The Regulatory Perspective. Encyclopedia; 2023; pp. 1345–57.
- FDA draft Guidance- Computer Software Assurance for Production and Quality System Software. 2022.
- EMA Concept Paper on the revision of Annex 11 of the guidelines on Good Manufacturing Practice for medicinal products – Computerised Systems.
- Consortium, D.T. Two Years of Twins https://www.digitaltwinconsortium.org: Digital Twin Consortium; 2022 [Available from: https://www.digitaltwinconsortium.org/wp-content/uploads/sites/3/2022/06/DTC-Year-2-Report.pdf.
- reference, O. Digital Twin Consortium. 2022.
- Hartman, C.R.; Ferguson, D.M.; Goh, J.Y. AI-Driven Innovations in Pharmaceutical Manufacturing: A New Paradigm for Process Optimization.: Journal of Manufacturing Science and Engineering; 2023; pp. 041005.
- Z.Mariam SNaMM. Unlocking the Future of Drug Development: Generative AI, Digital Twins, and Beyond BioMedInformatics; 2024; pp. 1441–56.
- Rathore, A.S.; Saxena, N.; Thakur, G.; Mishra, S. Artificial intelligence and machine learning applications in biopharmaceutical manufacturing.: Trends in Biotechnology; 2023; pp. 497–510.
- Joslyn, L.R.; Huang, W.; Miles, D.; Hosseini, I.; Ramanujan, S. “Digital twins elucidate critical role of T. NPJ Syst Biol Appl 2024, 10, 11. [Google Scholar] [CrossRef] [PubMed]
- Kuriakose, S.M.; Joseph, J.A.R.; Kollinal, R. The Rise of Digital Twins in Healthcare: A Mapping of the Research Landscape. Cureus 2024, 16, e65358. [Google Scholar] [CrossRef] [PubMed]
- Venkatesh, K.P.R.M.; Kvedar, J.C. Health digital twins as tools for precision medicine: considerations for computation, implementation, and regulation. Npj Digit Med 2022, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Regenmortel, M.H.V. Personalized medicine and digital twins: Emerging trends in biopharmaceutical development.: Trends in Biotechnology; 2023; pp. 12–21.
- Rani, S.; Bhambri, P.; Kumar, S.; Pareek, P.K.; Elngar, A.A. AI-driven digital twin and Industry 4.0 : a conceptual framework with applications. First edition. ed. Boca Raton, FL: CRC Press; 2024. pages cm. p.
- Schroeder, M.; Zhang, J.; Santamaria, F. Leveraging digital twin technology for predictive biomanufacturing. Journal of Biotechnology and Bioprocess Engineering 2022, 899–908. [Google Scholar]
- Zhang, H.; Ling, S.; Yao, W. The impact of digital twins on the future of biomanufacturing. Journal of Computational Science and Engineering 2021, 115–24. [Google Scholar]
- Feng, S.; Lee, T.J.; Wang, X. The rise of digital twins in pharmaceutical manufacturing: Current challenges and future prospects.: Pharmaceutical Technology; pp. 54–65.
- G. S. Use case scenarios for digital twin implementation based on ISO 23247.. NIST Advanced Manufacturing Series (NIST AMS) - 400-2 2021.

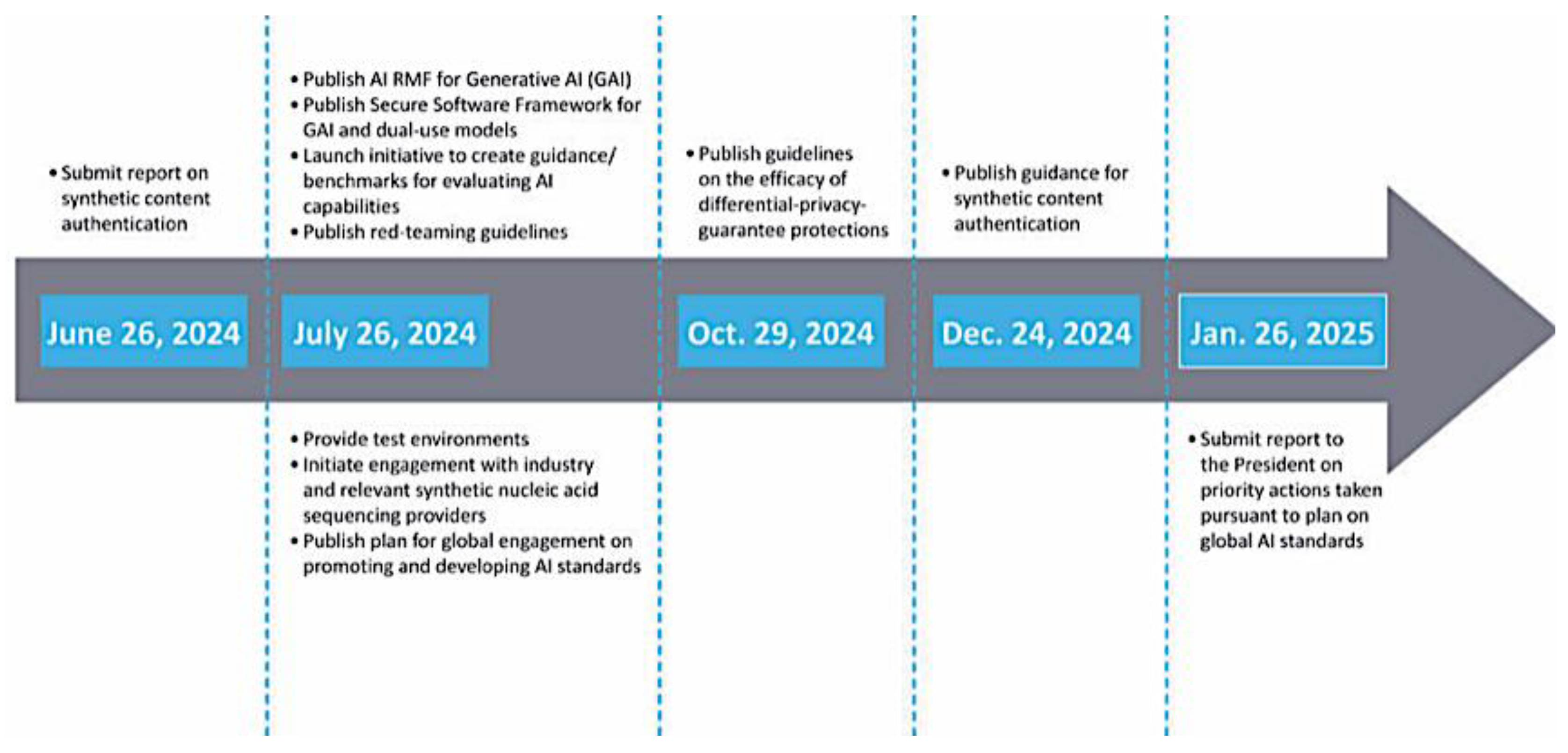
| Enhance AI Utilization in Drug Development Goals: Improve Safety, Efficacy, Efficiency Focus Areas: Data Quality, Transparency, Risk Management | ||
|---|---|---|
| FDA | NIST | Other Agencies |
| •Ensure AI tools meet •safety and efficacy standards •Review AI in Drug Development •Provide Guidelines for AI integration •Risk Management and Post-Market Surveillance •Stakeholder Engagement |
•Develop AI technology •Standard and Benchmarks •Promoting Performance metrics and Best Practices •Provide Technical guidance •Risk management framework •Governance and Policy |
•Ethics and Privacy •Data Protection •Global Partnership •International Coordination •Innovative Support |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




