Submitted:
29 October 2024
Posted:
30 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Experimental Animals
2.2. Experimental Design and Treatment
2.3. Laminar Tissue Sampling
2.4. RNA Isolation and cDNA Synthesis
2.5. Quantitative Real-Time Polymerase Chain Reaction (RT-qPCR)
2.6. Western Blot
2.7. Immunohistochemistry
2.8. Statistical Analysis
3. Results
3.1. Clinical Manifestation of Dairy Cow Laminitis
3.2. Apoptosis-Related Genes Expression in Laminar Tissue of Laminitis Dairy Cow
3.3. Apoptosis-Related Proteins Expression in Laminar Tissue of Laminitis Dairy Cow
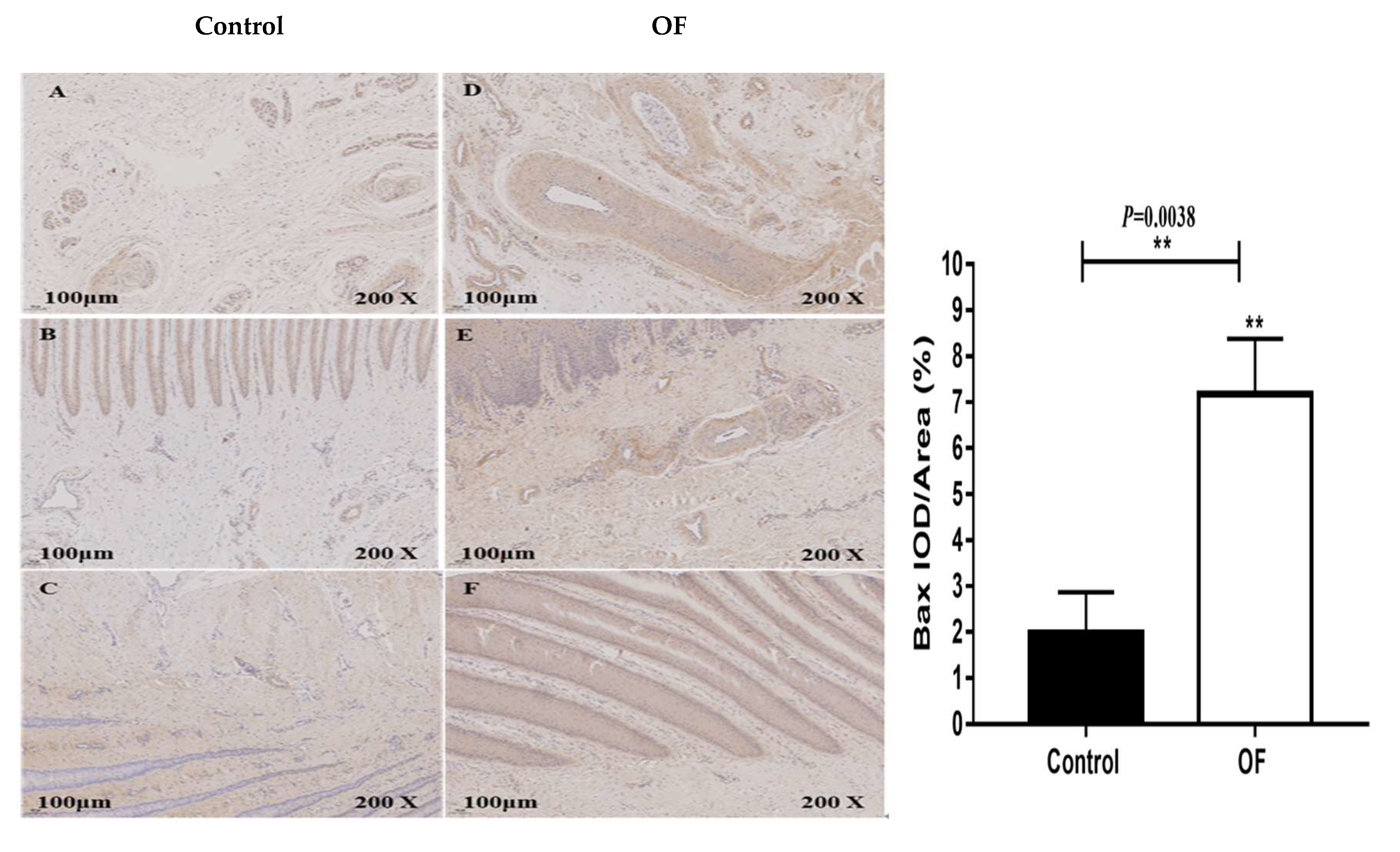
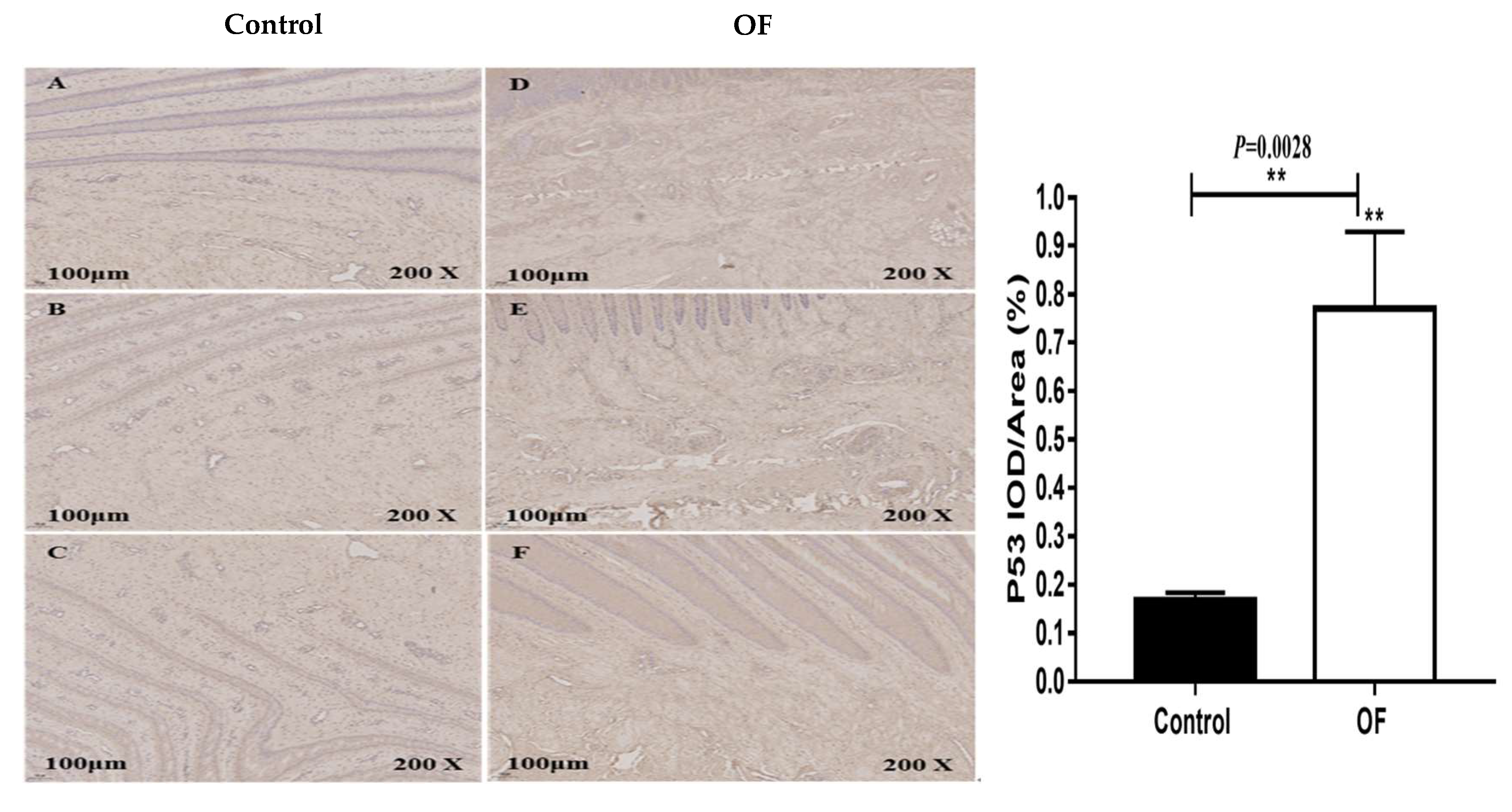
3.4. Immuno-Expression of Bax and P53 Proteins in Laminar Tissue of Laminitis Dairy Cow
4. Discussion
5. Conclusions
Author Contributions
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, X.; Ding, J.; Li, Y.; Song, Q.; Li, S.; Hayat, M.A.; Zhang, J.; Wang, H. The changes of inflammatory mediators and vasoactive substances in dairy cows’ plasma with pasture-associated laminitis. BMC Vet. Res. 2020, 16, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, S.A. Clinical, morphological and experimental studies of laminitis in cattle. Acta Vet. Scand. 1963, 4, 1–304. [Google Scholar]
- Greenough, P.R. Bovine laminitis and lameness: a hands on approach. Els. Health Sci. 2007. [Google Scholar]
- Randall, L.V.; Green, M.J.; Huxley, J.N. Use of statistical modelling to investigate the pathogenesis of claw horn disruption lesions in dairy cattle. Vet. J. 2018, 238, 41–48. [Google Scholar] [CrossRef] [PubMed]
- van der Spek, D.; van Arendonk, J.A.M.; Vallee, A.A.A.; Bovenhuis, H. Genetic parameters for claw disorders and the effect of preselecting cows for trimming. J. Dairy Sci. 2013, 96, 6070–6078. [Google Scholar] [CrossRef]
- Thoefner, M.B.; Pollitt, C.C.; Van-Eps, A.W.; Milinovich, G.J.; Trott, D.J.; Wattle, O.; Andersen, P.H. Acute bovine laminitis: A new induction model using alimentary oligofructose overload. J. Dairy Sci. 2004, 87, 2932–2940. [Google Scholar] [CrossRef]
- Danscher, A.M.; Enemark, J.M.D.; Telezhenko, E.; Capion, N.; Ekstrom, C.T.; Thoefner, M.B. Oligofructose overload induces lameness in cattle. J. Dairy Sci. 2009, 92, 607–616. [Google Scholar] [CrossRef]
- Thoefner, M.B.; Wattle, O.; Pollitt, C.C.; French, K.R.; Nielsen, S.S. Histopathology of oligofructose-induced acute laminitis in heifers. J. Dairy Sci. 2005, 88, 2774–2782. [Google Scholar] [CrossRef]
- Mendes, H.M.; Casagrande, F.P.; Lima, I.R.; Souza, C.H.; Gontijo, L.D.; Alves, G.E.; Vasconcelos, A.C.; Faleiros, R.R. Histopathology of dairy cows' hooves with signs of naturally acquired laminitis. Pesquisa. Vet. Brasil. 2013, 33, 613–619. [Google Scholar] [CrossRef]
- Sousa, R.D.S.; Oliveira, F.L.C.; Dias, M.R.B.; Minami, N.S.; Amaral, L.; Santos, J.A.A.D.; Barreto, J.R.A.; Minervino, A.H.H.; Ortolani, E.L. Characterization of oligofructose-induced acute rumen lactic acidosis and the appearance of laminitis in zebu cattle. Animals. 2020, 10, 429. [Google Scholar] [CrossRef]
- Leise, B.S.; Faleiros, R.R.; Watts, M.; Johnson, P.J.; Black, S.J.; Belknap, J.K. Laminar inflammatory gene expression in the carbohydrate overload model of equine laminitis. Equine. Vet. J. 2011, 43, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Dern, K.; van-Eps, A.; Wittum, T.; Watts, M.; Pollitt, C.; Belknap, J. Effect of continuous digital hypothermia on lamellar inflammatory signaling when applied at a clinically-relevant timepoint in the oligofructose laminitis model. J. Vet. Int. Med. 2018, 32, 450–458. [Google Scholar] [CrossRef] [PubMed]
- Raber, M.; Lischer, C.J.; Geyer, H.; Ossent, P. The bovine digital cushion – a descriptive anatomical study. Vet. J. 2004, 167, 167,258–264. [Google Scholar] [CrossRef] [PubMed]
- Boosman, R.; Nemeth, F.; Gruys, E. Bovine laminitis: clinical aspects, pathology and pathogenesis with reference to acute equine laminitis. Vet. Q. 1991, 13, 163–171. [Google Scholar] [CrossRef]
- Vermunt, J. “Subclinical” laminitis in dairy cattle. N. Z. Vet. J. 1992, 40, 133–138. [Google Scholar] [CrossRef]
- Alvergnas, M.; Strabel, T.; Rzewuska, K.; Sell-Kubiak, E. Claw disorders in dairy cattle: Effects on production, welfare and farm economics with possible prevention methods. Livest. Sci. 2019, 222, 54–64. [Google Scholar] [CrossRef]
- Potterton, S.L.; Bell, N.J.; Whay, H.R.; Berry, E.A.; Atkinson, O.C.; Dean, R.S.; Main, D.C.; Huxley, J.N. A descriptive review of the peer and non-peer reviewed literature on the treatment and prevention of foot lameness in cattle published between 2000 and 2011. Vet. J. 2012, 193, 612–616. [Google Scholar] [CrossRef]
- Nagata, S.; Tanaka, M. Programmed cell death and the immune system. Nat. Rev. Immunol. 2017, 17, 333–340. [Google Scholar] [CrossRef]
- Catunda, A.P.; Alves, G.E.; Leme, F.O.; Carvalho, A.M.; Leise, B.S.; Johnson, P.J.; Faleiros, R.R. Apoptosis in epithelial cells and its correlation with leukocyte accumulation in lamellar tissue from horses subjected to experimental sepsis-associated laminitis. Res. Vet. Sci. 2021, 136, 318–323. [Google Scholar] [CrossRef]
- Faleiros, R.R.; Stokes, A.M.; Eades, S.C.; Kim, D.Y.; Paulsen, D.B.; Moore, R.M. Assessment of apoptosis in epidermal lamellar cells in clinically normal horses and those with laminitis. Am. J. Vet. Res. 2004, 65, 578–585. [Google Scholar] [CrossRef]
- Mungall, B.A.; Kyaw-Tanner, M.; Pollitt, C.C. In vitro evidence for a bacterial pathogenesis of equine laminitis. Vet. Microbiol. 2001, 79, 209–223. [Google Scholar] [CrossRef] [PubMed]
- Olivetti, G.; Abbi, R.; Quaini, F.; Kajstura, J.; Cheng, W.; Nitahara, J.A.; Quaini, E.; Di Loreto, C.; Beltrami, C.A.; Krajewski, S.; Reed, J.C. Apoptosis in the failing human heart. New England J. Med. 1997, 336, 1131–1141. [Google Scholar] [CrossRef] [PubMed]
- Fliss, H.; Gattinger, D. Apoptosis in ischemic and reperfused rat myocardium. Circ. Res. 1996, 79, 949–956. [Google Scholar] [CrossRef] [PubMed]
- Gottlieb, R.A.; Burleson, K.O.; Kloner, R.A.; Babior, B.M.; Engler, R.L. Reperfusion injury induces apoptosis in rabbit cardiomyocytes. J. Clin. Invest. 1994, 94, 1621–1628. [Google Scholar] [CrossRef]
- Kohli, V.; Selzner, M.; Madden, J.F.; Bentley, R.C.; Clavien, P.A. Endothelial cell and hepatocyte deaths occur by apoptosis after ischemia-reperfusion injury in the rat liver1, 2. Transplant. 1999, 67, 1099–1105. [Google Scholar] [CrossRef]
- Morgan, S.J.; Hood, D.M.; Wagner, I.P.; Postl, S.P. Submural histopathologic changes attributable to peracute laminitis in horses. Ame. J. Vet. Res. 2003, 64, 829–834. [Google Scholar] [CrossRef]
- Mungall, B.A.; Pollitt, C.C. Thermolysin activates equine lamellar hoof matrix metalloproteinases. J. Comp. Pathol. 2002, 126, 9–12. [Google Scholar] [CrossRef]
- Ding, J.; Shi, M.; Wang, L.; Qi, D.; Tao, Z.; Hayat, M.A.; Liu, T.; Zhang, J.T.; Wang, H. Gene Expression of Metalloproteinases and Endogenous Inhibitors in the Lamellae of Dairy Heifers With Oligofructose-Induced Laminitis. Front. Vet. Sci. 2020, 7, 597827. [Google Scholar] [CrossRef]
- Sprecher, D.E.; Hostetler, D.E.; Kaneene, J.B. A lameness scoring system that uses posture and gait to predict dairy cattle reproductive performance. Theriogenol. 1997, 47, 1179–1187. [Google Scholar] [CrossRef]
- Edmonson, A.J.; Lean, I.J.; Weaver, L.D.; Farver, T.; Webster, G. A body condition scoring chart for Holstein dairy cows. J. Dairy Sci. 1989, 72, 68–78. [Google Scholar] [CrossRef]
- Ding, J.; Li, S.; Jiang, L.; Li, Y.; Zhang, X.; Song, Q.; Hayat, M.A.; Zhang, J.T.; Wang, H. Laminar inflammation responses in the oligofructose overload induced model of bovine laminitis. Front. Vet. Sci. 2020, 7, 351. [Google Scholar] [CrossRef] [PubMed]
- Hayat, M.A.; Ding, J.; Li, Y.U.; Zhang, X.; Zhang, J.I.; Li, S.; Wang, H.B. Determination of the activity of selected antioxidant enzymes during bovine laminitis, induced by oligofructose overload. Med. Weter. 2020, 76, 289–295. [Google Scholar] [CrossRef]
- Hayat, M.A.; Ding, J.; Zhang, X.; Liu, T.; Zhang, J.; Bokhari, S.G.; Akbar, H.; Wang, H. Enhanced Autophagy in Damaged Laminar Tissue of Acute Laminitis Induced by Oligofructose Overloading in Dairy Cows. Animals. 2023, 13, 2478. [Google Scholar] [CrossRef] [PubMed]
- Concha, C.; Carretta, M.D.; Alarcón, P.; Conejeros, I.; Gallardo, D.; Hidalgo, A.I.; Tadich, N.; Cáceres, D.D.; Hidalgo, M.A.; Burgos, R.A. Oxidative response of neutrophils to platelet-activating factor is altered during acute ruminal acidosis induced by oligofructose in heifers. J. Vet. Sci. 2014, 15, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Alarcon, P.; Hidalgo, A.I.; Manosalva, C.; Cristi, R.; Teuber, S.; Hidalgo, M.A.; Burgos, R.A. Metabolic disturbances in synovial fluid are involved in the onset of synovitis in heifers with acute ruminal acidosis. Sci. Reports. 2019, 9, 5452. [Google Scholar] [CrossRef]
- Snyder, E.; Credille, B. Diagnosis and treatment of clinical rumen acidosis. Vet. Clin. N. Am. Food A. 2017, 33, 451–461. [Google Scholar] [CrossRef]
- Khafipour, E.; Krause, D.; Plaizier, J. A grain-based subacute ruminal acidosis challenge causes translocation of lipopolysaccharide and triggers inflammation. J. Dairy Sci. 2009, 92, 1060–1070. [Google Scholar] [CrossRef]
- Abaker, J.A.; Xu, T.L.; Jin, D. Lipopolysaccharide derived from the digestive tract provokes oxidative stress in the liver of dairy cows fed a high-grain diet. J. Dairy Sci. 2017, 100, 666–678. [Google Scholar] [CrossRef]
- Zhao, C.; Liu, G.; Li, X.; Guan, Y.; Wang, Y.; Yuan, X.; Sun, G.; Wang, Z.; Li, X. Inflammatory mechanism of Rumenitis in dairy cows with subacute ruminal acidosis. BMC Vet. Res. 2018, 14, 1–8. [Google Scholar] [CrossRef]
- Wu, Z.; Deng, J.; Zhou, H.; Tan, W.; Lin, L.; Yang, J. Programmed cell death in sepsis associated acute kidney injury. Front. Med. 2022, 9, 883028. [Google Scholar] [CrossRef]
- Zhang, Z.; Bai, L.; Hou, L.; Deng, H.; Luan, S.; Liu, D.; Huang, M.; Zhao, L. Trends in targeting Bcl-2 anti-apoptotic proteins for cancer treatment. Europ. J. Med. Chem. 2022, 232, 114184. [Google Scholar] [CrossRef] [PubMed]
- Korsmeyer, S.J. BCL-2 gene family and the regulation of programmed cell death. Cancer Res. 1999, 59, 59,1693s–1700s. [Google Scholar] [CrossRef] [PubMed]
- Suen, D.F.; Norris, K.L.; Youle, R.J. Mitochondrial dynamics and apoptosis. Genes Dev. 2008, 22, 1577–1590. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, Y.; Karbowski, M.; Yamaguchi, H.; Kazi, A.; Wu, J.; Sebti, S.M.; Youle, R.J.; Wang, H.G. Loss of Bif-1 suppresses Bax/Bak conformational change and mitochondrial apoptosis. Mol. Cell. Biol. 2005, 25, 9369–9382. [Google Scholar] [CrossRef]
- Cuddeback, S.M.; Yamaguchi, H.; Komatsu, K.; Miyashita, T.; Yamada, M.; Wu, C.; Singh, S.; Wang, H.G. Molecular cloning and characterization of Bif-1: a novel Src homology 3 domain-containing protein that associates with Bax. J. Biol. Chem. 2001, 276, 20559–20565. [Google Scholar] [CrossRef]
- Dai, H.; Ma, N.; Chang, G.; Aabdin, Z.U.; Shen, X. Long-term high-concentrate diet feeding induces apoptosis of rumen epithelial cells and inflammation of rumen epithelium in dairy cows. Animal Biotechnol. 2020, 14, 1–8. [Google Scholar] [CrossRef]
- Du, X.; Chen, L.; Huang, D.; Peng, Z.; Zhao, C.; Zhang, Y.; Zhu, Y.; Wang, Z.; Li, X.; Liu, G. Elevated apoptosis in the liver of dairy cows with ketosis. Cellular Physiol. Biochem. 2017, 43, 568–578. [Google Scholar] [CrossRef]
- Harris, S.L.; Levine, A.J. The p53 pathway: positive and negative feedback loops. Oncogene. 2005, 24, 2899–2908. [Google Scholar] [CrossRef]
- Somal, A.; Aggarwal, A.; Upadhyay, R.C. Effect of thermal stress on expression profile of apoptosis related genes in peripheral blood mononuclear cells of transition Sahiwal cow. Iranian J. Vet. Res. 2015, 16, 16,137–141. [Google Scholar]
- Boatright, K.M.; Salvesen, G.S. Mechanisms of caspase activation. Cur. Opi. Cell Biol. 2003, 15, 725–731. [Google Scholar] [CrossRef]
- Deng, C.; Li, J.; Li, L.; Sun, F.; Xie, J. Effects of hypoxia ischemia on caspase-3 expression and neuronal apoptosis in the brain of neonatal mice. Exp. Therap. Med. 2019, 17, 4517–4521. [Google Scholar] [CrossRef] [PubMed]
- ul Aabdin, Z.; Cheng, X.; Dai, H.; Wang, Y.; Sahito, B.; Roy, A.C.; Memon, M.A.; Shen, X. High-concentrate feeding to dairy cows induces apoptosis via the NOD1/Caspase-8 pathway in mammary epithelial cells. Genes. 2020, 11, 107. [Google Scholar] [CrossRef] [PubMed]


| Genes | RefSeq accession No. | Primer sequences (5`-3`) |
|---|---|---|
| Bcl2 | NM_001166486.1 | Forward: AGGGGTCATGTGTGTGGAGAGC Reverse: GTGTGCAGGTGCCGGTTCAG |
| Bax | NM_173894.1 | Forward: CGGCCTCCTCTCCTACTTTGGG Reverse: TGGTGAGCGAGGCGGTGAG |
| P53 | NM_174201.2 | Forward: AGACCGCCGCACTGAGGAAG Reverse: TGTGGAGAGGAGCTGGTGTTGG |
| GAPDH | DQ402990 | Forward: GGGTCATAAGTCCCTCCACGA Reverse: GGTCATAAGTCCCTCCACGA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




