Submitted:
28 October 2024
Posted:
28 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Study Design
2.2. Data Collection
2.3. Statistical Analysis
3. Results
3.1. Baseline Characteristics
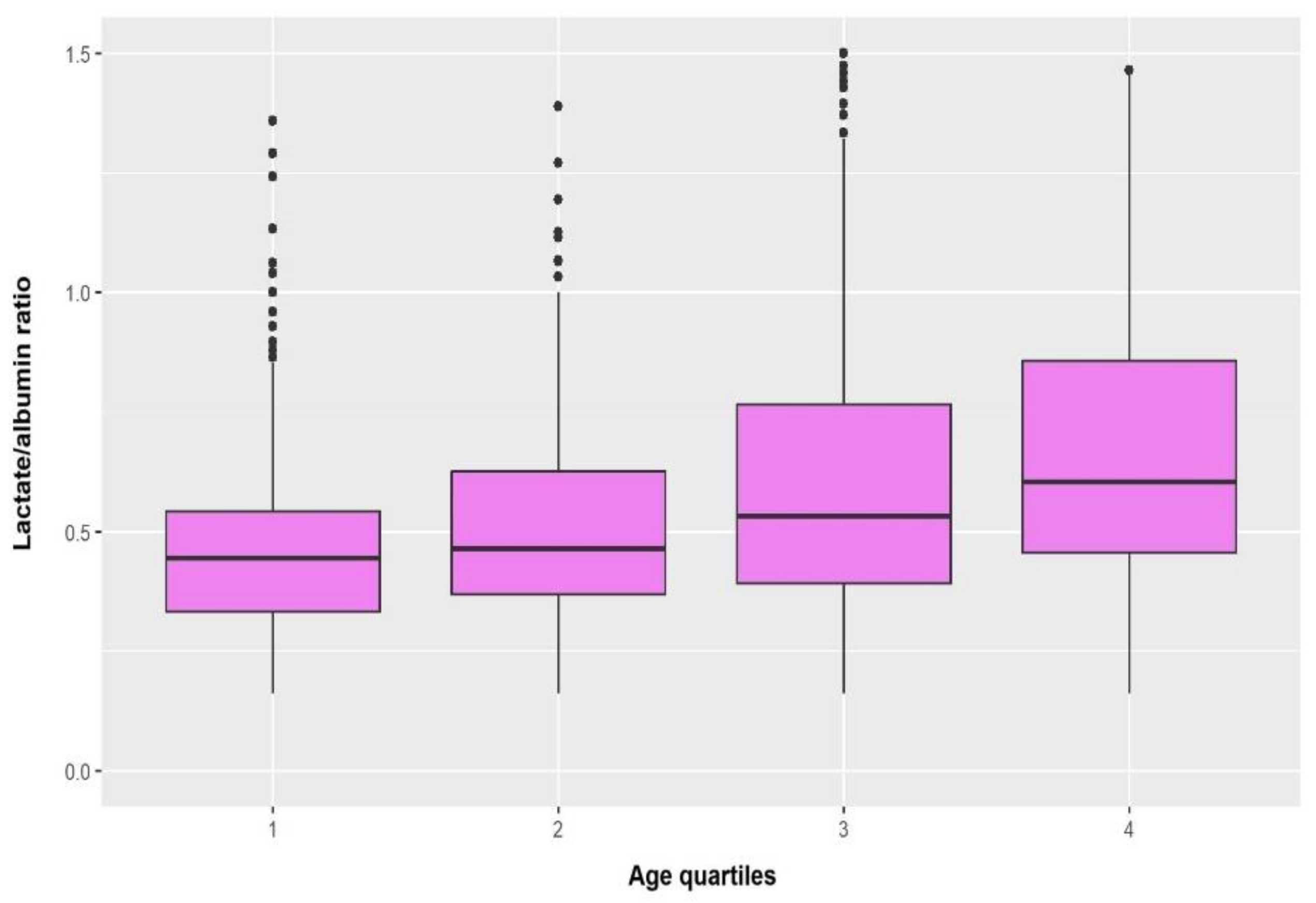
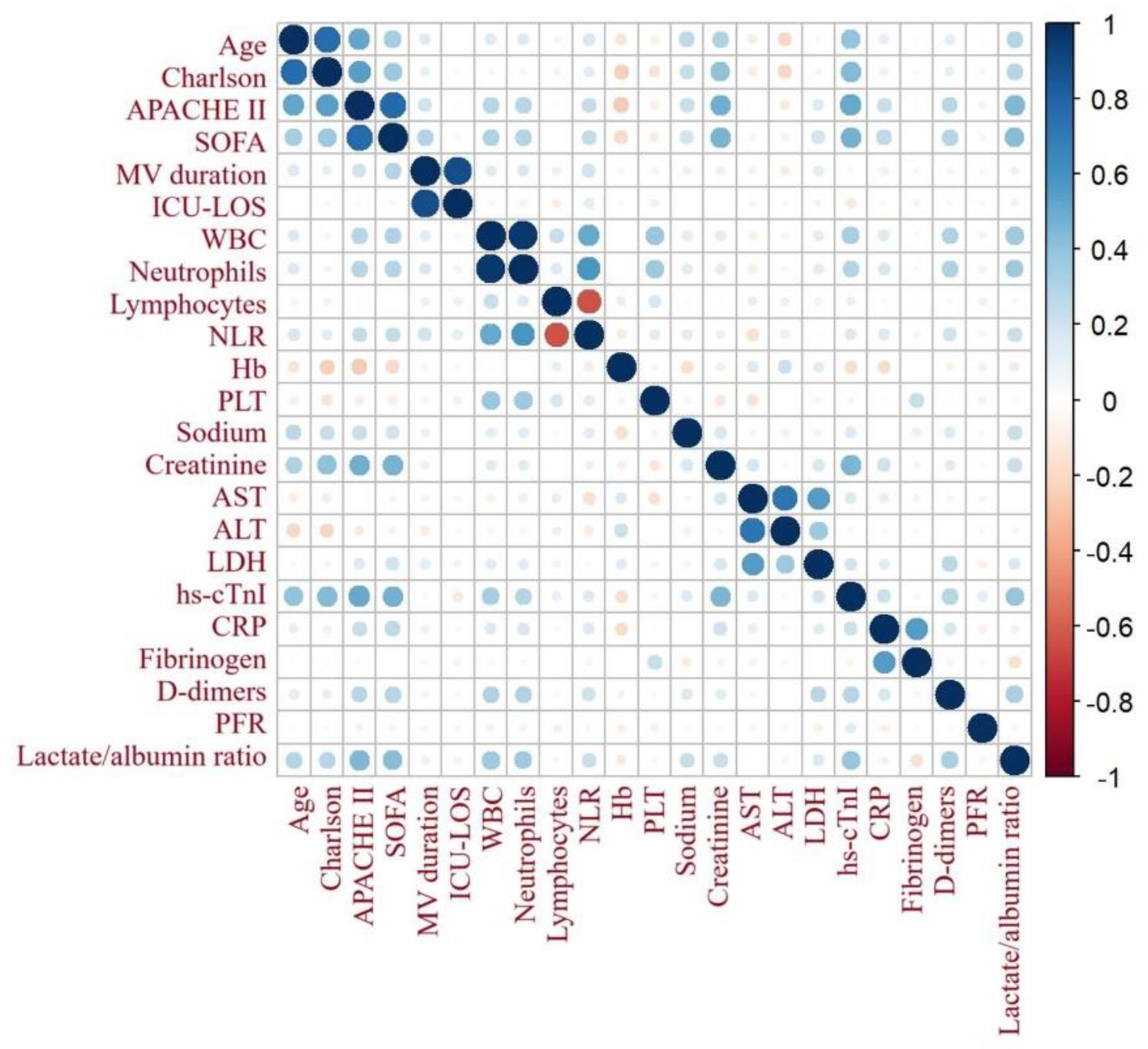
3.2. Correlations of Lactate/Albumin Ratio with Other Variables
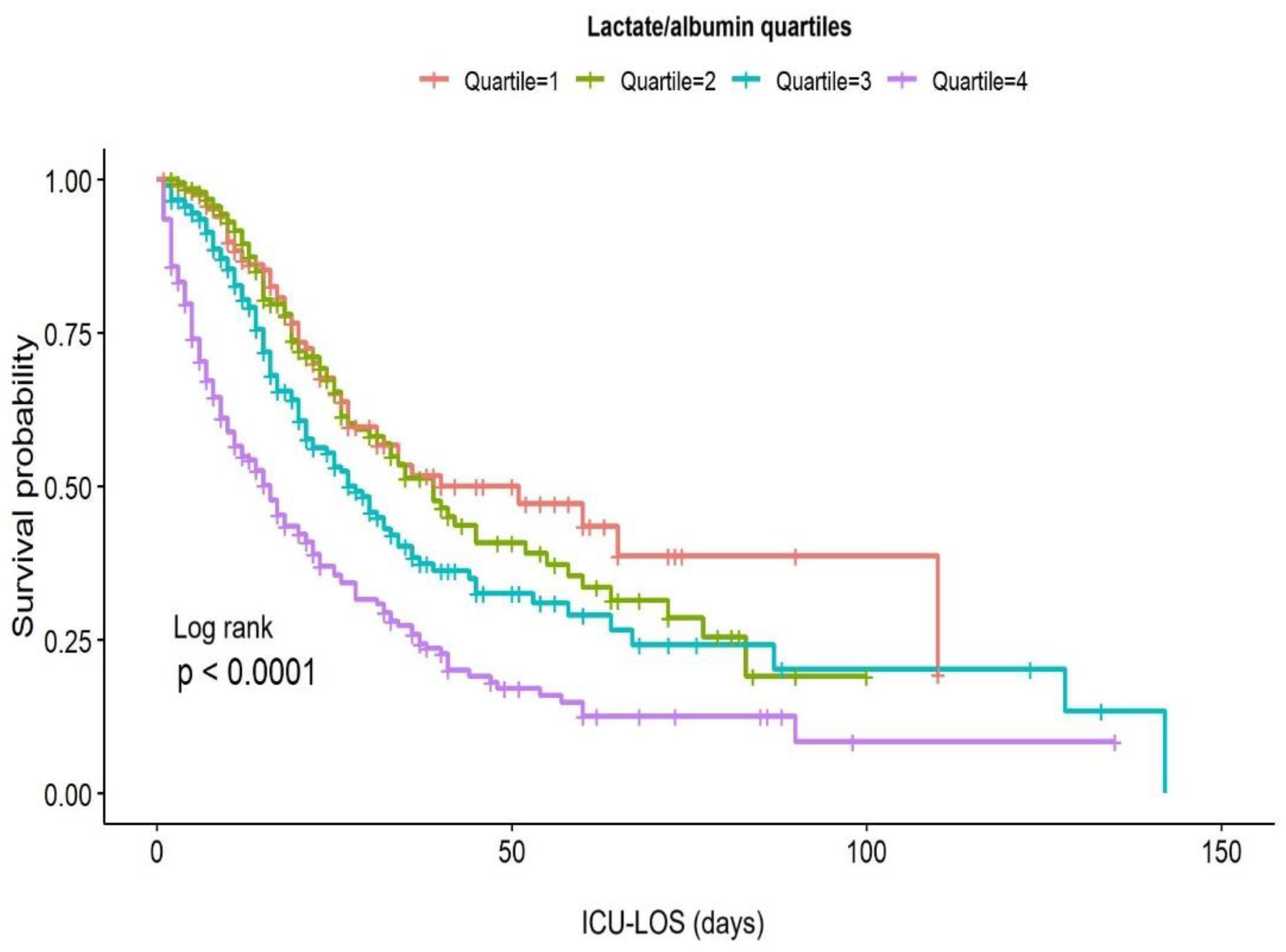
3.3. Survival Analysis
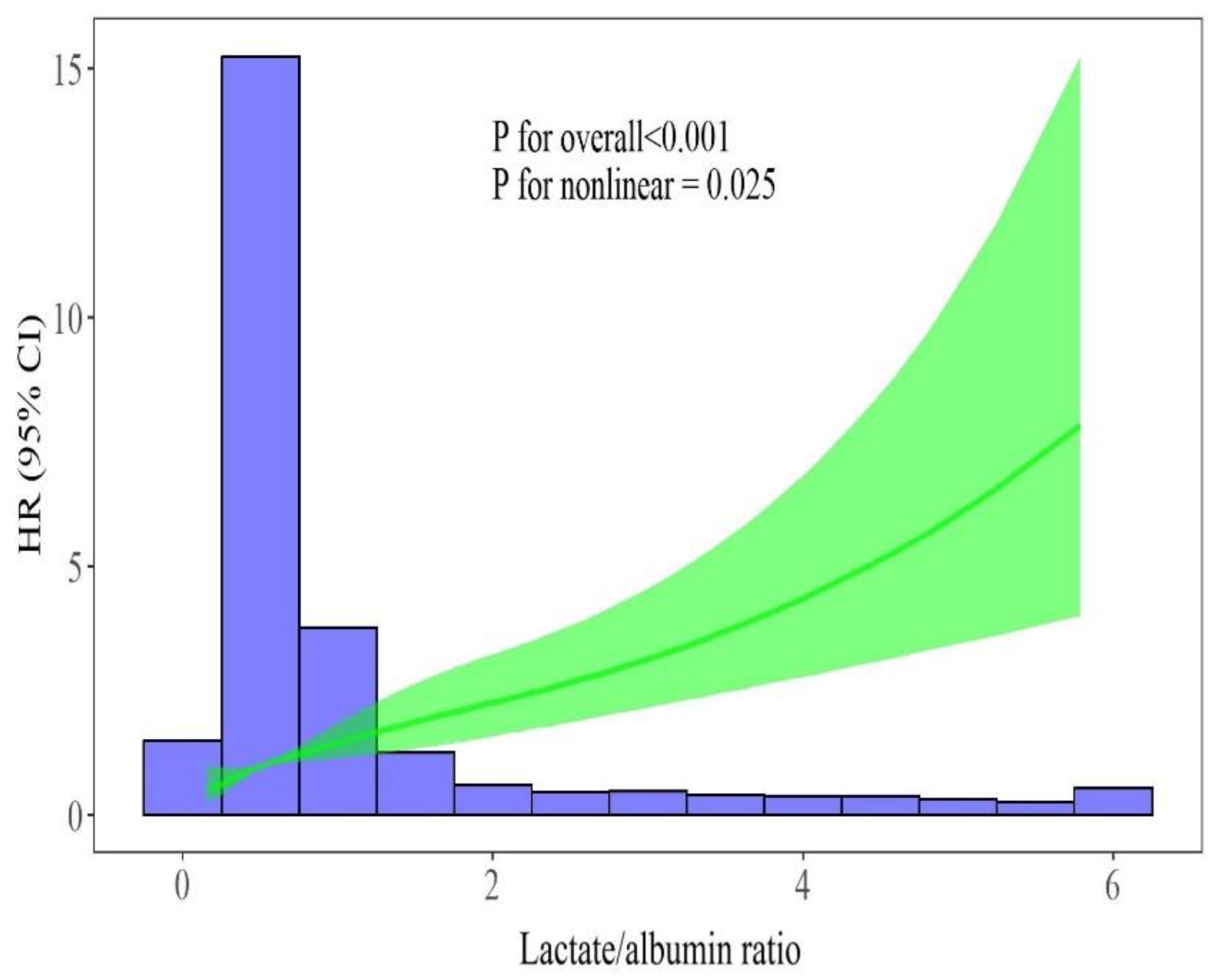
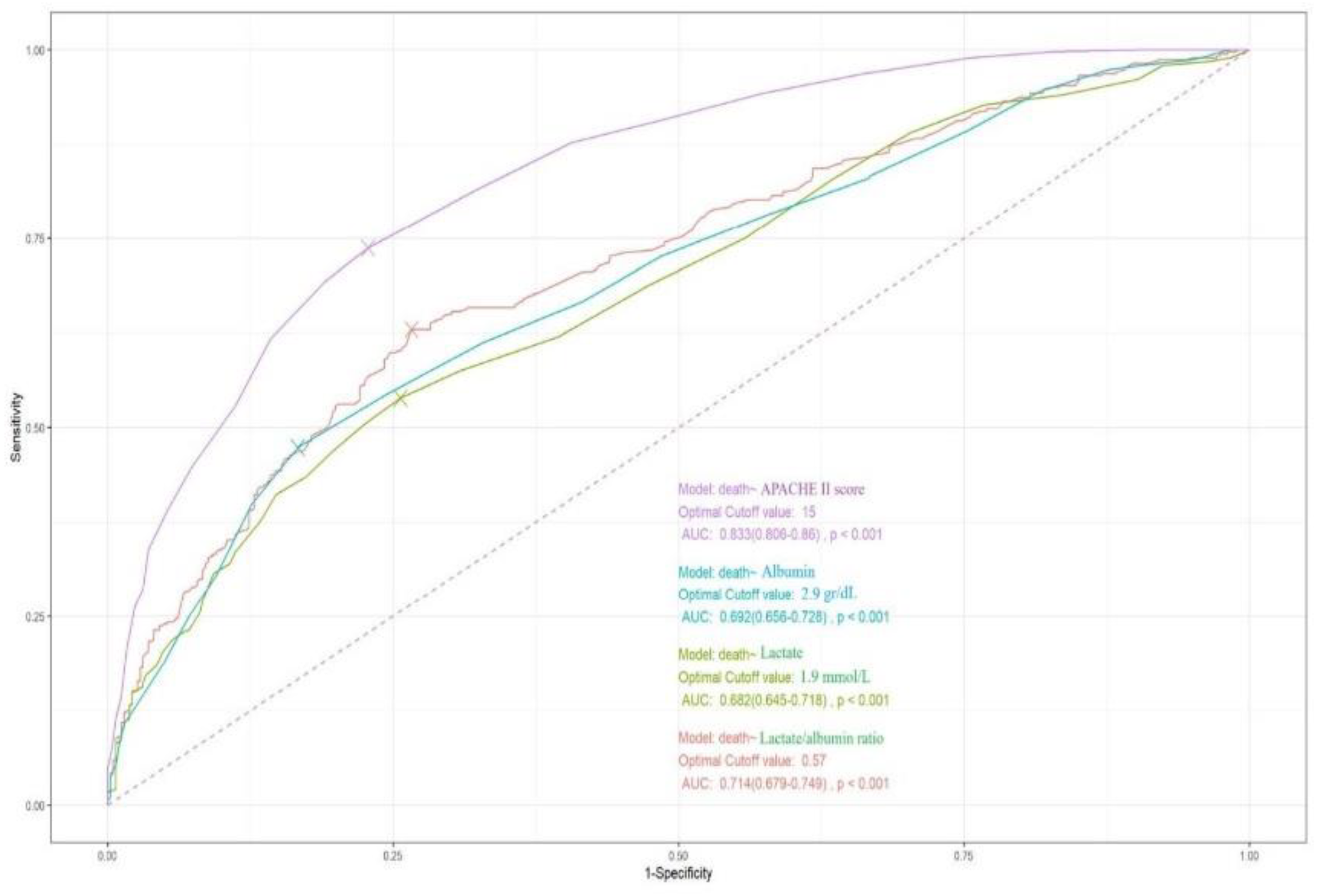
3.4. ROC Curves Analysis
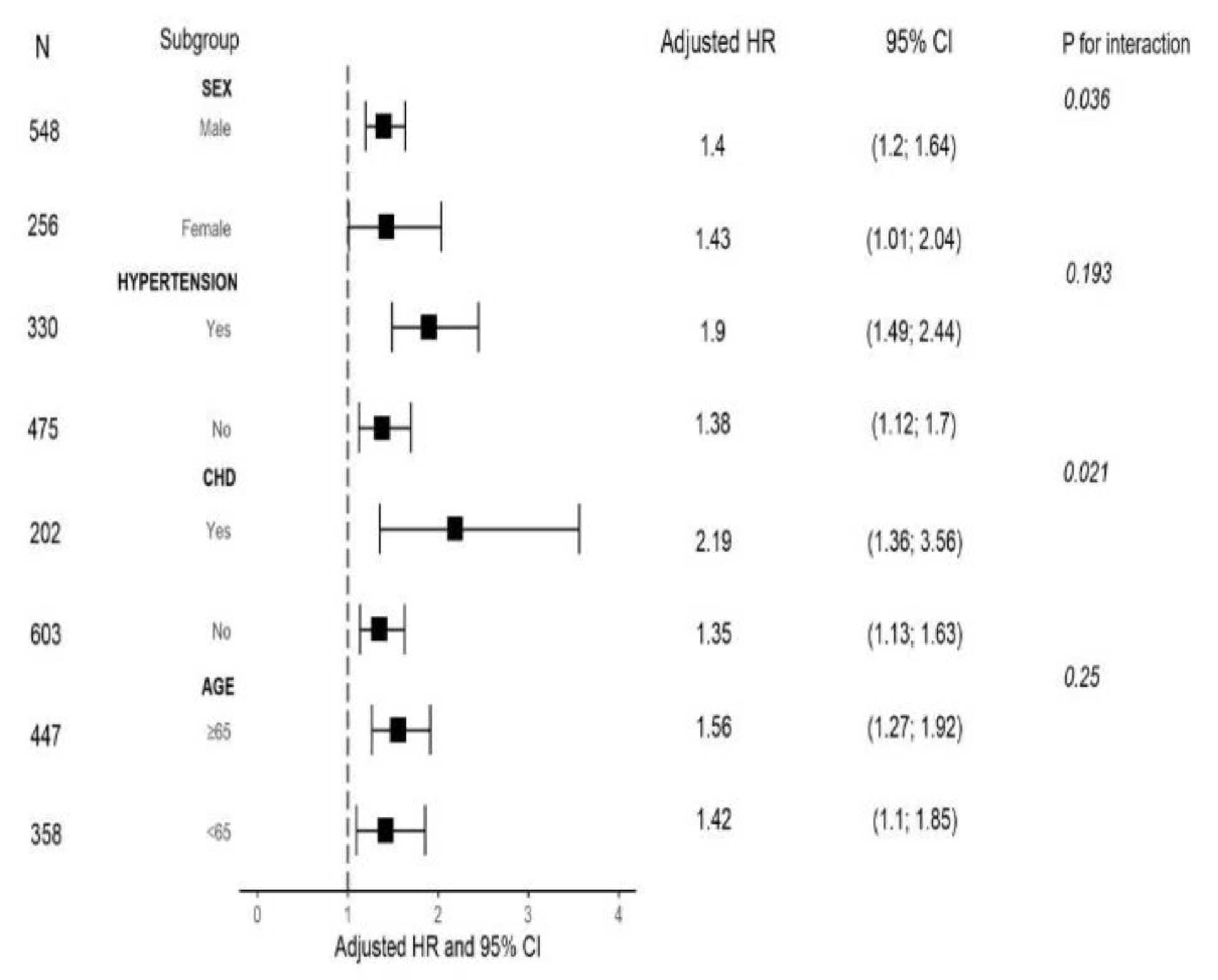
3.5. Subgroup Analyses
4. Discussion
Author Contributions
Funding
Conflicts of Interest
Declarations
References
- Rashkin, M.; Boxkin, C.; Baughman, R. Oxygen delivery in critically ill patients: relationship to blood lactate and survival. Chest 1985, 87, 580-84. [CrossRef]
- Bakker, J.; Vincent J-L. The oxygen-supply dependency phenomenon is associated with increased blood lactate levels. J Crit Care 1991, 6, 152-159. [CrossRef]
- Kraut, JA.; Madias, NE. Lactic Acidosis. N Engl J Med 2014, 371, 2309-2319. [CrossRef]
- Gibot, S. On the origins of lactate during sepsis. Critical Care 2012, 16, 151. [CrossRef]
- Routsi, C.; Bardouniotou, E.; Delivoria-loannidou, V.; Kazi, D.; Roussos, C.; Zakynthinos, S. Pulmonary lactate release in patients with acute lung injury is not attributable to lung tissue hypoxia. Crit Care Med 1999, 27, 2469-2473. [CrossRef]
- Michaeli, B.; Martinez, A.; Revelly, JP.; Cayeux, MC.; Chioléro, RL.; Tappy, L.; Berger, MM. Effects of endotoxin on lactate metabolism in humans. Critical Care 2012, 16:R139. [CrossRef]
- Bakker, J.; Coffernil, M.; Leon, M.; Gris, P.; Vincent, JL. Blood Lactate Levels Are Superior to Oxygen-Derived Variables in Predicting Outcome in Human Septic Shock. Chest 1991, 99, 956-62. [CrossRef]
- Bakker, J.; Nijsten, MW.; Jansen, TC. Clinical use of lactate monitoring in critically ill patients. Ann Intensive Care 2013, 3, 12. [CrossRef]
- Fleck, A.; Raines, G.; Hawker, F.; Trotter, J.; Wallace, PI.; Lendingham, IM.; Calman, KC. Increased vascular permeability: A major cause of hypoalbuminaemia in disease and injury. Lancet 1985, 1, 781–784. [CrossRef]
- Soeters, PB.; Wolfe, RR.; Shenkin, A. Hypoalbuminemia: Pathogenesis and clinical significance. J Parenter Enteral Nutr 2019, 43, 181–193. [CrossRef]
- Arnau-Barres, I.; Guerri-Fernandez, R.; Luque, S.; Sorli, L.; Vázquez O.; Miralles R. Serum albumin is a strong predictor of sepsis outcome in elderly patients. Eur J Clin Microbiol Infect Dis 2019, 38, 743–746. [CrossRef]
- Wang, B.; Chen, G.; Cao, Y.; Xue, J.; Li, J.; Wu, Y. Correlation of lactate/albumin ratio level to organ failure and mortality in severe sepsis and septic shock. J Crit Care 2015, 30, 271-5. [CrossRef]
- Shin, J.; Hwang, SY.; Jo, IJ.; Kim, WY.; Ryoo, SM.; Kang, GH.; Kim, K.; Jo, YH.; Chung, SP.; Joo, YS. et al. Prognostic value of the lactate/albumin ratio for predicting 28-day mortality in critically ill sepsis patients. Shock 2018, 50, 545-550. [CrossRef]
- Cakir, E.; Turan, O. Lactate/albumin ratio is more effective than lactate or albumin alone in predicting clinical outcomes in intensive care patients with sepsis. Scand J Clin Lab Invest 2021, 81, 225-229. [CrossRef]
- Lichtenauer, M.; Wernly, B.; Ohnewein, B.; Franz, M.; Kabisch, B.; Muessig, J.;IMasyuk, M.; Lauten, A.; Schulze, PC.; Hoppe, UC.; et al. The Lactate/Albumin Ratio: A Valuable Tool for Risk Stratification in Septic Patients Admitted to ICU. Int J Mol Sci 2017, 18, 1893. [CrossRef]
- Chen, Q.; Zhan, H.; Chen, J.; Mo, J.; Huang, S. Predictive value of lactate/albumin ratio for death and multiple organ dysfunction syndrome in patients with sepsis. J Med Biochem 2024, 43, 617-625. [CrossRef]
- Shadvar, K.; Nader-Djalal, N.; Vahed, N.; Sanaie, S.; Iranpour, A.; Mahmoodpoor, A.; Vahedian-Azimi, A.; Samim, A.; Rahimi-Bashar, F. Comparison of lactate/albumin ratio to lactate and lactate clearance for predicting outcomes in patients with septic shock admitted to intensive care unit: an observational study. Sci Rep 2022, 12, 1304. [CrossRef]
- Bou Chebl, R.; Jamali, S.; Sabra, M.; Safa, R.; Berbari, I.; Shami, A.; Makki, M.; Tamim, H.; Abou Dagher, G. Lactate/Albumin Ratio as a Predictor of In-Hospital Mortality in Septic Patients Presenting to the Emergency Department. Front Med (Lausanne) 2020, 7, 550182. [CrossRef]
- Gharipour, A.; Razavi, R.; Gharipour, M.; Mukasa, D. Lactate/albumin ratio: An early prognostic marker in critically ill patients. Am J Emerg Med 2020, 38, 2088-2095. [CrossRef]
- Karakike, E.; Giamarellos-Bourboulis, EJ.; Kyprianou, M.; Fleischmann-Struzek, C.; Pletz, MW.; Netea, MG.; Reinhart, K.; Kyriazopoulou, E. Coronavirus disease 2019 as cause of viral sepsis: a systematic review and meta-analysis. Crit. Care Med 2021, 49, 2042-2057. [CrossRef]
- Su, C.; Hoffman, KL.; Xu, Z.; Sanchez, E.; Siempos, II.; Harrington, JS.; Racanelli, AC.; Plataki, M.; Wang, F.; Schenck, EJ. Evaluation of Albumin Kinetics in Critically Ill Patients With Coronavirus Disease 2019 Compared to Those With Sepsis-Induced Acute Respiratory Distress Syndrome. Crit Care Explor 2021, 3, e0589. [CrossRef]
- Kokkoris, S.; Kanavou, A.; Katsaros, D.; Karageorgiou, S.; Kremmydas, P.; Gkoufa, A.; Ntaidou, T.; Giannopoulos, C.; Kardamitsi, MA.; Dimopoulou, G.; et al. Temporal trends in laboratory parameters in survivors and non-survivors of critical COVID-19 illness and the effect of dexamethasone treatment. Biomed Rep 2023, 20, 12. [CrossRef]
- Carpenè, G.; Onorato, D.; Nocini, R.; Fortunato, G.; Rizk, JG.; Henry, BM.; Lippi G. Blood lactate concentration in COVID-19: a systematic literature review. Clin Chem Lab Med 2021, 60, 332-337. [CrossRef]
- Vassiliou, AG.; Tsipilis, S.; Keskinidou, C.; Vrettou, CS.; Jahaj, E.; Gallos, P.; Routsi, C.; Orfanos, SE.; Kotanidou, A.; Dimopoulou, I. Lactate and Lactate-to-Pyruvate Ratio in Critically Ill COVID-19 Patients: A Pilot Study. J Pers Med 2022, 12, 171. [CrossRef]
- Iepsen, UW.; Plovsing, RR.; Tjelle, K.; Foss, NB.; Meyhoff, CS.; Ryrsø, CK.; Berg, RMG.; Secher, NH. The role of lactate in sepsis and COVID-19: Perspective from contracting skeletal muscle metabolism. Exp Physiol 2022, 107, 665-673. [CrossRef]
- Routsi, C.; Magira, E.; Kokkoris, S.; Siempos, I.; Vrettou, C.; Zervakis, D.; Ischaki, E.; Malahias, S.; Sigala, I.; Asimakos, A.; et al. Hospital resources may be an important aspect of mortality rate among critically ill patients with CO VID-19: the paradigm of Greece. J Clin Med 2020, 9, 3730. [CrossRef]
- Knaus, WA.; Draper, EA.; Wagner, DP.; Zimmerman, JE. APACHE II: a severity of disease classification system. Crit Care Med 1985, 13, 818–29.
- Vincent, JL.; Moreno, R.; Takala, J.; Willatts, S.; De Mendonça, A.; Bruining, H.; Reinhart, CK.; Suter, PM.; Thijs, LG. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med 1996, 22, 707-10. [CrossRef]
- Charlson, ME.; Pompei, P.; Ales, KL.; MacKenzie, CR. A new method of classifying prognostic comorbidity in longi tudinal studies: development and validation. J Chronic Dis 1987, 40, 373–83. [CrossRef]
- Cecconi, M.; De Backer, D.; Antonelli, M.; Beale, R.; Bakker, J.; Hofer, C.; Jaeschke, R.; Mebazaa, A.; Pinsky, MR.; Teboul, JL.; et al. Consensus on circulatory shock and hemodynamic monitoring. Task force of the European Society of Intensive Care Medicine. Intensive Care Med 2014, 40, 1795–1815. [CrossRef]
- Jeong, JH.; Heo, M.; Lee, SJ.; Jeong, YY.; Lee, JD.; Yoo, JW. Clinical Usefulness of Red Cell Distribution Width/Albumin Ratio to Discriminate 28-Day Mortality in Critically Ill Patients with Pneumonia Receiving Invasive Mechanical Ventilation, Compared with Lacate/Albumin Ratio: A Retrospective Cohort Study. Diagnostics (Basel) 2021, 11, 2344. [CrossRef]
- Ren, J.; Kang, Q.; Wang, F.; Yu, W. Association of lactate/albumin ratio with in-hospital mortality in ICU patients with acute respiratory failure: A retrospective analysis based on MIMIC-IV database. Medicine (Baltimore) 2023, 102, e35410. [CrossRef]
- Wang, HX.; Huang, XH.; Ma, LQ.; Yang, ZJ.; Wang, HL.; Xu, B.; Luo, MQ. Association between lactate-to-albumin ratio and short-time mortality in patients with acute respiratory distress syndrome. J Clin Anesth 2024, 99, 111632. [CrossRef]
- Guo, W.; Zhao, L.; Zhao, H.; Zeng, F.; Peng, C.; Guo, W.; Yan, H. The value of lactate/albumin ratio for predicting the clinical outcomes of critically ill patients with heart failure. Ann Transl Med 2021, 9, 118. [CrossRef]
- Liu, J.; Min, J.; Lu, J.; Zhong, L.; Luo, H. Association between lactate/albumin ratio and prognosis in critically ill patients with acute kidney injury undergoing continuous renal replacement therapy. Ren Fail 2024, 46, 2374451. [CrossRef]
- Dudoignon, E.; Quennesson, T.; De Tymowski, C.; Moreno, N.; Coutrot, M.; Chaussard, M.; Guillemet, L.; Abid, S.; Fratani, A.; Ressaire, Q.; et al. Usefulness of lactate albumin ratio at admission to predict 28-day mortality in critically ill severely burned patients: A retrospective cohort study. Burns 2022, 48, 1836-1844. [CrossRef]
| Characteristic | Overall N = 805 |
Survival N = 421 |
Non-survival N = 384 |
P-value |
|---|---|---|---|---|
| Demographics | ||||
| Sex, male, n (%) | 548 (68%) | 285 (68%) | 263 (68%) | 0.8 |
| Age, years | 67 (57, 76) | 61 (52, 70) | 73 (65, 79) | <0.001 |
| Age quartiles | <0.001 | |||
| 1 | 201 (25%) | 158 (38%) | 43 (11%) | |
| 2 | 192 (24%) | 127 (30%) | 65 (17%) | |
| 3 | 210 (26%) | 86 (20%) | 124 (32%) | |
| 4 | 202 (25%) | 50 (12%) | 152 (40%) | |
| Severity scores | ||||
| Charlson comorbidity index | 3 (2, 5) | 2 (1, 4) | 4 (3, 6) | <0.001 |
| APACHE II score | 14 (11, 19) | 11 (9, 14) | 18.0 (14, 23) | <0.001 |
| SOFA score | 7 (4, 9) | 6 (2, 7) | 8 (7, 10) | <0.001 |
| Outcomes | ||||
| CRRT, n (%) | 238 (30%) | 51 (12%) | 187 (49%) | <0.001 |
| MV duration, days | 11 (4, 24) | 8 (0, 23) | 14 (7, 25) | <0.001 |
| ICU-LOS, days | 15 (8, 29) | 15 (8, 33) | 15 (8, 26) | 0.056 |
| Laboratory tests | ||||
| WBC count, x 109/L | 10 (7, 15) | 9 (7, 13) | 11 (8, 16) | <0.001 |
| Neutrophil count, x 109/L | 8.7 (5.7, 12.9) | 8.0 (5.4, 11.9) | 9.7 (6.6, 14.4) | <0.001 |
| Lymphocyte count, x 109/L | 0.76 (0.51, 1.10) | 0.80 (0.55, 1.11) | 0.73 (0.47, 1.10) | 0.015 |
| NLR | 12 (7, 20) | 10 (6, 17) | 14 (8, 23) | <0.001 |
| Hb, g/dL | 12.4 (10.8, 13.7) | 12.7 (11.4, 13.8) | 11.9 (10.1, 13.5) | <0.001 |
| PLT count, x 109/L | 241 (182, 309) | 254 (199, 321) | 226 (164, 296) | <0.001 |
| Albumin, g/dL | 3.2 (2.8, 3.6) | 3.4 (3.1, 3.6) | 3.0 (2.6, 3.4) | <0.001 |
| Sodium, mEq/L | 140 (137, 143) | 139 (136, 142) | 141 (138, 145) | <0.001 |
| Creatinine, mg/dL | 0.9 (0.7, 1.3) | 0.8 (0.7, 1.0) | 1.1 (0.8, 1.6) | <0.001 |
| AST, IU/L | 38 (25, 66) | 37 (25, 60) | 39 (25, 72) | 0.2 |
| ALT, IU/L | 32 (19, 54) | 34 (21, 54) | 28 (17, 58) | 0.020 |
| LDH, IU/L | 456 (347, 624) | 437 (312, 573) | 488 (370, 669) | <0.001 |
| hs-cTnI, ng/L | 24 (10, 77) | 15 (7, 41) | 42 (17, 123) | <0.001 |
| CRP, mg/dL | 11 (5, 18) | 10 (4, 17) | 12 (7, 20) | <0.001 |
| Fibrinogen, mg/dL | 579 (474, 701) | 575 (485, 686) | 583 (459, 708) | >0.9 |
| D-dimers, mg/L | 1.6 (0.9, 3.9) | 1.2 (0.7, 2.7) | 2.2 (1.1, 5.0) | <0.001 |
| PaO2/FiO2 ratio | 126 (88, 187) | 136 (93, 206) | 118 (83, 173) | <0.001 |
| Lactate, mmol/L | 1.70 (1.30, 2.30) | 1.50 (1.20, 1.90) | 2.00 (1.45, 2.90) | <0.001 |
| Lactate/albumin ratio | 0.53 (0.39, 0.79) | 0.46 (0.35, 0.58) | 0.64 (0.46, 1.02) | <0.001 |
| Lactate/albumin ratio quartiles | <0.001 | |||
| 1 | 206 (26%) | 150 (36%) | 56 (15%) | |
| 2 | 198 (25%) | 123 (29%) | 75 (20%) | |
| 3 | 203 (25%) | 96 (23%) | 107 (28%) | |
| 4 | 198 (25%) | 52 (12%) | 146 (38%) | |
| Treatment | ||||
| Full vaccination, n (%) | 0 (0%) | 26 (6.2%) | 35 (9.1%) | 0.12 |
| Remdesivir, n (%) | 391 (49%) | 221 (52%) | 170 (44%) | 0.020 |
| Dexamethasone, n (%) | 637 (79%) | 328 (78%) | 309 (80%) | 0.4 |
| Tocilizumab, n (%) | 36 (4.5%) | 28 (6.7%) | 8 (2.1%) | 0.002 |
| MV on admission, n (%) | 588 (73%) | 252 (60%) | 336 (88%) | <0.001 |
| HFNC on admission, n (%) | 158 (20%) | 116 (28%) | 42 (11%) | <0.001 |
| Shock on admission, n (%) | 304 (38%) | 172 (41%) | 132 (34%) | 0.058 |
| Comorbidities | ||||
| Hypertension, n (%) | 330 (41%) | 159 (38%) | 171 (45%) | 0.051 |
| Diabetes mellitus, n (%) | 204 (25%) | 99 (24%) | 105 (27%) | 0.2 |
| Obesity, n (%) | 104 (13%) | 63 (15%) | 41 (11%) | 0.070 |
| Cardiovascular disease, n (%) | 202 (25%) | 81 (19%) | 121 (32%) | <0.001 |
| Chronic pulmonary disease, n (%) | 106 (13%) | 40 (9.5%) | 66 (17%) | 0.001 |
| Malignancy, n (%) | 85 (11%) | 24 (5.7%) | 61 (16%) | <0.001 |
| Chronic kidney disease, n (%) | 65 (8.1%) | 15 (3.6%) | 50 (13%) | <0.001 |
| Data are expressed as median (IQR), unless otherwise denoted. Abbreviations: IQR, interquartile range; APACHE, acute physiology and chronic health evaluation; SOFA, sequential organ failure assessment ; MV, mechanical ventilation; ICU-LOS, intensive care unit-length of stay; WBC, white blood cell; NLR, neutrophil to lymphocyte ratio; Hb, hemoglobin; PLT, platelets; AST, aspartate aminotransferase; ALT, alanine aminotransferase; LDH, lactate dehydrogenase; hs-cTnI, high sensitivity cardiac troponin I; CRP, C-reactive protein; CRRT, continuous renal replacement therapy; HFNC, high flow nasal cannula. | ||||
| Characteristic | Quartile 1 N = 206 |
Quartile 2 N = 198 |
Quartile 3 N = 203 |
Quartile 4 N = 198 |
P-value |
|---|---|---|---|---|---|
| Demographics | |||||
| Sex, male, n (%) | 134 (65%) | 141 (71%) | 139 (68%) | 134 (68%) | 0.6 |
| Age, years | 61 (52, 70) | 64 (54, 73) | 70 (61, 77) | 72 (63, 80) | <0.001 |
| Severity scores | |||||
| Charlson comorbidity index | 3 (1, 4) | 3 (1, 4) | 4 (2, 5) | 4 (3, 6) | <0.001 |
| APACHE II score | 11 (8, 16) | 13 (9, 16) | 14 (12, 18) | 19 (15, 24) | <0.001 |
| SOFA score | 6 (2, 7) | 6 (3, 8) | 7 (6, 8) | 8 (7, 11) | <0.001 |
| Outcomes | |||||
| ICU outcome, death, n (%) | 56 (27%) | 75 (38%) | 107 (53%) | 146 (74%) | <0.001 |
| CRRT, n (%) | 49 (24%) | 44 (22%) | 65 (32%) | 80 (40%) | <0.001 |
| MV duration, days | 9 (0, 20) | 13 (5, 26) | 14 (6, 26) | 10 (4, 23) | <0.001 |
| ICU-LOS, days | 14 (8, 25) | 17 (9, 33) | 17 (10, 31) | 12 (5, 28) | <0.001 |
| Laboratory tests | |||||
| WBC count, x 109/L | 8 (6, 11) | 9 (7, 13) | 11 (8, 15) | 14 (10, 20) | <0.001 |
| Neutrophil count, x 109/L | 6.5 (4.6, 9.5) | 8.2 (5.4, 11.9) | 9.3 (7.1, 13.7) | 12.4 (8.0, 17.4) | <0.001 |
| Lymphocyte count, x 109/L | 0.70 (0.49, 1.07) | 0.80 (0.54, 1.11) | 0.66 (0.48, 0.99) | 0.84 (0.54, 1.24) | 0.022 |
| NLR | 9 (5, 16) | 10 (6, 16) | 14 (8, 22) | 16 (8, 24) | <0.001 |
| Hb, g/dL | 12 (11, 14) | 13 (11, 14) | 12 (11, 14) | 12 (9, 13) | <0.001 |
| PLT count, x 109/L | 231 (180, 295) | 263 (203, 325) | 251 (194, 326) | 222 (159, 303) | <0.001 |
| Albumin, g/dL | 3.6 (3.3, 3.8) | 3.3 (3.1, 3.6) | 3.0 (2.8, 3.4) | 2.8 (2.4, 3.1) | <0.001 |
| Sodium, mEq/L | 139 (137, 141) | 140 (136, 142) | 140 (137, 143) | 143 (138, 147) | <0.001 |
| Creatinine, mg/dL | 0.8 (0.7, 1.1) | 0.8 (0.7, 1.0) | 0.9 (0.7, 1.3) | 1.2 (0.8, 1.9) | <0.001 |
| AST, IU/L | 41 (26, 64) | 38 (26, 65) | 36 (22, 59) | 38 (24, 95) | 0.12 |
| ALT, IU/L | 34 (21, 54) | 33 (21, 55) | 30 (19, 51) | 29 (15, 67) | 0.2 |
| LDH, IU/L | 430 (298, 550) | 455 (352, 591) | 469 (350, 634) | 478 (358, 842) | 0.001 |
| hs-cTnI, ng/L | 13 (7, 38) | 16 (8, 47) | 23 (12, 62) | 74 (27, 223) | <0.001 |
| CRP, mg/dL | 11 (4, 18) | 11 (5, 17) | 11 (5, 18) | 13 (6, 21) | 0.085 |
| Fibrinogen, mg/dL | 595 (513, 726) | 588 (506, 679) | 593 (485, 721) | 510 (375, 669) | <0.001 |
| D-dimers, mg/L | 1.1 (0.7, 2.2) | 1.3 (0.7, 2.7) | 2.2 (1.1, 4.3) | 2.5 (1.3, 10.0) | <0.001 |
| PaO2/FiO2 ratio | 121 (90, 183) | 121 (87, 180) | 132 (86, 192) | 140 (94, 200) | 0.3 |
| Lactate, mmol/L | 1.1 (0.9, 1.3) | 1.5 (1.4, 1.6) | 1.9 (1.7, 2.2) | 3.3 (2.7, 5.4) | <0.001 |
| Shock on admission, n (%) | 74 (36%) | 92 (46%) | 94 (46%) | 44 (22%) | <0.001 |
| Treatment | |||||
| Full vaccination, n (%) | 15 (7.3%) | 12 (6.1%) | 14 (6.9%) | 20 (10%) | 0.5 |
| Remdesivir, n (%) | 100 (49%) | 109 (55%) | 102 (50%) | 80 (40%) | 0.031 |
| Dexamethasone, n (%) | 150 (73%) | 166 (84%) | 182 (90%) | 139 (70%) | <0.001 |
| Tocilizumab, n (%) | 5 (2.4%) | 11 (5.6%) | 13 (6.4%) | 7 (3.5%) | 0.2 |
| MV on admission, n (%) | 104 (50%) | 139 (70%) | 169 (83%) | 176 (89%) | <0.001 |
| HFNC on admission, n (%) | 71 (34%) | 46 (23%) | 28 (14%) | 13 (6.6%) | <0.001 |
| Comorbidities | |||||
| Hypertension, n (%) | 84 (41%) | 77 (39%) | 85 (42%) | 84 (42%) | 0.9 |
| Diabetes, n (%) | 59 (29%) | 51 (26%) | 47 (23%) | 47 (24%) | 0.6 |
| Obesity, n (%) | 37 (18%) | 27 (14%) | 29 (14%) | 11 (5.6%) | 0.002 |
| Cardiovascular disease, n (%) | 40 (19%) | 42 (21%) | 54 (27%) | 66 (33%) | 0.006 |
| Chronic pulmonary disease, n (%) | 25 (12%) | 22 (11%) | 30 (15%) | 29 (15%) | 0.6 |
| Malignancy, n (%) | 23 (11%) | 19 (9.6%) | 18 (8.9%) | 25 (13%) | 0.6 |
| Chronic kidney disease, n (%) | 16 (7.8%) | 10 (5.1%) | 15 (7.4%) | 24 (12%) | 0.073 |
| Data are expressed as median (IQR), unless otherwise denoted. Abbreviations: IQR, interquartile range; APACHE, acute physiology and chronic health evaluation; SOFA, sequential organ failure assessment; MV, mechanical ventilation; ICU-LOS, intensive care unit-length of stay; WBC, white blood cell; NLR, neutrophil to lymphocyte ratio; Hb, hemoglobin; PLT, platelets; AST, aspartate aminotransferase; ALT, alanine aminotransferase; LDH, lactate dehydrogenase; hs-cTnI, high sensitivity cardiac troponin I; CRP, C-reactive protein; CRRT, continuous renal replacement therapy; HFNC, high flow nasal cannula. | |||||
| Univariate model | *Multivariate model | |||
|---|---|---|---|---|
| Variable | HR (95%CI) | P-value | HR (95%CI) | P-value |
| Lactate/albumin ratio | 1.60 (1.40-1.70) | <0.001 | 1.39 (1.27-1.52) | <0.001 |
|
*The model was adjusted for age, sex, NLR, Hb, PLT count, sodium, creatinine, AST, ALT, LDH, hs-cTnI, CRP, fibrinogen, d-dimers, PaO2/FiO2 ratio, presence of shock, vaccination status, CRRT, remdesivir, dexamethasone, hypertension, diabetes mellitus, obesity, coronary heart disease, chronic pulmonary disease, chronic kidney disease and active malignancy. Abbreviations: HR, hazard ratio; CI, confidence interval; NLR, neutrophil to lymphocyte ratio; Hb, hemoglobin; PLT, platelets; AST, aspartate aminotransferase; ALT, alanine aminotransferase; hs-cTnI, high sensitivity cardiac troponin I; CRP, C-reactive protein; CRRT, continuous renal replacement therapy. | ||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




