Submitted:
25 October 2024
Posted:
28 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
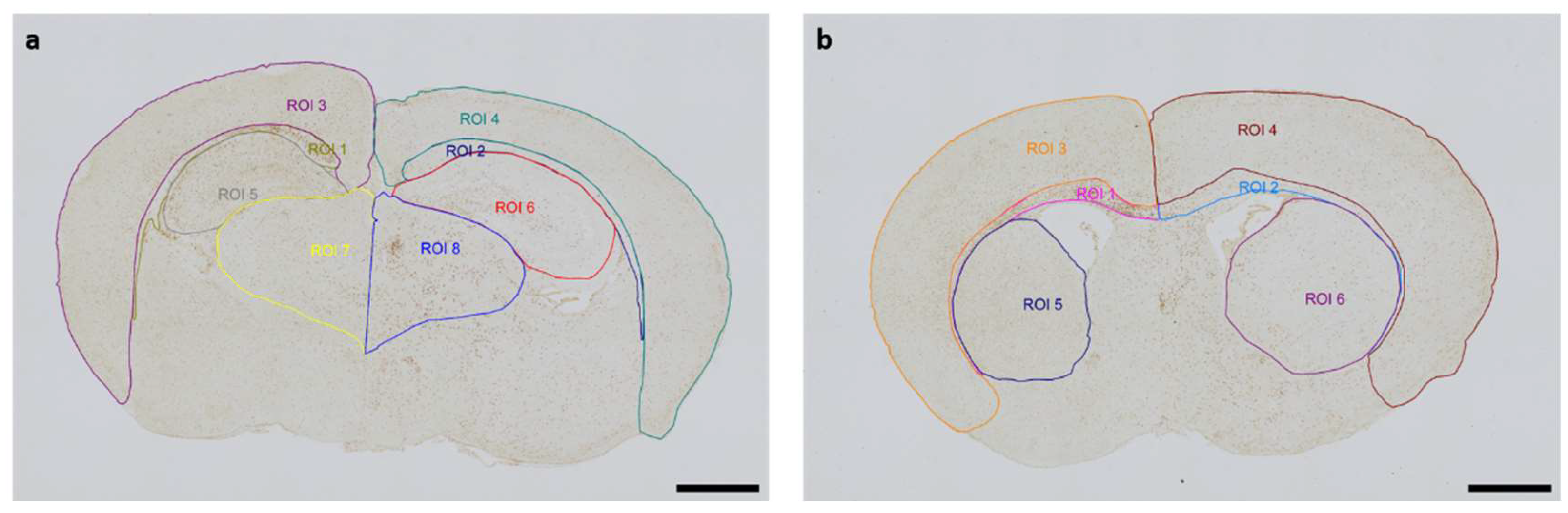
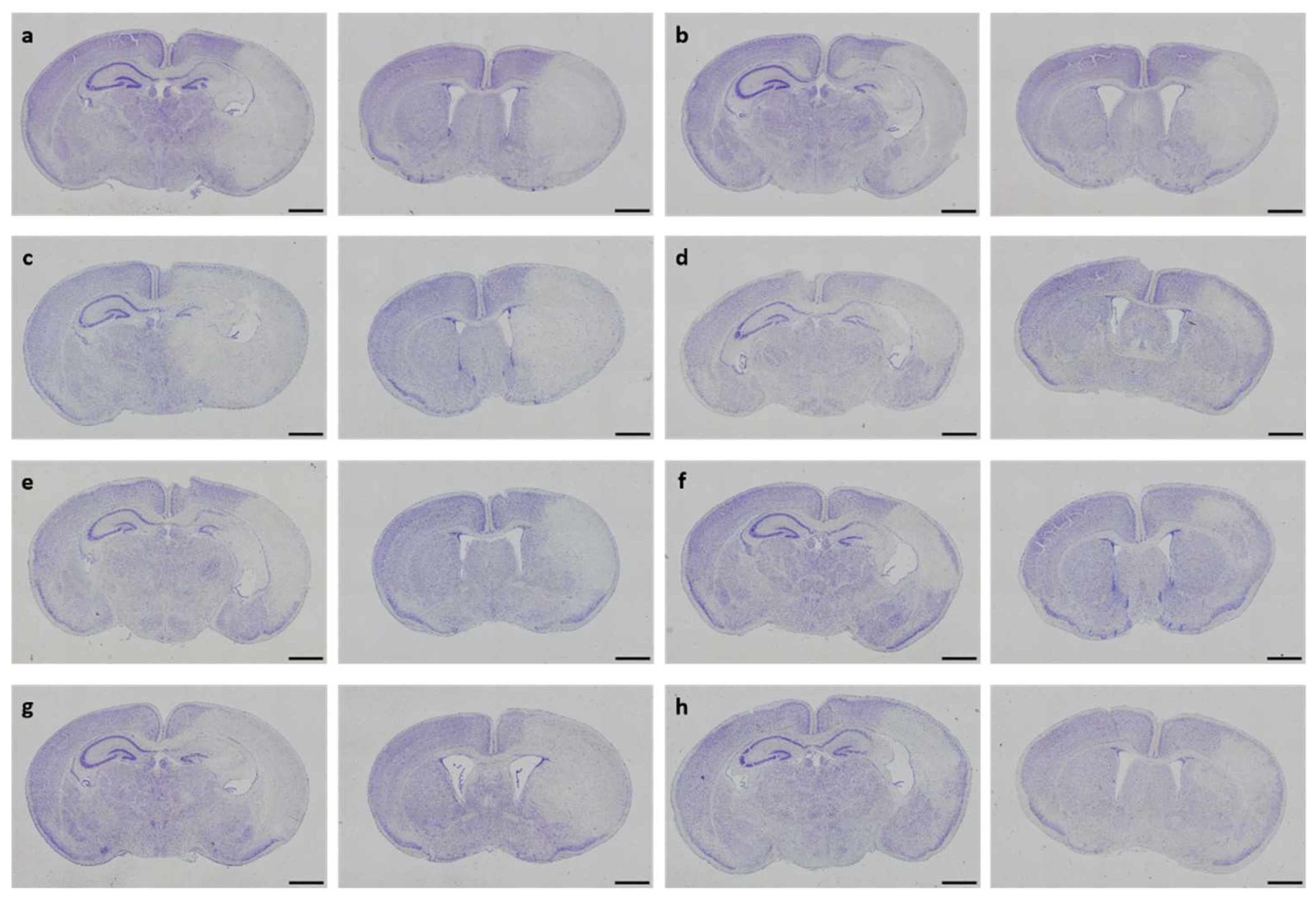
Neuropathological Injury Assessment
Immunohistochemical Analyses
Protein Fractionation and Western Blotting
ELISA of 4-Hydroxynonenal and 8-Hydroxydesoxyguanosin
Statistical Analysis
3. Results
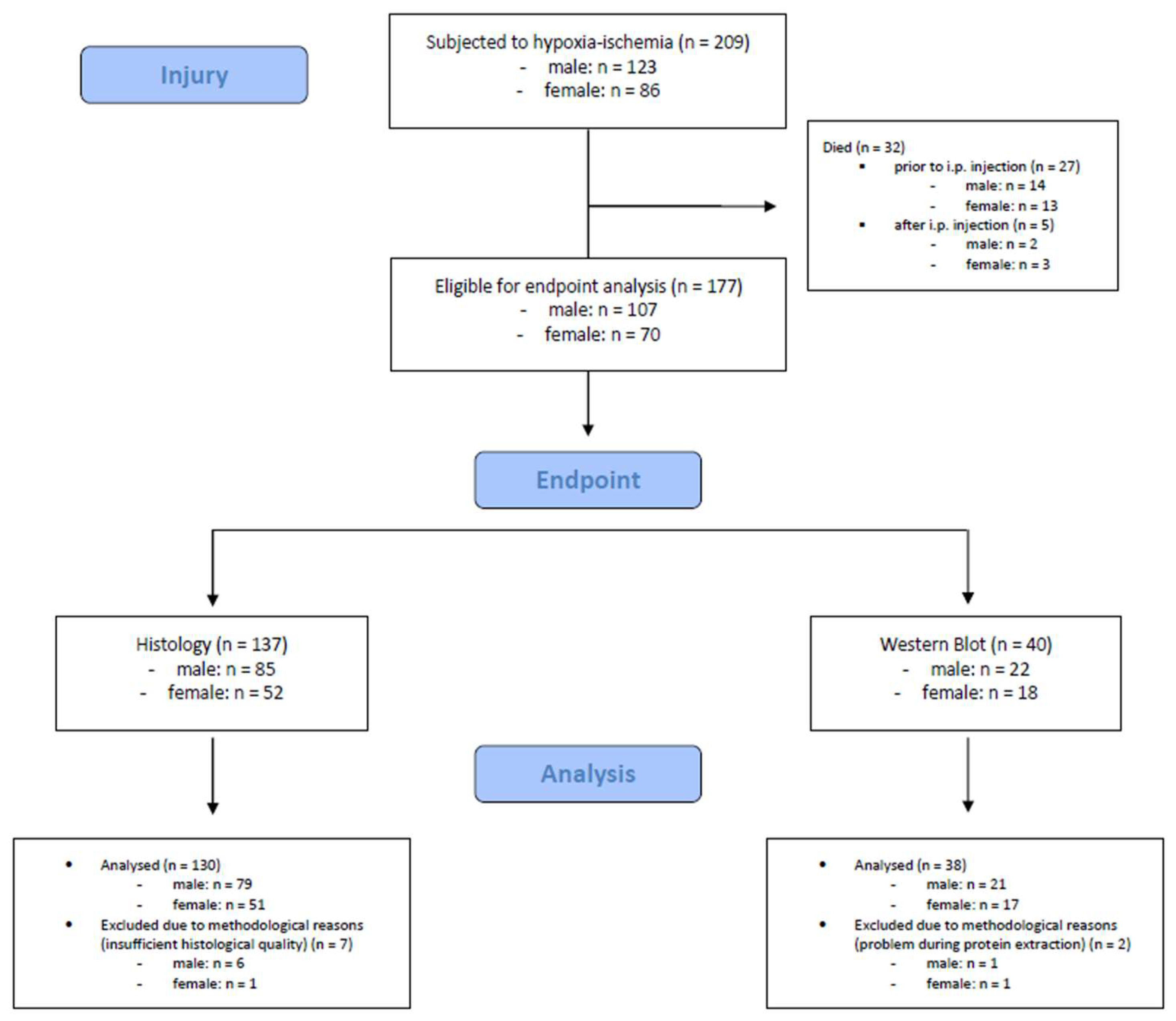
3.1. Study Population
3.1.1. Somatometry
3.2. Neuropathological Injury
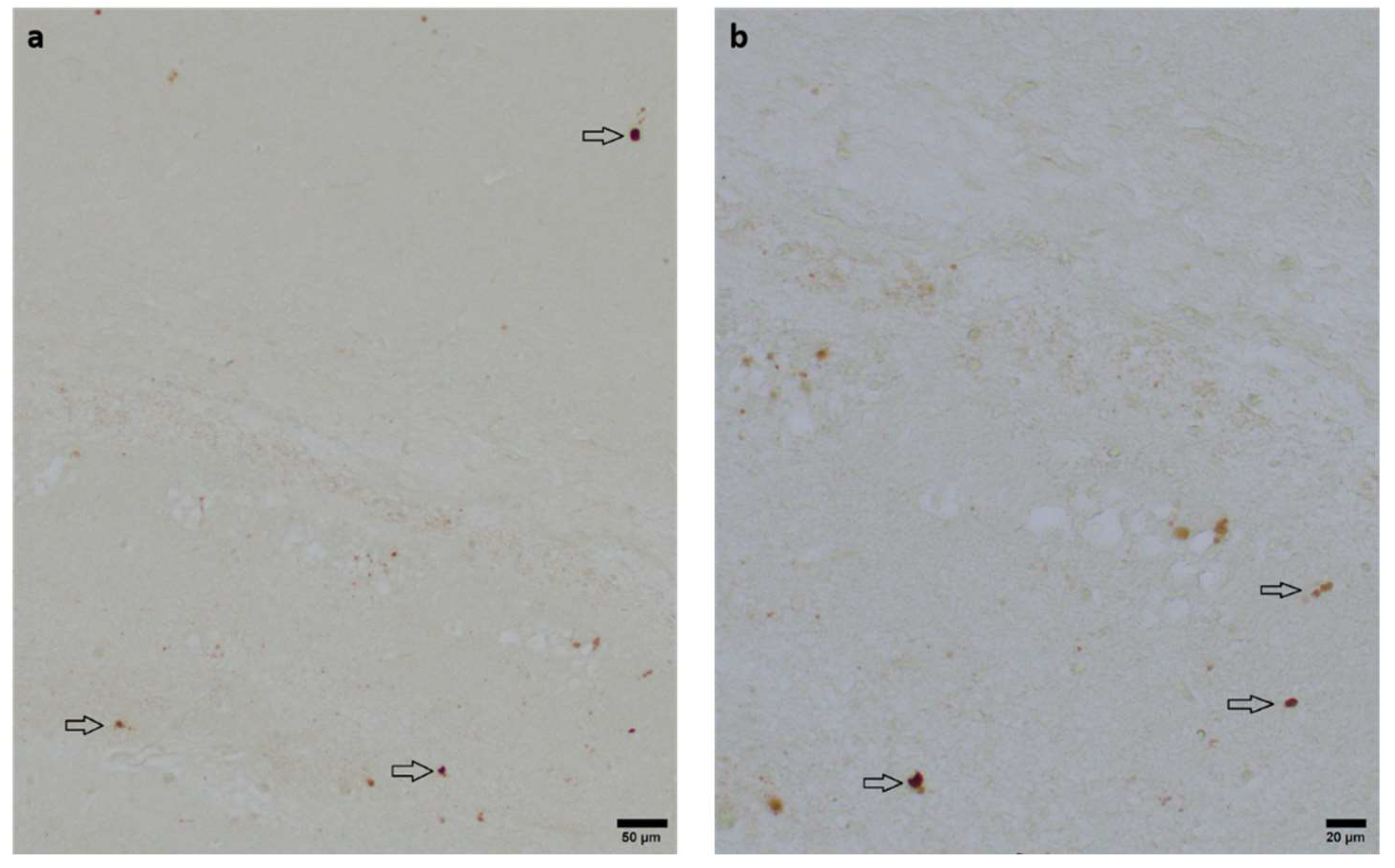
3.3. Anti-Apoptotic Potential of DHEA and DHEAS
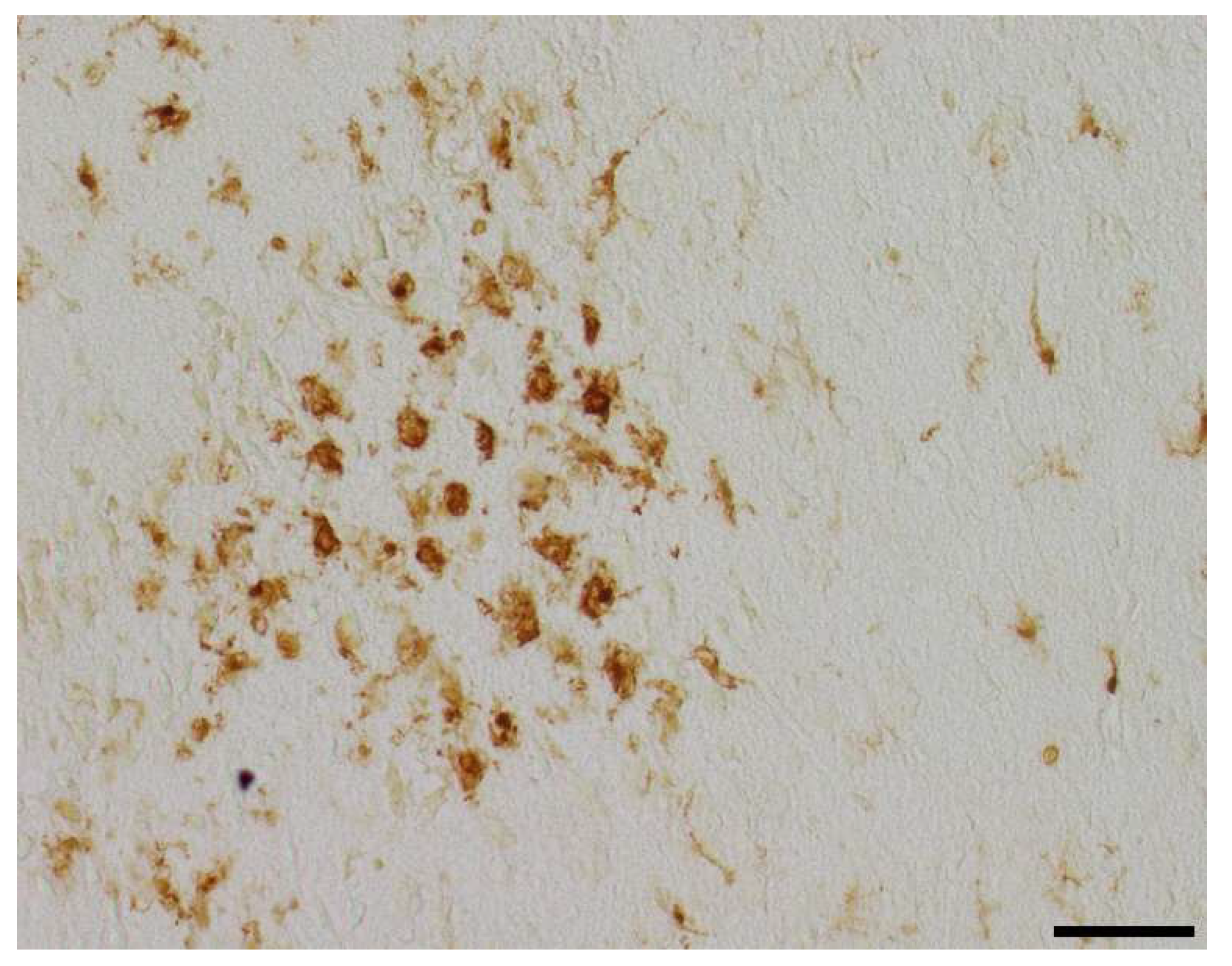
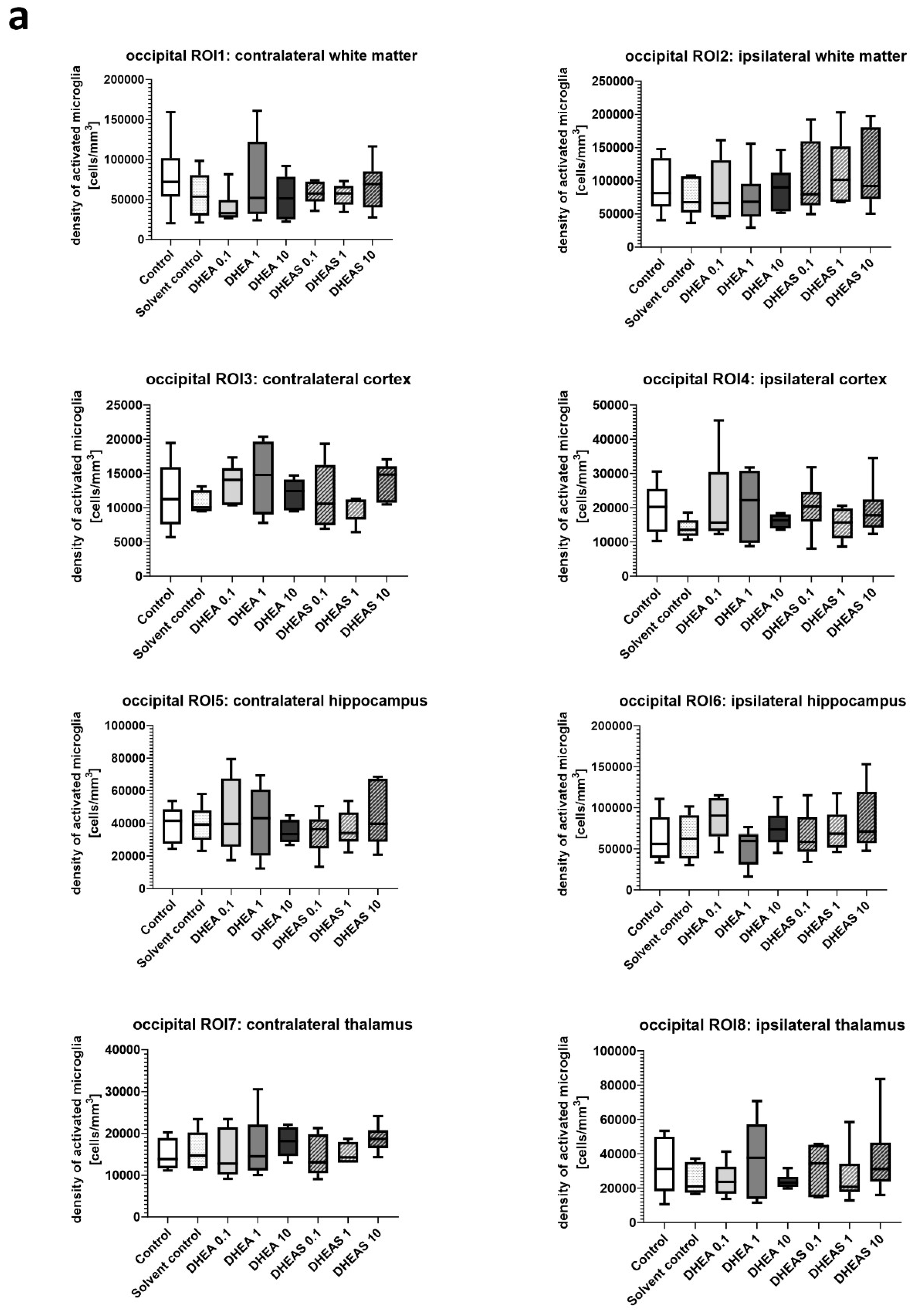
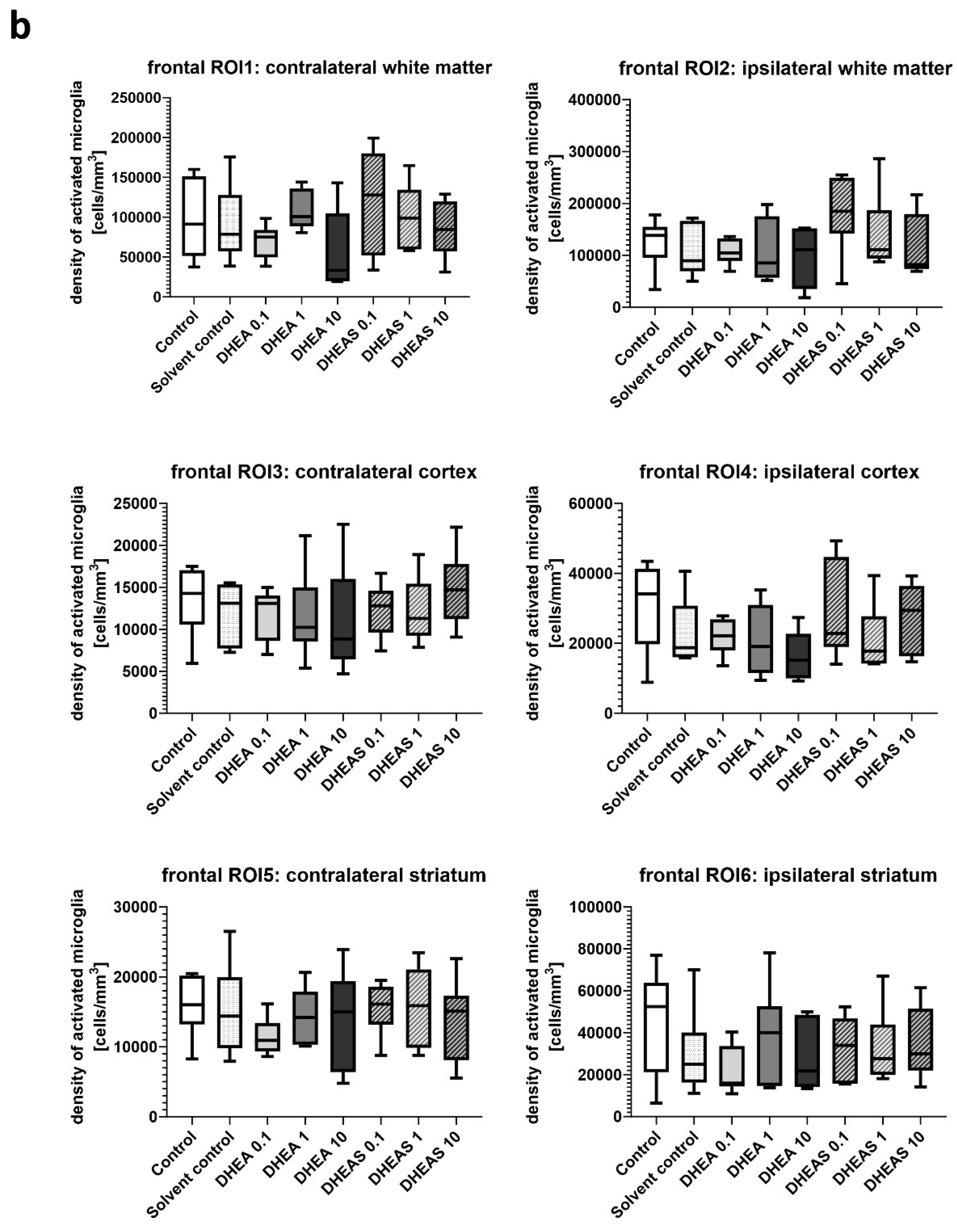
3.4. Microglial Cell Activation
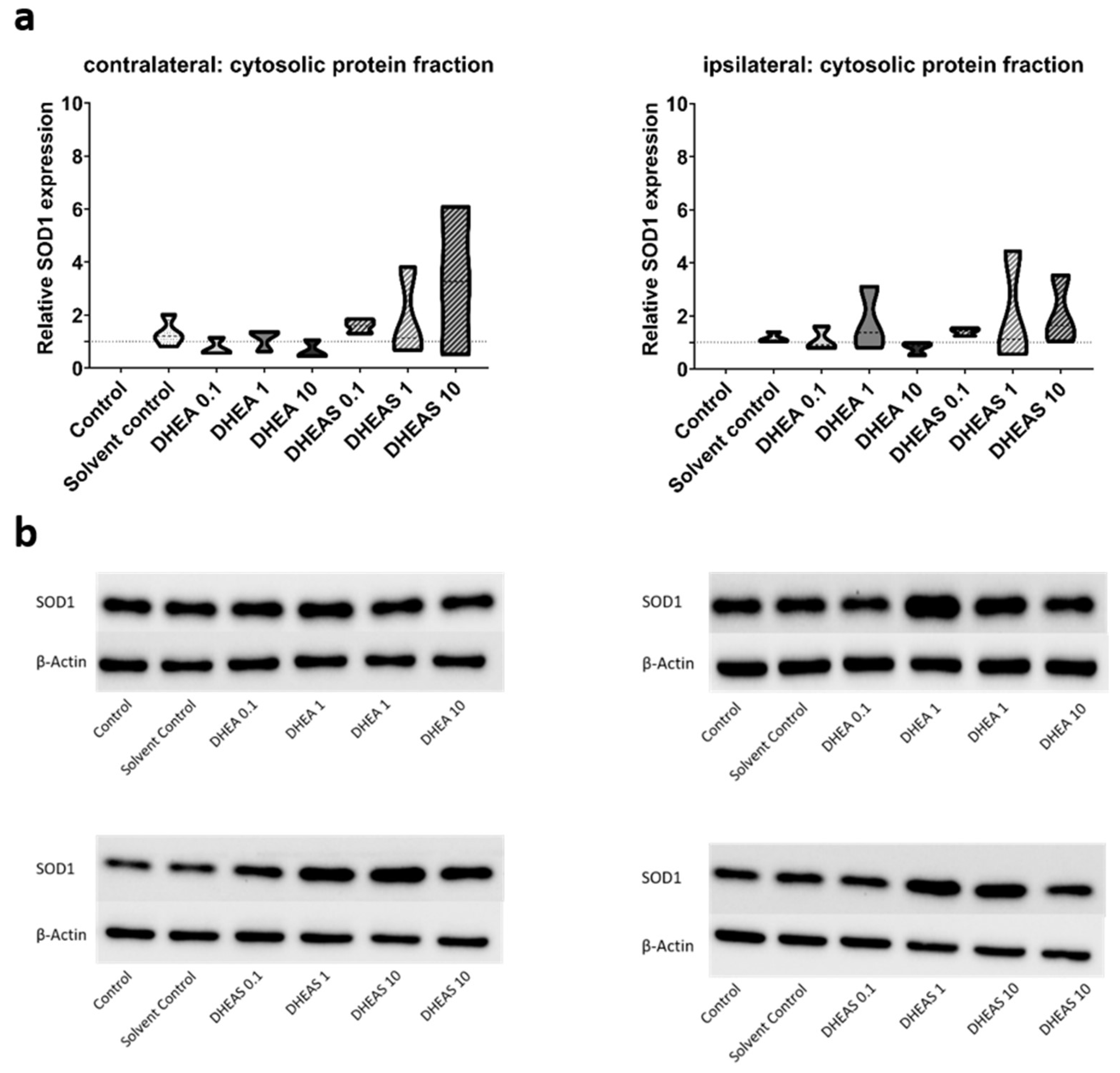
3.5. Anti-Oxidative Potential of DHEA and DHEAS
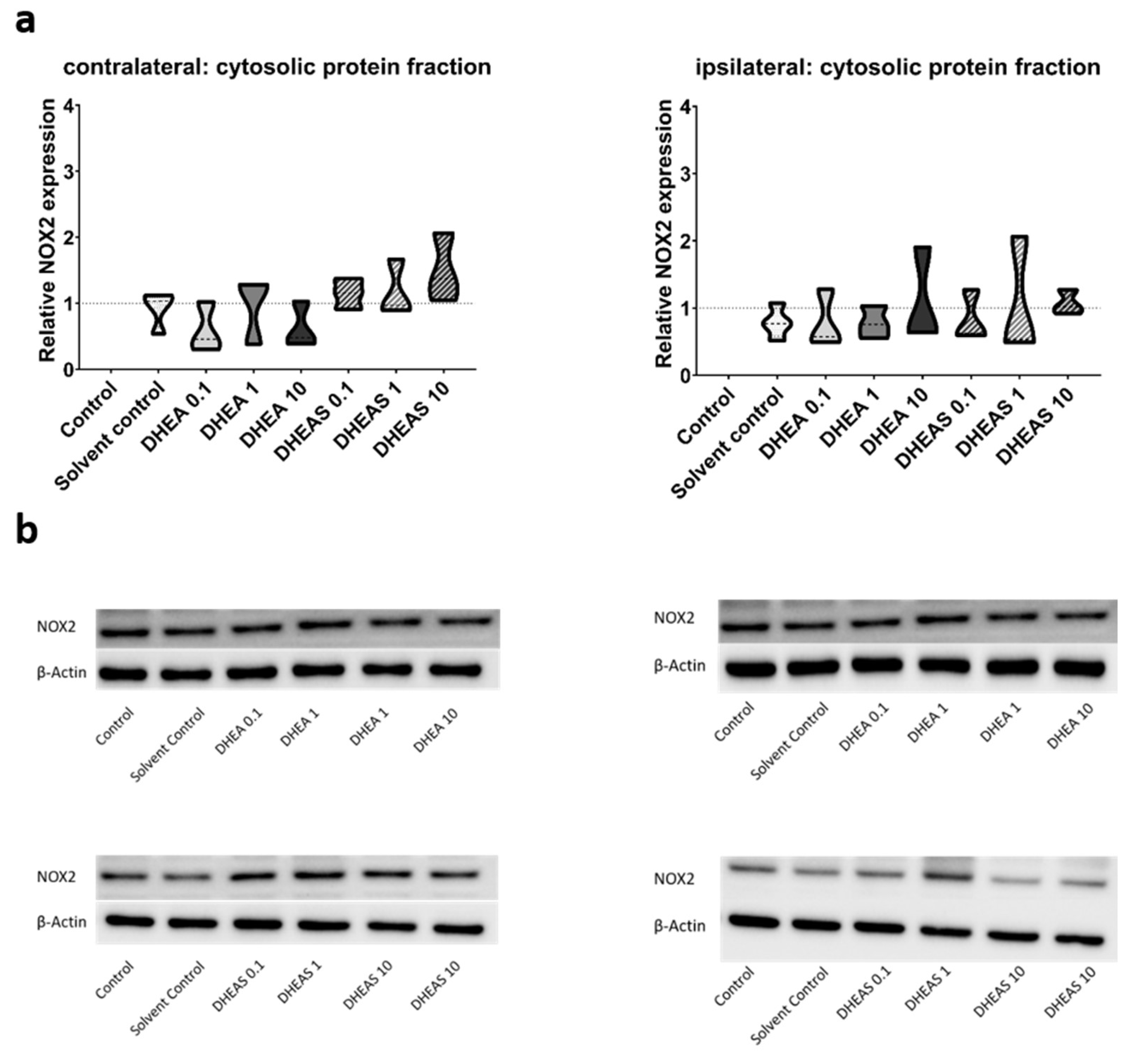
3.5.1. Upstream Markers of Oxidative Stress—Western Blot
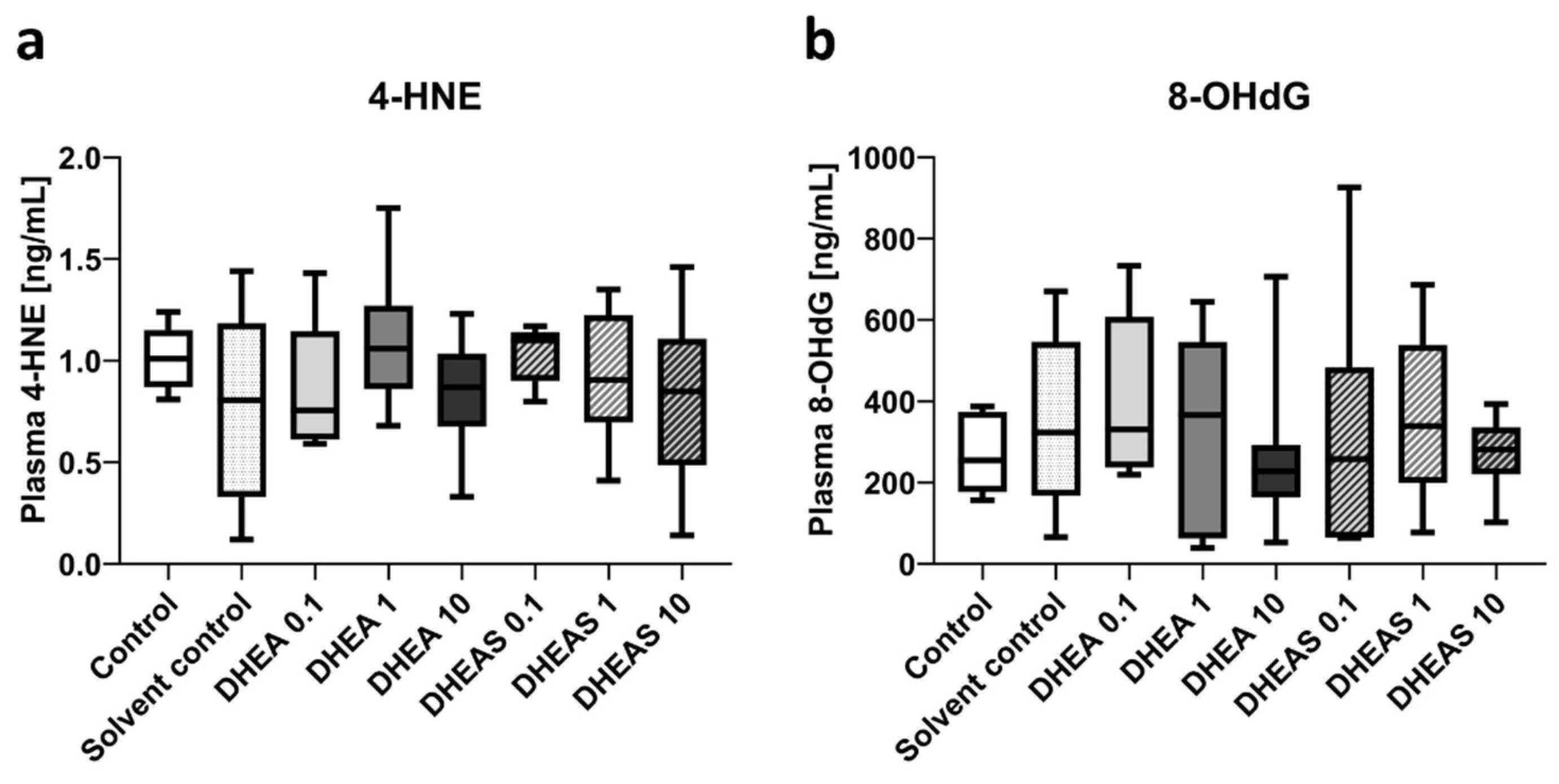
3.5.2. Downstream Markers of Oxidative Stress—ELISA
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hill MG, Reed KL, Brown RN. Perinatal asphyxia from the obstetric standpoint. Semin Fetal Neonatal Med 2021; 26(4):101259. [CrossRef]
- Juul SE, Ferriero DM. Pharmacologic neuroprotective strategies in neonatal brain injury. Clin Perinatol 2014; 41(1):119–31. [CrossRef]
- Golubnitschaja O, Yeghiazaryan K, Cebioglu M, Morelli M, Herrera-Marschitz M. Birth asphyxia as the major complication in newborns: moving towards improved individual outcomes by prediction, targeted prevention and tailored medical care. EPMA J 2011; 2(2):197–210. [CrossRef]
- Elsadek AE, FathyBarseem N, Suliman HA, Elshorbagy HH, Kamal NM, Talaat IM et al. Hepatic Injury in Neonates with Perinatal Asphyxia. Glob Pediatr Health 2021; 8:2333794X20987781. [CrossRef]
- UNICEF. Child Mortality Report 2023. United Nations Children’s Fund; 2024 [cited October 4th of 2024]. Available from: URL: https://childmortality.org/wp-content/uploads/2024/03/UNIGME-2023-Child-Mortality-Report.pdf.
- Villavicencio F, Perin J, Eilerts-Spinelli H, Yeung D, Prieto-Merino D, Hug L et al. Global, regional, and national causes of death in children and adolescents younger than 20 years: an open data portal with estimates for 2000-21. Lancet Glob Health 2024; 12(1):e16-e17. [CrossRef]
- Admasu FT, Melese BD, Amare TJ, Zewude EA, Denku CY, Dejenie TA. The magnitude of neonatal asphyxia and its associated factors among newborns in public hospitals of North Gondar Zone, Northwest Ethiopia: A cross-sectional study. PLoS One 2022; 17(3):e0264816. [CrossRef]
- Gillam-Krakauer M, Gowen Jr CW. StatPearls: Birth Asphyxia. Treasure Island (FL); 2024.
- Lai M-C, Yang S-N. Perinatal hypoxic-ischemic encephalopathy. J Biomed Biotechnol 2011; 2011:609813. [CrossRef]
- Gopagondanahalli KR, Li J, Fahey MC, Hunt RW, Jenkin G, Miller SL et al. Preterm Hypoxic-Ischemic Encephalopathy. Front Pediatr 2016; 4:114. [CrossRef]
- Cotten CM, Shankaran S. Hypothermia for hypoxic-ischemic encephalopathy. Expert Rev Obstet Gynecol 2010; 5(2):227–39.
- Antonucci R, Porcella A, Pilloni MD. Perinatal asphyxia in the term newborn. Journal of Pediatric and Neonatal Individualized Medicine 2014; 3(2):e030269.
- Distefano G, Praticò AD. Actualities on molecular pathogenesis and repairing processes of cerebral damage in perinatal hypoxic-ischemic encephalopathy. Ital J Pediatr 2010; 36:63. [CrossRef]
- Douglas-Escobar M, Weiss MD. Hypoxic-ischemic encephalopathy: a review for the clinician. JAMA Pediatr 2015; 169(4):397–403.
- Greco P, Nencini G, Piva I, Scioscia M, Volta CA, Spadaro S et al. Pathophysiology of hypoxic-ischemic encephalopathy: a review of the past and a view on the future. Acta Neurol Belg 2020; 120(2):277–88. [CrossRef]
- Morales P, Bustamante D, Espina-Marchant P, Neira-Peña T, Gutiérrez-Hernández MA, Allende-Castro C et al. Pathophysiology of perinatal asphyxia: can we predict and improve individual outcomes? EPMA J 2011; 2(2):211–30.
- Ruscher K, Wieloch T. The involvement of the sigma-1 receptor in neurodegeneration and neurorestoration. J Pharmacol Sci 2015; 127(1):30–5. [CrossRef]
- Griesmaier E, Posod A, Gross M, Neubauer V, Wegleiter K, Hermann M et al. Neuroprotective effects of the sigma-1 receptor ligand PRE-084 against excitotoxic perinatal brain injury in newborn mice. Experimental neurology 2012; 237(2):388–95. [CrossRef]
- Posod A, Pinzer K, Urbanek M, Wegleiter K, Keller M, Kiechl-Kohlendorfer U et al. The common antitussive agent dextromethorphan protects against hyperoxia-induced cell death in established in vivo and in vitro models of neonatal brain injury. Neuroscience 2014; 274:260–72. [CrossRef]
- Yang Z-J, Carter EL, Torbey MT, Martin LJ, Koehler RC. Sigma receptor ligand 4-phenyl-1-(4-phenylbutyl)-piperidine modulates neuronal nitric oxide synthase/postsynaptic density-95 coupling mechanisms and protects against neonatal ischemic degeneration of striatal neurons. Experimental neurology 2010; 221(1):166–74. [CrossRef]
- Ajmo CT, Vernon DOL, Collier L, Pennypacker KR, Cuevas J. Sigma receptor activation reduces infarct size at 24 hours after permanent middle cerebral artery occlusion in rats. Curr Neurovasc Res 2006; 3(2):89–98. [CrossRef]
- Takahashi H, Kirsch JR, Hashimoto K, London ED, Koehler RC, Traystman RJ. PPBP 4-phenyl-1-(4-phenylbutyl) piperidine decreases brain injury after transient focal ischemia in rats. Stroke 1996; 27(11):2120–3. Available from: URL: https://pubmed.ncbi.nlm.nih.gov/8898825/.
- Quinn T, Greaves R, Badoer E, Walker D. DHEA in Prenatal and Postnatal Life: Implications for Brain and Behavior: Elsevier. Vitam Horm; 2018. (Vitamins and Hormones; vol 108).
- Kroboth PD, Salek FS, Pittenger AL, Fabian TJ, Frye RF. DHEA and DHEA-S: a review. J Clin Pharmacol 1999; 39(4):327–48.
- Rabe T, Ahrendt HJ, Albring C, Bachmann A, Bitzer, Blume-Peytavi U et al. Dehydroepiandrosterone and its Sulfate Joint: Statement by the German Society for Gynecological Endocrinology and Reproductive Medicine [DGGEF] and the German Professional Association of Gynecologists (BVF). Journal of Reproductive Medicine and Endocrinology 2015; (2 (4)):18–341.
- Stárka L, Dušková M, Hill M. Dehydroepiandrosterone: a neuroactive steroid. The Journal of steroid biochemistry and molecular biology 2015; 145:254–60.
- Yilmaz C, Karali K, Fodelianaki G, Gravanis A, Chavakis T, Charalampopoulos I et al. Neurosteroids as regulators of neuroinflammation. Front Neuroendocrinol 2019; 55:100788. [CrossRef]
- Lazaridis I, Charalampopoulos I, Alexaki V-I, Avlonitis N, Pediaditakis I, Efstathopoulos P et al. Neurosteroid dehydroepiandrosterone interacts with nerve growth factor (NGF) receptors, preventing neuronal apoptosis. PLoS Biol 2011; 9(4):e1001051. [CrossRef]
- Borowicz KK, Piskorska B, Banach M, Czuczwar SJ. Neuroprotective actions of neurosteroids. Front Endocrinol (Lausanne) 2011; 2:50. [CrossRef]
- Maninger N, Wolkowitz OM, Reus VI, Epel ES, Mellon SH. Neurobiological and neuropsychiatric effects of dehydroepiandrosterone (DHEA) and DHEA sulfate (DHEAS). Front Neuroendocrinol 2009; 30(1):65–91.
- Kimonides VG, Khatibi NH, Svendsen CN, Sofroniew MV, Herbert J. Dehydroepiandrosterone (DHEA) and DHEA-sulfate (DHEAS) protect hippocampal neurons against excitatory amino acid-induced neurotoxicity. Proc Natl Acad Sci U S A 1998; 95(4):1852–7. [CrossRef]
- Cardounel A, Regelson W, Kalimi M. Dehydroepiandrosterone protects hippocampal neurons against neurotoxin-induced cell death: mechanism of action. Proceedings of the Society for Experimental Biology and Medicine. Society for Experimental Biology and Medicine (New York, N.Y.) 1999; 222(2):145–9. Available from: URL: https://pubmed.ncbi.nlm.nih.gov/10564538/. [CrossRef]
- Tao T, Liu G-J, Shi X, Zhou Y, Lu Y, Gao Y-Y et al. DHEA Attenuates Microglial Activation via Induction of JMJD3 in Experimental Subarachnoid Haemorrhage. J Neuroinflammation 2019; 16(1):243. [CrossRef]
- Li Z, Cui S, Zhang Z, Zhou R, Ge Y, Sokabe M et al. DHEA-neuroprotection and -neurotoxicity after transient cerebral ischemia in rats. J Cereb Blood Flow Metab 2009; 29(2):287–96. [CrossRef]
- Lapchak PA, Chapman DF, Nunez SY, Zivin JA. Dehydroepiandrosterone sulfate is neuroprotective in a reversible spinal cord ischemia model: possible involvement of GABA(A) receptors. Stroke 2000; 31(8):1953-6; discussion 1957.
- Li H, Klein G, Sun P, Buchan AM. Dehydroepiandrosterone (DHEA) reduces neuronal injury in a rat model of global cerebral ischemia. Brain Res 2001; 888(2):263–6. [CrossRef]
- Marx CE, Jarskog LF, Lauder JM, Gilmore JH, Lieberman JA, Morrow AL. Neurosteroid modulation of embryonic neuronal survival in vitro following anoxia. Brain Res 2000; 871(1):104–12. [CrossRef]
- Yabuki Y, Shinoda Y, Izumi H, Ikuno T, Shioda N, Fukunaga K. Dehydroepiandrosterone administration improves memory deficits following transient brain ischemia through sigma-1 receptor stimulation. Brain Res 2015; 1622:102–13. [CrossRef]
- Zhang L, Li Bs, Ma W, Barker JL, Chang YH, Zhao W et al. Dehydroepiandrosterone (DHEA) and its sulfated derivative (DHEAS) regulate apoptosis during neurogenesis by triggering the Akt signaling pathway in opposing ways. Brain Res Mol Brain Res 2002; 98(1-2):58–66. [CrossRef]
- Alexaki VI, Fodelianaki G, Neuwirth A, Mund C, Kourgiantaki A, Ieronimaki E et al. DHEA inhibits acute microglia-mediated inflammation through activation of the TrkA-Akt1/2-CREB-Jmjd3 pathway. Mol Psychiatry 2018; 23(6):1410–20.
- Clark BJ, Prough RA, Klinge CM. Mechanisms of Action of Dehydroepiandrosterone. Vitam Horm 2018; 108:29–73.
- Vannucci RC, Connor JR, Mauger DT, Palmer C, Smith MB, Towfighi J et al. Rat model of perinatal hypoxic-ischemic brain damage. J. Neurosci. Res. 1999; 55(2):158–63. Available from: URL: https://pubmed.ncbi.nlm.nih.gov/9972818/.
- Hagberg H, Wilson MA, Matsushita H, Zhu C, Lange M, Gustavsson M et al. PARP-1 gene disruption in mice preferentially protects males from perinatal brain injury. J Neurochem 2004; 90(5):1068–75. [CrossRef]
- Dimauro I, Pearson T, Caporossi D, Jackson MJ. A simple protocol for the subcellular fractionation of skeletal muscle cells and tissue. BMC Res Notes 2012; 5:513. [CrossRef]
- Cox B, Emili A. Tissue subcellular fractionation and protein extraction for use in mass-spectrometry-based proteomics. Nat Protoc 2006; 1(4):1872–8. [CrossRef]
- Crowley LC, Waterhouse NJ. Detecting Cleaved Caspase-3 in Apoptotic Cells by Flow Cytometry. Cold Spring Harb Protoc 2016; 2016(11). [CrossRef]
- Tsang CK, Liu Y, Thomas J, Zhang Y, Zheng XFS. Superoxide dismutase 1 acts as a nuclear transcription factor to regulate oxidative stress resistance. Nat Commun 2014; 5:3446. [CrossRef]
- Ganguly U, Kaur U, Chakrabarti SS, Sharma P, Agrawal BK, Saso L et al. Oxidative Stress, Neuroinflammation, and NADPH Oxidase: Implications in the Pathogenesis and Treatment of Alzheimer’s Disease. Oxid Med Cell Longev 2021; 2021:7086512.
- Li Y, Zhao T, Li J, Xia M, Li Y, Wang X et al. Oxidative Stress and 4-hydroxy-2-nonenal (4-HNE): Implications in the Pathogenesis and Treatment of Aging-related Diseases. J Immunol Res 2022; 2022.
- Valavanidis A, Vlachogianni T, Fiotakis C. 8-hydroxy-2’ -deoxyguanosine (8-OHdG): A critical biomarker of oxidative stress and carcinogenesis. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev 2009; 27(2):120–39. [CrossRef]
- Li Z, Zhou R, Cui S, Xie G, Cai W, Sokabe M et al. Dehydroepiandrosterone sulfate prevents ischemia-induced impairment of long-term potentiation in rat hippocampal CA1 by up-regulating tyrosine phosphorylation of NMDA receptor. Neuropharmacology 2006; 51(5):958–66. [CrossRef]
- Karishma KK, Herbert J. Dehydroepiandrosterone (DHEA) stimulates neurogenesis in the hippocampus of the rat, promotes survival of newly formed neurons and prevents corticosterone-induced suppression. Eur J Neurosci 2002; 16(3):445–53.
- Yuan C, Gao J, Guo J, Bai L, Marshall C, Cai Z et al. Dimethyl sulfoxide damages mitochondrial integrity and membrane potential in cultured astrocytes. PLoS One 2014; 9(9):e107447. [CrossRef]
- Kurata K, Takebayashi M, Morinobu S, Yamawaki S. beta-estradiol, dehydroepiandrosterone, and dehydroepiandrosterone sulfate protect against N-methyl-D-aspartate-induced neurotoxicity in rat hippocampal neurons by different mechanisms. J Pharmacol Exp Ther 2004; 311(1):237–45.
- Milman A, Zohar O, Maayan R, Weizman R, Pick CG. DHEAS repeated treatment improves cognitive and behavioral deficits after mild traumatic brain injury. Eur Neuropsychopharmacol 2008; 18(3):181–7. [CrossRef]
- Aragno M, Parola S, Brignardello E, Mauro A, Tamagno E, Manti R et al. Dehydroepiandrosterone prevents oxidative injury induced by transient ischemia/reperfusion in the brain of diabetic rats. Diabetes 2000; 49(11):1924–31. [CrossRef]
- Ferriero DM. Neonatal brain injury. N Engl J Med 2004; 351(19):1985–95.
| Treatment group | Number of animals, n | Total injury score, median (25th; 75th percentile) |
p-value |
|---|---|---|---|
| Control | 16 | 3.5 (1.0; 10.5) | |
| Solvent control | 16 | 8.3 (0.9; 12.0) | |
| DHEA 0.1 µg/g bw | 15 | 5.0 (0.5; 13.0) | |
| DHEA 1 µg/g bw | 17 | 9.5 (1.5; 12.0) | |
| DHEA 10 µg/g bw | 16 | 5.0 (1.1; 7.9) | |
| DHEAS 0.1 µg/g bw | 16 | 7.5 (1.3; 12.8) | |
| DHEAS 1 µg/g bw | 17 | 4.0 (1.3; 11.0) | |
| DHEAS 10 µg/g bw | 17 | 6.5 (1.5; 12.5) | 0.8291 |
| Section plane | Hemisphere | Brain region | Treatment group | Number of positive cells, median (25th; 75th percentile) |
p-value |
|---|---|---|---|---|---|
| Occipital | Contralateral | White matter | |||
| Control | 11 (4; 12) | ||||
| Solvent control | 6 (4; 10) | ||||
| DHEA 0.1 µg/g bw | 6 (4; 11) | ||||
| DHEA 1 µg/g bw | 13 (11; 16) | ||||
| DHEA 10 µg/g bw | 10 (6; 14) | ||||
| DHEAS 0.1 µg/g bw | 11 (3; 23) | ||||
| DHEAS 1 µg/g bw | 7 (6; 10) | ||||
| DHEAS 10 µg/g bw | 8 (6; 11) | 0.2681 | |||
| Ipsilateral | White matter | ||||
| Control | 125 (12; 181) | ||||
| Solvent control | 16 (4; 162) | ||||
| DHEA 0.1 µg/g bw | 19 (11; 111) | ||||
| DHEA 1 µg/g bw | 182 (10; 282) | ||||
| DHEA 10 µg/g bw | 27 (10; 141) | ||||
| DHEAS 0.1 µg/g bw | 121 (7; 248) | ||||
| DHEAS 1 µg/g bw | 25 (9; 228) | ||||
| DHEAS 10 µg/g bw | 83 (17; 230) | 0.8361 | |||
| Contralateral | Cortex | ||||
| Control | 16 (13; 25) | ||||
| Solvent control | 16 (9; 20) | ||||
| DHEA 0.1 µg/g bw | 23 (14; 33) | ||||
| DHEA 1 µg/g bw | 24 (13; 32) | ||||
| DHEA 10 µg/g bw | 15 (9; 24) | ||||
| DHEAS 0.1 µg/g bw | 20 (13; 24) | ||||
| DHEAS 1 µg/g bw | 20 (16; 26) | ||||
| DHEAS 10 µg/g bw | 19 (15; 23) | 0.5901 | |||
| Ipsilateral | Cortex | ||||
| Control | 160 (37; 276) | ||||
| Solvent control | 37 (10; 202) | ||||
| DHEA 0.1 µg/g bw | 39 (25; 107) | ||||
| DHEA 1 µg/g bw | 286 (23; 376) | ||||
| DHEA 10 µg/g bw | 34 (24; 220) | ||||
| DHEAS 0.1 µg/g bw | 158 (18; 446) | ||||
| DHEAS 1 µg/g bw | 39 (27; 169) | ||||
| DHEAS 10 µg/g bw | 93 (25; 366) | 0.9011 | |||
| Contralateral | Hippocampus | ||||
| Control | 4 (3; 5) | ||||
| Solvent control | 3 (2; 5) | ||||
| DHEA 0.1 µg/g bw | 5 (4; 5) | ||||
| DHEA 1 µg/g bw | 5 (3; 6) | ||||
| DHEA 10 µg/g bw | 4 (2; 6) | ||||
| DHEAS 0.1 µg/g bw | 5 (3; 9) | ||||
| DHEAS 1 µg/g bw | 4 (4; 6) | ||||
| DHEAS 10 µg/g bw | 4 (3; 6) | 0.8821 | |||
| Ipsilateral | Hippocampus | ||||
| Control | 69 (23; 87) | ||||
| Solvent control | 35 (5; 96) | ||||
| DHEA 0.1 µg/g bw | 48 (25; 109) | ||||
| DHEA 1 µg/g bw | 81 (4; 107) | ||||
| DHEA 10 µg/g bw | 41 (8; 115) | ||||
| DHEAS 0.1 µg/g bw | 53 (8; 159) | ||||
| DHEAS 1 µg/g bw | 51 (7; 112) | ||||
| DHEAS 10 µg/g bw | 70 (49; 135) | 0.9511 | |||
| Contralateral | Thalamus | ||||
| Control | 6 (5; 15) | ||||
| Solvent control | 6 (4; 11) | ||||
| DHEA 0.1 µg/g bw | 7 (4; 12) | ||||
| DHEA 1 µg/g bw | 6 (5; 10) | ||||
| DHEA 10 µg/g bw | 15 (6; 25) | ||||
| DHEAS 0.1 µg/g bw | 9 (6; 12) | ||||
| DHEAS 1 µg/g bw | 7 (4; 8) | ||||
| DHEAS 10 µg/g bw | 7 (4; 16) | 0.6801 | |||
| Ipsilateral | Thalamus | ||||
| Control | 43 (15; 113) | ||||
| Solvent control | 16 (6; 64) | ||||
| DHEA 0.1 µg/g bw | 12 (6; 29) | ||||
| DHEA 1 µg/g bw | 35 (8; 65) | ||||
| DHEA 10 µg/g bw | 10 (6; 41) | ||||
| DHEAS 0.1 µg/g bw | 15 (4; 38) | ||||
| DHEAS 1 µg/g bw | 8 (5; 22) | ||||
| DHEAS 10 µg/g bw | 23 (10; 39) | 0.3811 | |||
| Frontal | Contralateral | White matter | |||
| Control | 6 (4; 10) | ||||
| Solvent control | 6 (5; 10) | ||||
| DHEA 0.1 µg/g bw | 8 (6; 9) | ||||
| DHEA 1 µg/g bw | 11 (7; 14) | ||||
| DHEA 10 µg/g bw | 4 (4; 8) | ||||
| DHEAS 0.1 µg/g bw | 5 (3; 10) | ||||
| DHEAS 1 µg/g bw | 5 (4; 18) | ||||
| DHEAS 10 µg/g bw | 8 (5; 13) | 0.4941 | |||
| Ipsilateral | White matter | ||||
| Control | 22 (9; 52) | ||||
| Solvent control | 9 (4; 30) | ||||
| DHEA 0.1 µg/g bw | 8 (7; 27) | ||||
| DHEA 1 µg/g bw | 35 (12; 62) | ||||
| DHEA 10 µg/g bw | 6 (4; 17) | ||||
| DHEAS 0.1 µg/g bw | 16 (3; 67) | ||||
| DHEAS 1 µg/g bw | 7 (2; 30) | ||||
| DHEAS 10 µg/g bw | 14 (6; 29) | 0.4091 | |||
| Contralateral | Cortex | ||||
| Control | 16 (13; 30) | ||||
| Solvent control | 17 (9; 20) | ||||
| DHEA 0.1 µg/g bw | 23 (17; 32) | ||||
| DHEA 1 µg/g bw | 16 (10; 22) | ||||
| DHEA 10 µg/g bw | 15 (10; 17) | ||||
| DHEAS 0.1 µg/g bw | 15 (11; 22) | ||||
| DHEAS 1 µg/g bw | 16 (14; 18) | ||||
| DHEAS 10 µg/g bw | 14 (9; 24) | 0.4681 | |||
| Ipsilateral | Cortex | ||||
| Control | 89 (24; 231) | ||||
| Solvent control | 26 (9; 107) | ||||
| DHEA 0.1 µg/g bw | 23 (16; 93) | ||||
| DHEA 1 µg/g bw | 145 (24; 204) | ||||
| DHEA 10 µg/g bw | 13 (9; 68) | ||||
| DHEAS 0.1 µg/g bw | 54 (9; 184) | ||||
| DHEAS 1 µg/g bw | 21 (15; 85) | ||||
| DHEAS 10 µg/g bw | 59 (24; 92) | 0.3511 | |||
| Contralateral | Striatum | ||||
| Control | 6 (4; 10) | ||||
| Solvent control | 6 (3; 8) | ||||
| DHEA 0.1 µg/g bw | 9 (7; 13) | ||||
| DHEA 1 µg/g bw | 9 (4; 11) | ||||
| DHEA 10 µg/g bw | 8 (6; 11) | ||||
| DHEAS 0.1 µg/g bw | 6 (4; 9) | ||||
| DHEAS 1 µg/g bw | 5 (4; 9) | ||||
| DHEAS 10 µg/g bw | 5 (4; 6) | 0.3051 | |||
| Ipsilateral | Striatum | ||||
| Control | 92 (8; 198) | ||||
| Solvent control | 13 (5; 104) | ||||
| DHEA 0.1 µg/g bw | 12 (9; 117) | ||||
| DHEA 1 µg/g bw | 130 (15; 288) | ||||
| DHEA 10 µg/g bw | 10 (4; 15) | ||||
| DHEAS 0.1 µg/g bw | 15 (4; 72) | ||||
| DHEAS 1 µg/g bw | 9 (5; 85) | ||||
| DHEAS 10 µg/g bw | 16 (10; 69) | 0.2961 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




