Submitted:
24 October 2024
Posted:
25 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Ethical Aspects
2.2. Patients' Selections
2.3. Data Collection
2.4. Statistical Analysis
3. Results
3.1. Demographic Data
3.2. Higher Incidence of Side Effects in DMF Treated Patients
3.3. Lymphopenia Detected in Patients Treated with DRF

3.4. Higher Incidence of Infections in DMF Treated Patients
4. Discussion
5. Conclusions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dobson R, Giovannoni G. Multiple sclerosis - a review. Eur J Neurol. 2019 Jan;26(1):27-40. [CrossRef] [PubMed]
- Oh J, Vidal-Jordana A, Montalban X. Multiple sclerosis: clinical aspects. Curr Opin Neurol. 2018 Dec;31(6):752-759. [CrossRef] [PubMed]
- Popova EV, Boyko AN, Orlova EV. Dimetilfumarat v terapii remittiruyushchego rasseyannogo skleroza [Dimethylfumarate in the treatment of relapsing-remitting multiple sclerosis]. Zh Nevrol Psikhiatr Im S S Korsakova. 2016;116(10 Pt 2):68-72. Russian. [CrossRef] [PubMed]
- Wang Y, Bhargava P. Diroximel fumarate to treat multiple sclerosis. Drugs Today (Barc). 2020 Jul;56(7):431-437. [CrossRef] [PubMed]
- Biogen. Tecfdera® (dimethyl fumarate) prescribing information and patient information. 2023. https://www.tecfdera.com/conte nt/dam/commercial/multiple-sclerosis/tecfdera/pat/en_us/pdf/ full-prescribing-info.pdf. Revised Dec 2023.
- Tastan B, Arioz BI, Tufekci KU, Tarakcioglu E, Gonul CP, Genc K, Genc S. Dimethyl Fumarate Alleviates NLRP3 Inflammasome Activation in Microglia and Sickness Behavior in LPS-Challenged Mice. Front Immunol. 2021 Nov 10;12:737065. [CrossRef] [PubMed]
- Kornberg MD, Bhargava P, Kim PM, Putluri V, Snowman AM, Putluri N, Calabresi PA, Snyder SH. Dimethyl fumarate targets GAPDH and aerobic glycolysis to modulate immunity. Science. 2018 Apr 27;360(6387):449-453. [CrossRef] [PubMed]
- Biogen. Vumerity® (diroximel fumarate) prescribing information and patient information. 2023. https://www.vumerity.com/conte nt/dam/commercial/vumerity/pat/en_us/pdf/vumerity-prescribin g-information.pdf. Revised Dec 2023.
- R. T. Naismith et al., “Diroximel Fumarate Demonstrates an Improved Gastrointestinal Tolerability Profile Compared with Dimethyl Fumarate in Patients with Relapsing–Remitting Multiple Sclerosis: Results from the Randomized, Double-Blind, Phase III EVOLVE-MS-2 Study,” CNS Drugs, vol. 34, no. 2, pp. 185–196, Feb. 2020. [CrossRef]
- World Medical Association. World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA. 2013 Nov 27; 310(20): 2191-4. [CrossRef] [PubMed]
- J. Sabin et al., “Tolerability and safety of dimethyl fumarate in relapsing multiple sclerosis: a prospective observational multicenter study in a real-life Spanish population,” J Neurol, vol. 267, no. 8, pp. 2362–2371, Aug. 2020. [CrossRef]
- J. Liseno, B. Lager, C. Miller, S. L. Shankar, J. P. Mendoza, and J. B. Lewin, “Multiple Sclerosis Patients Treated With Diroximel Fumarate in the Real-World Setting Have High Rates of Persistence and Adherence,” Neurol Ther, vol. 10, no. 1, pp. 349–360, Jun. 2021. [CrossRef]
- S. Sainz de la Maza et al., “Early predictive risk factors for dimethyl fumarate-associated lymphopenia in patients with multiple sclerosis,” Mult Scler Relat Disord, vol. 59, Mar. 2022. [CrossRef]
- Jaboob A, Asmi AA, Islam MM, Rezvi S, Redha I, Al-Khabouri J, Al-Zakwani I, Al-Qassabi A, Al-Abri H, Gujjar AR. Frequency of Dimethyl Fumarate-Induced Lymphopenia among Omani Patients with Multiple Sclerosis. Sultan Qaboos Univ Med J. 2024 Feb;24(1):44-51. [CrossRef] [PubMed]
- Ravn J, Jensen HB, Kant M, Andersen PB, Góra MK, Sejbaek T. Risk factors for development of lymphopenia in dimethyl fumarate-treated patients with multiple sclerosis. Mult Scler Relat Disord. 2022 Nov;67:104081. [CrossRef] [PubMed]
- Singer BA, Wray S, Gudesblatt M, Bumstead B, Ziemssen T, Bonnell A, Scaramozza M, Levin S, Shanmugasundaram M, Chen H, Mendoza JP, Lewin JB, Shankar SL. Lymphopenia is Not the Primary Therapeutic Mechanism of Diroximel Fumarate in Relapsing-Remitting Multiple Sclerosis: Subgroup Analyses of the EVOLVE-MS-1 Study. Neurol Ther. 2024 Jun 27. [CrossRef] [PubMed]
- Dempsey JP, Wu L, Balshi A, Jun C, Baber U, Sloane JA. Worsening of lymphopenia in patients with multiple sclerosis when switched from dimethyl fumarate to diroximel fumarate. Mult Scler Relat Disord. 2024 Sep;89:105737. [CrossRef] [PubMed]
- Schneider M, Kramer J, Banks A, Moses H. New onset lymphopenia in patients with relapsing multiple sclerosis switching from long-standing dimethyl fumarate treatment to diroximel fumarate: A case series. Mult Scler. 2024 Sep;30(10):1379-1382. [CrossRef] [PubMed]
| N | Overall | Treatment | p-value2 | ||
|---|---|---|---|---|---|
| DRF, N = 30 | DMF, N = 30 | ||||
| Demography | |||||
| Sex (% Female) | 60 | 39 (65%) | 21 (70%) | 18 (60%) | 0.42 |
| Age1 | 60 | 40 (33, 46) | 45 (38, 48) | 36 (33, 41) | 0.015 |
| Side effects | |||||
| GI symptoms (%) | 60 | 23 (38%) | 2 (6.7%) | 19 (63%) | <0.001 |
| Flushing (%) | 60 | 22 (37%) | 9 (30%) | 13 (43%) | 0.28 |
| Security | |||||
| Infections (%) | 60 | 26 (43%) | 9 (30%) | 17 (57%) | 0.037 |
| Efficacy | |||||
| Outbreak (%) | 60 | 4 (6.7%) | 1 (3.3%) | 3 (10%) | 0.61 |
| EDSS1 | 60 | 1.50 (1.00, 2.00) | 1.50 (1.00, 2.00) | 1.25 (1.00, 2.00) | 0.24 |
| 1 Median (IQR) | |||||
| Treatment Received | |||||
|---|---|---|---|---|---|
| N | Overall | DRF | DMF | p-value | |
|
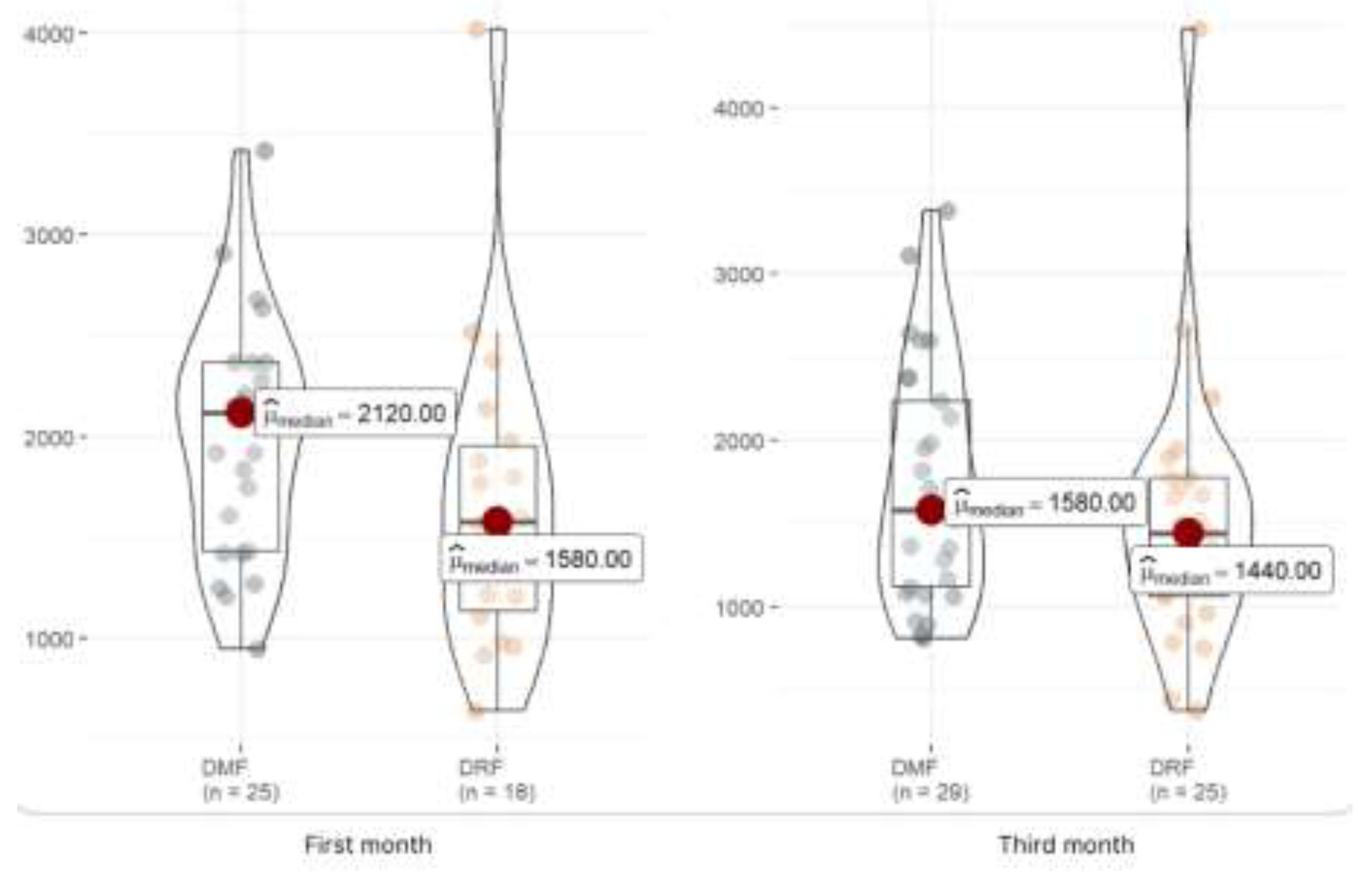
Lymphocytes M1 (x 103/µL) |
43 |
1,840 (1,345, 2,245) |
1,580 (1,135, 1,955) |
2,120 (1,430, 2,370) |
0.058 |
|
Lymphocytes M3 (x 103/µL) |
54 |
1,520 (1,080, 1,948) |
1,440 (1,060, 1,770) |
1,580 (1,120, 2,230) |
0.21 |
| Categories M1 | 43 | 0.42 | |||
| Mild | 42 (98%) | 17 (94%) | 25 (100%) | ||
| Moderate | 1 (2.3%) | 1 (5.6%) | 0 (0%) | ||
| Severe | 0 (0%) | 0 (0%) | 0 (0%) | ||
| Categories M3 | 54 | 0.040 | |||
| Mild | 50 (93%) | 21 (84%) | 29 (100%) | ||
| Moderate | 2 (3.7%) | 2 (8.0%) | 0 (0%) | ||
| Severe | 2 (3.7%) | 2 (8.0%) | 0 (0%) | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




