Submitted:
20 October 2024
Posted:
22 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1 Cell Lines and Plasmids
2.2 Peptides
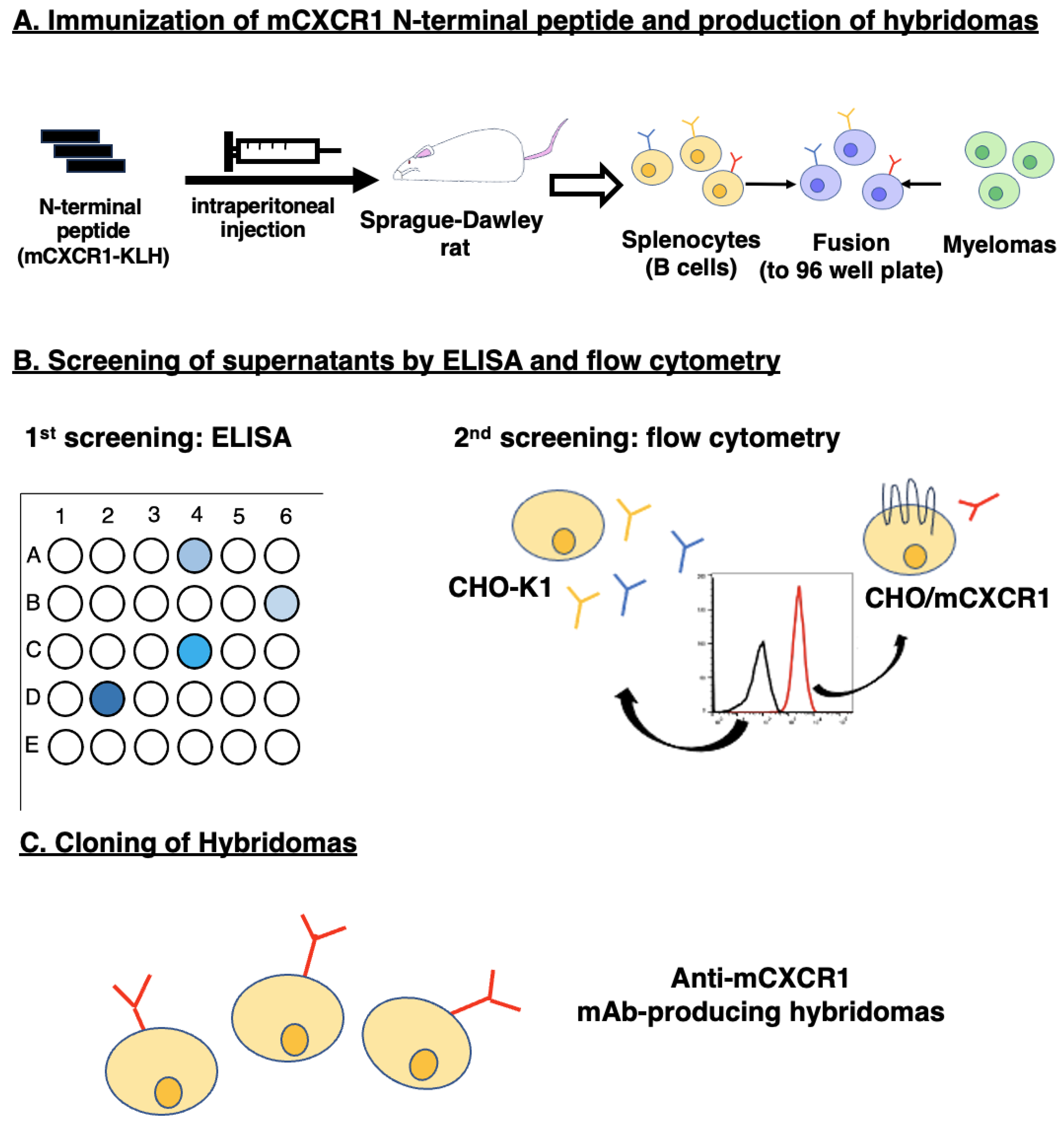
2.3. Production of Hybridomas
2.4. Aantibodies
2.5. ELISA
2.6. Flow Cytometric Analysis
2.7. Determination of Dissociation Constant (KD) by Flow Cytometry
3. Results
3.1. Development of Anti-mCXCR1 mAbs Using N-Terminal Peptide Immunization
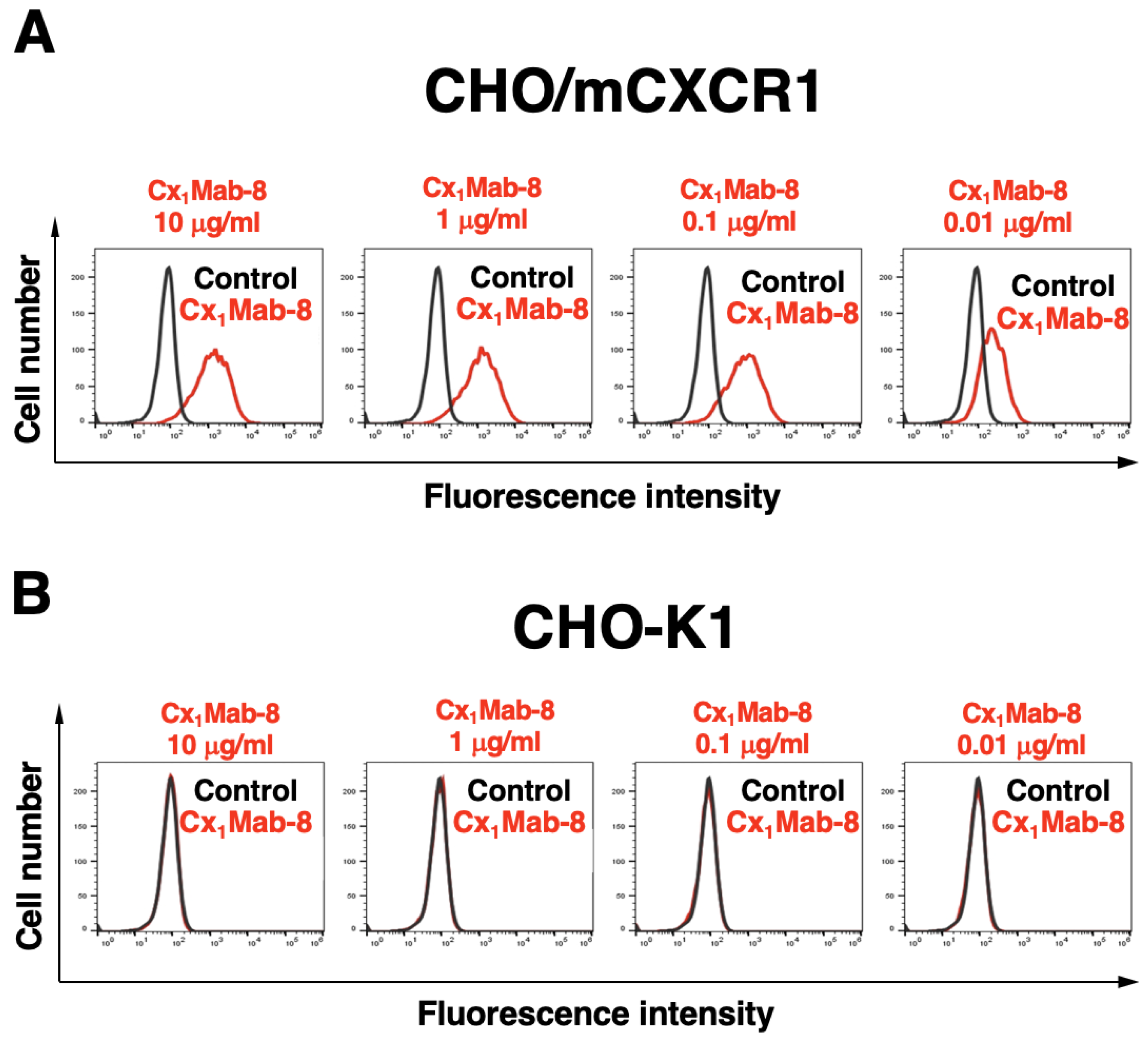
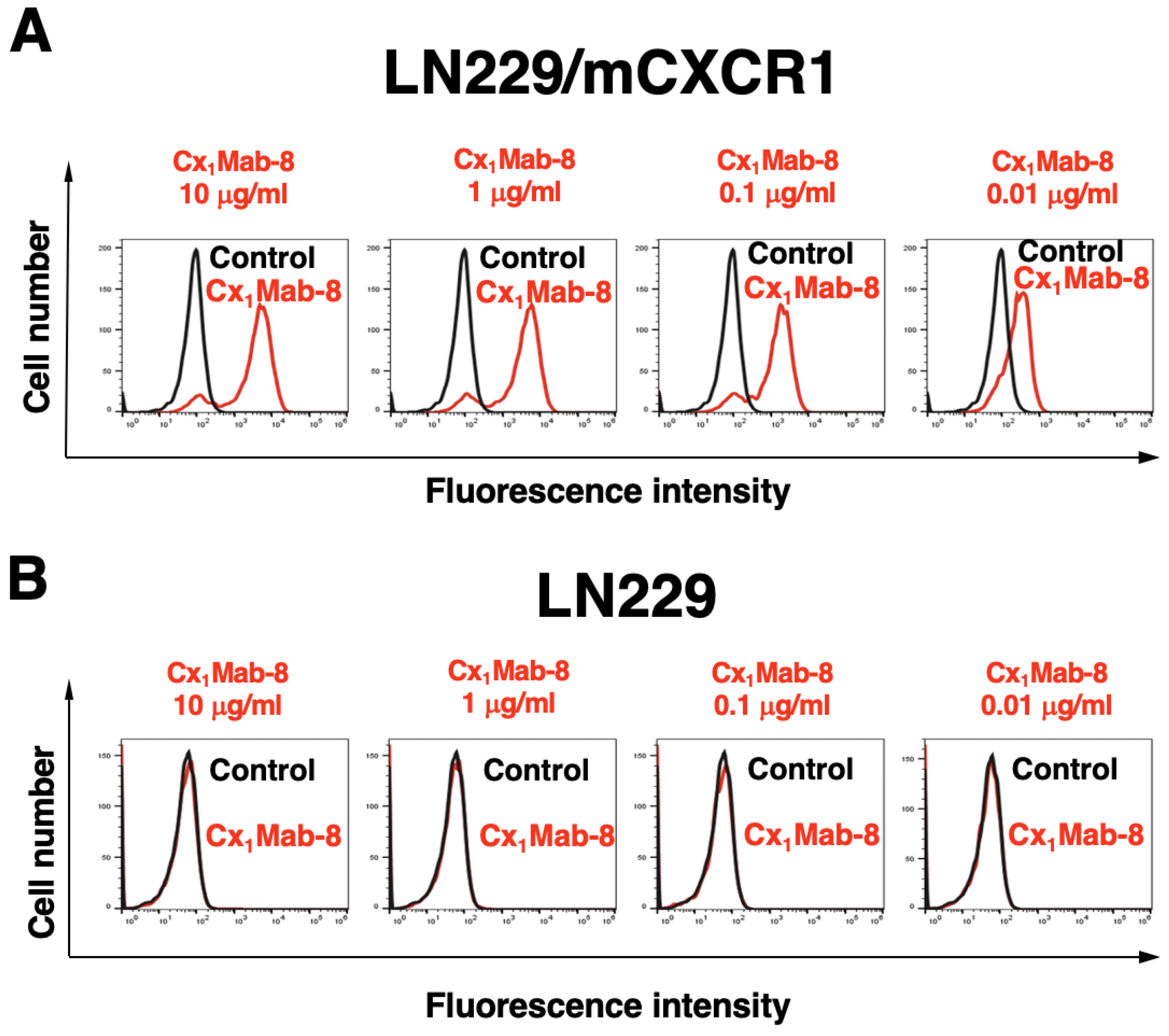
3.2. Flow Cytometric Analysis using Cx1Mab-8
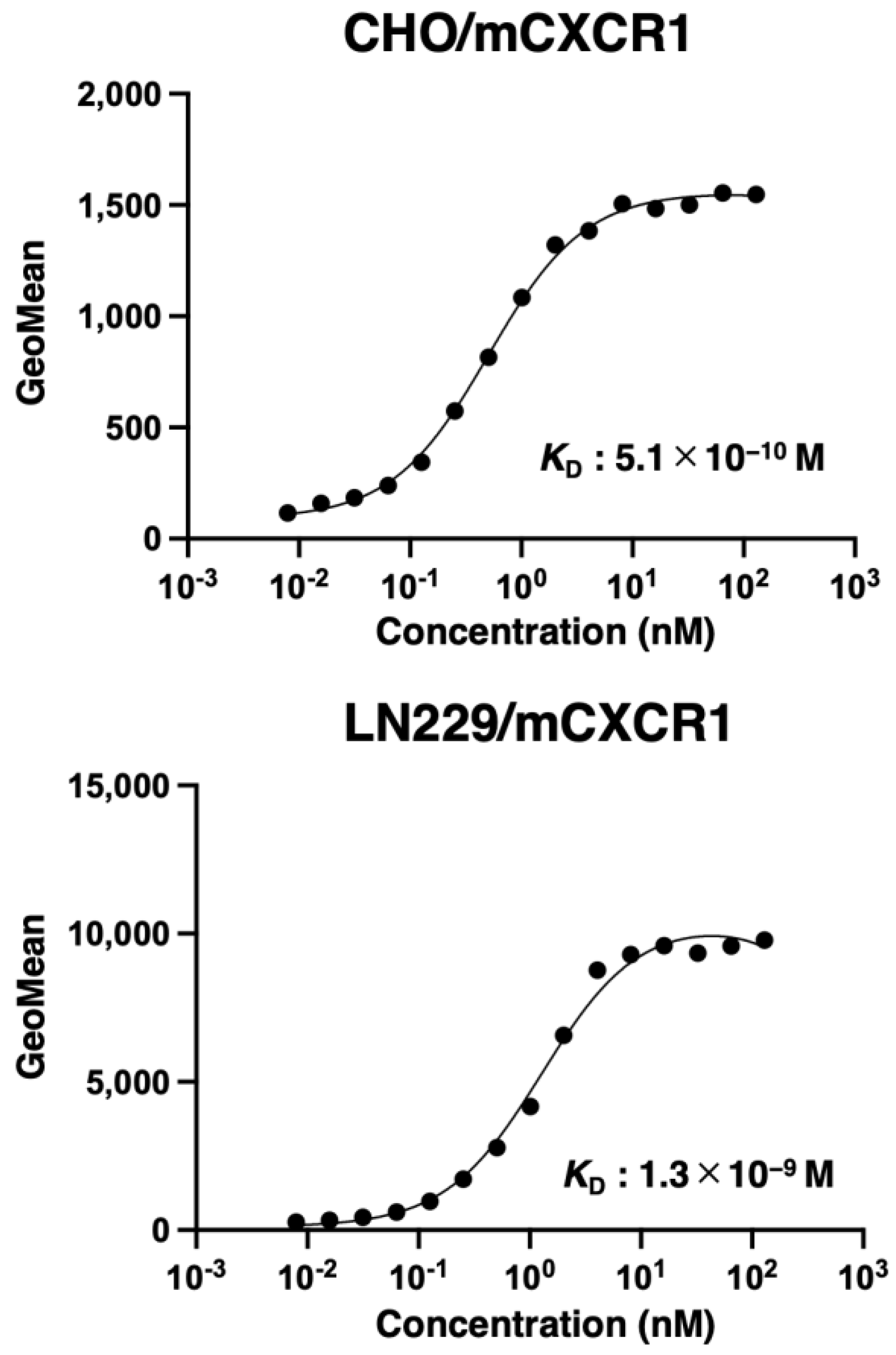
3.3. Determination of the Binding Affinity of Cx1Mab-8 Using Flow Cytometry
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Russo, R.C.; Garcia, C.C.; Teixeira, M.M.; Amaral, F.A. The CXCL8/IL-8 chemokine family and its receptors in inflammatory diseases. Expert Rev Clin Immunol 2014;10(5): 593-619.
- Thelen, M.; Peveri, P.; Kernen, P.; et al. Mechanism of neutrophil activation by NAF, a novel monocyte-derived peptide agonist. Faseb j 1988;2(11): 2702-2706.
- Sallusto, F.; Baggiolini, M. Chemokines and leukocyte traffic. Nat Immunol 2008;9(9): 949-952.
- Ha, H.; Debnath, B.; Neamati, N. Role of the CXCL8-CXCR1/2 Axis in Cancer and Inflammatory Diseases. Theranostics 2017;7(6): 1543-1588.
- McClelland, S.; Maxwell, P.J.; Branco, C.; et al. Targeting IL-8 and Its Receptors in Prostate Cancer: Inflammation, Stress Response, and Treatment Resistance. Cancers (Basel) 2024;16(16).
- Rajagopalan, L.; Rajarathnam, K. Ligand selectivity and affinity of chemokine receptor CXCR1. Role of N-terminal domain. J Biol Chem 2004;279(29): 30000-30008.
- Park, S.H.; Das, B.B.; Casagrande, F.; et al. Structure of the chemokine receptor CXCR1 in phospholipid bilayers. Nature 2012;491(7426): 779-783.
- Ishimoto, N.; Park, J.H.; Kawakami, K.; et al. Structural basis of CXC chemokine receptor 1 ligand binding and activation. Nat Commun 2023;14(1): 4107.
- Li, G.; Tanaka, T.; Suzuki, H.; Kaneko, M.K.; Kato, Y. Cx1Mab-1: A Novel Anti-mouse CXCR1 Monoclonal Antibody for Flow Cytometry. Monoclon Antib Immunodiagn Immunother 2024;in press.
- Ouchida, T.; Isoda, Y.; Tanaka, T.; et al. Cx(3)Mab-4: A Novel Anti-Mouse CXCR3 Monoclonal Antibody for Flow Cytometry. Monoclon Antib Immunodiagn Immunother 2024.
- Ouchida, T.; Suzuki, H.; Tanaka, T.; Kaneko, M.K.; Kato, Y. Cx(4)Mab-1: A Novel Anti-Mouse CXCR4 Monoclonal Antibody for Flow Cytometry. Monoclon Antib Immunodiagn Immunother 2023.
- Ouchida, T.; Isoda, Y.; Nakamura, T.; et al. Establishment of a Novel Anti-Mouse CCR1 Monoclonal Antibody C(1)Mab-6. Monoclon Antib Immunodiagn Immunother 2024.
- Tateyama, N.; Asano, T.; Suzuki, H.; et al. Epitope Mapping of Anti-Mouse CCR3 Monoclonal Antibodies Using Flow Cytometry. Antibodies (Basel) 2022;11(4).
- Saito, M.; Harigae, Y.; Li, G.; et al. C(3)Mab-2: An Anti-Mouse CCR3 Monoclonal Antibody for Immunocytochemistry. Monoclon Antib Immunodiagn Immunother 2022;41(1): 45-49.
- sano, T.; Suzuki, H.; Tanaka, T.; et al. C(3)Mab-3: A Monoclonal Antibody for Mouse CC Chemokine Receptor 3 for Flow Cytometry. Monoclon Antib Immunodiagn Immunother 2022;41(2): 74-79.
- Suzuki, H.; Tanaka, T.; Li, G.; et al. Development of a Sensitive Anti-Mouse CCR5 Monoclonal Antibody for Flow Cytometry Monoclon Antib Immunodiagn.Immunother 2024;in press.
- Suzuki, H.; Saito, M.; Asano, T.; et al. C(8)Mab-3: An Anti-Mouse CCR8 Monoclonal Antibody for Immunocytochemistry. Monoclon Antib Immunodiagn Immunother 2022;41(2): 110-114.
- Saito, M.; Tanaka, T.; Asano, T.; et al. C(8)Mab-2: An Anti-Mouse C-C Motif Chemokine Receptor 8 Monoclonal Antibody for Immunocytochemistry. Monoclon Antib Immunodiagn Immunother 2022;41(2): 115-119.
- Saito, M.; Suzuki, H.; Tanaka, T.; et al. Development of an Anti-Mouse CCR8 Monoclonal Antibody (C(8)Mab-1) for Flow Cytometry and Immunocytochemistry. Monoclon Antib Immunodiagn Immunother 2022;41(6): 333-338.
- Tanaka, T.; Li, G.; Asano, T.; et al. Development of a Novel Anti-Mouse CCR2 Monoclonal Antibody (C(2)Mab-6) by N-Terminal Peptide Immunization. Monoclon Antib Immunodiagn Immunother 2022;41(2): 80-86.
- Asano, T.; Suzuki, H.; Goto, N.; et al. Establishment of Novel Anti-Mouse CCR3 Monoclonal Antibodies (C(3)Mab-6 and C(3)Mab-7) by N-terminal Peptide Immunization. Monoclon Antib Immunodiagn Immunother 2022;41(2): 94-100.
- Takei, J.; Suzuki, H.; Asano, T.; et al. Development of a Novel Anti-Mouse CCR4 Monoclonal Antibody (C(4)Mab-1) by N-Terminal Peptide Immunization. Monoclon Antib Immunodiagn Immunother 2022;41(2): 87-93.
- Ubukata, R.; Suzuki, H.; Tanaka, T.; et al. Development of Sensitive Anti-Mouse CCR5 Monoclonal Antibodies Using the N-Terminal Peptide Immunization. Monoclon Antib Immunodiagn Immunother 2024;43(4): 112-118.
- Kobayashi, H.; Asano, T.; Suzuki, H.; et al. Establishment of a Sensitive Monoclonal Antibody Against Mouse CCR9 (C(9)Mab-24) for Flow Cytometry. Monoclon Antib Immunodiagn Immunother 2023;42(1): 15-21.
- Kitamura, K.; Suzuki, H.; Kaneko, M.K.; Kato, Y. Cx(6)Mab-1: A Novel Anti-Mouse CXCR6 Monoclonal Antibody Established by N-Terminal Peptide Immunization. Monoclon Antib Immunodiagn Immunother 2022;41(3): 133-141.
- Hirose, M.; Suzuki, H.; Ubukata, R.; et al. Development of specific anti-mouse atypical chemokine receptor 4 monoclonal antibodies. Biochem Biophys Rep 2024;40: 101824.
- Isoda, Y.; Tanaka, T.; Suzuki, H.; et al. Epitope Mapping of an Anti-Mouse CXCR6 Monoclonal Antibody (Cx(6)Mab-1) Using the 2 × Alanine Scanning Method. Monoclon Antib Immunodiagn Immunother 2022;41(5): 275-278.
- Prado, G.N.; Suetomi, K.; Shumate, D.; et al. Chemokine signaling specificity: essential role for the N-terminal domain of chemokine receptors. Biochemistry 2007;46(31): 8961-8968.
- Catusse, J.; Liotard, A.; Loillier, B.; Pruneau, D.; Paquet, J.L. Characterization of the molecular interactions of interleukin-8 (CXCL8), growth related oncogen alpha (CXCL1) and a non-peptide antagonist (SB 225002) with the human CXCR2. Biochem Pharmacol 2003;65(5): 813-821.
- Dominguez, C.; McCampbell, K.K.; David, J.M.; Palena, C. Neutralization of IL-8 decreases tumor PMN-MDSCs and reduces mesenchymalization of claudin-low triple-negative breast cancer. JCI Insight 2017;2(21).
- Bilusic, M.; Heery, C.R.; Collins, J.M.; et al. Phase I trial of HuMax-IL8 (BMS-986253), an anti-IL-8 monoclonal antibody, in patients with metastatic or unresectable solid tumors. J Immunother Cancer 2019;7(1): 240.
- Rovai, L.E.; Herschman, H.R.; Smith, J.B. The murine neutrophil-chemoattractant chemokines LIX, KC, and MIP-2 have distinct induction kinetics, tissue distributions, and tissue-specific sensitivities to glucocorticoid regulation in endotoxemia. J Leukoc Biol 1998;64(4): 494-502.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



