Submitted:
13 October 2024
Posted:
18 October 2024
You are already at the latest version
Abstract

Keywords:
INTRODUCTION
METHODS
- Eligibility criteria:
- Category I: PLHIV, including children on ART in Delhi, initially received INR 1,000 monthly, increased to INR 2,496 as of August 7, 2023. Eligibility criteria include regular ART for last 01 year, three years of residency in Delhi, and a family income of less than INR 100,000 annually.
- Category II: Double orphans children living with HIV (double OCI) in community-based care in Delhi, received INR 2,050 per month initially, increased to INR 5,116 as of August 7, 2023.
- Category III: Destitute children living with HIV (DCI) in institutional care in Delhi on ART, received INR 2,050 per month initially, increased to INR 5,116 as of August 7, 2023.
- Category IV: HIV-negative double orphan children affected by HIV (DO-ChABH) in community-based care in Delhi, received INR 1,750 per month initially, increased to INR 4,368 as of August 7, 2023.
- 2.
- Implementation Process:
- (i)
- Identification of eligible beneficiaries: Eleven government ART clinics in Delhi are responsible for identifying individuals who meet the eligibility criteria for the CTS.
- (ii)
- Document collection: The ART clinics gather necessary documents based on the category of the persons seeking assistance:
RESULTS
Cumulative Performance of the Scheme
ART Adherence Rates in Beneficiaries
Year-on-Year Performance of the Scheme
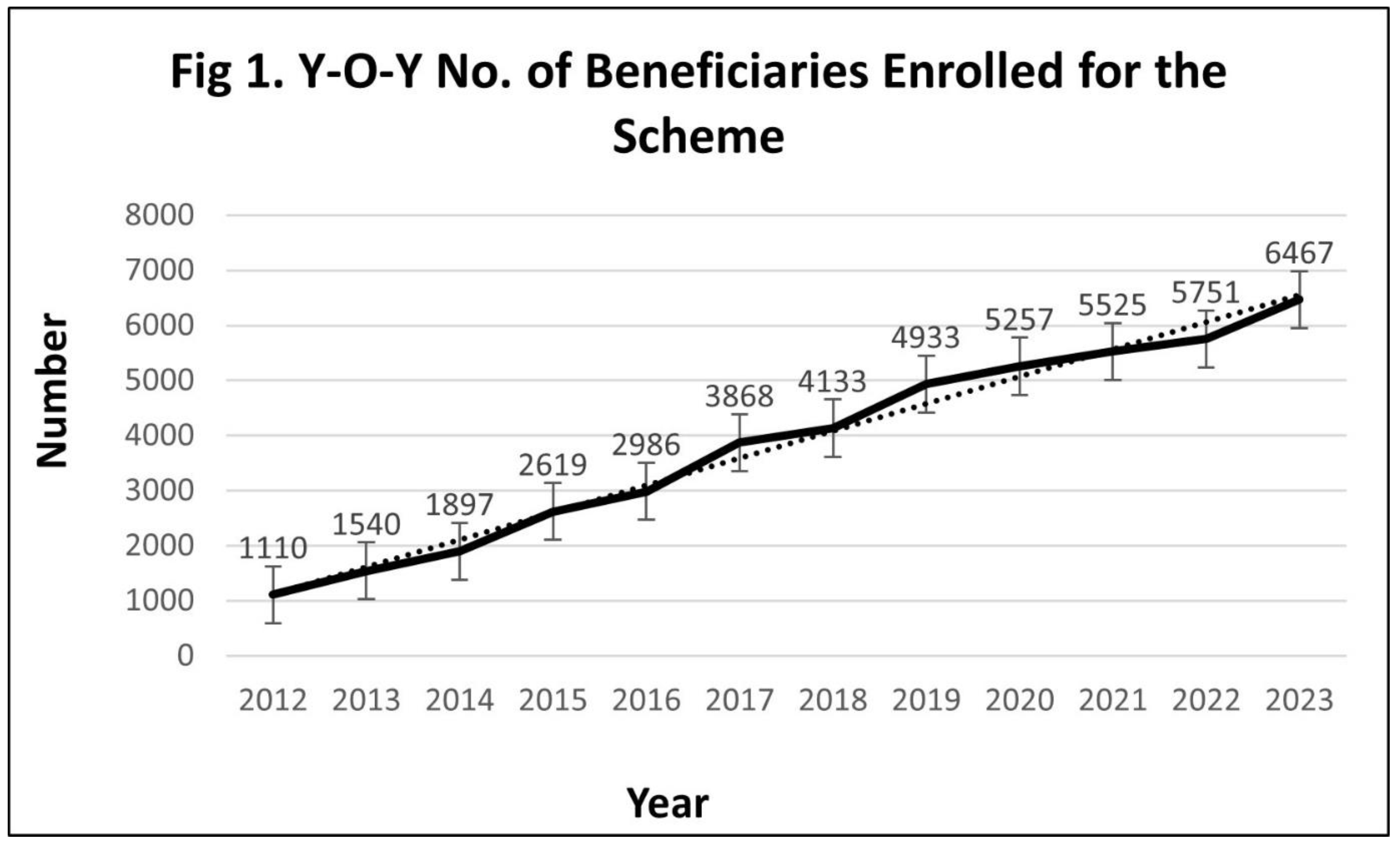
Implementation Challenges
- (i)
- Lack of Information, Education & Communication: Insufficient dissemination of CTS at ART clinics likely contributed to low enrollment rates.
- (ii)
- Lack of family centric approach at ART clinics: While ART clinics primarily focus on providing medical treatment to PLHIV, a broader approach is necessary to address the social and family aspects, especially concerning orphaned children. Developing a clear protocol for identifying and documenting children orphaned due to the death of a registered PLHIV is essential.
- (iii)
- Difficulty in Obtaining Income Proof: Some beneficiaries reported delays in acquiring income proof from the Revenue Department, which is an essential document required to enroll for the scheme.
- (iv)
- Interruption in Cash Transfers: Beneficiaries experience interruptions in cash transfers in case they fail to submit the income proof every six months. Also, payments cease for beneficiaries whose subsequent income surpasses INR 1,00,000.
- (v)
- Failure to Provide ‘Adherence to ART’ Certificate Every Quarter: Beneficiaries face discontinuation of cash transfers if they fail to provide quarterly regular treatment certificates from ART clinics. This measure aims to ensure beneficiaries maintain 100% compliance with treatment.
- (vi)
- Complex documentation requirement: The requirement for intricate documentation, including proof of the death of both parents and confirmation that at least one parent died of HIV, serves as a barrier to enrollment for HIV orphans.
- (vii)
- Rise in Cost of Living: Despite the financial assistance provided, numerous beneficiaries have expressed concerns about the inadequacy of the disbursement amount, particularly in light of the escalating cost of living due to inflation.
- (viii)
-
Delay in Payment Release:The current differentiated ART delivery involving quarterly visits for stable patients and monthly visits for new or unstable patients, coupled with subsequent procedural steps for payment release, has led to delays in disbursing financial support to beneficiaries.
- (i)
- Improved Eligibility Screening: ART center counsellors have been provided comprehensive training to refine their assessment skills in determining the eligibility of all PLHIV and HIV orphans seeking treatment.
- (ii)
- Maintain Post-Enrollment Eligibility:
- ∙
- While initial income proof from the Revenue Department remains a requirement for enrollment, subsequent verification can now be facilitated through the beneficiary's self-declaration.
- ∙
- Certificates for regular treatment for PLHIV will be dependent on quarterly biometric attendance records, ensuring more accurate monitoring and assessment of treatment adherence.
- (iii)
- Special Provisions for Illness Cases: In instances where beneficiaries are incapacitated to visit ART clinics due to illness or bedridden conditions, a designated relative or friend may be authorized to collect antiretroviral medicines for the month. However, it is imperative that the beneficiary personally attends the ART clinic in the ensuing months. The verification of treatment adherence is diligently monitored by the Nodal Officer of the ART clinic.
- (iv)
- Revision in eligibility criteria for HIV orphan: Delhi Government made the decision to include double orphan children living with HIV in the scheme regardless of their parental HIV status, thereby eliminating the previous requirement that at least one parent should have died of HIV. This change was motivated by the recognition that most children acquire HIV through Mother-to-Child Transmission, underscoring the need to provide support and assistance to all double orphan children infected with HIV irrespective of their parental HIV status.
- (v)
- Tracking Affected Family Members: ART clinics were encouraged to develop mechanisms to track affected families in case of the death of PLHIV. This is essential to consider affected children for financial support through the scheme.
- (vi)
- Streamlined Monthly Payment Release: Delhi Government has instituted a policy mandating the automatic release of payments to beneficiaries by the 10th of each month, eliminating the need for certificates confirming attendance at ART clinics. This adjustment is aimed at reducing delays associated with procedural checks, thereby ensuring timely support for beneficiaries.
- (vii)
- Additional Measures: Crucially, the scheme guarantees that no recovery will be sought from the beneficiary or their family for payments disbursed under the scheme in the event that the beneficiary is deemed ineligible during quarterly screenings or due to circumstances such as decease within the quarter or ineligibility for any other reason.
DISCUSSION
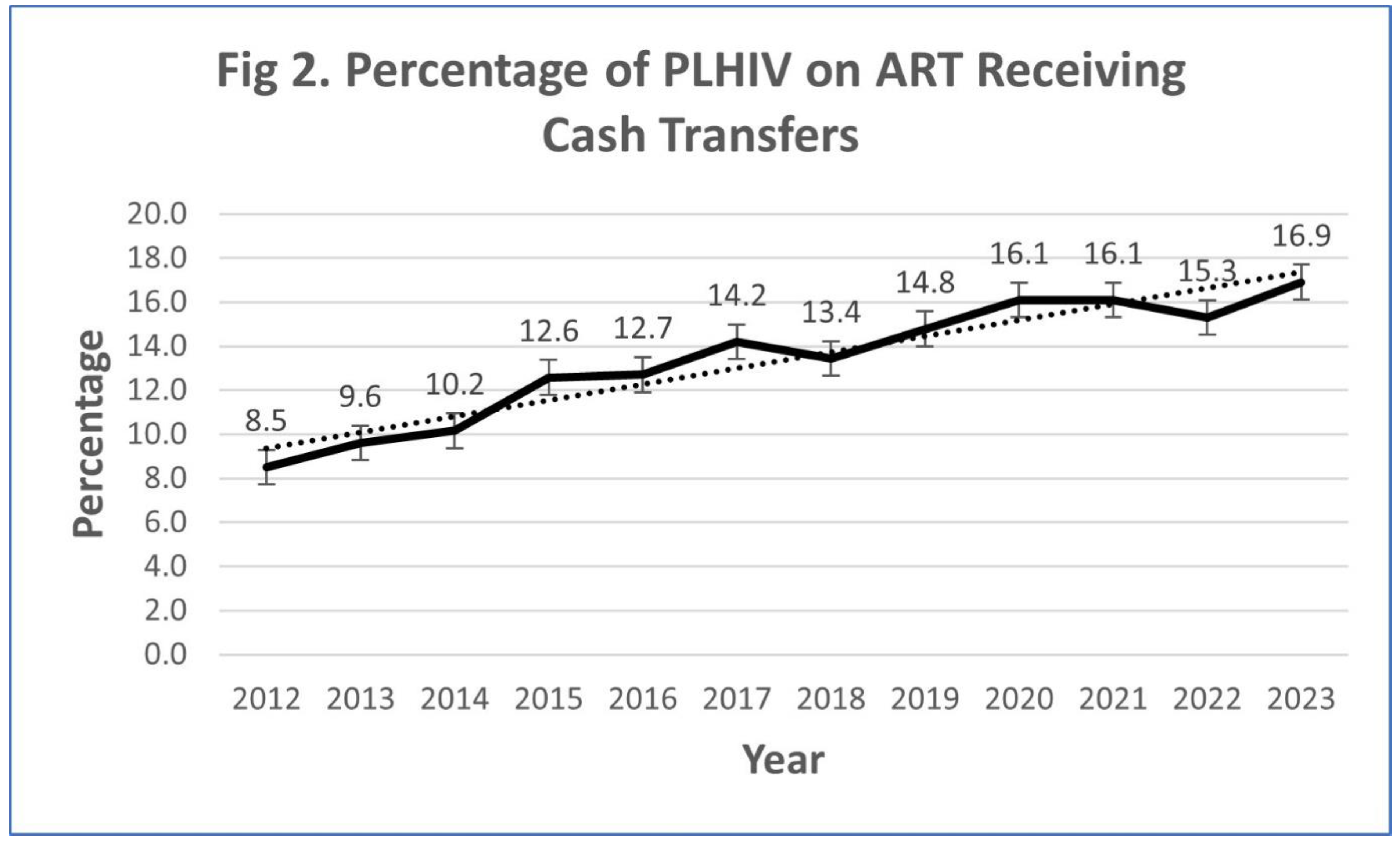
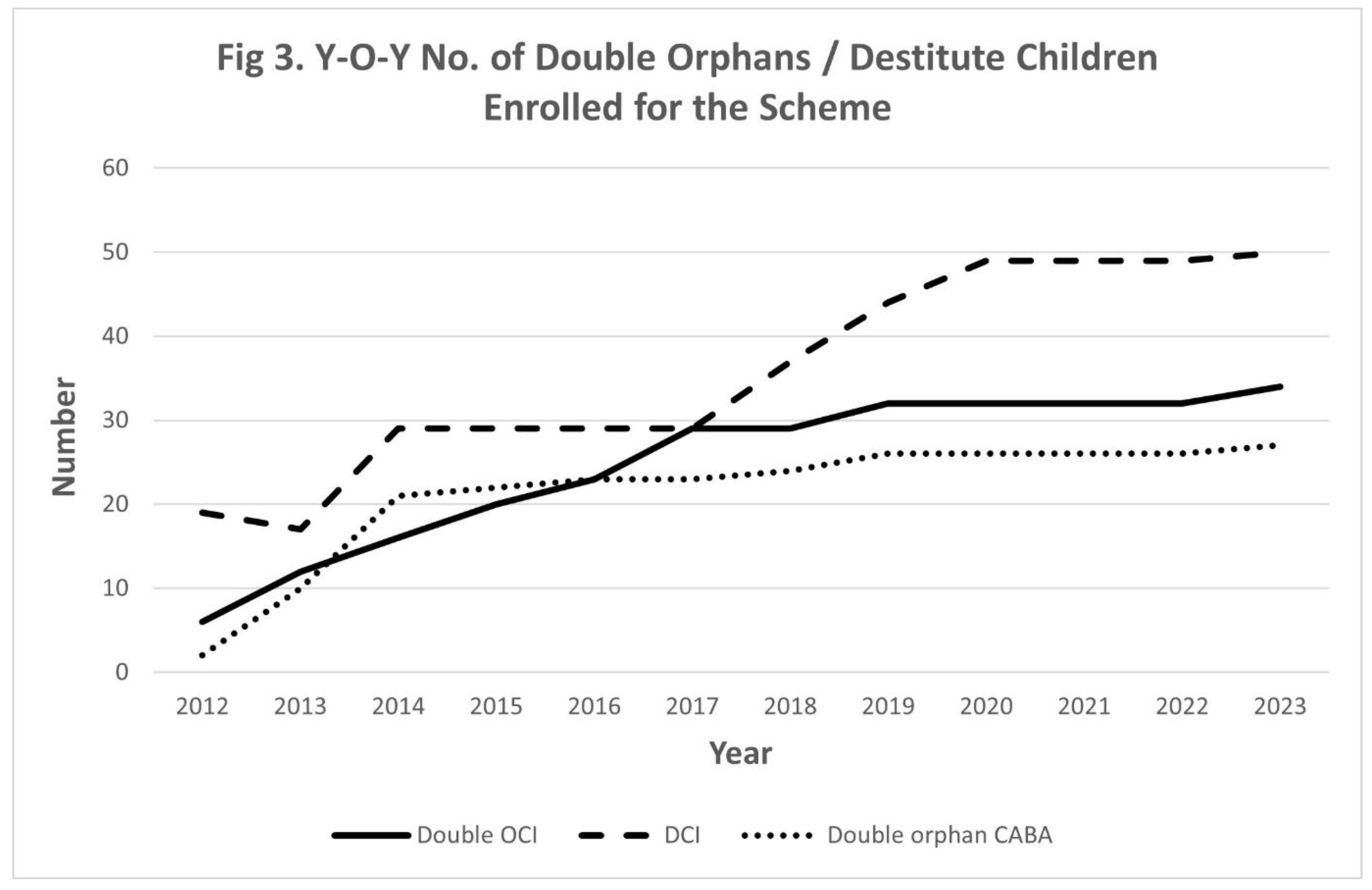
Persistent Challenges
- Double Orphan Children Infected (OCI): 34 enrolled, 1 (2.9%) died.
- Double Children Infected (DCI): 50 enrolled, 8 (16%) died.
- Double Orphan Children Affected by HIV (ChABH): 27 enrolled, all alive.
- Magnitude of the Problem Remains Unknown: Accurately estimating the number of HIV orphans remains a significant challenge due to under-reporting and the absence of comprehensive community-based surveys. Although a mapping study conducted by DSACS between 2010-2012 [3] identified some orphaned children, the lack of a systematic, extensive survey by bodies such as the National Commission for Protection of Child Rights (NCPCR) or the Delhi Commission for Protection of Child Rights (DCPCR) highlights a critical data gap. Updated data on the scope of HIV-related orphanhood in Delhi is essential for more targeted and effective interventions to support this vulnerable population.
- Lack of Focus on Single Orphan Children Affected by HIV: Current CTS provisions do not adequately address the unique needs of single orphan children affected by HIV (SO-ChABH). These children are grouped under Category I, receiving the same financial assistance as children living with HIV under parental care. However, given the specific vulnerabilities of single orphans, the scheme should provide tailored financial support, especially for HIV-infected paternal orphans. Adjusting the policy to better accommodate their needs will help improve their welfare and inclusion in the scheme.
- Enrollment Criteria for HIV-Positive Children on ART: The stipulation that HIV-positive children must be on antiretroviral therapy (ART) for at least one year before qualifying for Category I financial support is problematic. Given the high mortality rate among children without early ART, this requirement may exclude many children who need immediate financial assistance. Revising the eligibility to include all HIV-positive children on ART, regardless of duration, would be a more appropriate and life-saving policy adjustment.
Study Limitations
Financial Implications
Conclusion and Future Directions
Funding
Author contribution
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgements
Conflicts of Interest
Abbreviations
References
- National AIDS Control Organization (NACO, 2022). India HIV Estimates 2021 Fact Sheet. Ministry of Health & Family Welfare, Government of India.
- https://naco.gov.in/sites/default/files/India%20HIV%20Estimates%202021%20_Fact%20Sheets__Final_Shared_24_08_2022.
- World Health Organization (WHO, 2016). Consolidated guidelines on the use of antiretroviral drugs for treating and preventing HIV infection. Recommendations for a public health approach. 2nd Edition.
- https://www.who. 9789.
- Gupta, A.K.; Rawat, N.; Rai, K.; Rana, S.; & Sabyasachi, C.; & Sabyasachi, C. Orphan and vulnerable children infected or affected by HIV/AIDS in Delhi – situational analysis and state government's initiative of household economic strengthening. Vulnerable Children and Youth Studies 2013, 8, 161–170. [Google Scholar] [CrossRef]
- . [CrossRef]
- Packel, L. , Njau, P., Fahey, C.; Ramadhani, A.; Dow, W.H.; Jewell, N.P.; & McCoy, S. Optimizing the efficiency and implementation of cash transfers to improve adherence to antiretroviral therapy: study protocol for a cluster randomized controlled trial. Trials 2020, 21, 963. [Google Scholar] [PubMed]
- . [CrossRef]
- Yotebieng, M.; Thirumurthy, H.; Moracco, K.E.; Edmonds, A.; Tabala, M.; Kawende, B.; Wenzi, L.K.; Okitolonda, E.W.; & Behets, F.; & Behets, F. Conditional cash transfers to increase retention in PMTCT care, antiretroviral adherence, and postpartum virological suppression: a randomized controlled trial. J Acquir Immune Defic Syndr 2016, 72, S124–9, 10.1097/QAI.0000000000001062. [Google Scholar] [PubMed]
- DOI: 10.1097/QAI.0000000000001062.
- McCoy, S.I.; Njau, P.F.; Fahey, C.; Kapologwe, N; Kadiyala, S. ; Jewell, N.P.; Dow, W.H.; & Padian,N.S. Cash vs. food assistance to improve adherence to antiretroviral therapy among HIV-infected adults in Tanzania. AIDS 2017, 31, 815–825. [Google Scholar] [PubMed]
- DOI: 10.1097/QAD.0000000000001406.
- Linnemayr, S.; Stecher, C.; & Mukasa, B.; & Mukasa, B. Behavioral economic incentives to improve adherence to antiretroviral medication. AIDS 2017, 31, 719–726, 10.1097/QAD.0000000000001387. [Google Scholar] [PubMed]
- DOI: 10.1097/QAD.0000000000001387.
- Guimarães NS, Magno L, de Paula AA, Silliman M, Anderle RVR, Rasella D, Macinko J, de Souza LE, Dourado I. The effects of cash transfer programmes on HIV/AIDS prevention and care outcomes: a systematic review and meta-analysis of intervention studies. Lancet HIV, 2023, 10, e394–e403. [CrossRef]
- Ministry of External Affair. Annual Report 2013-14. https://www.mea.gov.in/Uploads/PublicationDocs/23873_EXTERNAL_AFFAIR__Eng__AR_2013-14.
- National AIDS Control Organization (NACO, 2018). National AIDS Control Programme Care Support & Treatment Services- National technical guidelines on anti-retroviral treatment. Ministry of Health & Family Welfare, Government of India.
- https://naco.gov.in/sites/default/files/NACO%20%20National%20Technical%20Guidelines%20on%20ART_October%202018%20%281%29.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



