Submitted:
19 November 2024
Posted:
20 November 2024
You are already at the latest version
Abstract
Background: Chronic heart failure (CHF) often necessitates the use of high doses of diuretics, which can lead to side effects such as worsening renal function and electrolyte imbalances. Methods: A total of 102 patients (median age 75 years) were included in the study and divided into two groups based on the level of diuretic therapy. The diuretic group received the following average doses: furosemide — 39.1 ± 22.1 mg, torasemide — 7.4 ± 3 mg, and spironolactone — 42 ± 12.4 mg. Biochemical analyses were performed on days 1 and 7 of hospitalization, with measurements of sodium, potassium, glucose, urea, and plasma osmolality (eOSM). Results: By day 7, the plasma osmolality in the diuretic group increased from 300 [297; 304] to 302.2 [298.3; 305.8] mOsm/L (p = 0.039). Urea levels in the diuretic group rose to 7.95 [5.65; 9.90] mmol/L by day 7, while remaining stable in the non-diuretic group at 5.90 [5.05; 7.50] mmol/L (p = 0.012). The proportion of urea to plasma osmolality (PropUrea/eOSM) in the diuretic group increased to 2.63% [1.89; 3.28] compared to 2.00% [1.70; 2.50] in the non-diuretic group (p = 0.011). The proportion of sodium to plasma osmolality (PropNa/eOSM) in the diuretic group decreased to 46.46% [46.02; 46.74], compared to 46.68% [46.33; 46.89] in the non-diuretic group (p = 0.050). Analysis of PropNa/eOSM and PropUrea/eOSM on day 7 without considering salt intake yielded statistically significant results, though they were less robust compared to models including salt intake. For example, in the PropUrea/eOSM model, the odds ratio (OR) was 3.01 (95% CI 1.74, 5.89; p < 0.001), and in the PropNa/eOSM model, the OR was 0.19 (95% CI 0.07, 0.43; p < 0.001). Conclusion: The study confirmed that high salt intake and active diuretic therapy significantly influence the biochemical parameters of patients with chronic heart failure (CHF), contributing to the development of the aestivation phenomenon. This is characterized by an increase in nonionic osmolytes such as urea, which may represent a physiological adaptation to stress. Patients receiving diuretics and following a high-salt diet exhibited significant changes in plasma osmolality and urea levels, indicating a physiological adaptation that may worsen prognosis by increasing the risk of sarcopenia and cachexia. Further research is needed to better understand these mechanisms and develop strategies to mitigate the negative effects.
Keywords:
1. Introduction
2. Materials and Methods
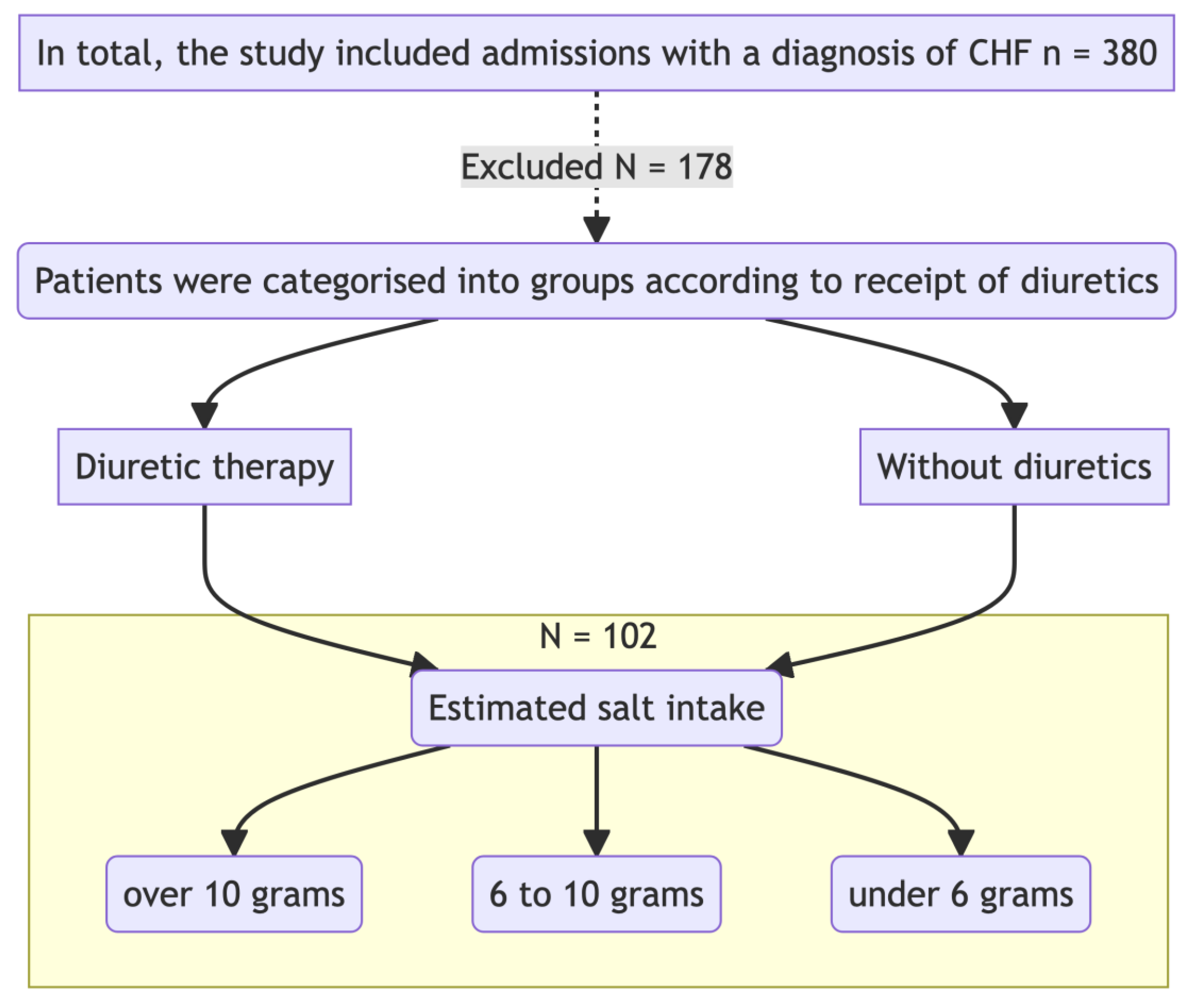
Study Design
Inclusion and Exclusion Criteria
Calculation of Plasma Osmolarity
Methods for Determining Salt Intake
Definition of Heart Failure
Determination of Kidney Function and Verification of Chronic Kidney Disease (CKD)
- The presence of any clinical markers of kidney damage was confirmed twice, with a time interval of at least 3 months between tests.
- Detected decrease in GFR (>60 ml/min/1.73 m²), albuminuria, or any other clinical markers of kidney damage confirmed over a period of 3 months.
- Persistent GFR <60 ml/min/1.73 m² regardless of the dynamics of other markers.
- Diagnosis of irreversible markers (signs) of structural kidney changes confirmed by biopsy or imaging studies.
Sample Size Calculation
Statistical Analysis
3. Results
Clinical Characteristics of the Group
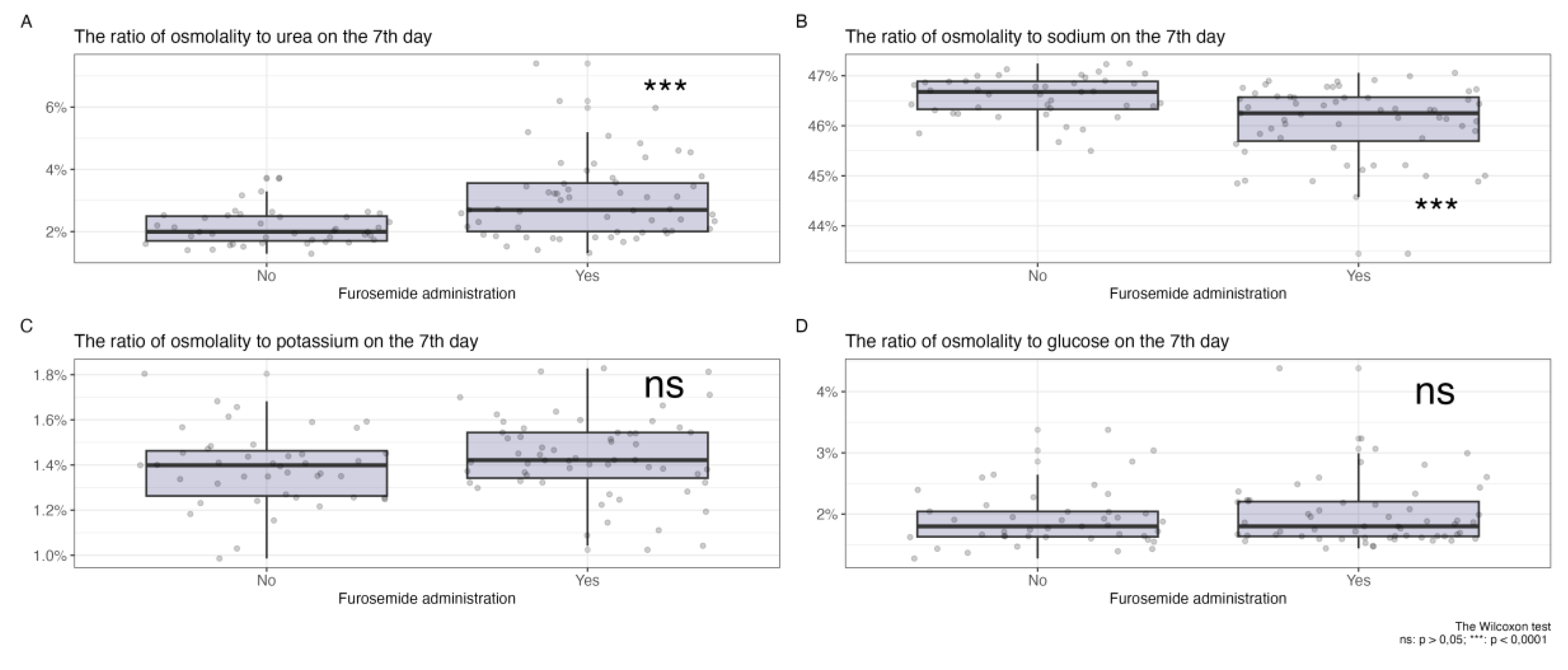
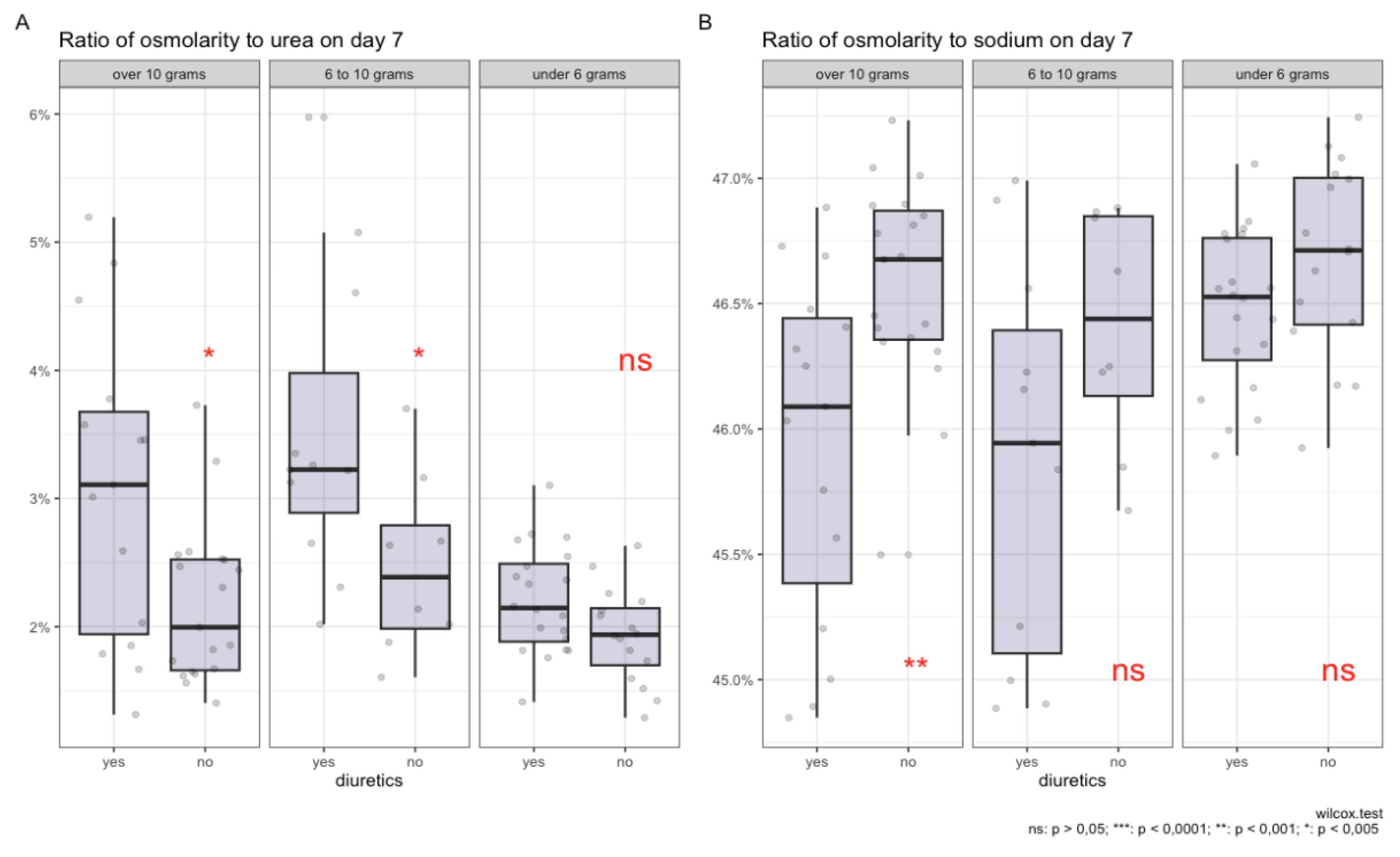
Changes in Plasma Osmolyte Ratios on Day 7 of Diuretic Therapy
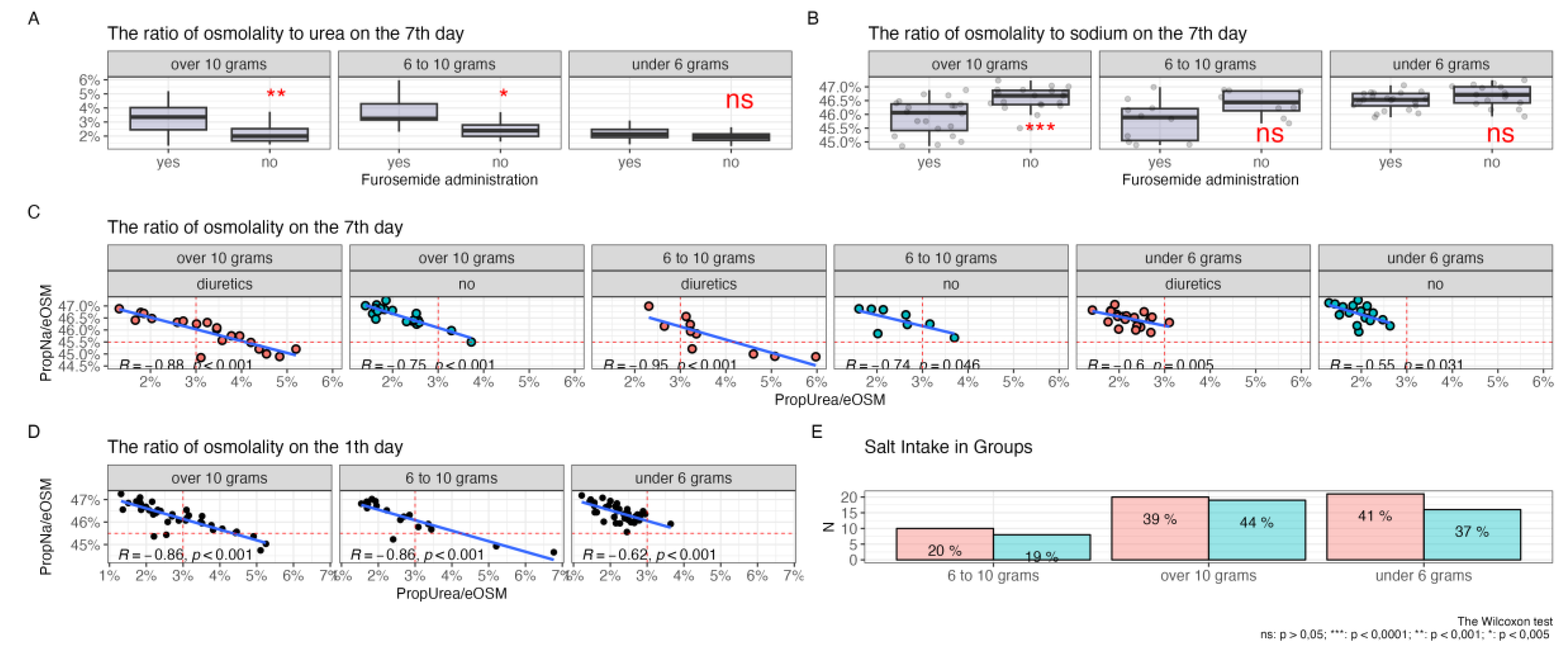
Relationship Between Plasma Osmolytes and Sodium Intake Levels
Propensity Score Matching
Modelling Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Peacock, W.F.; et al. Impact of Intravenous Loop Diuretics on Outcomes of Patients Hospitalized with Acute Decompensated Heart Failure: Insights from the ADHERE Registry. Cardiology 2008, 113, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Felker, G.M.; et al. Diuretic Strategies in Patients with Acute Decompensated Heart Failure. New England Journal of Medicine 2011, 364, 797–805. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; et al. Obesity and the Response to Intensified Diuretic Treatment in Decompensated Heart Failure: A DOSE Trial Substudy. Journal of Cardiac Failure 2012, 18, 837–844. [Google Scholar] [CrossRef] [PubMed]
- Storey, K.B.; Storey, J.M. Aestivation: Signaling and Hypometabolism. Journal of Experimental Biology 2012, 215, 1425–1433. [Google Scholar] [CrossRef] [PubMed]
- Nihlén, S.; et al. The Contribution of Plasma Urea to Total Osmolality during Iatrogenic Fluid Reduction in Critically Ill Patients. Function 2021, 3, zqab055. [Google Scholar] [CrossRef]
- Delles, C.; Rossitto, G. Of Snails, Earthworms, and Men: Insights into Strategies to Preserve Water. Function 2021, 3, zqab071. [Google Scholar] [CrossRef]
- Haussinger, D.; Lang, F.; Gerok, W. Regulation of Cell Function by the Cellular Hydration State. American Journal of Physiology-Endocrinology and Metabolism 1994, 267, E343–E355. [Google Scholar] [CrossRef]
- Lerchl, K.; et al. Agreement Between 24-Hour Salt Ingestion and Sodium Excretion in a Controlled Environment. Hypertension 2015, 66, 850–857. [Google Scholar] [CrossRef]
- Titze, J.; et al. Reduced Osmotically Inactive Na Storage Capacity and Hypertension in the Dahl Model. American Journal of Physiology-Renal Physiology 2002, 283, F134–F141. [Google Scholar] [CrossRef]
- Titze, J.; et al. Osmotically Inactive Skin Na+ Storage in Rats. American Journal of Physiology-Renal Physiology 2003, 285, F1108–F1117. [Google Scholar] [CrossRef]
- Zuo, X.; et al. Sarcopenia and Cardiovascular Diseases: A Systematic Review and Meta-Analysis. Journal of Cachexia, Sarcopenia and Muscle 2023, 14, 1183–1198. [Google Scholar] [CrossRef] [PubMed]
- Russian Society of Cardiology (RSC). 2020 Clinical Practice Guidelines for Chronic Heart Failure. Russian Journal of Cardiology 2020, 25, 4083. [Google Scholar] [CrossRef]
- Levey, A.S.; et al. A New Equation to Estimate Glomerular Filtration Rate. Annals of Internal Medicine 2009, 150, 604. [Google Scholar] [CrossRef] [PubMed]
- Clinical recommendations. Chronic kidney disease (CKD). Nephrology (Saint-Petersburg). 2021, 25, 10–82. (In Russia) [Google Scholar]
- Austin, P.C. An Introduction to Propensity Score Methods for Reducing the Effects of Confounding in Observational Studies. Multivariate Behavioral Research 2011, 46, 399–424. [Google Scholar] [CrossRef] [PubMed]
- Ho, D.E.; et al. MatchIt: Nonparametric Preprocessing for Parametric Causal Inference. Journal of Statistical Software 2011, 42. [Google Scholar] [CrossRef]
- Kovarik, J.J.; et al. Adaptive Physiological Water Conservation Explains Hypertension and Muscle Catabolism in Experimental Chronic Renal Failure. Acta Physiologica (Oxford, England) 2021, 232, e13629. [Google Scholar] [CrossRef]
- Wilkie, M.P.; et al. The African Lungfish (Protopterus dolloi): Ionoregulation and Osmoregulation in a Fish out of Water. Physiological and Biochemical Zoology 2007, 80, 99–112. [Google Scholar] [CrossRef]
- Kitada, K.; et al. High Salt Intake Reprioritizes Osmolyte and Energy Metabolism for Body Fluid Conservation. The Journal of Clinical Investigation 2017, 127, 1944–1959. [Google Scholar] [CrossRef]
- Perucca, J.; et al. Sex Difference in Urine Concentration Across Differing Ages, Sodium Intake, and Level of Kidney Disease. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology 2007, 292, R700–705. [Google Scholar] [CrossRef]
- Ishikawa, S.; et al. Loop Diuretics Are Associated with Greater Risk of Sarcopenia in Patients with Non-Dialysis-Dependent Chronic Kidney Disease. PLOS ONE 2018, 13, e0192990. [Google Scholar] [CrossRef]
- Hultström, M.; et al. Dehydration Is Associated with Production of Organic Osmolytes and Predicts Physical Long-Term Symptoms After COVID-19: A Multicenter Cohort Study. Journal 2021.
- Baumgartner, C. , Wolf, P. , Beiglböck, H., Pfleger, L., Fellinger, P., Heitzinger, G., Metz, M., Leutner, M., Kautzky-Willer, A., Krššák, M., et al. Potential role of skeletal muscle glycerophosphocholine in response to altered fluid balance in humans: an in vivo nuclear magnetic resonance study. American Journal of Physiology-Endocrinology and Metabolism 2023, 324, E339–E346. [Google Scholar] [CrossRef]
- Rossitto, G. , Maiolino, G. , Lerco, S., Ceolotto, G., Blackburn, G., Mary, S., Antonelli, G., Berton, C., Bisogni, V., Cesari, M., et al. High sodium intake, glomerular hyperfiltration, and protein catabolism in patients with essential hypertension. Cardiovasc Res 2020, 117, 1372–1381. [Google Scholar] [CrossRef]
- Wiig, H.; Luft, F.C.; Titze, J.M. The Interstitium Conducts Extrarenal Storage of Sodium and Represents a Third Compartment Essential for Extracellular Volume and Blood Pressure Homeostasis. Acta Physiologica 2018, 222, e13006. [Google Scholar] [CrossRef] [PubMed]
- Artyukov, I.; et al. The First Observation of Osmotically Neutral Sodium Accumulation in the Myocardial Interstitium. Scientific Reports 2021, 11, 22025. [Google Scholar] [CrossRef]
- Sonaglioni A, Lonati C, Tescaro L, Nicolosi GL, Proietti M, Lombardo M, Harari S. Prevalence and clinical outcome of main echocardiographic and hemodynamic heart failure phenotypes in a population of hospitalized patients 70 years old and older. Aging Clin Exp Res. 2022, 34l, 1081–1094. [CrossRef]
| Inclusion Criteria | Exclusion Criteria |
|---|---|
| Presence of an established diagnosis of CHF (NYHA I- IV Functional class) | Unlikely cooperation with the patient during the trial, low adherence to therapy for social, psychological, economic, and other reasons, incapacity |
| Stable therapy for 3 months prior to inclusion in the study | Patients with cancer who are on radiation therapy or chemotherapy |
| Stable therapy with ACE inhibitors/ARBs, beta-blockers for 3 weeks prior to hospitalization | Need to be in the intensive care unit at the time of admission |
| Signing voluntary informed consent | Any severe, decompensated, or unstable somatic disease or condition that, in the opinion of the investigator, threatens the life of the patient or worsens the prognosis (including anemia, autoimmune, endocrinological, etc.) |
| Constant salt intake according to the food diary during the week before hospitalization | Abuse of alcohol, drugs, or medicines |
| No loop diuretic therapy prior to inclusion in the study for 2 weeks | Taking thiazide or thiazide-like diuretics |
| Characteristic | Patients Receiving Diuretics, N = 59 1 | Patients Not Receiving Diuretics, N = 431 | p-value 2 |
|---|---|---|---|
| Age in years, median [Q25;Q75] | 81 (72, 84) | 73 (67, 79) | 0.011 |
| Gender | >0.9 | ||
| Female (%) | 34 (58%) | 25 (58%) | |
| Male (%) | 25 (42%) | 18 (42%) | |
| Sodium 1 day (mMol/L) | 139.0 (137.2, 140.6) | 138.8 (137.2, 140.7) | >0.9 |
| Potassium 1 day (mMol/L) | 4.01 (3.90, 4.32) | 3.97 (3.71, 4.29) | 0.3 |
| Urea 1 day (mMol/L) | 7.4 (5.6, 10.0) | 6.6 (5.5, 8.0) | 0.15 |
| Glucose 1 day (mMol/L) | 5.81 (5.39, 6.99) | 5.91 (5.39, 7.09) | 0.7 |
| Sodium day 7 (mMol/L) | 139.50 (138.50, 141.25) | 139.80 (138.45, 141.20) | 0.9 |
| Potassium day 7 (mMol/L) | 4.36 (4.08, 4.70) | 4.12 (3.77, 4.42) | 0.042 |
| Urea day 7 (mMol/L) | 8.2 (6.1, 10.8) | 5.9 (5.1, 7.5) | <0.001 |
| Glucose day 7 (mMol/L) | 5.47 (4.94, 6.60) | 5.33 (4.87, 6.25) | 0.5 |
| Functional class of CHF (NYHA) | <0.001 | ||
| NYHA I | 5 (8.5%) | 18 (42%) | |
| NYHA II | 27 (46%) | 25 (58%) | |
| NYHA III | 26 (44%) | 0 (0%) | |
| NYHA IV | 1 (1.7%) | 0 (0%) | |
| Ejection fraction (%) | 51 (47, 52) | 52 (50, 52) | 0.049 |
| Estimated osmolarity on day 1 (mOsm/L) | 300 (297, 304) | 299 (296, 302) | 0.5 |
| Estimated osmolarity on day 7 (mOsm/L) | 302.2 (298.3, 305.8) | 300.2 (295.9, 303.5) | 0.039 |
| Estimated glomerular filtration rate (ml/min/1.73 m2) | 50 (40, 59) | 56 (48, 65) | 0.021 |
| Albumin (g/l) | 39.2 (37.1, 40.8) | 39.2 (37.0, 41.4) | 0.8 |
| Total protein (g/l) | 67.8 (65.1, 71.0) | 68.5 (64.8, 71.5) | >0.9 |
| ALT (U/L) | 18 (12, 29) | 17 (13, 23) | 0.5 |
| AST (U/L) | 20 (16, 29) | 20 (18, 25) | 0.8 |
| HDL (mMol/L) | 1.31 (1.17, 1.45) | 1.26 (1.06, 1.34) | 0.2 |
| LDL (mMol/L) | 2.80 (2.18, 3.29) | 2.63 (2.20, 3.27) | >0.9 |
| Triglycerides (mMol/L) | 1.30 (0.91, 1.71) | 1.27 (0.85, 1.84) | 0.9 |
| Cholesterol (mMol/L) | 4.50 (3.64, 5.40) | 4.60 (3.50, 5.45) | >0.9 |
| CRP (mg/L) | 5 (3, 10) | 5 (2, 8) | 0.3 |
| Diabetes mellitus | 0.7 | ||
| No (%) | 39 (66%) | 30 (70%) | |
| Yes (%) | 20 (34%) | 13 (30%) | |
| CKD (stage) | 0.025 | ||
| C1 | 0 (0%) | 1 (2.3%) | |
| C2 | 1 (1.7%) | 5 (12%) | |
| C3a | 24 (41%) | 23 (53%) | |
| C3b | 16 (27%) | 4 (9.3%) | |
| C4 | 5 (8.5%) | 1 (2.3%) | |
| no | 13 (22%) | 9 (21%) |
| Characteristic | Patients Receiving Diuretics, N = 311 | Patients Not Receiving Diuretics, N = 431 | p-value2 |
|---|---|---|---|
| Age (years) | 73 (65, 77) | 73 (67, 79) | 0.6 |
| Ejection fraction (%) | 52.0 (50.0, 53.0) | 52.0 (50.0, 52.0) | 0.8 |
| Estimated glomerular filtration rate (ml/min/1.73 m2) | 55 (49, 66) | 56 (48, 65) | 0.7 |
| CKD (stage) | 0.2 | ||
| C1 | 0 (0%) | 1 (2.3%) | |
| C2 | 1 (3.2%) | 5 (12%) | |
| C3a | 13 (42%) | 23 (53%) | |
| C3b | 4 (13%) | 4 (9.3%) | |
| C4 | 0 (0%) | 1 (2.3%) | |
| нет | 13 (42%) | 9 (21%) |
| Characteristic | Patients Receiving Diuretics, N = 281 | Patients Not Receiving Diuretics, N = 431 | p-value2 |
|---|---|---|---|
| Sodium day 1 (mMol/L) | 138.90 (137.33, 140.53) | 138.80 (137.20, 140.70) | >0.9 |
| Potassium day 1 (mMol/L) | 3.97 (3.90, 4.07) | 3.97 (3.71, 4.29) | >0.9 |
| Glucose day 1 (mMol/L) | 5.81 (5.40, 6.67) | 5.91 (5.39, 7.09) | 0.6 |
| Urea day 1 (mMol/L) | 6.45 (5.15, 8.10) | 6.60 (5.50, 7.95) | 0.6 |
| Sodium day 7 (mMol/L) | 139.40 (138.58, 140.93) | 139.80 (138.45, 141.20) | 0.8 |
| Potassium day 7 (mMol/L) | 4.23 (4.02, 4.46) | 4.12 (3.77, 4.42) | 0.7 |
| Glucose day 7 (mMol/L) | 5.26 (4.78, 6.52) | 5.33 (4.87, 6.25) | 0.9 |
| Urea day 7 (mMol/L) | 7.95 (5.65, 9.90) | 5.90 (5.05, 7.50) | 0.012 |
| Urea to osmolarity ratio (%) | 2.63 (1.89, 3.28) | 2.00 (1.70, 2.50) | 0.011 |
| Sodium to osmolarity ratio (%) | 46.46 (46.02, 46.74) | 46.68 (46.33, 46.89) | 0.050 |
| Potassium to osmolarity ratio (%) | 1.40 (1.32, 1.49) | 1.40 (1.26, 1.46) | 0.8 |
| Glucose to osmolarity ratio (%) | 1.74 (1.59, 2.19) | 1.80 (1.63, 2.04) | 0.9 |
| Model 1 | Model 2 | Model 3 | Model 4 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Переменные | OR1 | 95% CI1 | p-value | OR1 | 95% CI1 | p-value | OR1 | 95% CI1 | p-value | OR1 | 95% CI1 | p-value |
| PropK/eOSM | 6.45 | 0.62, 78.3 | 0.12 | |||||||||
| Salt intake | 0.5 | 0.6 | 0.2 | 0.3 | ||||||||
| over 10 grams | — | — | — | — | — | — | — | — | ||||
| 6 to 10 grams | 1.68 | 0.68, 4.20 | 1.51 | 0.62, 3.71 | 2.65 | 0.97, 7.71 | 2.29 | 0.86, 6.39 | ||||
| under 6 grams | 1.70 | 0.57, 5.30 | 1.54 | 0.53, 4.69 | 1.39 | 0.40, 4.88 | 1.54 | 0.46, 5.38 | ||||
| PropGlucose/eOSM. | 1.32 | 0.60, 3.13 | 0.5 | |||||||||
| PropUrea/eOSM | 3.52 | 1.94, 7.26 | <0.001 | |||||||||
| PropNa/eOSM | 0.16 | 0.06, 0.39 | <0.001 | |||||||||
| AIC | 143 | 145 | 124 | 126 | ||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




