Submitted:
14 October 2024
Posted:
15 October 2024
You are already at the latest version
Abstract
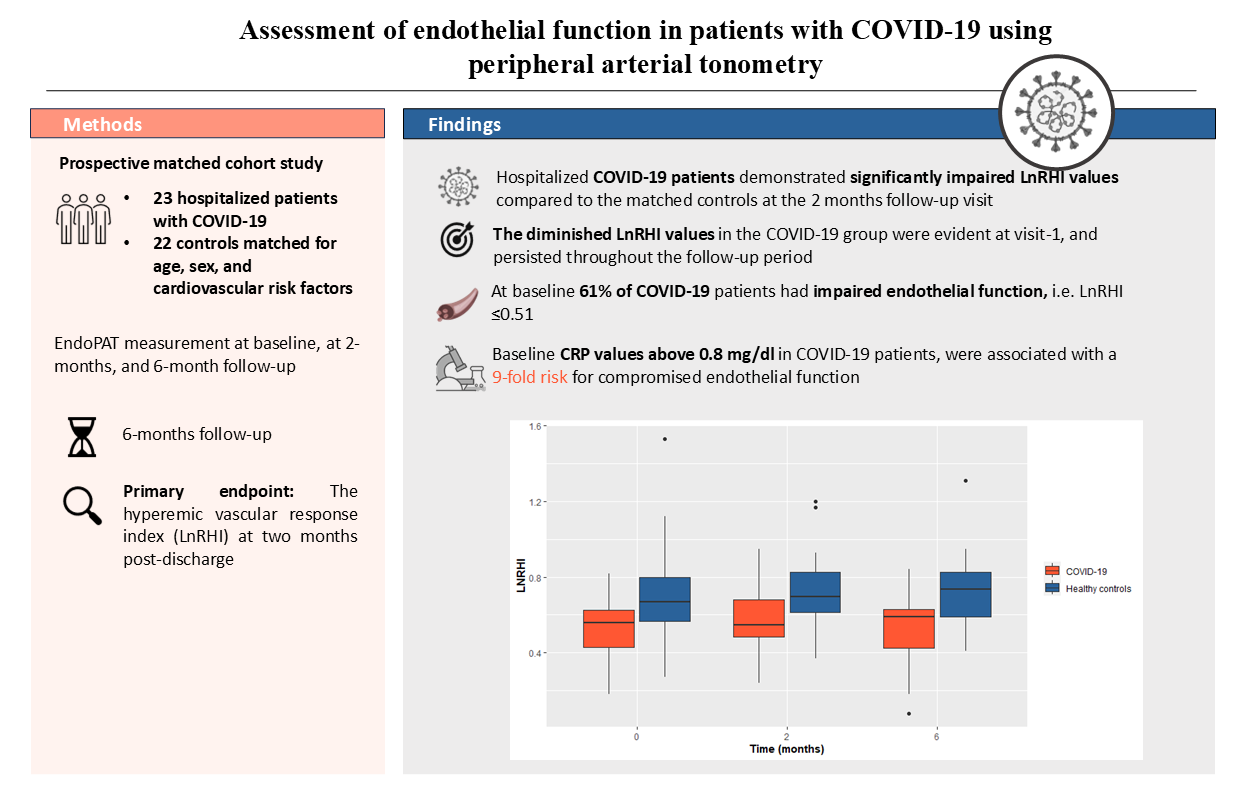
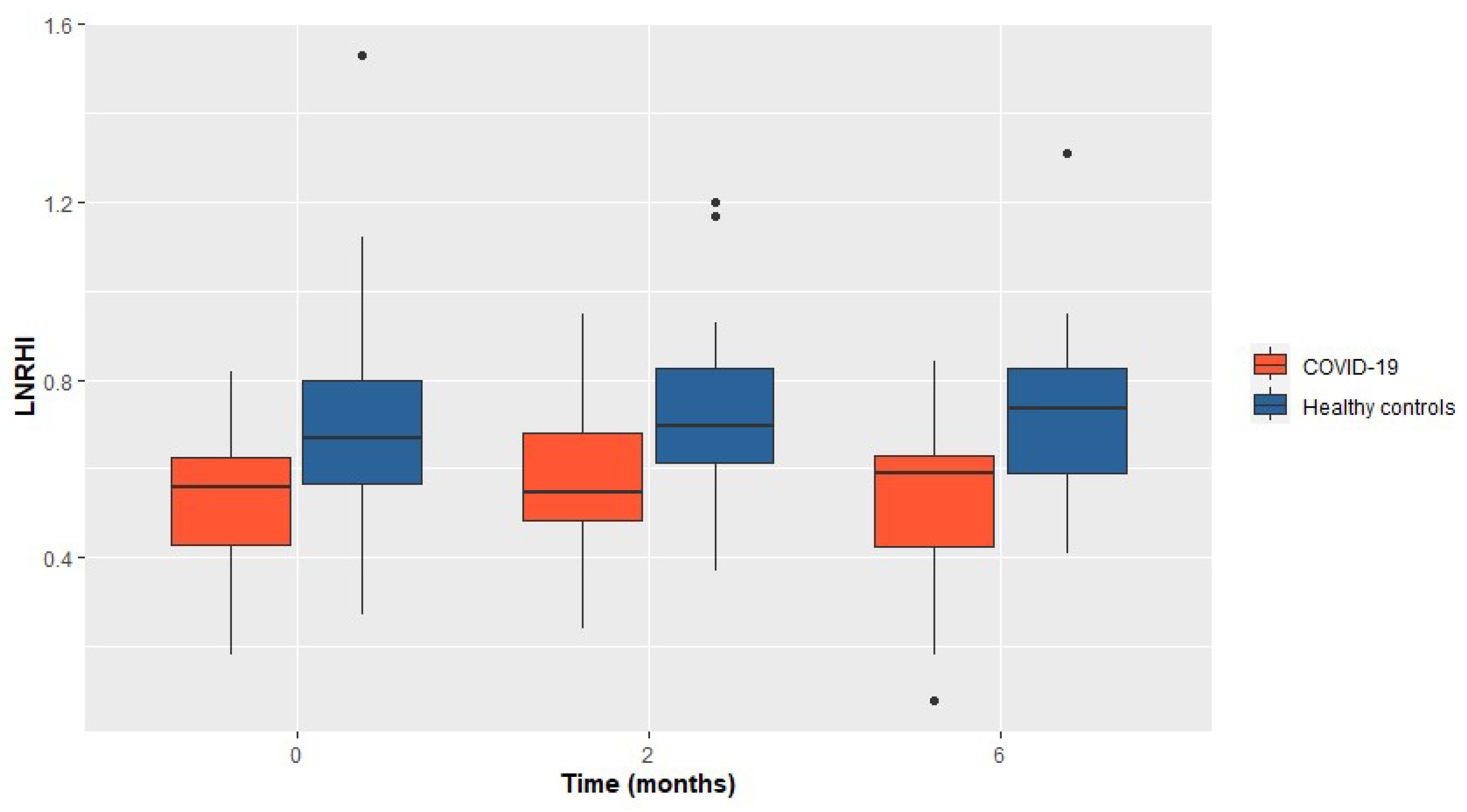
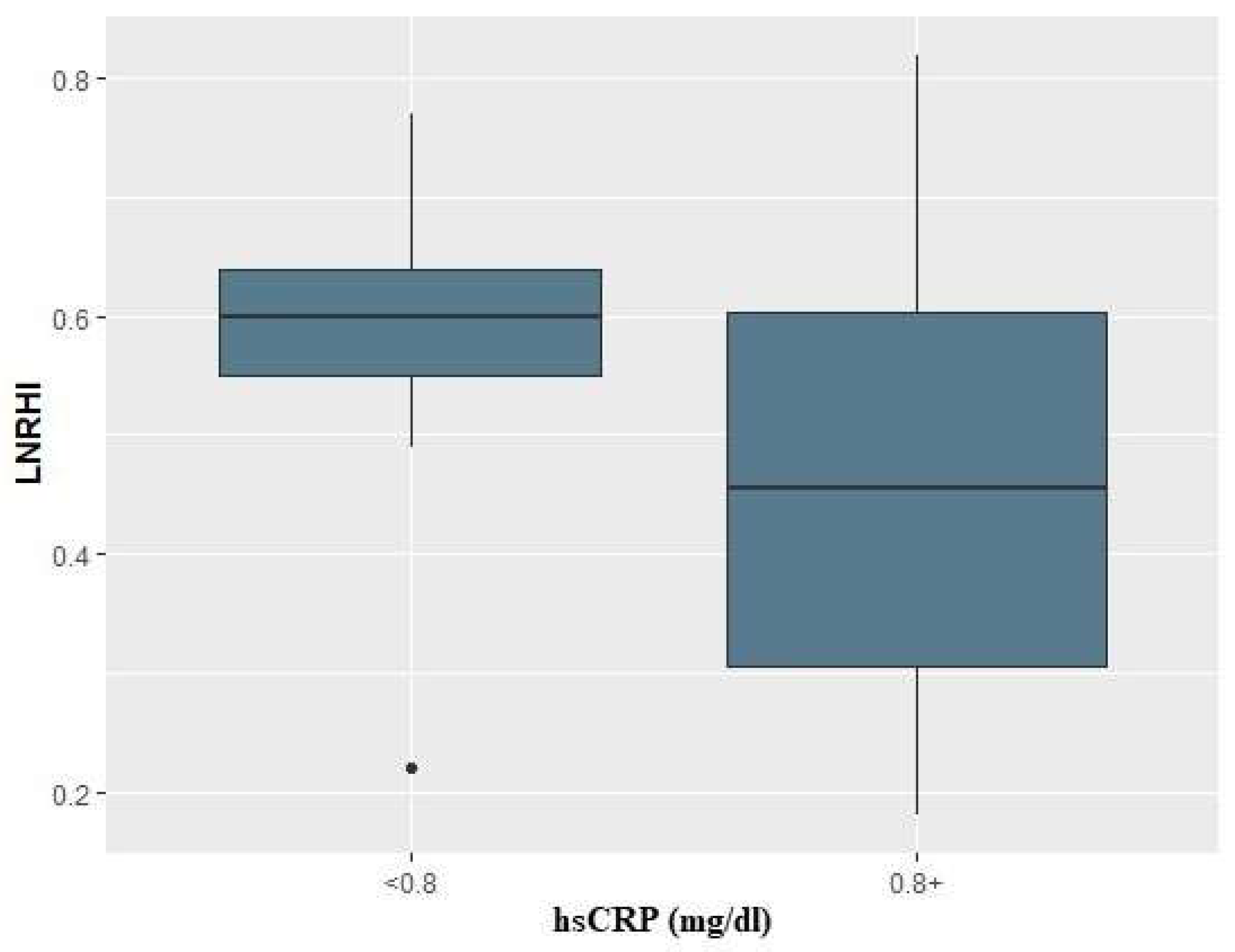
There is increasing evidence that COVID-19 induces endothelial dysfunction that may precede thrombotic and cardiovascular complications. The aim of this study is to evaluate the endothelial function using peripheral arterial tonometry (EndoPAT). The primary endpoint is the hyperemic vascular response index (LnRHI) at two months post-discharge. Secondary endpoints include the LnRHI during hospitalization and at six months follow-up, the proportion of patients with endothelial dysfunction (LnRHI≤0.51), and the incidence of thrombotic events, cardiovascular complications, and mortality during the follow-up period. The study included 23 COVID-19 patients and 22 healthy matched controls. The patients exhibited a significant reduction in the LnRHI at two months post-discharge compared to the controls (median=0.55 [IQR: 0.49-0.68] vs median=0.70 [IQR: 0.62-0.83]; p=0.012). The difference in the LnRHI between patients and controls was evident from hospitalization and persisted at two and six months without significant temporal changes. The proportion of COVID-19 patients with endothelial dysfunction (LnRHI≤0.51) was 61% during hospitalization, and 55% at six months. There was no significant difference in thrombotic or cardiovascular events, nor in mortality. This study demonstrates that COVID-19 adversely affects endothelial function, as evidenced by a reduction in the hyperemic vascular response index, and the endothelial dysfunction may also persist.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Statistical Analysis
3. Results
3.1. Baseline Characteristics
3.2. Primary Endpoint and Secondary Outcomes
4. Discussion
4.1. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhou P, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 2020;579:270–273.
- Evans, P.C.; Rainger, G.E.; Mason, J.C.; Guzik, T.J.; Osto, E.; Stamataki, Z.; Neil, D.; Hoefer, I.E.; Fragiadaki, M.; Waltenberger, J.; et al. Endothelial dysfunction in COVID-19: A position paper of the ESC Working Group for Atherosclerosis and Vascular Biology, and the ESC Council of Basic Cardiovascular Science. Cardiovasc. Res. 2020, 116, 2177–2184. [Google Scholar] [CrossRef] [PubMed]
- Maria Chiara Pelle; Isabella Zaffina; Stefania Lucà; Valentina Forte; Vincenzo Trapanese; Melania Melina; Federica Giofrè; Franco Arturi Endothelial Dysfunction in COVID-19: Potential Mechanisms and Possible Therapeutic Options Life 2022, 12(10), 1605. [CrossRef]
- Verdecchia P; Cavallini C; Spanevello A; et al. The pivotal link between ACE2 deficiency and SARS-CoV-2 infection. Eur J Intern Med. 2020;76:14–20.
- Hassan M. Otifi; and Balkur K. Adiga Endothelial Dysfunction in Covid-19 Infection Am J Med Sci 2022;363(4):281–287.
- Esper RJ; Nordaby RA; Vilari~no JO; et al. Endothelial dysfunction: a comprehensive appraisal. Cardiovasc Diabetol. 2006;5(1):1–18.
- Andrea L. Axtell; Fatemeh A. Gomari; John P. Cooke Assessing Endothelial Vasodilator Function with the Endo-PAT 2000 J Vis Exp.2010; (44): 2167. Published online 2010 Oct 15. [CrossRef]
- M. Moerland; A. J. Kales; L. Schrier; M. G. J. van Dongen; D. Bradnock; J. Burggraaf Evaluation of the EndoPAT as a Tool to Assess Endothelial Function International Journal of Vascular Medicine 2012 Article ID 904141. [CrossRef]
- Naomi M. Hamburg; Michelle J. Keyes; Martin G. Larson; Ramachandran S. Vasan; Renate Schnabel; Moira M. Pryde; Gary F. Mitchell; Jacob Sheffy; Joseph A. Vita; Emelia J. Benjamin Cross-Sectional Relations of Digital Vascular Function to Cardiovascular Risk Factors in the Framingham Heart Study Circulation 2008;117:2467–2474. [CrossRef]
- Andreas J. Flammer et al. The Assessment of Endothelial Function From Research Into Clinical Practice Circulation 2012;126:753–767 2012. [CrossRef]
- World Health Organization. COVID-19 Clinical management: living guidance, 25 January 2021. World Health Organization. WHO. 2021;(January).
- Knight SR; Ho A; Pius R; et al. Risk stratification of patients admitted to hospital with covid-19 using the ISARIC WHO Clinical Characterisation Protocol: Development and validation of the 4C Mortality Score. BMJ 2020;370. [CrossRef]
- Zsuzsanna Varga et al. Endothelial cell infection and endotheliitis in COVID-19 The Lancet Published:April 20, 2020. [CrossRef]
- Ambrosino P.; Calcaterra I.L.; Mosella M.; FormisanoR.; D’Anna S.E.; Bachetti T.; Marcuccio G.; Galloway B.; Mancini F.P.; Papa A.; et al. Endothelial Dysfunction in COVID-19: A Unifying Mechanism and a Potential Therapeutic Target. Biomedicines 2022, 10, 812. [CrossRef]
- Haffke M.; Freitag H.; Rudolf G.; et al. Endothelial dysfunction and altered endothelial biomarkers in patients with post-COVID-19 syndrome and chronic fatigue syndrome (ME/CFS). J Transl Med 20, 138 (2022).
- Mejia-Renteria H; Travieso A; Sagir A; Martínez-Gómez E; Carrascosa-Granada A; Toya T; Núñez-Gil IJ; Estrada V; Lerman A; Escaned J. In-vivo evidence of systemic endothelial vascular dysfunction in COVID-19. Int J Cardiol. 2021 Dec 15;345:153-155. Epub 2021 Oct 24. PMID: 34706286; PMCID: PMC8542397. [CrossRef]
- O Mohammad; D Connell; H Muhammad; J Chalmers; F Khan Focused longitudinal observational study to improve knowledge of COVID-19 (Follow COVID19). Heart 2024;110(Suppl 2):A1–A14. [CrossRef]
- Gouzi F.; Filippe A.; Pastre J.; et al. Recovery of Endothelium-dependent vascular relaxation impairment in convalescent COVID-19 patients: Insight from a pilot study. Respir Med Res. 2023;84. [CrossRef]
- Cimino, Giuliana; Enrico Vizzardi; Emiliano Calvi; Edoardo Pancaldi; Greta Pascariello; Nicola Bernardi; Angelica Cersosimo; Ludovica Amore; Riccardo M. Inciardi; Riccardo Raddino; and Marco Metra Endothelial Dysfunction in COVID-19 Patients Assessed With Endo-PAT2000 Monaldi Archives for Chest Disease 2022 92 (4). [CrossRef]
- Ambrosino P; Sanduzzi Zamparelli S; Mosella M; Formisano R; Molino A; Spedicato GA; Papa A; Motta A; Di Minno MND; Maniscalco M. Clinical assessment of endothelial function in convalescent COVID-19 patients: a meta-analysis with meta-regressions. Ann Med. 2022 Dec;54(1):3234-3249. PMID: 36382632; PMCID: PMC9673781. [CrossRef]
- E. Oikonomou et al. Endothelial dysfunction in acute and long standing COVID−19: A prospective cohort study Vascular Pharmacology, Volume 144,2022,106975,ISSN 1537-1891. [CrossRef]
- Ambrosino, P.; Calcaterra, I.; Molino, A.; Moretta, P.; Lupoli, R.; Spedicato, G.A.; Papa, A.; Motta, A.; Maniscalco, M.; Di Minno, M.N.D. Persistent Endothelial Dysfunction in Post-Acute COVID-19 Syndrome: A Case-Control Study. Biomedicines 2021, 9, 957. [Google Scholar] [CrossRef] [PubMed]
- Riou, M.; et al. Reduced Flow-Mediated Dilatation Is Not Related to COVID-19 Severity Three Months after Hospitalization for SARS-CoV-2 Infection. J. Clin. Med. 2021, 10, 1318. [Google Scholar] [CrossRef] [PubMed]
- H. Fogarty; L. Townsend; H. Morrin; et al. Persistent endotheliopathy in thepathogenesis of long COVID syndrome, J. Thromb. Haemost. 19 (10) (2021)2546–2553.
| COVID-19 N=23 |
Control N=22 |
*P-value | |
|---|---|---|---|
| Age (years) | 65 (57-74) | 63 (55-72) | 0.90 |
| Female | 7 (30%) | 7 (32%) | 1 |
| Height (cm) | 172 (164-178) | 178 (166-181) | 0.12 |
| Weight (kg) | 82 (70-101) | 87 (71-91) | 0.40 |
| Active smoker | 10 (43%) | 8 (36%) | 0.60 |
| Hypertension | 14 (61%) | 12 (55%) | 0.70 |
| Dyslipidemia | 11 (48%) | 11 (50%) | 0.90 |
| Diabetes mellitus | 8 (35%) | 2 (9.1%) | 0.071 |
| Atrial fibrillation | 2 (8.7%) | 0 (0%) | 0.50 |
| Chronic kidney disease | 1 (4.3%) | 2 (9.1%) | 0.60 |
| Chronic medical treatment | |||
| ASA | 2 (8.7%) | 3 (14%) | 0.70 |
| P2Y12i | 1 (4.3%) | 0 (0%) | >0.90 |
| NOAC | 4 (17%) | 0 (0%) | 0.11 |
| Warfarin | 1 (4.3%) | 0 (0%) | >0.90 |
| B-Blockers | 5 (22%) | 7 (41%) | 0.40 |
| ACEI/ARB | 10 (43.5%) | 9 (18%) | 0.65 |
| MRA | 1 (4.3%) | 0 (0%) | >0.90 |
| Diuretics | 6 (26%) | 2 (9.1%) | 0.20 |
| Statins | 11 (48%) | 11 (50%) | 0.90 |
| Antidiabetics | 8 (35%) | 2 (9.1%) | 0.071 |
| COVID-19 N=23 |
Control N=22 |
*P-value | |
|---|---|---|---|
| LnRHI, baseline | 0.56 (0.43-0.63) | 0.67 (0.57-0.80) | 0.007 |
| LnRHI, 2 months | 0.55 (0.49-0.68) | 0.70 (0.62-0.83) | 0.012 |
| LnRHI, 6 months | 0.59 (0.43-0.63) | 178 (166-181) | 0.007 |
| LnRHI≤0.51 at baseline | 14 (61) | 4 (18.2) | 0.074 |
| LnRHI≤0.51 at 2 months | 12 (55) | 3 (13.6) | 0.051 |
| LnRHI≤0.51 at 6 months | 12 (55) | 3 (13.6) | 0.051 |
| Thrombotic complications | 2 (8.7) | 0 | 0.48 |
| Cardiovascular events | 0 | 0 | - |
| Deaths | 0 | 0 | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




