Submitted:
14 October 2024
Posted:
15 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Surgical Procedure
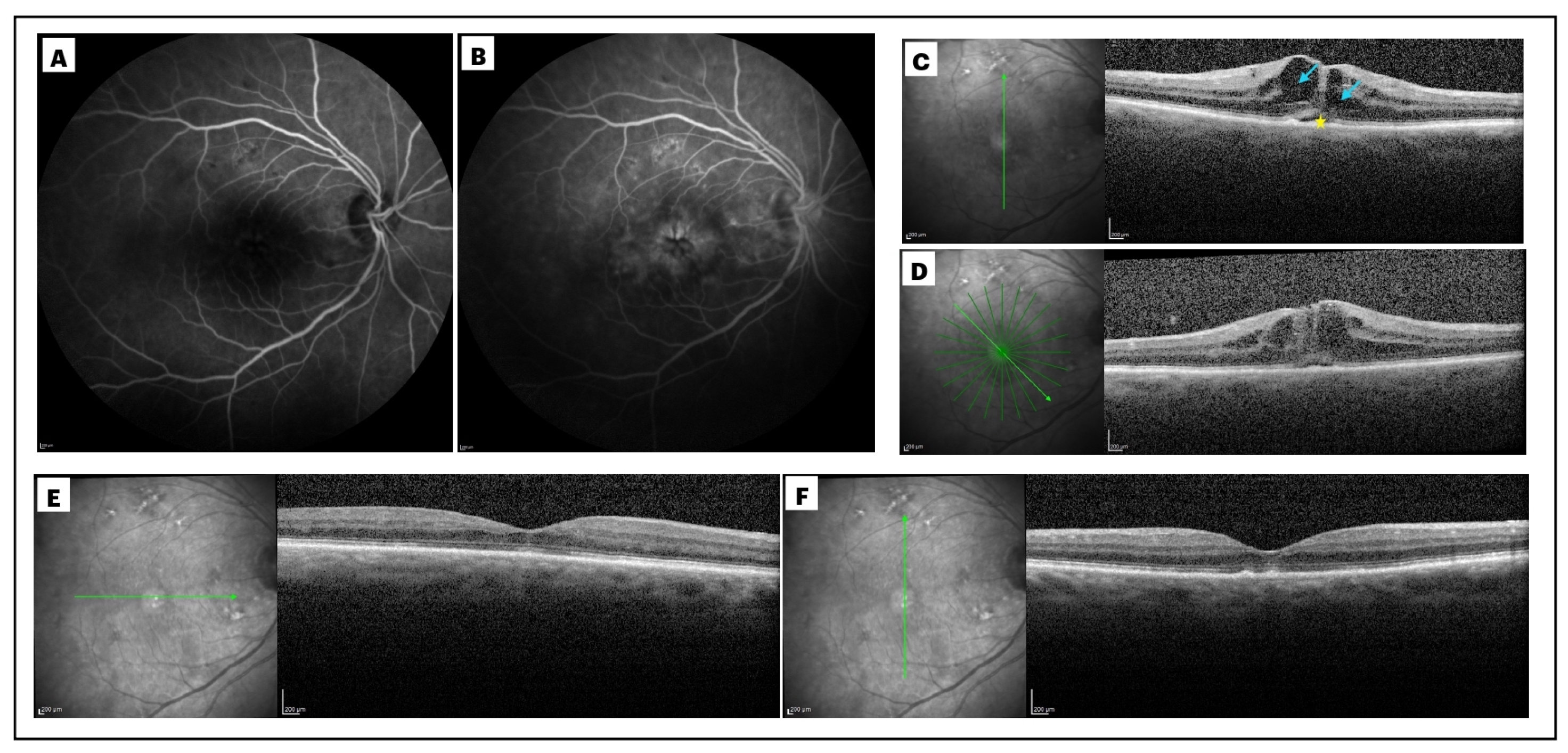
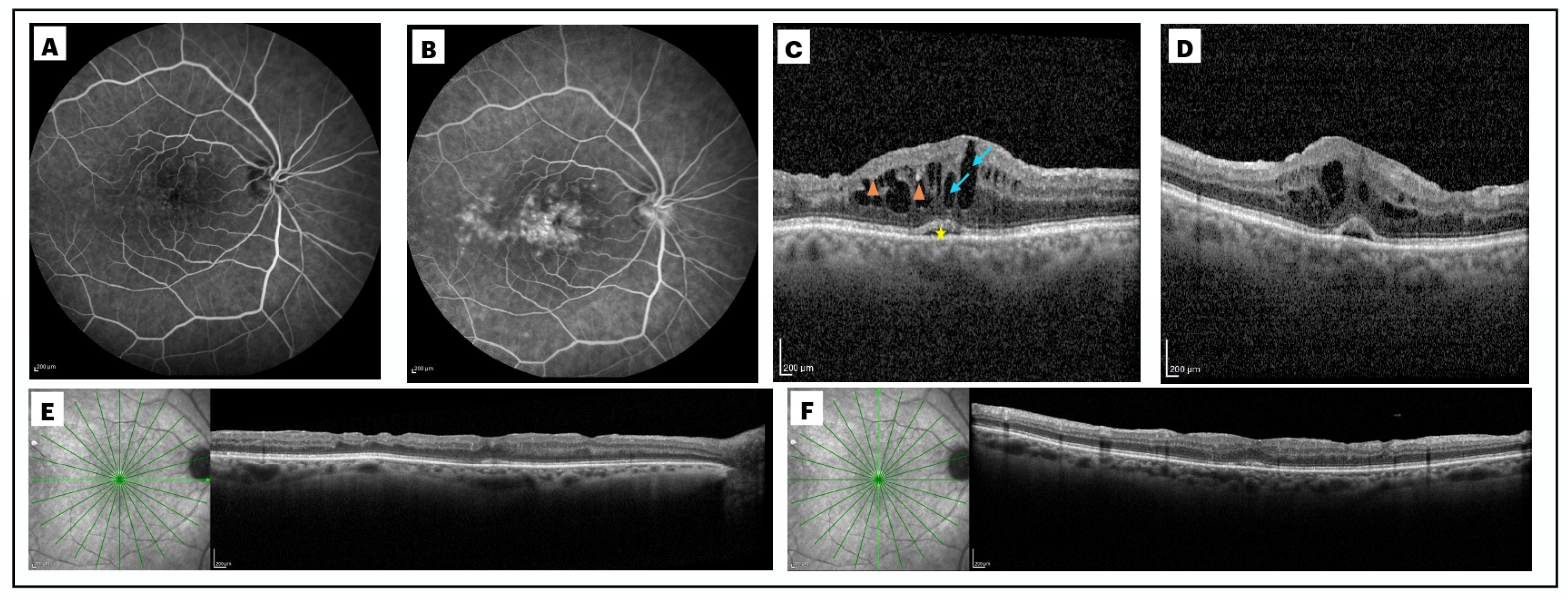
2.2. Definition and Diagnosis of Post-Surgical Macular Edema
2.3. Treatment of Post-Surgical Macular Edema
2.4. Analysis of the Biomarkers
2.5. Statistical Analysis
3. Results
3.1. Treatment Exposure
3.2. Analysis of the Correlations
3.3. Safety
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Romano, V.; Angi, M.; Scotti, F.; Del Grosso, R.; Romano, D.; Semeraro, F.; Vinciguerra, P.; Costagliola, C.; Romano, M.R. Inflammation and Macular Oedema after Pars Plana Vitrectomy. Mediators of Inflammation 2013, 2013, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Zur, D.; Loewenstein, A. Postsurgical Cystoid Macular Edema. In Developments in Ophthalmology; Coscas, G., Loewenstein, A., Cunha-Vaz, J., Soubrane, G., Eds.; S. Karger AG, 2017; Vol. 58, pp. 178–190 ISBN 978-3-318-06032-4.
- Iuliano, L.; Cisa Di Gresy, G.; Fogliato, G.; Corbelli, E.; Bandello, F.; Codenotti, M. Increased Risk of Postsurgical Macular Edema in High Stage Idiopathic Epiretinal Membranes. Eye and Vis 2021, 8, 29. [Google Scholar] [CrossRef]
- Bernabei, F.; Marcireau, I.; Frongia, F.; Azan, F.; Vagge, A.; Peiretti, E.; Guerrier, G.; Rothschild, P.-R. Risk Factors of Cystoid Macular Edema After Pars Plana Vitrectomy for Pseudophakic Retinal Detachment. Ophthalmol Ther 2023, 12, 1737–1745. [Google Scholar] [CrossRef] [PubMed]
- Bellocq, D.; Korobelnik, J.-F.; Burillon, C.; Voirin, N.; Dot, C.; Souied, E.; Conrath, J.; Milazzo, S.; Massin, P.; Baillif, S.; et al. Effectiveness and Safety of Dexamethasone Implants for Post-Surgical Macular Oedema Including Irvine-Gass Syndrome: The EPISODIC Study. Br J Ophthalmol 2015, 99, 979–983. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.J.; Johnston, R.L.; Buscombe, C.; Sallam, A.B.; Mohamed, Q.; Yang, Y.C. Risk Factors and Incidence of Macular Edema after Cataract Surgery. Ophthalmology 2016, 123, 316–323. [Google Scholar] [CrossRef]
- Yao, H.; Yang, Z.; Cheng, Y.; Shen, X. Macular Changes Following Cataract Surgery in Eyes with Early Diabetic Retinopathy: An OCT and OCT Angiography Study. Front. Med. 2023, 10, 1290599. [Google Scholar] [CrossRef] [PubMed]
- Parisi, G.; Fallico, M.; Avitabile, T.; Longo, A.; Ortisi, E.; Russo, A.; Petrillo, F.; Maugeri, A.; Barchitta, M.; Bonfiglio, V.; et al. Intravitreal Dexamethasone Implant for Postoperative Macular Oedema Secondary to Vitrectomy for Epiretinal Membrane and Retinal Detachment: A Systematic Review and Meta-Analysis. Journal of Ophthalmology 2021, 2021, 1–11. [Google Scholar] [CrossRef]
- Chatziralli, I.; Dimitriou, E.; Theodossiadis, G.; Chatzirallis, A.; Kazantzis, D.; Theodossiadis, P. Treatment of Macular Edema after Pars Plana Vitrectomy for Idiopathic Epiretinal Membrane Using Intravitreal Dexamethasone Implant: Long-Term Outcomes. Ophthalmologica 2019, 242, 16–21. [Google Scholar] [CrossRef]
- Freissinger, S.; Vounotrypidis, E.; Wolf, A.; Kortuem, K.U.; Shajari, M.; Sakkias, F.; Herold, T.; Priglinger, S.G.; Mayer, W.J. Evaluation of Functional Outcomes and OCT-Biomarkers after Intravitreal Dexamethasone Implant for Postoperative Cystoid Macular Edema in Vitrectomized Eyes. Journal of Ophthalmology 2020, 2020, 1–9. [Google Scholar] [CrossRef]
- Pignatelli, F.; Nacucchi, A.; Niro, A.; Gigliola, S.; Passidomo, F.; Donghia, R.; Addabbo, G. Intravitreal Dexamethasone Implant at the Time of Silicone Oil Removal to Treat Persistent Macular Edema after Rhegmatogenous Retinal Detachment Repair. JCM 2023, 12, 1697. [Google Scholar] [CrossRef]
- Markan, A.; Agarwal, A.; Arora, A.; Bazgain, K.; Rana, V.; Gupta, V. Novel Imaging Biomarkers in Diabetic Retinopathy and Diabetic Macular Edema. Ophthalmol Eye Dis 2020, 12, 251584142095051. [Google Scholar] [CrossRef] [PubMed]
- Costanzo, E.; Giannini, D.; De Geronimo, D.; Fragiotta, S.; Varano, M.; Parravano, M. Prognostic Imaging Biomarkers in Diabetic Macular Edema Eyes Treated with Intravitreal Dexamethasone Implant. JCM 2023, 12, 1303. [Google Scholar] [CrossRef]
- Visioli, G.; Alisi, L.; Mastrogiuseppe, E.; Albanese, G.M.; Romano, E.; Iannetti, L.; Armentano, M.; Giovannetti, F.; Gharbiya, M. OCT Biomarkers as Predictors of Visual Improvement in Diabetic Macular Edema Eyes Receiving Dexamethasone Implants. Int J Retin Vitr 2023, 9, 35. [Google Scholar] [CrossRef]
- Metrangolo, C.; Donati, S.; Mazzola, M.; Fontanel, L.; Messina, W.; D’alterio, G.; Rubino, M.; Radice, P.; Premi, E.; Azzolini, C. OCT Biomarkers in Neovascular Age-Related Macular Degeneration: A Narrative Review. Journal of Ophthalmology 2021, 2021, 1–16. [Google Scholar] [CrossRef]
- Castro-Navarro, V.; Monferrer-Adsuara, C.; Navarro-Palop, C.; Montero-Hernández, J.; Cervera-Taulet, E. Optical Coherence Tomography Biomarkers in Patients with Macular Edema Secondary to Retinal Vein Occlusion Treated with Dexamethasone Implant. BMC Ophthalmol 2022, 22, 191. [Google Scholar] [CrossRef] [PubMed]
- Guigou, S.; Pommier, S.; Meyer, F.; Hajjar, C.; Merite, P.Y.; Parrat, E.; Rouhette, H.; Rebollo, O.; Matonti, F. Efficacy and Safety of Intravitreal Dexamethasone Implant in Patients with Diabetic Macular Edema. Ophthalmologica 2015, 233, 169–175. [Google Scholar] [CrossRef]
- Leisser, C.; Hirnschall, N.; Findl, O. Effect of Phacoemulsification on Outcomes after Vitrectomy with Membrane Peeling Regarding New Intraretinal Cystoid Changes and Transient Macular Edema. Ophthalmologica 2021, 244, 150–158. [Google Scholar] [CrossRef] [PubMed]
- Park, S.W.; Kim, H.K.; Zaidi, M.H.; Byon, I.S.; Lee, J.E.; Nguyen, Q.D. Cystoid Macular Edema after Vitrectomy and after Phacovitrectomy for Epiretinal Membrane. Canadian Journal of Ophthalmology 2024, 59, e596–e602. [Google Scholar] [CrossRef]
- Leisser, C.; Hirnschall, N.; Hackl, C.; Döller, B.; Varsits, R.; Ullrich, M.; Kefer, K.; Karl, R.; Findl, O. Risk Factors for Postoperative Intraretinal Cystoid Changes after Peeling of Idiopathic Epiretinal Membranes among Patients Randomized for Balanced Salt Solution and Air-tamponade. Acta Ophthalmologica 2018, 96. [Google Scholar] [CrossRef]
- Frisina, R.; Pinackatt, S.J.; Sartore, M.; Monfardini, A.; Baldi, A.; Cesana, B.M.; Semeraro, F.; Bratu, A.; Parolini, B. Cystoid Macular Edema after Pars Plana Vitrectomy for Idiopathic Epiretinal Membrane. Graefes Arch Clin Exp Ophthalmol 2015, 253, 47–56. [Google Scholar] [CrossRef]
- Kontou, E.P.; Karakosta, C.; Kounas, K.; Iatropoulos, I.; Tsinopoulos, I.; Kozombolis, V.; Stavrakas, P. Macular Edema Following Silicone Oil Tamponade for Retinal Detachment: A Literature Review. Cureus 2023. [Google Scholar] [CrossRef] [PubMed]
- Merad, M.; Vérité, F.; Baudin, F.; Ghezala, I.B.; Meillon, C.; Bron, A.M.; Arnould, L.; Eid, P.; Creuzot-Garcher, C.; Gabrielle, P.-H. Cystoid Macular Edema after Rhegmatogenous Retinal Detachment Repair with Pars Plana Vitrectomy: Rate, Risk Factors, and Outcomes. JCM 2022, 11, 4914. [Google Scholar] [CrossRef]
- Pole, C.; Chehaibou, I.; Govetto, A.; Garrity, S.; Schwartz, S.D.; Hubschman, J.-P. Macular Edema after Rhegmatogenous Retinal Detachment Repair: Risk Factors, OCT Analysis, and Treatment Responses. Int J Retin Vitr 2021, 7, 9. [Google Scholar] [CrossRef]
- Govetto, A.; Su, D.; Farajzadeh, M.; Megerdichian, A.; Platner, E.; Ducournau, Y.; Virgili, G.; Hubschman, J.P. Microcystoid Macular Changes in Association With Idiopathic Epiretinal Membranes in Eyes With and Without Glaucoma: Clinical Insights. American Journal of Ophthalmology 2017, 181, 156–165. [Google Scholar] [CrossRef]
- Cicinelli, M.V.; Post, M.; Brambati, M.; Rabiolo, A.; Pignatelli, F.; Szaflik, J.P.; Bandello, F.; Coppola, M. ASSOCIATED FACTORS AND SURGICAL OUTCOMES OF MICROCYSTOID MACULAR EDEMA AND CONE BOUQUET ABNORMALITIES IN EYES WITH EPIRETINAL MEMBRANE. Retina 2022, 42, 1455–1464. [Google Scholar] [CrossRef]
- Burggraaff, M.C.; Trieu, J.; De Vries-Knoppert, W.A.E.J.; Balk, L.; Petzold, A. The Clinical Spectrum of Microcystic Macular Edema. Invest. Ophthalmol. Vis. Sci. 2014, 55, 952. [Google Scholar] [CrossRef] [PubMed]
- Benhamou, N.; Massin, P.; Haouchine, B.; Audren, F.; Tadayoni, R.; Gaudric, A. Intravitreal Triamcinolone for Refractory Pseudophakic Macular Edema. American Journal of Ophthalmology 2003, 135, 246–249. [Google Scholar] [CrossRef] [PubMed]
- Williams, G.A.; Haller, J.A.; Kuppermann, B.D.; Blumenkranz, M.S.; Weinberg, D.V.; Chou, C.; Whitcup, S.M. Dexamethasone Posterior-Segment Drug Delivery System in the Treatment of Macular Edema Resulting from Uveitis or Irvine-Gass Syndrome. American Journal of Ophthalmology 2009, 147, 1048–1054.e2. [Google Scholar] [CrossRef] [PubMed]
- Barone, A.; Russo, V.; Prascina, F.; Noci, N.D. SHORT-TERM SAFETY AND EFFICACY OF INTRAVITREAL BEVACIZUMAB FOR PSEUDOPHAKIC CYSTOID MACULAR EDEMA. Retina 2009, 29, 33–37. [Google Scholar] [CrossRef]
- Fallico, M.; Maugeri, A.; Romano, G.L.; Bucolo, C.; Longo, A.; Bonfiglio, V.; Russo, A.; Avitabile, T.; Barchitta, M.; Agodi, A.; et al. Epiretinal Membrane Vitrectomy With and Without Intraoperative Intravitreal Dexamethasone Implant: A Systematic Review With Meta-Analysis. Front. Pharmacol. 2021, 12, 635101. [Google Scholar] [CrossRef]
- Pignatelli, F.; Niro, A.; Fallico, M.; Passidomo, F.; Gigliola, S.; Nacucchi, A.; Bonfiglio, V.; Reibaldi, M.; Addabbo, G.; Avitabile, T. Mid-Term Safety and Effectiveness of Macular Peeling One Month after Intravitreal Dexamethasone Implant for Tractional Diabetic Macular Edema. Sci Rep 2023, 13, 5990. [Google Scholar] [CrossRef] [PubMed]
- Zhu, R.; Xiao, S.; Zhang, W.; Li, J.; Yang, M.; Zhang, Y.; Gu, X.; Yang, L. Comparison of Hyperreflective Foci in Macular Edema Secondary to Multiple Etiologies with Spectral-Domain Optical Coherence Tomography: An Observational Study. BMC Ophthalmol 2022, 22, 352. [Google Scholar] [CrossRef]
- Zur, D.; Iglicki, M.; Busch, C.; Invernizzi, A.; Mariussi, M.; Loewenstein, A.; Busch, C.; Cebeci, Z.; Chhablani, J.K.; Chaikitmongkol, V.; et al. OCT Biomarkers as Functional Outcome Predictors in Diabetic Macular Edema Treated with Dexamethasone Implant. Ophthalmology 2018, 125, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Vujosevic, S.; Torresin, T.; Bini, S.; Convento, E.; Pilotto, E.; Parrozzani, R.; Midena, E. Imaging Retinal Inflammatory Biomarkers after Intravitreal Steroid and Anti- VEGF Treatment in Diabetic Macular Oedema. Acta Ophthalmologica 2017, 95, 464–471. [Google Scholar] [CrossRef] [PubMed]
- Hatano, M.; Higashijima, F.; Yoshimoto, T.; Ogata, T.; Ohta, M.; Kobayashi, Y.; Wakuta, M.; Yanai, R.; Kimura, K. Evaluation of Microaneurysms as Predictors of Therapeutic Response to Anti-VEGF Therapy in Patients with DME. PLoS ONE 2022, 17, e0277920. [Google Scholar] [CrossRef]
- Grigsby, J.G.; Cardona, S.M.; Pouw, C.E.; Muniz, A.; Mendiola, A.S.; Tsin, A.T.C.; Allen, D.M.; Cardona, A.E. The Role of Microglia in Diabetic Retinopathy. Journal of Ophthalmology 2014, 2014, 1–15. [Google Scholar] [CrossRef]
- Zeng, H. Microglial Activation in Human Diabetic Retinopathy. Arch Ophthalmol 2008, 126, 227. [Google Scholar] [CrossRef]
- Kim, K.T.; Kim, D.Y.; Chae, J.B. Association between Hyperreflective Foci on Spectral-Domain Optical Coherence Tomography and Early Recurrence of Diabetic Macular Edema after Intravitreal Dexamethasone Implantation. Journal of Ophthalmology 2019, 2019, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Das, R.; Spence, G.; Hogg, R.E.; Stevenson, M.; Chakravarthy, U. Disorganization of Inner Retina and Outer Retinal Morphology in Diabetic Macular Edema. JAMA Ophthalmol 2018, 136, 202. [Google Scholar] [CrossRef]
- Zur, D.; Iglicki, M.; Sala-Puigdollers, A.; Chhablani, J.; Lupidi, M.; Fraser-Bell, S.; Mendes, T.S.; Chaikitmongkol, V.; Cebeci, Z.; Dollberg, D.; et al. Disorganization of Retinal Inner Layers as a Biomarker in Patients with Diabetic Macular Oedema Treated with Dexamethasone Implant. Acta Ophthalmologica 2020, 98. [Google Scholar] [CrossRef]
- Rajesh, B.; Zarranz-Ventura, J.; Fung, A.T.; Busch, C.; Sahoo, N.K.; Rodriguez-Valdes, P.J.; Sarao, V.; Mishra, S.K.; Saatci, A.O.; Udaondo Mirete, P.; et al. Safety of 6000 Intravitreal Dexamethasone Implants. Br J Ophthalmol 2020, 104, 39–46. [Google Scholar] [CrossRef] [PubMed]
| RRD | SO | ERM | P-value * | ||
|---|---|---|---|---|---|
| N° (%); patients / eyes | 36 (75) | 10 (20.8) | 2 (4.2) | ||
| Laterality: Right/Left | 16/20 | 6/4 | 1/1 | 0.29 | |
| Age, year (mean±SD) | 64.6±4.5 | 63.8±3.6 | 58.5±0.7 | 0.17 | |
| Gender, N° (%) | |||||
| Male | 25 (69.4) | 6 (60) | 1 (50) | ||
| Female | 11 (30.6) | 4 (40) | 1 (50) | ||
| Type 2 Diabetes (controlled with therapy); N° (%) | 3 (8%) | 1 (10%) | 0 (0%) | 0.45 | |
| Time between cataract surgery and PPV; years, mean ± SD (range) | 4.1± 2.48 (1-11) | 3 ± 1.5 (2-6) | 2 ± 0.7 (1-2.5) | 0.20 | |
| Macula attached at the time of surgery (RRD group); n° (%) | 31 (86.1%) | - | - | ||
| PVR present: n° (%) | 14 (38.8%) | - | - | ||
| Mild VH associated to RRD: n° (%) | 2 (5.5%) | - | - | ||
| Endotamponade agent used (RRD group) | |||||
| Gas (SF6 24%; C3F8 14%) | 22 (64.7%) | ||||
| Silicone Oil 1000 cst | 10 (29.4%) | ||||
| Densiron | 2 (5.8%) | ||||
| BCVA Baseline* (logMAR) | 1.44±0.99 | |
| BCVA 1 Month (logMAR) | 0.56±0.40 | <0.001 |
| BCVA 3 Months (logMAR) | 0.44±0.48 | <0.001 |
| BCVA 6 Months (logMAR) | 0.37±0.35 | <0.001 |
| BCVA 9 Months (logMAR) | 0.38±0.39 | <0.001 |
| BCVA 12 Months (logMAR) | 0.32±0.37 | <0.001 |
| CMT 1 month* (µm) | 347±123.5 | |
| CMT 3 months (µm) | 304±93.6 | 0.002 |
| CMT 6 months (µm) | 289±79.7 | 0.002 |
| CMT 9 months (µm) | 322±104.5 | 0.048 |
| CMT 12 months (µm) | 290±80.4 | 0.003 |
| IRC | DRIL | DROL | HRF | SRF | |
| RRD n(%) | 20 (41.7) | 7 (14.6) | 8 (16.7) | 7 (14.6) | 7 (14.6) |
| SO n(%) | 10 (20.8) | 4 (8.3) | 2 (4.2) | 2 (4.2) | 4 (8.3) |
| ERM n(%) | 2 (4.2) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (2.1) |
| p | 0.02 | 0.29 | 0.75 | 0.78 | 0.30 |
| IRC | DRIL | DROL | HRF | SRF | |
| RRD n(%) | 20 (41.7) | 7 (14.6) | 8 (16.7) | 7 (14.6) | 7 (14.6) |
| SO n(%) | 10 (20.8) | 4 (8.3) | 2 (4.2) | 2 (4.2) | 4 (8.3) |
| ERM n(%) | 2 (4.2) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (2.1) |
| p | 0.02 | 0.29 | 0.75 | 0.78 | 0.30 |
| RRD | SO | ERM | p | |
| DEX-i at 3 months, n (%) | 10 (20.8) | 5 (10.4) | 1 (2.1) | 0.37 |
| DEX-i at 6 months, n (%) | 3 (6.4) | 2 (4.3) | 0 (0.0) | 0.52 |
| DEX-i at 9 months, n (%) | 11 (22.9) | 5 (10.4) | 0 (0.0) | 0.31 |
| DEX-i at 12 months, n (%) | 4 (8.3) | 0 (0.0) | 0 (0.0) | 0.49 |
| CMT 1 month | CMT 3 Months | CMT 6 Months | CMT 9 Months | CMT 12 Months | ||
|
CMT 1 month |
Spearman’s Rho p |
— — |
||||
| CMT 3 Months | Spearman’s Rho | 0.592 *** | — | |||
| p | <0.001 | — | ||||
| CMT 6 Months | Spearman’s Rho | 0.174 | 0.274 | — | ||
| p | 0.237 | 0.060 | — | |||
| CMT 9 Months | Spearman’s Rho | 0.145 | 0.541 *** | 0.578 *** | — | |
| p | 0.326 | <0.001 | <0.001 | — | ||
|
CMT 12 Months |
Spearman’s Rho p |
0.117 0.428 |
0.573 *** <0.001 |
0.454 ** 0.001 |
0.662 *** <0.001 |
— — |
| (A) DEX-i at 3 months as dependent variable | |||||
| Parameters | β | SE (β) | p | OR | |
| Age | 0.10 | 0.07 | 0.17 | 1.11 | |
| Gender (male) | -3.67e-16 | 0.66 | 0.99 | 0.99 | |
| Disease | |||||
| SO—RRD | 0.95 | 0.73 | 0.19 | 2.6 | |
| ERM—RRD | 0.95 | 1.46 | 0.51 | 2.6 | |
| CMT 1 month | 0.01 | 0.003 | 0.005 | 1.01 | |
| IRC (No) | 1.69 | 0.83 | 0.04 | 5.44 | |
| DRIL (No) | 2.27 | 0.78 | 0.004 | 9.66 | |
| HRF (No) | 1.76 | 0.79 | 0.02 | 5.80 | |
| DROL (No) | 1.44 | 0.74 | 0.05 | 4.20 | |
| SRF (No) | 0.95 | 0.68 | 0.16 | 2.60 | |
| (B) DEX-i at 3 months as dependent variable | |||||
| Parameters | β | SE (β) | p | OR | |
| Age | 0.09 | 0.10 | 0.35 | 1.10 | |
| Gender (male) | -0.18 | 0.87 | 0.83 | 0.83 | |
| Disease | |||||
| SO—RRD | 0.49 | 0.98 | 0.61 | 1.64 | |
| ERM—RRD | 0.77 | 2.44 | 0.75 | 2.17 | |
| CMT 1 month | 0.008 | 0.005 | 0.09 | 1.0 | |
| IRC (No) | -0.33 | 1.26 | 0.78 | 0.71 | |
| DRIL (No) | 1.62 | 0.92 | 0.08 | 5.08 | |
| HRF (No) | 1.33 | 1.03 | 0.19 | 3.80 | |
| (A) DEX-i at 6 months as dependent variable | |||||
| Parameters | β | SE (β) | p | OR | |
| Age | 0.04 | 0.10 | 0.69 | 1.03 | |
| Gender (male) | 0.39 | 0.97 | 0.68 | 1.5 | |
| Disease | |||||
| SO—RRD | 0.98 | 0.99 | 0.32 | 2.66 | |
| ERM—RRD | -15.2 | 2797.4 | 0.99 | 2.51è-7 | |
| IRC (No) | -0.28 | 0.96 | 0.76 | 0.75 | |
| DRIL (No) | -17.7 | 3242.4 | 0.99 | 2.04e-8 | |
| HRF (No) | 1.23 | 1.00 | 0.21 | 3.42 | |
| DROL (No) | 0.06 | 1.18 | 0.96 | 1.06 | |
| SRF (No) | -0.22 | 1.17 | 0.84 | 0.80 | |
| (A) DEX-i at 9 months as dependent variable | |||||
| Parameters | β | SE (β) | p | OR | |
| Age | 0.02 | 0.07 | 0.74 | 1.02 | |
| Gender (male) | 0.42 | 0.64 | 0.51 | 1.5 | |
| Disease | |||||
| SO—RRD | 0.87 | 0.73 | 0.23 | 2.27 | |
| ERM—RRD | -15.7 | 1696.7 | 0.99 | 1.45e-7 | |
| CMT 3 months | 0.006 | 0.003 | 0.07 | 1.00 | |
| CMT 6 months | 0.007 | 0.004 | 0.11 | 1.00 | |
| IRC (No) | 0.14 | 0.65 | 0.82 | 1.15 | |
| DRIL (No) | 0.67 | 0.70 | 0.33 | 1.97 | |
| HRF (No) | 1.76 | 0.79 | 0.02 | 5.80 | |
| DROL (No) | 2.02 | 0.78 | 0.01 | 7.51 | |
| SRF (No) | 0.95 | 0.68 | 0.16 | 2.60 | |
| (B) DEX-i at 9 months as dependent variable | |||||
| Parameters | β | SE (β) | p | OR | |
| Age | -0.02 | 0.09 | 0.85 | 0.97 | |
| Gender (male) | 0.01 | 0.83 | 0.99 | 1.01 | |
| Disease | |||||
| SO—RRD | 1.01 | 0.84 | 0.22 | 2.75 | |
| ERM—RRD | -14.9 | 1696.7 | 0.99 | 3.23e-7 | |
| HRF (No) | 1.71 | 0.90 | 0.04 | 5.53 | |
| DROL (No) | 1.96 | 0.87 | 0.02 | 7.11 | |
| (A) DEX-i at 12 months as dependent variable | |||||
| Parameters | β | SE (β) | p | OR | |
| Age | -0.20 | 0.15 | 0.18 | 0.81 | |
| Gender (male) | -0.33 | 1.19 | 0.77 | 0.71 | |
| Disease | |||||
| SO—RRD | -17.5 | 3400.7 | 0.99 | 2.39e-8 | |
| ERM—RRD | -17.5 | 7604.2 | 0.99 | 2.39e-8 | |
| CMT 3 months | -0.01 | 0.01 | 0.21 | 0.98 | |
| CMT 6 months | 0.007 | 0.004 | 0.10 | 1.00 | |
| CMT 9 months | 0.005 | 0.004 | 0.20 | 1.00 | |
| IRC (No) | -1.97 | 1.20 | 0.10 | 0.14 | |
| DRIL (No) | -17.4 | 3242.4 | 0.99 | 2.62e-8 | |
| HRF (No) | -17.4 | 3584.6 | 0.99 | 2.78e-8 | |
| DROL (No) | 0.26 | 1.21 | 0.83 | 1.29 | |
| SRF (No) | -17.4 | 3104.4 | 0.99 | 2.54e-8 | |
| IOP Baseline (mmHg) | 15.6±3.9 | |
| IOP 1 Month (mmHg) | 15.6±2.8 | 0.90 |
| IOP 3 Months (mmHg) | 15.0±2.4 | 0.32 |
| IOP 6 Months (mmHg) | 15.9±3.1 | 0.31 |
| IOP 9 Months (mmHg) | 15.8±2.1 | 0.71 |
| IOP 12 Months (mmHg) | 15.6±1.9 | 0.80 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




