Submitted:
13 October 2024
Posted:
14 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
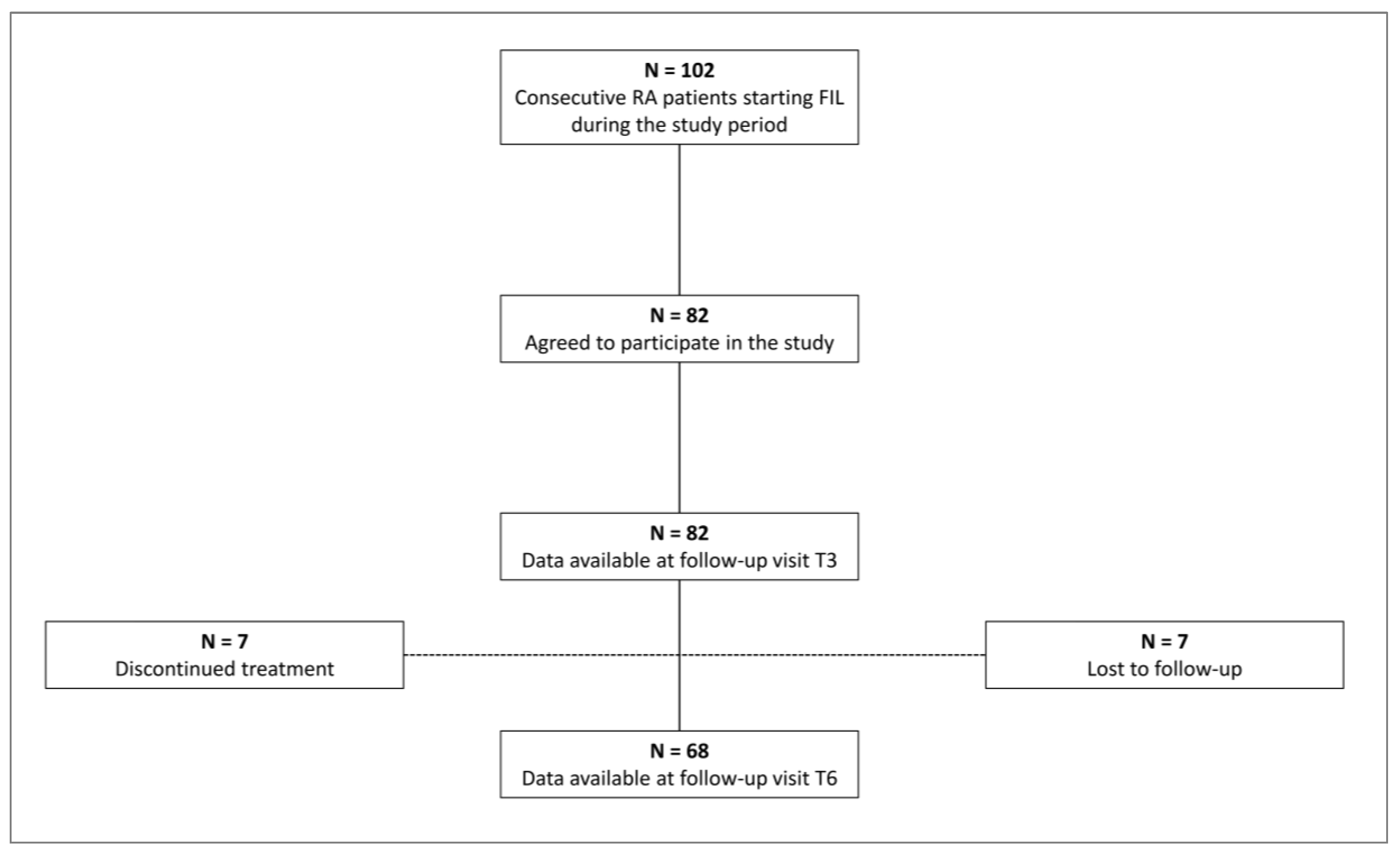
Study Design
Participants
Data Collection
Ethical Approval
Data Analysis
3. Results
Baseline Characteristics
Effectiveness of Filgotinib in Real Life
4. Discussion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Tanaka Y, Luo Y, O’Shea JJ, et al. Janus kinase-targeting therapies in rheumatology: a mechanisms-based approach. Nat Rev Rheumatol. 2022;18:133–45. [CrossRef]
- Hu X, Li J, Fu M, et al. The JAK/STAT signaling pathway: from bench to clinic. Signal Transduct Target Ther. 2021;6:402. [CrossRef]
- Malemud CJ. The role of the JAK/STAT signal pathway in rheumatoid arthritis. Ther Adv Musculoskelet Dis. 2018;10:117–27.
- Leonard WJ. Role of Jak kinases and STATs in cytokine signal transduction. Int J Hematol. 2001;73:271–7. [CrossRef]
- Namour F, Anderson K, Nelson C, et al. Filgotinib: A Clinical Pharmacology Review. Clin Pharmacokinet. 2022;61:819–32. [CrossRef]
- Genovese MC, Kalunian K, Gottenberg J-E, et al. Effect of Filgotinib vs Placebo on Clinical Response in Patients With Moderate to Severe Rheumatoid Arthritis Refractory to Disease-Modifying Antirheumatic Drug Therapy: The FINCH 2 Randomized Clinical Trial. JAMA. 2019;322:315–25.
- Westhovens R, Taylor PC, Alten R, et al. Filgotinib (GLPG0634/GS-6034), an oral JAK1 selective inhibitor, is effective in combination with methotrexate (MTX) in patients with active rheumatoid arthritis and insufficient response to MTX: results from a randomised, dose-finding study (DARWIN 1). Ann Rheum Dis. 2017;76:998–1008.
- Kavanaugh A, Kremer J, Ponce L, et al. Filgotinib (GLPG0634/GS-6034), an oral selective JAK1 inhibitor, is effective as monotherapy in patients with active rheumatoid arthritis: results from a randomised, dose- finding study (DARWIN 2). Ann Rheum Dis. 2017;76:1009–19.
- Westhovens R, Rigby WFC, van der Heijde D, et al. Filgotinib in combination with methotrexate or as monotherapy versus methotrexate monotherapy in patients with active rheumatoid arthritis and limited or no prior exposure to methotrexate: the phase 3, randomised controlled FINCH 3 trial. Ann Rheum Dis. 2021;80:727–38.
- Combe B, Kivitz A, Tanaka Y, et al. Filgotinib versus placebo or adalimumab in patients with rheumatoid arthritis and inadequate response to methotrexate: a phase III randomised clinical trial. Ann Rheum Dis. 2021;80:848–58.
- Ytterberg SR, Bhatt DL, Mikuls TR, et al. Cardiovascular and Cancer Risk with Tofacitinib in Rheumatoid Arthritis. N Engl J Med. 2022;386:316–26.
- Kremer JM, Bingham CO, Cappelli LC, et al. Postapproval Comparative Safety Study of Tofacitinib and Biological Disease-Modifying Antirheumatic Drugs: 5-Year Results from a United States-Based Rheumatoid Arthritis Registry. ACR Open Rheumatol. 2021;3:173–84. [CrossRef]
- Khosrow-Khavar F, Kim SC, Lee H, et al. Tofacitinib and risk of cardiovascular outcomes: results from the Safety of TofAcitinib in Routine care patients with Rheumatoid Arthritis (STAR-RA) study. Ann Rheum Dis. 2022;81:798–804. [CrossRef]
- Khosrow-Khavar F, Desai RJ, Lee H, et al. Tofacitinib and Risk of Malignancy: Results From the Safety of Tofacitinib in Routine Care Patients With Rheumatoid Arthritis (STAR-RA) Study. Arthritis Rheumatol. 2022;74:1648–59. [CrossRef]
- Palleria C, Iannone L, Leporini C, et al. Implementing a simple pharmacovigilance program to improve reporting of adverse events associated with biologic therapy in rheumatology: Preliminary results from the Calabria Biologics Pharmacovigilance Program (CBPP). PLoS One. 2018;13:e0205134.
- Fuchs HA, Brooks RH, Callahan LF, et al. A simplified twenty-eight-joint quantitative articular index in rheumatoid arthritis. Arthritis Rheum. 1989;32:531–7.
- Smolen JS, Breedveld FC, Schiff MH, et al. A simplified disease activity index for rheumatoid arthritis for use in clinical practice. Rheumatology (Oxford). 2003;42:244–57.
- Besancenot R, Roos-Weil D, Tonetti C, et al. JAK2 and MPL protein levels determine TPO-induced megakaryocyte proliferation vs differentiation. Blood. 2014;124:2104–15. [CrossRef]
- Sopjani M, Morina R, Uka V, et al. JAK2-mediated Intracellular Signaling. Curr Mol Med. 2021;21:417– 25. [CrossRef]
- Park SO, Wamsley HL, Bae K, et al. Conditional deletion of Jak2 reveals an essential role in hematopoiesis throughout mouse ontogeny: implications for Jak2 inhibition in humans. PLoS One. 2013;8:e59675. [CrossRef]
- Winthrop KL, Curtis JR, Lindsey S, et al. Herpes Zoster and Tofacitinib: Clinical Outcomes and the Risk of Concomitant Therapy. Arthritis Rheumatol. 2017;69:1960–8.
- Smolen JS, Genovese MC, Takeuchi T, et al. Safety Profile of Baricitinib in Patients with Active Rheumatoid Arthritis with over 2 Years Median Time in Treatment. J Rheumatol. 2019;46:7–18.
- Molander V, Bower H, Frisell T, et al. Risk of venous thromboembolism in rheumatoid arthritis, and its association with disease activity: a nationwide cohort study from Sweden. Ann Rheum Dis. 2021;80:169–75. [CrossRef]
- Chen C-P, Kung P-T, Chou W-Y, et al. Effect of introducing biologics to patients with rheumatoid arthritis on the risk of venous thromboembolism: a nationwide cohort study. Sci Rep. 2021;11:17009. [CrossRef]
- Sivaraman P, Cohen SB. Malignancy and Janus Kinase Inhibition. Rheum Dis Clin North Am. 2017;43:79– 93.
- Avina-Zubieta JA, Thomas J, Sadatsafavi M, et al. Risk of incident cardiovascular events in patients with rheumatoid arthritis: a meta-analysis of observational studies. Ann Rheum Dis. 2012;71:1524–9.
- De Cock D, Hyrich K. Malignancy and rheumatoid arthritis: Epidemiology, risk factors and management. Best Pract Res Clin Rheumatol. 2018;32:869–86. [CrossRef]
- Tan YY, Papez V, Chang WH, et al. Comparing clinical trial population representativeness to real-world populations: an external validity analysis encompassing 43 895 trials and 5 685 738 individuals across 989 unique drugs and 286 conditions in England. Lancet Healthy Longev. 2022;3:e674–89. [CrossRef]
- EMA. Measures to minimise risk of serious side effects with Janus kinase inhibitors for chronic inflammatory disorders. https://www.ema.europa.eu/en/medicines/human/referrals/janus-kinase- inhibitors-jaki.
- Benucci M, Bardelli M, Cazzato M, et al. ReLiFiRa (Real Life Filgotinib in Rheumatoid Arthritis): Retrospective Study of Efficacy and Safety in Common Clinical Practice. J Pers Med. 2023;13:1303. [CrossRef]
- Nagy G, Roodenrijs NM, Welsing PM, et al. EULAR definition of difficult-to-treat rheumatoid arthritis. Ann Rheum Dis. 2021;80:31–5.
| Age, years | 62 ± 13 |
| Female gender, n (%) | 63 (76.8) |
| Disease duration, years | 13 ± 9 |
| BMI category | |
| Underweight, n (%) | 4 (4.9) |
| Normal weight, n (%) | 25 (30.5) |
| Overweight, n (%) | 38 (46.3) |
| Obesity, n (%) | 15 (18.3) |
| Smoking | |
| Current smoker, n (%) | 12 (14.6) |
| Former smoker, n (%) | 7 (8.5) |
| History of ASCVD, n (%) | 4 (4.9) |
| Type 2 diabetes, n (%) | 12 (14.6) |
| High blood pressure, n (%) | 43 (52.4) |
| Dyslipidemia, n (%) | 28 (34.1) |
| RF positive, n (%) | 59 (72.0) |
| ACPA positive, n (%) | 50 (61.0) |
| bDMARDs failure, n (%) | 50 (61.0) |
| N° of previous bDMARDs, n [range] | 1 [1 – 6] |
| Received three or more bDMARDs, n (%) | 18 (22) |
| JAKi failure, n (%) | 11 (13.4) |
| Current treatment with steroids | |
| ≤ 5 mg/d prednisone-equivalent, n (%) | 34 (41.5) |
| > 5 mg/d prednisone-equivalent, n (%) | 10 (12.2) |
| FIL monotherapy, n (%) | 57 (69.5) |
| MTX combination therapy, n (%) | 24 (29.3) |
| T0 (n = 82) |
T3 (n = 82) |
T6 (n = 68) |
P value T0 vs T3 |
P value T0 vs T6 |
P value T3 vs T6 |
|
|---|---|---|---|---|---|---|
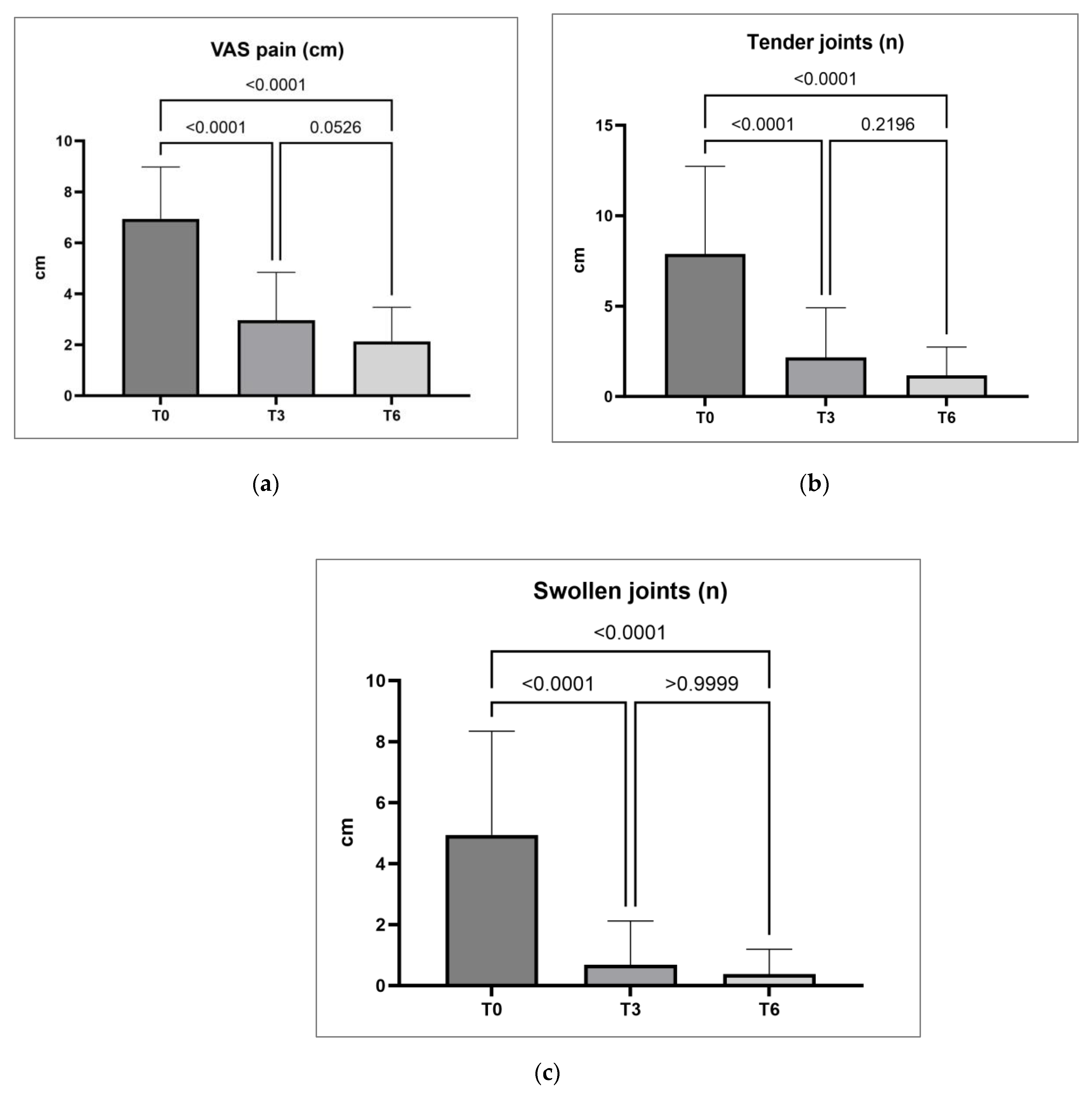
| VAS pain, mm | 7 ± 2 | 3 ± 2 | 2 ± 1 | < 0.0001 | < 0.0001 | 0.05 |
| Tender joints, n | 8 ± 5 | 2 ± 3 | 1 ± 2 | < 0.0001 | < 0.0001 | 0.22 |
| Swollen joints, n | 5 ± 3 | 1 ± 1 | 0 ± 1 | < 0.0001 | < 0.0001 | > 0.99 |
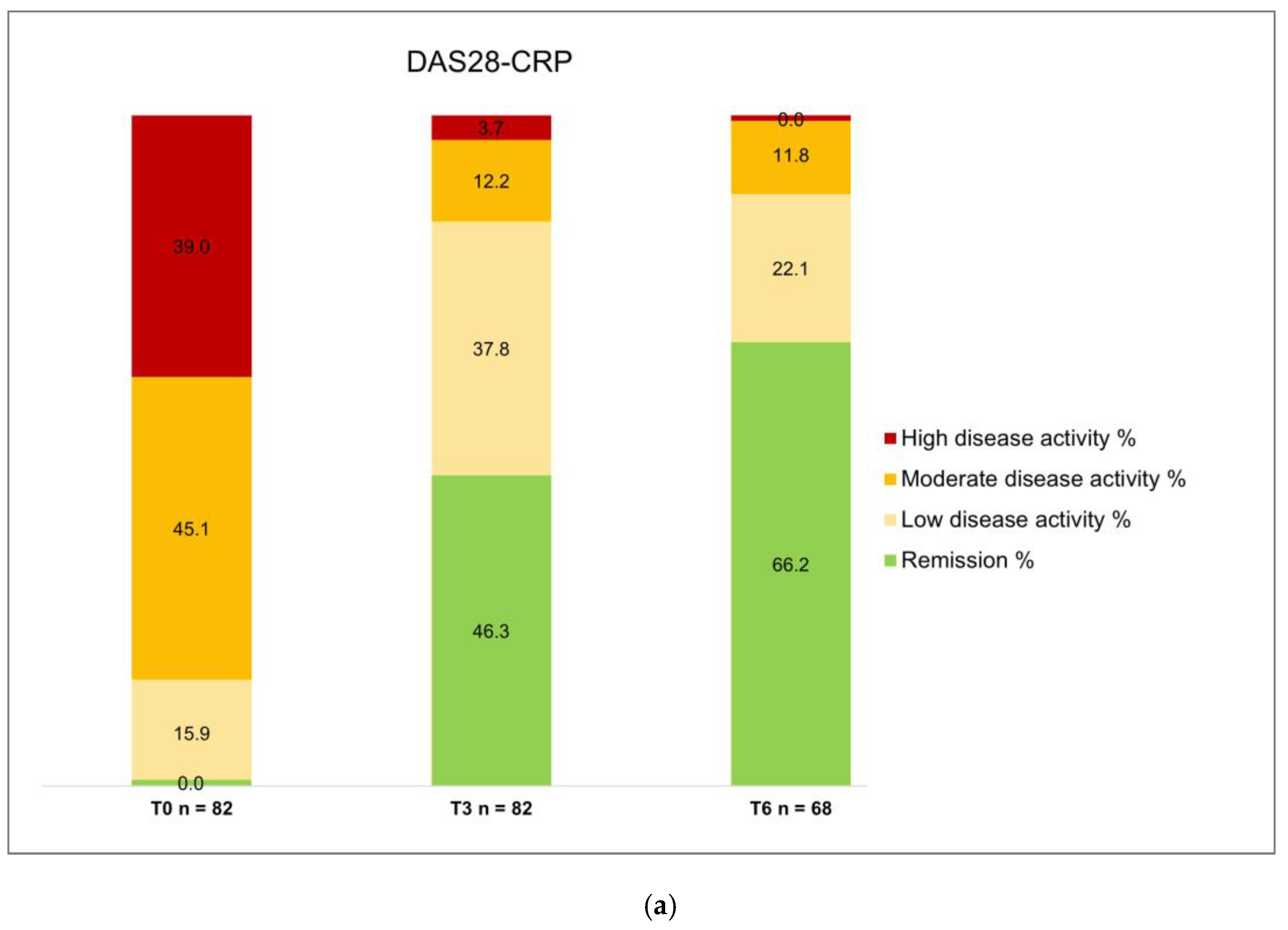
| DAS28-CRP categories | ||||||
| Remission, n (%) | 0.0 (0.0) | 38 (46.3) | 45 (66.2) | < 0.0001 | < 0.0001 | 0.021 |
| Low disease activity, n (%) | 13 (15.9) | 31 (37.8) | 15 (22.1) | 0.002 | 0.40 | 0.05 |
| Moderate disease activity, n (%) | 37 (45.1) | 10 (12.2) | 8 (11.8) | < 0.0001 | < 0.0001 | > 0.99 |
| High disease activity, n (%) | 32 (39.0) | 3 (3.7) | 0 (0.0) | < 0.0001 | < 0.0001 | 0.25 |
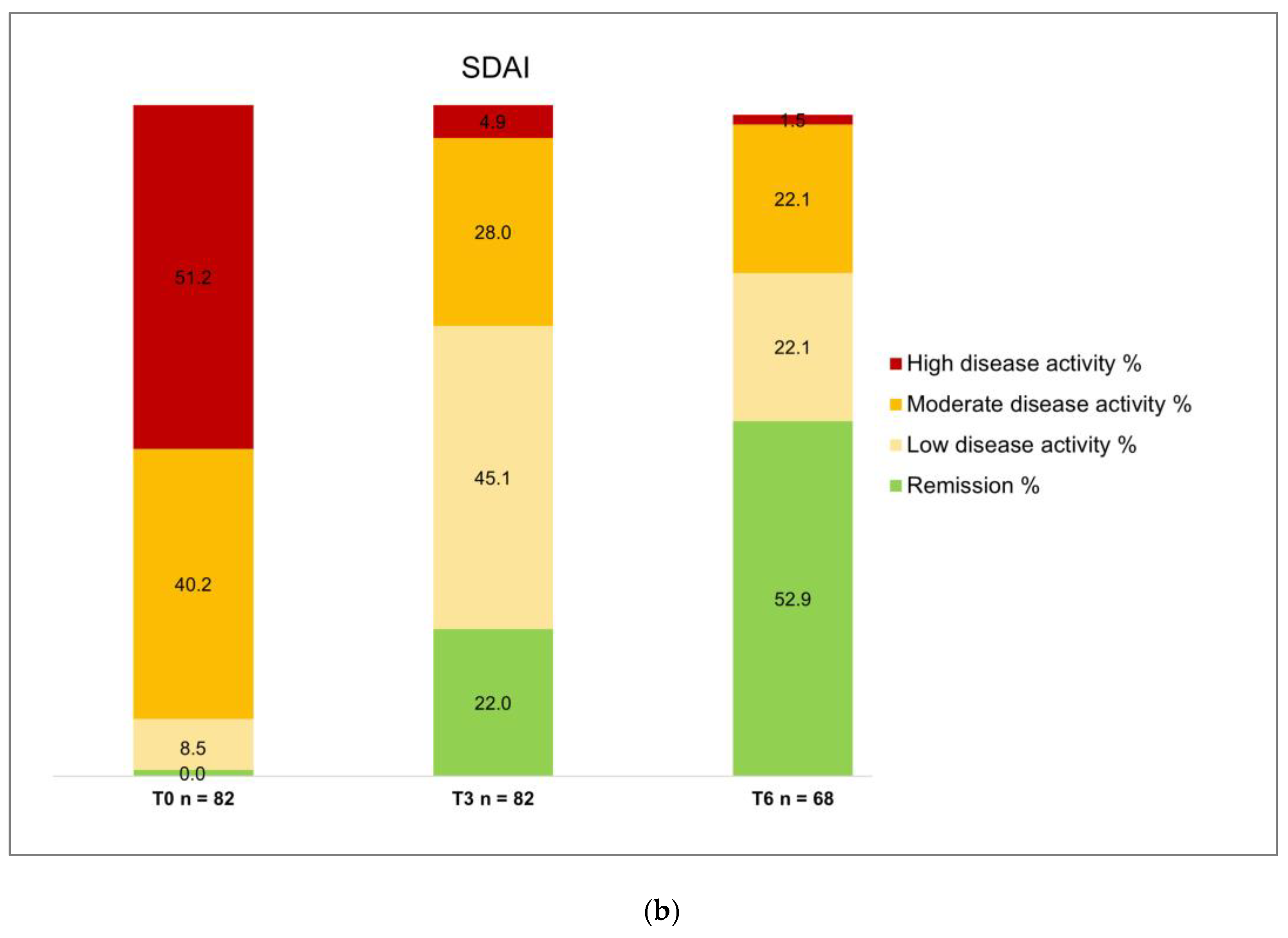
| SDAI categories | ||||||
| Remission, n (%) | 0 (0.0) | 18 (22.0) | 36 (52.9) | < 0.0001 | < 0.001 | 0.0001 |
| Low disease activity, n (%) | 7 (8.5) | 37 (45.1) | 15 (22.1) | < 0.0001 | 0.022 | 0.003 |
| Moderate disease activity, n (%) | 33 (40.2) | 23 (28.0) | 15 (22.1) | 0.138 | 0.02 | 0.45 |
| High disease activity, n (%) | 42 (51.2) | 4 (4.9) | 1 (1.5) | < 0.0001 | < 0.0001 | 0.38 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




