Submitted:
11 October 2024
Posted:
11 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
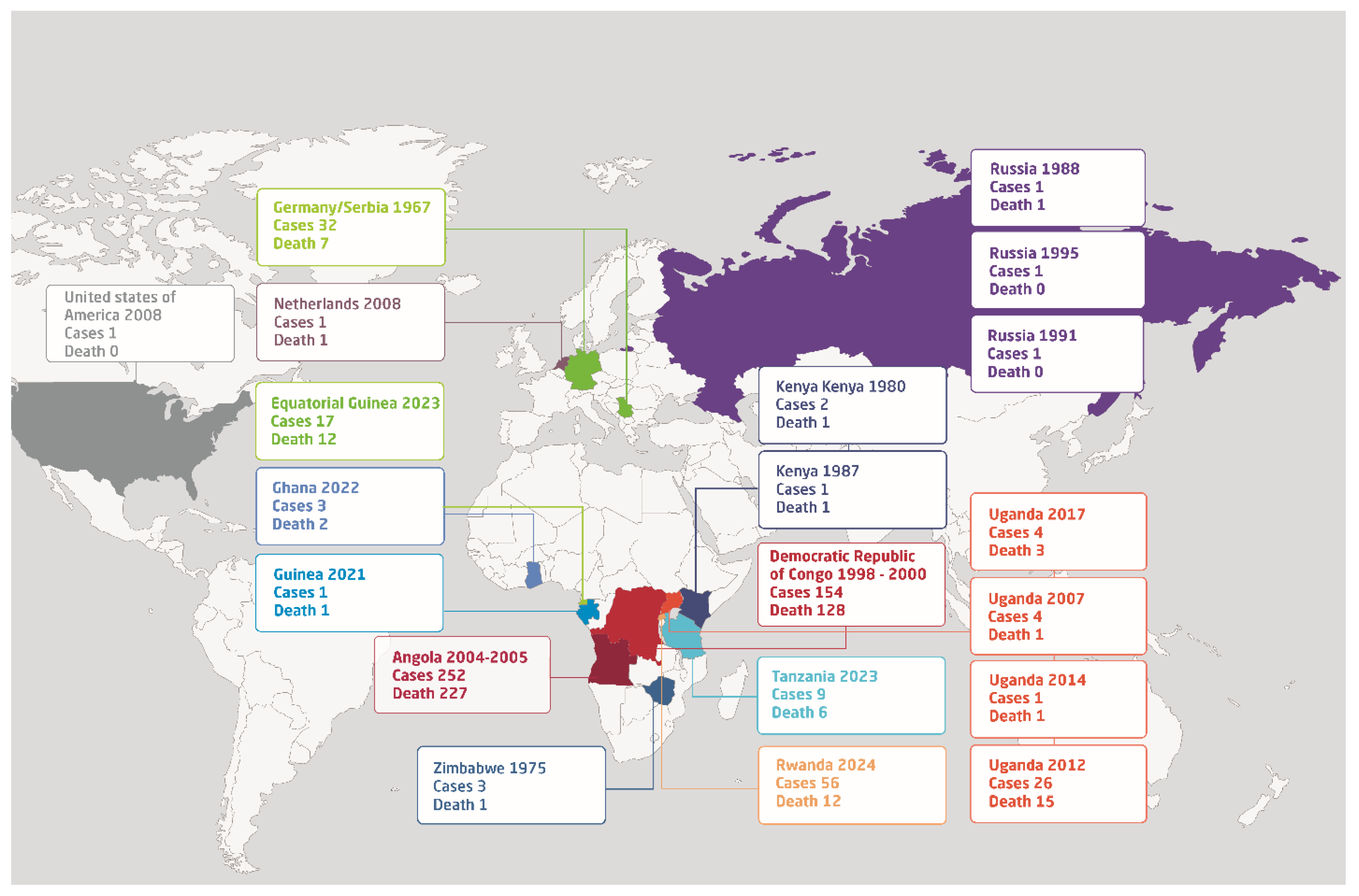
2. Historical Epidemiology
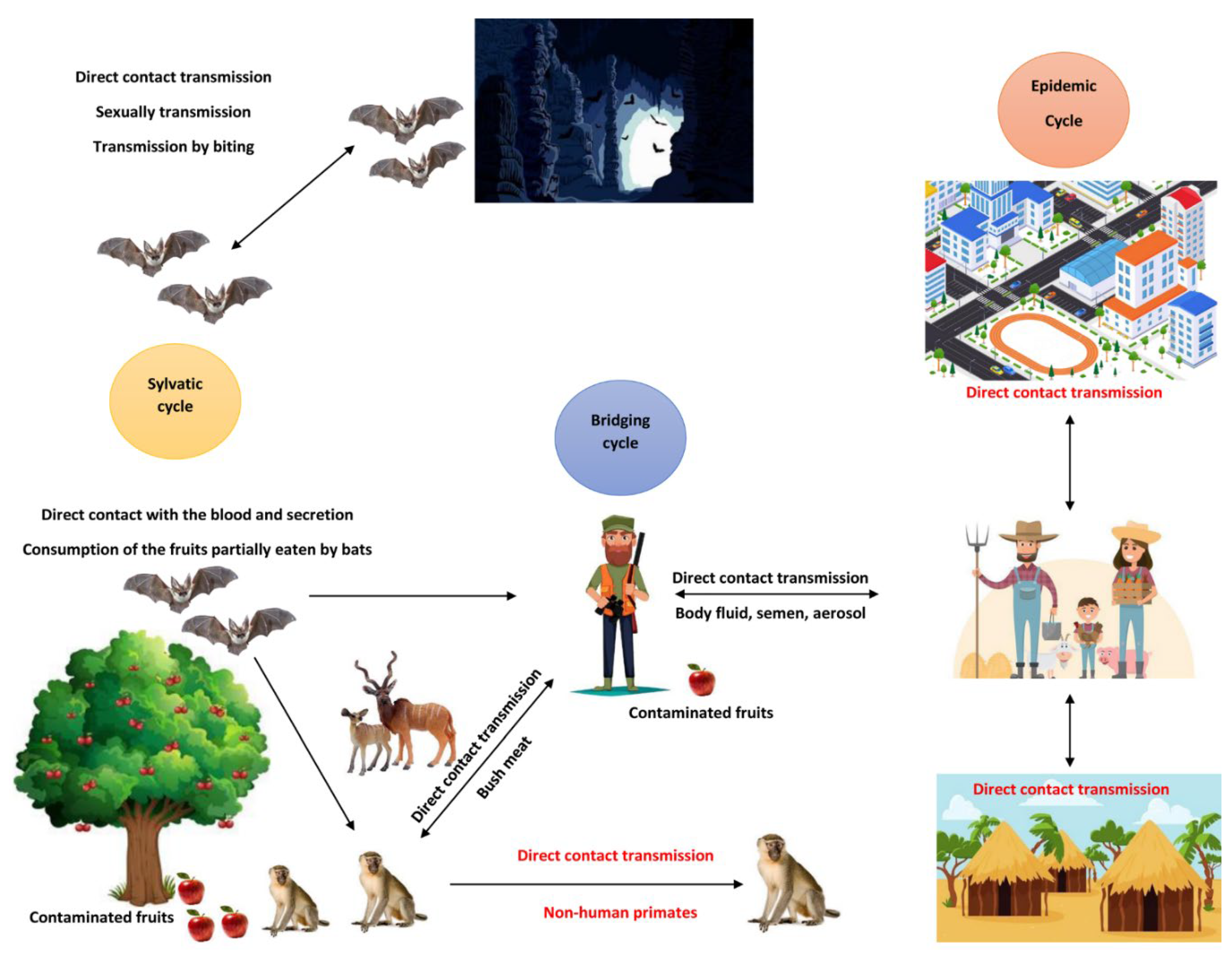
3. Natural Reservoirs and Routes of Transmission
4. Transmission Dynamics
5. Clinical Presentations
6. Diagnosis
7. Case Management
8. Integrated Multisectoral One Health Strategy for Prevention and Control of MVD
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shifflett, K.; Marzi, A. Marburg Virus Pathogenesis – Differences and Similarities in Humans and Animal Models. Virol J 2019, 16, 165. [Google Scholar] [CrossRef] [PubMed]
- Brauburger, K.; Hume, A.J.; Mühlberger, E.; Olejnik, J. Forty-Five Years of Marburg Virus Research. Viruses 2012, 4, 1878–1927. [Google Scholar] [CrossRef] [PubMed]
- The World Health Organization (WHO) Marburg Virus Disease. Available online: https://www.who.int/health-topics/marburg-virus-disease (accessed on 9 October 2024).
- Srivastava, S.; Sharma, D.; Kumar, S.; Sharma, A.; Rijal, R.; Asija, A.; Adhikari, S.; Rustagi, S.; Sah, S.; Al-qaim, Z.H.; et al. Emergence of Marburg Virus: A Global Perspective on Fatal Outbreaks and Clinical Challenges. Front. Microbiol. 2023, 14. [Google Scholar] [CrossRef]
- The Global Alliance for Vaccines and Immunizations (GAVI) The next Pandemic: Marburg? Available online: https://www.gavi.org/vaccineswork/next-pandemic/marburg (accessed on 10 October 2024).
- The World Health Organization (WHO) Prioritizing Diseases for Research and Development in Emergency Contexts Available online:. Available online: https://www.who.int/activities/prioritizing-diseases-for-research-and-development-in-emergency-contexts (accessed on 10 October 2024).
- The World Health Organization (WHO) Pathogens Prioritization: A Scientific Framework for Epidemic and Pandemic Research Preparedness. 2024.
- Ristanović, E.S.; Kokoškov, N.S.; Crozier, I.; Kuhn, J.H.; Gligić, A.S. A Forgotten Episode of Marburg Virus Disease: Belgrade, Yugoslavia, 1967. Microbiology and Molecular Biology Reviews 2020, 84, 10–1128. [Google Scholar] [CrossRef] [PubMed]
- Schuh, A.J.; Amman, B.R.; Guito, J.C.; Graziano, J.C.; Sealy, T.K.; Kirejczyk, S.G.M.; Towner, J.S. Natural Reservoir Rousettus Aegyptiacus Bat Host Model of Orthonairovirus Infection Identifies Potential Zoonotic Spillover Mechanisms. Sci Rep 2022, 12, 20936. [Google Scholar] [CrossRef]
- Guito, J.C.; Prescott, J.B.; Arnold, C.E.; Amman, B.R.; Schuh, A.J.; Spengler, J.R.; Sealy, T.K.; Harmon, J.R.; Coleman-McCray, J.D.; Kulcsar, K.A.; et al. Asymptomatic Infection of Marburg Virus Reservoir Bats Is Explained by a Strategy of Immunoprotective Disease Tolerance. Current Biology 2021, 31, 257–270.e5. [Google Scholar] [CrossRef]
- Timen, A.; Koopmans, M.P.G.; Vossen, A.C.T.M.; van Doornum, G.J.J.; Günther, S.; van den Berkmortel, F.; Verduin, K.M.; Dittrich, S.; Emmerich, P.; Osterhaus, A.D.M.E.; et al. Response to Imported Case of Marburg Hemorrhagic Fever, the Netherlands. Emerg Infect Dis 2009, 15, 1171–1175. [Google Scholar] [CrossRef]
- CDC, C. Transmission of Colorado Tick Fever Virus by Blood Transfusion - Montana. Morbidity and Mortality Weekly Report 1975. [Google Scholar]
- Amman, B.R.; Jones, M.E.B.; Sealy, T.K.; Uebelhoer, L.S.; Schuh, A.J.; Bird, B.H.; Coleman-McCray, J.D.; Martin, B.E.; Nichol, S.T.; Towner, J.S. ORAL SHEDDING OF MARBURG VIRUS IN EXPERIMENTALLY INFECTED EGYPTIAN FRUIT BATS (ROUSETTUS AEGYPTIACUS). Journal of Wildlife Diseases 2015, 51, 113–124. [Google Scholar] [CrossRef]
- Paweska, J.T.; Vuren, P.J. van; Masumu, J.; Leman, P.A.; Grobbelaar, A.A.; Birkhead, M.; Clift, S.; Swanepoel, R.; Kemp, A. Virological and Serological Findings in Rousettus Aegyptiacus Experimentally Inoculated with Vero Cells-Adapted Hogan Strain of Marburg Virus. PLOS ONE 2012, 7, e45479. [Google Scholar] [CrossRef]
- Paweska, J.T.; Jansen van Vuren, P.; Fenton, K.A.; Graves, K.; Grobbelaar, A.A.; Moolla, N.; Leman, P.; Weyer, J.; Storm, N.; McCulloch, S.D.; et al. Lack of Marburg Virus Transmission From Experimentally Infected to Susceptible In-Contact Egyptian Fruit Bats. The Journal of Infectious Diseases 2015, 212, S109–S118. [Google Scholar] [CrossRef] [PubMed]
- Amman, B.R.; Carroll, S.A.; Reed, Z.D.; Sealy, T.K.; Balinandi, S.; Swanepoel, R.; Kemp, A.; Erickson, B.R.; Comer, J.A.; Campbell, S.; et al. Seasonal Pulses of Marburg Virus Circulation in Juvenile Rousettus Aegyptiacus Bats Coincide with Periods of Increased Risk of Human Infection. PLOS Pathogens 2012, 8, e1002877. [Google Scholar] [CrossRef] [PubMed]
- Mehedi, M.; Groseth, A.; Feldmann, H.; Ebihara, H. Clinical Aspects of Marburg Hemorrhagic Fever. Future Virology 2011, 6, 1091–1106. [Google Scholar] [CrossRef] [PubMed]
- Hennessen, W. Epidemiology of “Marburg Virus” Disease. In Marburg Virus Disease; Martini, G.A., Siegert, R., Eds.; Springer: Berlin, Heidelberg, 1971; pp. 161–165. ISBN 978-3-662-01593-3. [Google Scholar]
- Martini, G.A.; Schmidt, H.A. [Spermatogenic transmission of the “Marburg virus”. (Causes of “Marburg simian disease”)]. Klin Wochenschr 1968, 46, 398–400. [Google Scholar] [CrossRef]
- Gear, J.S.; Cassel, G.A.; Gear, A.J.; Trappler, B.; Clausen, L.; Meyers, A.M.; Kew, M.C.; Bothwell, T.H.; Sher, R.; Miller, G.B.; et al. Outbreake of Marburg Virus Disease in Johannesburg. Br Med J 1975, 4, 489–493. [Google Scholar] [CrossRef]
- Borchert, M.; Muyembe-Tamfum, J.J.; Colebunders, R.; Libande, M.; Sabue, M.; Van der Stuyft, P. Short Communication: A Cluster of Marburg Virus Disease Involving an Infant. Tropical Medicine & International Health 2002, 7, 902–906. [Google Scholar] [CrossRef]
- Bausch, D.G.; Borchert, M.; Grein, T.; Roth, C.; Swanepoel, R.; Libande, M.L.; Talarmin, A.; Bertherat, E.; Muyembe-Tamfum, J.-J.; Tugume, B.; et al. Risk Factors for Marburg Hemorrhagic Fever, Democratic Republic of the Congo. Emerg Infect Dis 2003, 9, 1531–1537. [Google Scholar] [CrossRef]
- Alves, D.A.; Glynn, A.R.; Steele, K.E.; Lackemeyer, M.G.; Garza, N.L.; Buck, J.G.; Mech, C.; Reed, D.S. Aerosol Exposure to the Angola Strain of Marburg Virus Causes Lethal Viral Hemorrhagic Fever in Cynomolgus Macaques. Vet Pathol 2010, 47, 831–851. [Google Scholar] [CrossRef]
- The World Health Organization (WHO) Marburg Virus Disease. Available online: https://www.who.int/news-room/fact-sheets/detail/marburg-virus-disease (accessed on 10 October 2024).
- Mitu, R.A.; Islam, Md.R. The Current Pathogenicity and Potential Risk Evaluation of Marburg Virus to Cause Mysterious “Disease X”—An Update on Recent Evidences. Environ. Health. Insights 2024, 18, 11786302241235809. [Google Scholar] [CrossRef]
- Chauhan, L.; Matthews, E.; Piquet, A.L.; Henao-Martinez, A.; Franco-Paredes, C.; Tyler, K.L.; Beckham, D.; Pastula, D.M. Nervous System Manifestations of Arboviral Infections. Curr Trop Med Rep 2022, 9, 107–118. [Google Scholar] [CrossRef]
- Grolla, A.; Lucht, A.; Dick, D.; Strong, J.E.; Feldmann, H. Laboratory Diagnosis of Ebola and Marburg Hemorrhagic Fever. Bull Soc Pathol Exot 2005, 98, 205–209. [Google Scholar] [PubMed]
- Saijo, M.; Niikura, M.; Ikegami, T.; Kurane, I.; Kurata, T.; Morikawa, S. Laboratory Diagnostic Systems for Ebola and Marburg Hemorrhagic Fevers Developed with Recombinant Proteins. Clin Vaccine Immunol 2006, 13, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Kurosaki, Y.; Grolla, A.; Fukuma, A.; Feldmann, H.; Yasuda, J. Development and Evaluation of a Simple Assay for Marburg Virus Detection Using a Reverse Transcription-Loop-Mediated Isothermal Amplification Method. J Clin Microbiol 2010, 48, 2330–2336. [Google Scholar] [CrossRef]
- Gaudinski, M.R.; Coates, E.E.; Novik, L.; Widge, A.; Houser, K.V.; Burch, E.; Holman, L.A.; Gordon, I.J.; Chen, G.L.; Carter, C.; et al. Safety, Tolerability, Pharmacokinetics, and Immunogenicity of the Therapeutic Monoclonal Antibody mAb114 Targeting Ebola Virus Glycoprotein (VRC 608): An Open-Label Phase 1 Study. The Lancet 2019, 393, 889–898. [Google Scholar] [CrossRef] [PubMed]
- Kortepeter, M.G.; Dierberg, K.; Shenoy, E.S.; Cieslak, T.J. Marburg Virus Disease: A Summary for Clinicians. International Journal of Infectious Diseases 2020, 99, 233–242. [Google Scholar] [CrossRef]
- Warren, T.K.; Wells, J.; Panchal, R.G.; Stuthman, K.S.; Garza, N.L.; Van Tongeren, S.A.; Dong, L.; Retterer, C.J.; Eaton, B.P.; Pegoraro, G. Protection against Filovirus Diseases by a Novel Broad-Spectrum Nucleoside Analogue BCX4430. Nature 2014, 508, 402–405. [Google Scholar] [CrossRef]
- Bixler, S.L.; Bocan, T.M.; Wells, J.; Wetzel, K.S.; Van Tongeren, S.A.; Dong, L.; Garza, N.L.; Donnelly, G.; Cazares, L.H.; Nuss, J. Efficacy of Favipiravir (T-705) in Nonhuman Primates Infected with Ebola Virus or Marburg Virus. Antiviral Research 2018, 151, 97–104. [Google Scholar] [CrossRef]
- Porter, D.P.; Weidner, J.M.; Gomba, L.; Bannister, R.; Blair, C.; Jordan, R.; Wells, J.; Wetzel, K.; Garza, N.; Van Tongeren, S.; et al. Remdesivir (GS-5734) Is Efficacious in Cynomolgus Macaques Infected With Marburg Virus. The Journal of Infectious Diseases 2020, 222, 1894–1901. [Google Scholar] [CrossRef] [PubMed]
- Muvunyi, C.M.; Bigirimana, N.; Tuyishime, A.; Mukagatare, I.; Ngabonziza, J.C.; Ahmed, A. Initiatives and Strategies to Strengthen the National, Regional, and International Global Health Security: A Case Study of Rwanda Biomedical Centre. 2024. Available online: https://ssrn.com/abstract=4957490. [CrossRef]
- Gashema, P.; Musafiri, T.; Ndahimana, F.; Iradukunda, H.; Saramba, E.; Nyakatswau, S.T.; Gahamanyi, N.; Iradukunda, P.G.; Ahmed, A.; Dzinamarira, T.; et al. Mpox in East Africa: Learning from COVID-19 and Ebola to Strengthen Public Health Responses. Viruses 2024, 16, 1578. [Google Scholar] [CrossRef]
- Remera, E.; Rwagasore, E.; Muvunyi, C.M.; Ahmed, A. Emergence of the First Molecularly Confirmed Outbreak of Rift Valley Fever among Humans in Rwanda, Calls for Institutionalizing the One Health Strategy. IJID One Health 2024, 4. [Google Scholar] [CrossRef]
- Remera, E.; Rwagasore, E.; Nsekuye, O.; Semakula, M.; Gashegu, M.; Rutayisire, R.; Ishema, L.; Musanabaganwa, C.; Butera, Y.; Nsanzimana, S.; et al. Rift Valley Fever Epizootic, Rwanda, 2022. Emerg Infect Dis 2024, 30, 2191–2193. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.; Ali, Y.; Ibrahim, N.A.; Mohamed, S.I.; Zinsstag, J.; Siddig, E.E.; Mohamed, N.S.; Muvunyi, C.M. One Health Response for Rift Valley Fever Outbreak in Sudan. 2024.
- Nsengimana, I.; Juma, J.; Roesel, K.; Gasana, M.N.; Ndayisenga, F.; Muvunyi, C.M.; Hakizimana, E.; Hakizimana, J.N.; Eastwood, G.; Chengula, A.A.; et al. Genomic Epidemiology of Rift Valley Fever Virus Involved in the 2018 and 2022 Outbreaks in Livestock in Rwanda. Viruses 2024, 16, 1148. [Google Scholar] [CrossRef] [PubMed]
- Wirsiy, F.S.; Nkfusai, C.N.; Bain, L.E. The SPIN Framework to Control and Prevent the Marburg Virus Disease Outbreak in Equatorial Guinea. Pan Afr Med J 2023, 44, 110. [Google Scholar] [PubMed]
- Brainard, J.; Hooper, L.; Pond, K.; Edmunds, K.; Hunter, P.R. Risk Factors for Transmission of Ebola or Marburg Virus Disease: A Systematic Review and Meta-Analysis. International Journal of Epidemiology 2016, 45, 102–116. [Google Scholar] [CrossRef]
- Ahmed, A. Urgent Call for a Global Enforcement of the Public Sharing of Health Emergencies Data: Lesson Learned from Serious Arboviral Disease Epidemics in Sudan. Int Health 2020, 12, 238–240. [Google Scholar] [CrossRef]
- Manno, D. Developing a Vaccine against Marburg Virus Disease. The Lancet 2023, 401, 251–253. [Google Scholar] [CrossRef]
- O’Donnell, K.L.; Feldmann, F.; Kaza, B.; Clancy, C.S.; Hanley, P.W.; Fletcher, P.; Marzi, A. Rapid Protection of Nonhuman Primates against Marburg Virus Disease Using a Single Low-Dose VSV-Based Vaccine. eBioMedicine 2023, 89. [Google Scholar] [CrossRef]
- The World Health Organization (WHO) Marburg Virus Disease. Available online: https://www.who.int/news-room/fact-sheets/detail/marburg-virus-disease (accessed on 10 October 2024).
- CDC About Marburg. Available online: https://www.cdc.gov/marburg/about/index.html (accessed on 10 October 2024).
| Phase | Duration | Characteristics | Symptoms |
|---|---|---|---|
| Generalized Phase | Days 1 to 4 | Abrupt onset of nonspecific symptoms | High fever (39–40°C), severe headache, chills, myalgia, prostration, malaise |
| Early Phase | Days 5 to 13 | 50–75% of patients experience gastrointestinal symptoms | Anorexia, abdominal discomfort, severe nausea, vomiting, diarrhea; maculopapular rash; symptoms of hemorrhagic fever (petechiae, mucosal and gastrointestinal bleeding, hemorrhage from venipuncture sites) |
| Convalescence Phase | After Day 13 | Survivors may skip the most severe symptoms and may not progress to this phase | Neurological symptoms may present (disorientation, agitation, seizures, coma); recovery symptoms |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




