Submitted:
08 October 2024
Posted:
10 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
Noise-Induced Hearing Loss
2. Auditory Structures and Functions Impacted by Noise
2.1. Mechanisms of TTS
2.2. Mechanisms of PTS
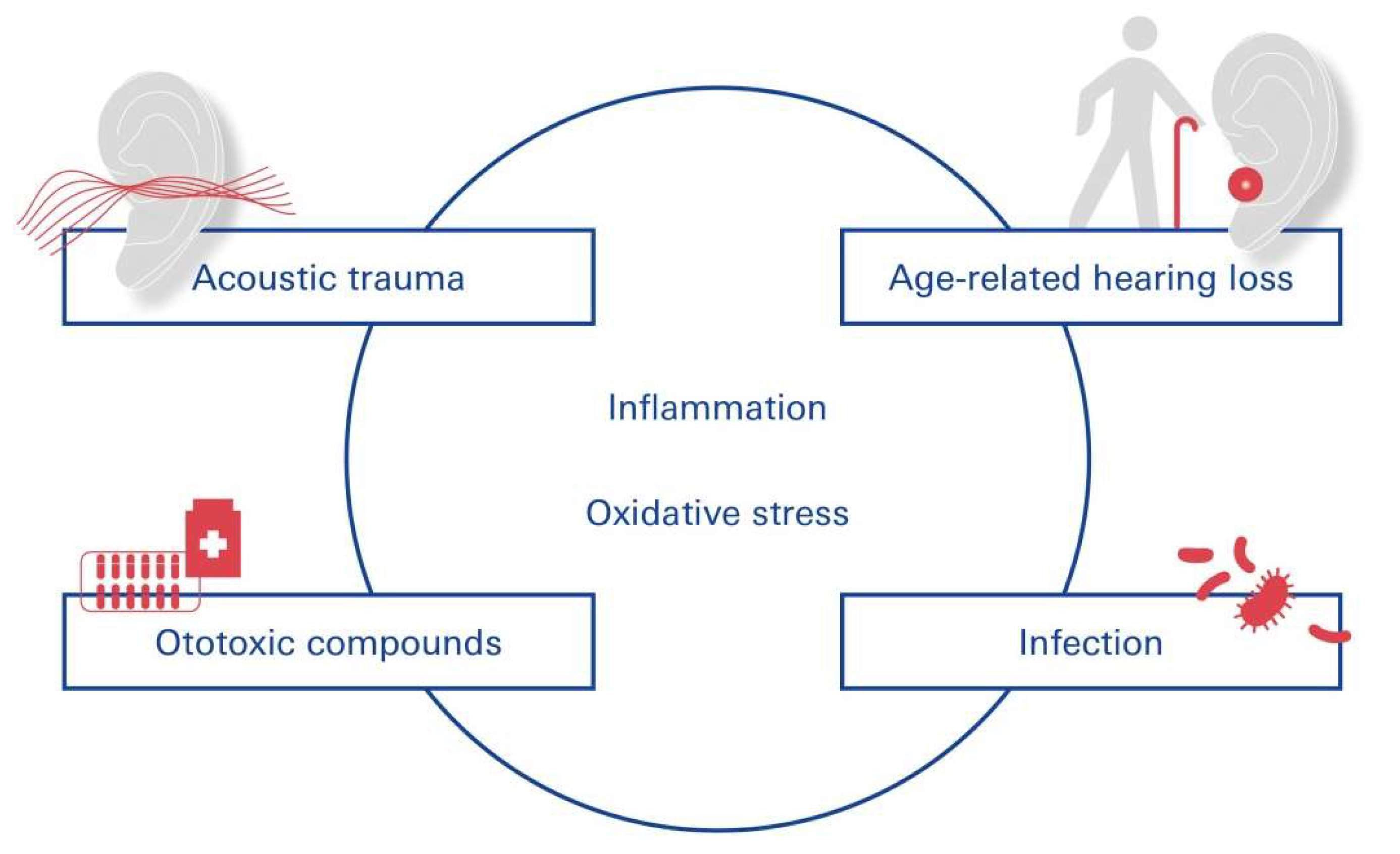
2.3. TTS, PTS, and Free Radical Impact
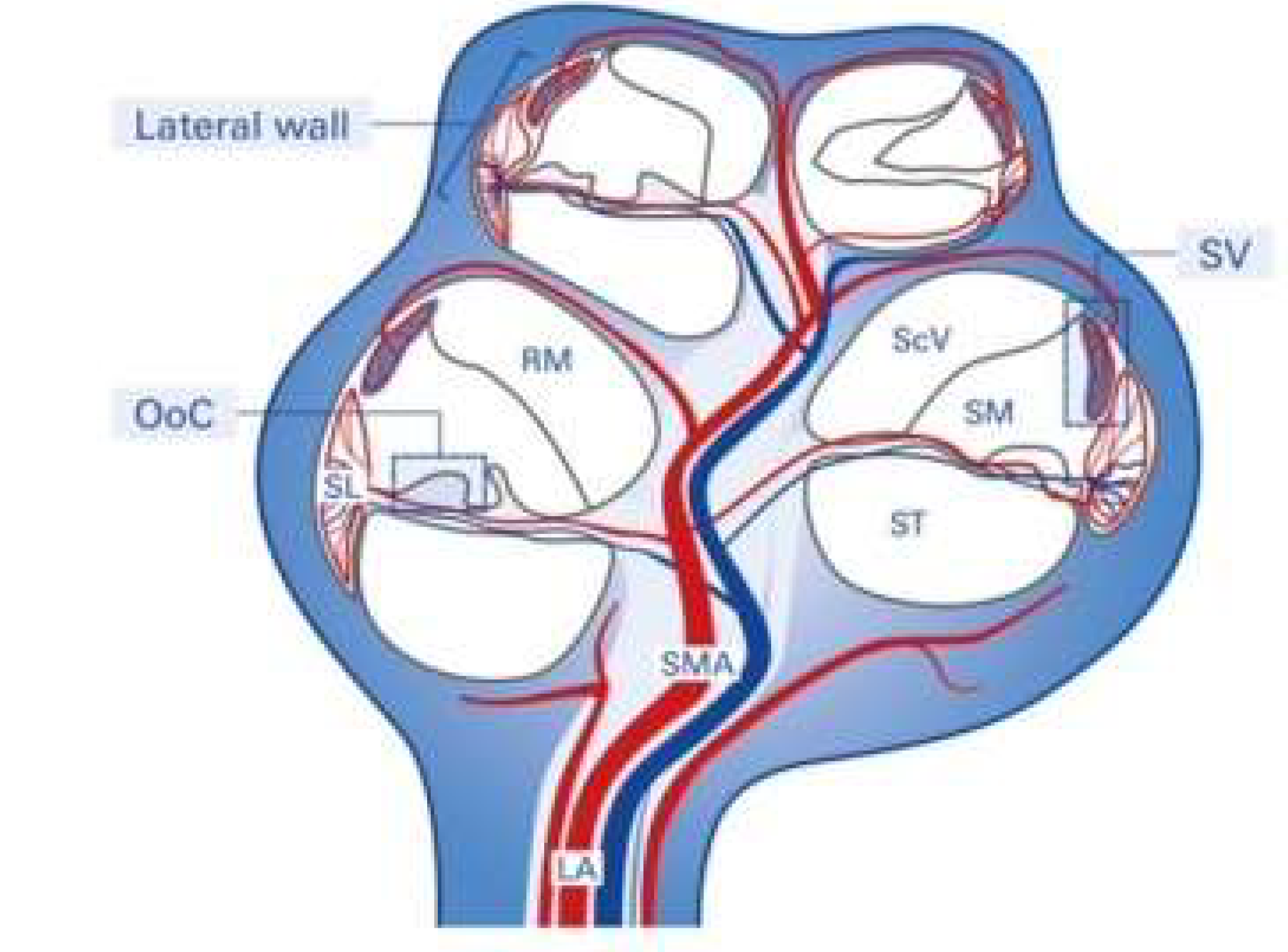
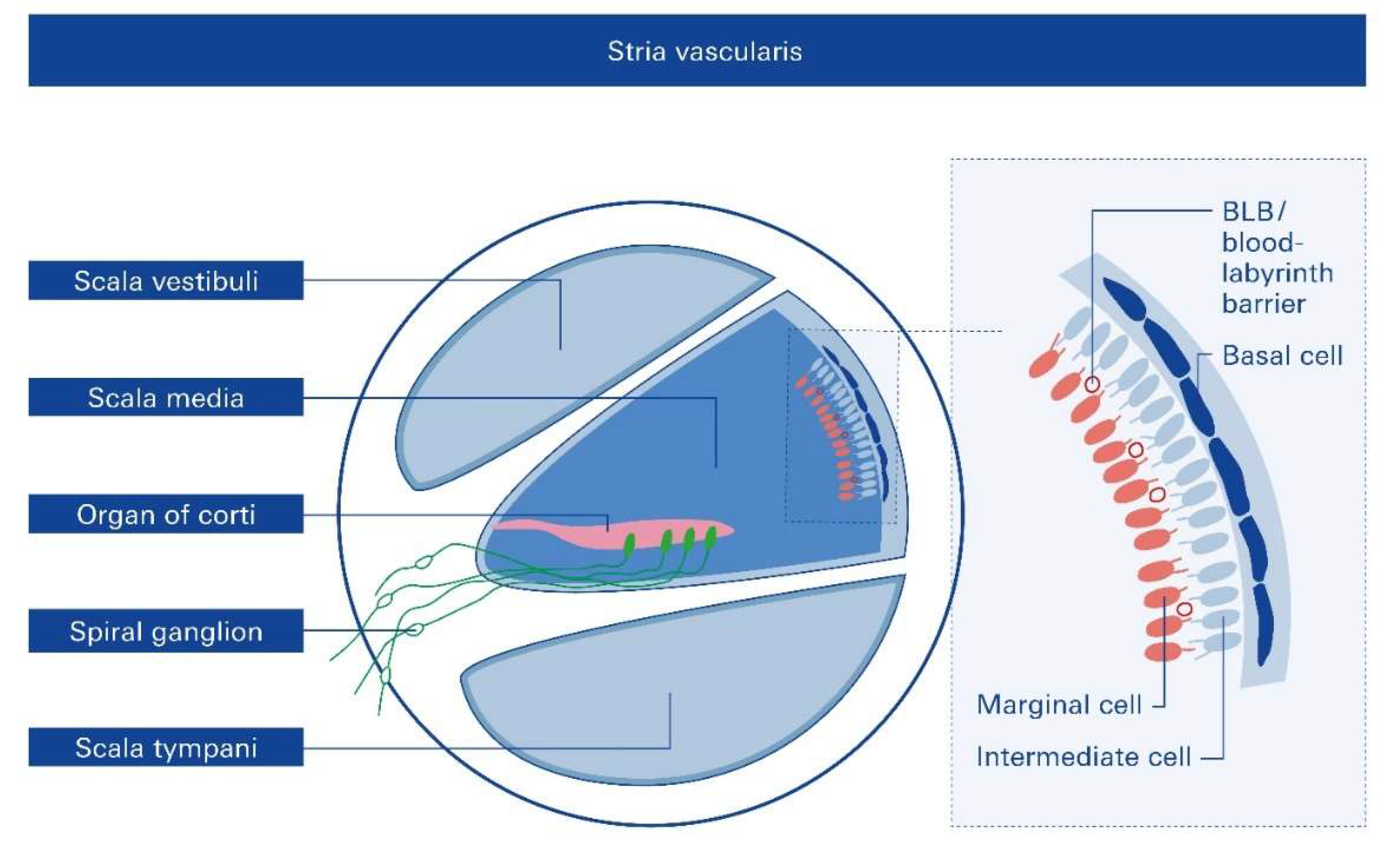
3. The Role of Stria Vascularis in NIHL
4. The Blood-Labyrinth Barrier (BLB) – Functional Contribution to SNHL Pathologies
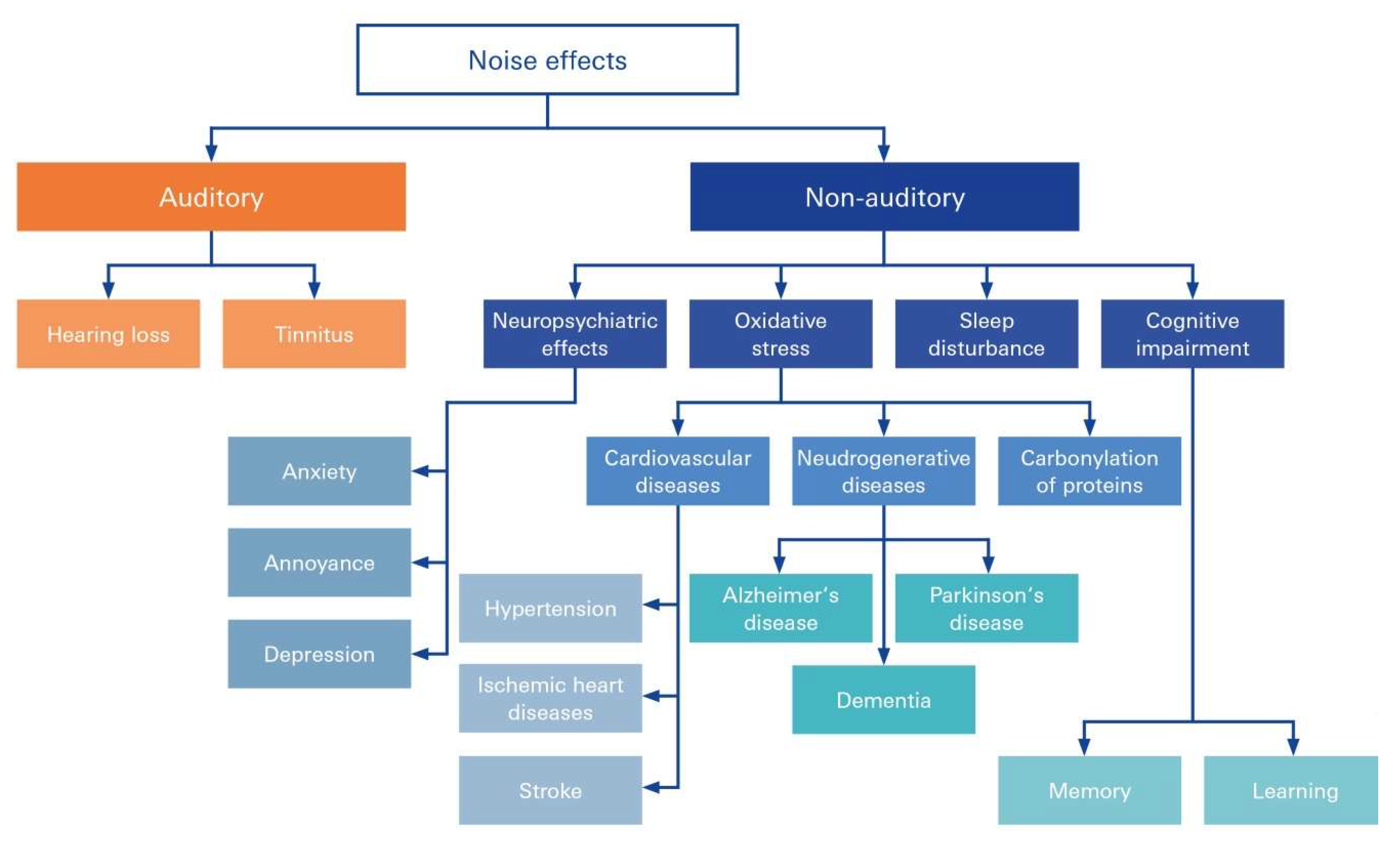
5. Spectrum of NIHL-Associated Disorders
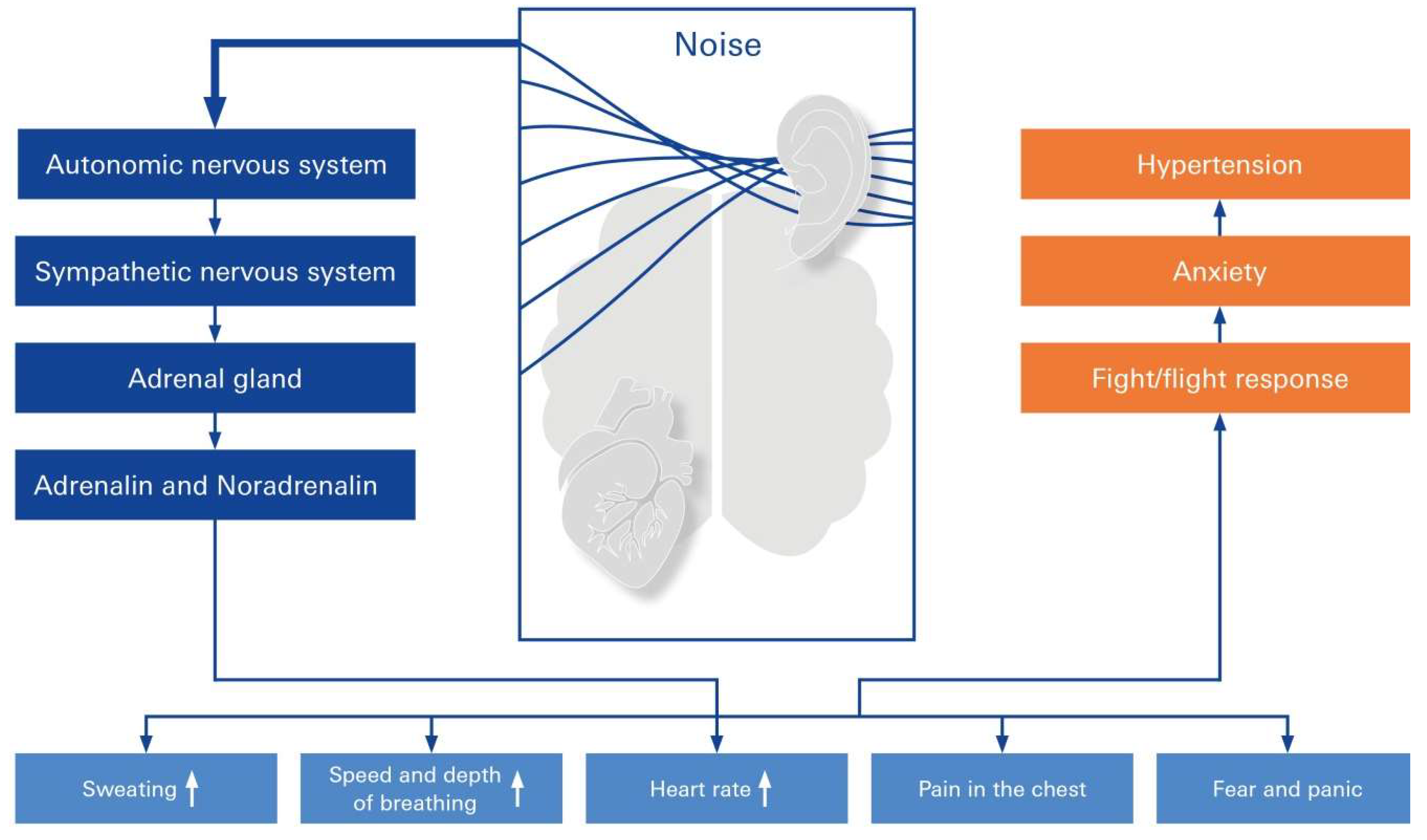
6. Noise above the Permissive Threshold May Beget Autonomic Changes and Hypertension
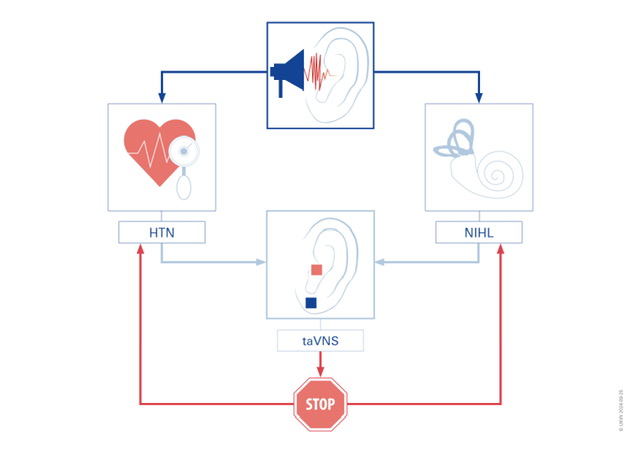
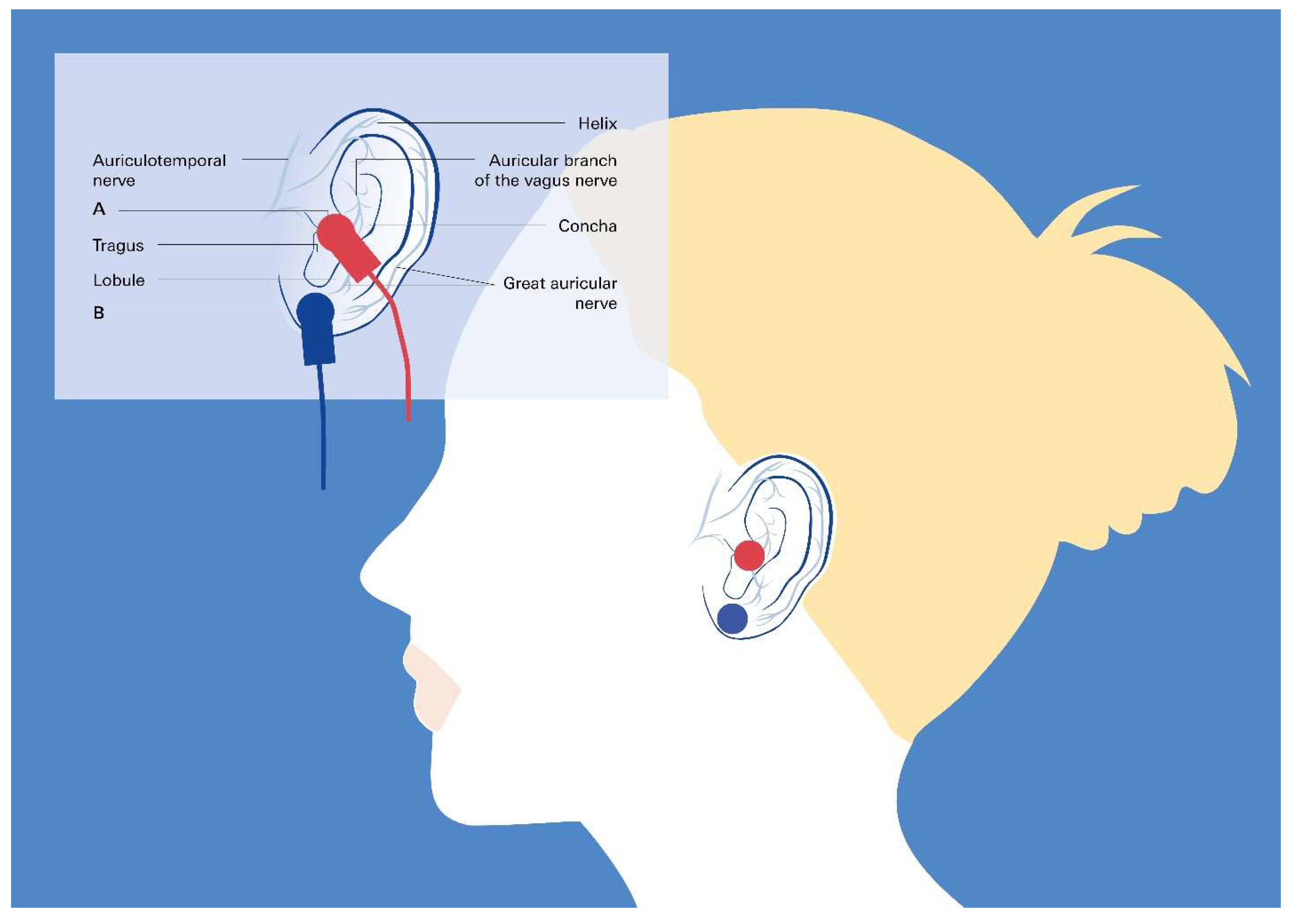
7. A New Method for Preventing and Treating NIHL and Hypertension Using Vagus Nerve Stimulation
8. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Ghadri JR, Wittstein IS, Prasad A, Sharkey S, Dote K, Akashi YJ, et al. International Expert Consensus Document on Takotsubo Syndrome (Part I): Clinical Characteristics, Diagnostic Criteria, and Pathophysiology. European heart journal. 2018;39(22):2032-46. Epub 2018/06/01.
- Sharkey SW, Lesser JR, Maron MS, Maron BJ. Why not just call it tako-tsubo cardiomyopathy: a discussion of nomenclature. J Am Coll Cardiol. 2011;57(13):1496-7.
- Song BG, Yang HS, Hwang HK, Kang GH, Park YH, Chun WJ, et al. The impact of stressor patterns on clinical features in patients with tako-tsubo cardiomyopathy: experiences of two tertiary cardiovascular centers. Clin Cardiol. 2012;35(11):E6-13. Epub 20121001.
- Lyon AR, Bossone E, Schneider B, Sechtem U, Citro R, Underwood SR, et al. Current state of knowledge on Takotsubo syndrome: a Position Statement from the Taskforce on Takotsubo Syndrome of the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2016;18(1):8-27. Epub 20151109.
- Elgendy AY, Elgendy IY, Mansoor H, Mahmoud AN. Clinical presentations and outcomes of Takotsubo syndrome in the setting of subarachnoid hemorrhage: A systematic review and meta-analysis. European heart journal Acute cardiovascular care. 2018;7(3):236-45. Epub 2016/11/18.
- Eitel I, von Knobelsdorff-Brenkenhoff F, Bernhardt P, Carbone I, Muellerleile K, Aldrovandi A, et al. Clinical characteristics and cardiovascular magnetic resonance findings in stress (takotsubo) cardiomyopathy. Jama. 2011;306(3):277-86.
- Templin C, Ghadri JR, Diekmann J, Napp LC, Bataiosu DR, Jaguszewski M, et al. Clinical Features and Outcomes of Takotsubo (Stress) Cardiomyopathy. N Engl J Med. 2015;373(10):929-38.
- Ghadri JR, Cammann VL, Napp LC, Jurisic S, Diekmann J, Bataiosu DR, et al. Differences in the Clinical Profile and Outcomes of Typical and Atypical Takotsubo Syndrome: Data From the International Takotsubo Registry. JAMA Cardiol. 2016;1(3):335-40.
- Couch LS, Fiedler J, Chick G, Clayton R, Dries E, Wienecke LM, et al. Circulating microRNAs predispose to takotsubo syndrome following high-dose adrenaline exposure. Cardiovasc Res. 2022;118(7):1758-70.
- Jaguszewski M, Osipova J, Ghadri JR, Napp LC, Widera C, Franke J, et al. A signature of circulating microRNAs differentiates takotsubo cardiomyopathy from acute myocardial infarction. European heart journal. 2014;35(15):999-1006. Epub 2013/09/21.
- Nagai M, Shityakov S, Smetak M, Hunkler H, Bär C, Schlegel N, et al. Blood Biomarkers in Takotsubo Syndrome Point to an Emerging Role for Inflammaging in Endothelial Pathophysiology. Biomolecules. 2023;2023:995.
- Frank N, Nagai M, Förster C. Exploration of transcutaneous vagus nerve stimulation as a treatment option for adjuvant cancer and heart failure therapy. Exploration of Neuroprotective Therapy. 2023:363-97.
- Nagai M, Shityakov S, Smetak M, Hunkler HJ, Bar C, Schlegel N, et al. Blood Biomarkers in Takotsubo Syndrome Point to an Emerging Role for Inflammaging in Endothelial Pathophysiology. Biomolecules. 2023;13(6). Epub 2023/06/28.
- Ittner C, Burek M, Stork S, Nagai M, Forster CY. Increased Catecholamine Levels and Inflammatory Mediators Alter Barrier Properties of Brain Microvascular Endothelial Cells in vitro. Frontiers in cardiovascular medicine. 2020;7:73. Epub 2020/05/21. [CrossRef]
- Karnati S, Garikapati V, Liebisch G, Van Veldhoven PP, Spengler B, Schmitz G, et al. Quantitative lipidomic analysis of mouse lung during postnatal development by electrospray ionization tandem mass spectrometry. PloS one. 2018;13(9):e0203464. Epub 2018/09/08.
- Nagai M, Dote K, Kato M, Sasaki S, Oda N, Kagawa E, et al. The Insular Cortex and Takotsubo Cardiomyopathy. Curr Pharm Des. 2017;23(6):879-88.
- Templin C, Hanggi J, Klein C, Topka MS, Hiestand T, Levinson RA, et al. Altered limbic and autonomic processing supports brain-heart axis in Takotsubo syndrome. European heart journal. 2019;40(15):1183-7. Epub 2019/03/05.
- Nagai M, Kishi K, Kato S. Insular cortex and neuropsychiatric disorders: a review of recent literature. Eur Psychiatry. 2007;22(6):387-94. Epub 20070409.
- George MS, Ketter TA, Parekh PI, Horwitz B, Herscovitch P, Post RM. Brain activity during transient sadness and happiness in healthy women. Am J Psychiatry. 1995;152(3):341-51.
- Reiman EM, Lane RD, Ahern GL, Schwartz GE, Davidson RJ, Friston KJ, et al. Neuroanatomical correlates of externally and internally generated human emotion. Am J Psychiatry. 1997;154(7):918-25.
- Damasio AR, Grabowski TJ, Bechara A, Damasio H, Ponto LL, Parvizi J, et al. Subcortical and cortical brain activity during the feeling of self-generated emotions. Nat Neurosci. 2000;3(10):1049-56.
- Gu X, Hof PR, Friston KJ, Fan J. Anterior insular cortex and emotional awareness. J Comp Neurol. 2013;521(15):3371-88.
- Ulrich-Lai YM, Herman JP. Neural regulation of endocrine and autonomic stress responses. Nat Rev Neurosci. 2009;10(6):397-409.
- Béjean S, Sultan-Taïeb H. Modeling the economic burden of diseases imputable to stress at work. Eur J Health Econ. 2005;6(1):16-23.
- de Kloet ER, Joëls M, Holsboer F. Stress and the brain: from adaptation to disease. Nat Rev Neurosci. 2005;6(6):463-75.
- Romier C, Bernassau JM, Cambillau C, Darbon H. Solution structure of human corticotropin releasing factor by 1H NMR and distance geometry with restrained molecular dynamics. Protein Eng. 1993;6(2):149-56.
- Trainer PJ, Faria M, Newell-Price J, Browne P, Kopelman P, Coy DH, et al. A comparison of the effects of human and ovine corticotropin-releasing hormone on the pituitary-adrenal axis. J Clin Endocrinol Metab. 1995;80(2):412-7.
- Hillhouse EW, Grammatopoulos DK. The molecular mechanisms underlying the regulation of the biological activity of corticotropin-releasing hormone receptors: implications for physiology and pathophysiology. Endocr Rev. 2006;27(3):260-86. Epub 20060216.
- Henckens MJ, Deussing JM, Chen A. Region-specific roles of the corticotropin-releasing factor-urocortin system in stress. Nat Rev Neurosci. 2016;17(10):636-51. Epub 20160902.
- Carrasco GA, Van de Kar LD. Neuroendocrine pharmacology of stress. Eur J Pharmacol. 2003;463(1-3):235-72.
- McEwen BS, Sapolsky RM. Stress and cognitive function. Curr Opin Neurobiol. 1995;5(2):205-16.
- McEwen, BS. Protective and damaging effects of stress mediators. N Engl J Med. 1998;338(3):171-9.
- De Kloet ER, Derijk R. Signaling pathways in brain involved in predisposition and pathogenesis of stress-related disease: genetic and kinetic factors affecting the MR/GR balance. Ann N Y Acad Sci. 2004;1032:14-34.
- Joëls M, Karst H, Alfarez D, Heine VM, Qin Y, van Riel E, et al. Effects of chronic stress on structure and cell function in rat hippocampus and hypothalamus. Stress. 2004;7(4):221-31.
- Willner, P. Chronic mild stress (CMS) revisited: consistency and behavioural-neurobiological concordance in the effects of CMS. Neuropsychobiology. 2005;52(2):90-110. Epub 20050719.
- Malta MB, Martins J, Novaes LS, Dos Santos NB, Sita L, Camarini R, et al. Norepinephrine and Glucocorticoids Modulate Chronic Unpredictable Stress-Induced Increase in the Type 2 CRF and Glucocorticoid Receptors in Brain Structures Related to the HPA Axis Activation. Mol Neurobiol. 2021;58(10):4871-85. Epub 20210702.
- Allen AP, Kennedy PJ, Cryan JF, Dinan TG, Clarke G. Biological and psychological markers of stress in humans: focus on the Trier Social Stress Test. Neuroscience and biobehavioral reviews. 2014;38:94-124. Epub 2013/11/19.
- Wallstrom S, Ulin K, Maatta S, Omerovic E, Ekman I. Impact of long-term stress in Takotsubo syndrome: Experience of patients. European journal of cardiovascular nursing. 2016;15(7):522-8. Epub 2015/11/18.
- Chan C, Elliott J, Troughton R, Frampton C, Smyth D, Crozier I, et al. Acute myocardial infarction and stress cardiomyopathy following the Christchurch earthquakes. PLoS One. 2013;8(7):e68504. Epub 20130702.
- Sharkey SW, Windenburg DC, Lesser JR, Maron MS, Hauser RG, Lesser JN, et al. Natural history and expansive clinical profile of stress (tako-tsubo) cardiomyopathy. J Am Coll Cardiol. 2010;55(4):333-41.
- Tagawa M, Nakamura Y, Ishiguro M, Satoh K, Chinushi M, Kodama M, et al. Transient left ventricular apical ballooning developing after the Central Niigata Prefecture Earthquake: two case reports. J Cardiol. 2006;48(3):153-8.
- Ghadri JR, Sarcon A, Diekmann J, Bataiosu DR, Cammann VL, Jurisic S, et al. Happy heart syndrome: role of positive emotional stress in takotsubo syndrome. Eur Heart J. 2016;37(37):2823-9. Epub 20160302.
- Scheitz JF, Mochmann HC, Witzenbichler B, Fiebach JB, Audebert HJ, Nolte CH. Takotsubo cardiomyopathy following ischemic stroke: a cause of troponin elevation. J Neurol. 2012;259(1):188-90. Epub 20110618.
- Modi S, Baig W. Radiotherapy-induced Tako-tsubo cardiomyopathy. Clin Oncol (R Coll Radiol). 2009;21(4):361-2. Epub 20090218.
- S YH, Settergren M, Henareh L. Sepsis-induced myocardial depression and takotsubo syndrome. Acute Card Care. 2014;16(3):102-9. Epub 20140623.
- Brezina P, Isler CM. Takotsubo cardiomyopathy in pregnancy. Obstet Gynecol. 2008;112(2 Pt 2):450-2.
- Riera M, Llompart-Pou JA, Carrillo A, Blanco C. Head injury and inverted Takotsubo cardiomyopathy. J Trauma. 2010;68(1):E13-5.
- Stöllberger C, Wegner C, Finsterer J. Seizure-associated Takotsubo cardiomyopathy. Epilepsia. 2011;52(11):e160-7. Epub 20110721.
- Hjalmarsson C, Oras J, Redfors B. A case of intracerebral hemorrhage and apical ballooning: An important differential diagnosis in ST-segment elevation. Int J Cardiol. 2015;186:90-2. Epub 20150318.
- Murakami T, Yoshikawa T, Maekawa Y, Ueda T, Isogai T, Sakata K, et al. Gender Differences in Patients with Takotsubo Cardiomyopathy: Multi-Center Registry from Tokyo CCU Network. PLoS One. 2015;10(8):e0136655. Epub 20150828.
- Krishnamoorthy P, Garg J, Sharma A, Palaniswamy C, Shah N, Lanier G, et al. Gender Differences and Predictors of Mortality in Takotsubo Cardiomyopathy: Analysis from the National Inpatient Sample 2009-2010 Database. Cardiology. 2015;132(2):131-6. Epub 20150707.
- Yoshizawa M, Itoh T, Morino Y, Taniai S, Ishibashi Y, Komatsu T, et al. Gender Differences in the Circadian and Seasonal Variations in Patients with Takotsubo Syndrome: A Multicenter Registry at Eight University Hospitals in East Japan. Intern Med. 2021;60(17):2749-55. Epub 20210322.
- Angelini P, Tobis JM. Is high-dose catecholamine administration in small animals an appropriate model for takotsubo syndrome? Circ J. 2015;79(4):897. Epub 20150305.
- Duma D, Collins JB, Chou JW, Cidlowski JA. Sexually dimorphic actions of glucocorticoids provide a link to inflammatory diseases with gender differences in prevalence. Sci Signal. 2010;3(143):ra74. Epub 20101012.
- Lindheim SR, Legro RS, Bernstein L, Stanczyk FZ, Vijod MA, Presser SC, et al. Behavioral stress responses in premenopausal and postmenopausal women and the effects of estrogen. Am J Obstet Gynecol. 1992;167(6):1831-6.
- Kudielka BM, Schmidt-Reinwald AK, Hellhammer DH, Kirschbaum C. Psychological and endocrine responses to psychosocial stress and dexamethasone/corticotropin-releasing hormone in healthy postmenopausal women and young controls: the impact of age and a two-week estradiol treatment. Neuroendocrinology. 1999;70(6):422-30.
- Ueyama T, Hano T, Kasamatsu K, Yamamoto K, Tsuruo Y, Nishio I. Estrogen attenuates the emotional stress-induced cardiac responses in the animal model of Tako-tsubo (Ampulla) cardiomyopathy. J Cardiovasc Pharmacol. 2003;42 Suppl 1:S117-9.
- Ueyama T, Ishikura F, Matsuda A, Asanuma T, Ueda K, Ichinose M, et al. Chronic estrogen supplementation following ovariectomy improves the emotional stress-induced cardiovascular responses by indirect action on the nervous system and by direct action on the heart. Circ J. 2007;71(4):565-73.
- Rivera CM, Grossardt BR, Rhodes DJ, Brown RD, Jr., Roger VL, Melton LJ, 3rd, et al. Increased cardiovascular mortality after early bilateral oophorectomy. Menopause. 2009;16(1):15-23.
- Brenner R, Weilenmann D, Maeder MT, Jörg L, Bluzaite I, Rickli H, et al. Clinical characteristics, sex hormones, and long-term follow-up in Swiss postmenopausal women presenting with Takotsubo cardiomyopathy. Clin Cardiol. 2012;35(6):340-7. Epub 20120409.
- Zhao D, Guallar E, Ouyang P, Subramanya V, Vaidya D, Ndumele CE, et al. Endogenous Sex Hormones and Incident Cardiovascular Disease in Post-Menopausal Women. J Am Coll Cardiol. 2018;71(22):2555-66.
- Barros RP, Machado UF, Gustafsson JA. Estrogen receptors: new players in diabetes mellitus. Trends Mol Med. 2006;12(9):425-31. Epub 20060804.
- Zhu Y, Bian Z, Lu P, Karas RH, Bao L, Cox D, et al. Abnormal vascular function and hypertension in mice deficient in estrogen receptor beta. Science. 2002;295(5554):505-8.
- Komesaroff PA, Esler MD, Sudhir K. Estrogen supplementation attenuates glucocorticoid and catecholamine responses to mental stress in perimenopausal women. J Clin Endocrinol Metab. 1999;84(2):606-10.
- Sung BH, Ching M, Izzo JL, Jr., Dandona P, Wilson MF. Estrogen improves abnormal norepinephrine-induced vasoconstriction in postmenopausal women. J Hypertens. 1999;17(4):523-8.
- Sader MA, Celermajer DS. Endothelial function, vascular reactivity and gender differences in the cardiovascular system. Cardiovasc Res. 2002;53(3):597-604.
- Forster C, Kietz S, Hultenby K, Warner M, Gustafsson JA. Characterization of the ERbeta-/-mouse heart. Proceedings of the National Academy of Sciences of the United States of America. 2004;101(39):14234-9. Epub 2004/09/18.
- Dimberg U, Petterson M. Facial reactions to happy and angry facial expressions: evidence for right hemisphere dominance. Psychophysiology. 2000;37(5):693-6.
- Drača, S. A possible relationship between Takotsubo cardiomyopathy and female sex steroid-related modulation of functional cerebral asymmetry. Med Hypotheses. 2015;84(3):238-40. Epub 20150114.
- Singh K, Carson K, Shah R, Sawhney G, Singh B, Parsaik A, et al. Meta-analysis of clinical correlates of acute mortality in takotsubo cardiomyopathy. Am J Cardiol. 2014;113(8):1420-8. Epub 20140201.
- Pelliccia F, Parodi G, Greco C, Antoniucci D, Brenner R, Bossone E, et al. Comorbidities frequency in Takotsubo syndrome: an international collaborative systematic review including 1109 patients. Am J Med. 2015;128(6):654.e11-9. Epub 20150204.
- Pelliccia F, Sinagra G, Elliott P, Parodi G, Basso C, Camici PG. Takotsubo: One, no one and one hundred thousand diseases. Int J Cardiol. 2018;261:35.
- Jänig, W. The Integrative Action of the Autonomic Nervous System: Neurobiology of Homeostasis. UK: Cambridge University Press; 2006.
- Li Y, Jiang W, Li ZZ, Zhang C, Huang C, Yang J, et al. Repetitive restraint stress changes spleen immune cell subsets through glucocorticoid receptor or β-adrenergic receptor in a stage dependent manner. Biochem Biophys Res Commun. 2018;495(1):1108-14. Epub 20171123.
- Duma D, Jewell CM, Cidlowski JA. Multiple glucocorticoid receptor isoforms and mechanisms of post-translational modification. J Steroid Biochem Mol Biol. 2006;102(1-5):11-21. Epub 20061027.
- Rhen T, Cidlowski JA. Antiinflammatory action of glucocorticoids--new mechanisms for old drugs. N Engl J Med. 2005;353(16):1711-23.
- Nagai M, Forster CY, Dote K. Sex Hormone-Specific Neuroanatomy of Takotsubo Syndrome: Is the Insular Cortex a Moderator? Biomolecules. 2022;12(1). Epub 2022/01/22.
- Walsh CP, Bovbjerg DH, Marsland AL. Glucocorticoid resistance and β2-adrenergic receptor signaling pathways promote peripheral pro-inflammatory conditions associated with chronic psychological stress: A systematic review across species. Neurosci Biobehav Rev. 2021;128:117-35. Epub 20210608.
- Walsh CP, Ewing LJ, Cleary JL, Vaisleib AD, Farrell CH, Wright AGC, et al. Development of glucocorticoid resistance over one year among mothers of children newly diagnosed with cancer. Brain Behav Immun. 2018;69:364-73. Epub 20171218.
- Capitanio JP, Mendoza SP, Lerche NW, Mason WA. Social stress results in altered glucocorticoid regulation and shorter survival in simian acquired immune deficiency syndrome. Proc Natl Acad Sci U S A. 1998;95(8):4714-9.
- Cole SW, Mendoza SP, Capitanio JP. Social stress desensitizes lymphocytes to regulation by endogenous glucocorticoids: insights from in vivo cell trafficking dynamics in rhesus macaques. Psychosom Med. 2009;71(6):591-7. Epub 20090624.
- Cole SW, Capitanio JP, Chun K, Arevalo JM, Ma J, Cacioppo JT. Myeloid differentiation architecture of leukocyte transcriptome dynamics in perceived social isolation. Proc Natl Acad Sci U S A. 2015;112(49):15142-7. Epub 20151123.
- Oakley RH, Cidlowski JA. The biology of the glucocorticoid receptor: new signaling mechanisms in health and disease. J Allergy Clin Immunol. 2013;132(5):1033-44. Epub 20130929.
- Gonzalez Ramirez C, Villavicencio Queijeiro A, Jimenez Morales S, Barcenas Lopez D, Hidalgo Miranda A, Ruiz Chow A, et al. The NR3C1 gene expression is a potential surrogate biomarker for risk and diagnosis of posttraumatic stress disorder. Psychiatry research. 2020;284:112797. Epub 2020/01/27.
- Ueyama T, Kasamatsu K, Hano T, Yamamoto K, Tsuruo Y, Nishio I. Emotional stress induces transient left ventricular hypocontraction in the rat via activation of cardiac adrenoceptors: a possible animal model of ‘tako-tsubo’ cardiomyopathy. Circ J. 2002;66(7):712-3.
- Kume T, Kawamoto T, Okura H, Toyota E, Neishi Y, Watanabe N, et al. Local release of catecholamines from the hearts of patients with tako-tsubo-like left ventricular dysfunction. Circ J. 2008;72(1):106-8.
- Ortak J, Khattab K, Barantke M, Wiegand UK, Bänsch D, Ince H, et al. Evolution of cardiac autonomic nervous activity indices in patients presenting with transient left ventricular apical ballooning. Pacing Clin Electrophysiol. 2009;32 Suppl 1:S21-5.
- Vaccaro A, Despas F, Delmas C, Lairez O, Lambert E, Lambert G, et al. Direct evidences for sympathetic hyperactivity and baroreflex impairment in Tako Tsubo cardiopathy. PLoS One. 2014;9(3):e93278. Epub 20140325.
- Pelliccia F, Kaski JC, Crea F, Camici PG. Pathophysiology of Takotsubo Syndrome. Circulation. 2017;135(24):2426-41.
- Ferrucci L, Semba RD, Guralnik JM, Ershler WB, Bandinelli S, Patel KV, et al. Proinflammatory state, hepcidin, and anemia in older persons. Blood. 2010;115(18):3810-6. Epub 20100115.
- Cohen HJ, Pieper CF, Harris T, Rao KM, Currie MS. The association of plasma IL-6 levels with functional disability in community-dwelling elderly. J Gerontol A Biol Sci Med Sci. 1997;52(4):M201-8. [CrossRef]
- Newman AB, Sanders JL, Kizer JR, Boudreau RM, Odden MC, Zeki Al Hazzouri A, et al. Trajectories of function and biomarkers with age: the CHS All Stars Study. Int J Epidemiol. 2016;45(4):1135-45. Epub 20160606.
- Gerli R, Monti D, Bistoni O, Mazzone AM, Peri G, Cossarizza A, et al. Chemokines, sTNF-Rs and sCD30 serum levels in healthy aged people and centenarians. Mech Ageing Dev. 2000;121(1-3):37-46. [CrossRef]
- Pallauf K, Rimbach G. Autophagy, polyphenols and healthy ageing. Ageing Res Rev. 2013;12(1):237-52. Epub 20120406.
- Salminen A, Kaarniranta K, Kauppinen A. Inflammaging: disturbed interplay between autophagy and inflammasomes. Aging (Albany NY). 2012;4(3):166-75.
- O’Donovan A, Pantell MS, Puterman E, Dhabhar FS, Blackburn EH, Yaffe K, et al. Cumulative inflammatory load is associated with short leukocyte telomere length in the Health, Aging and Body Composition Study. PLoS One. 2011;6(5):e19687. Epub 20110513.
- Shiels PG, McGlynn LM, MacIntyre A, Johnson PC, Batty GD, Burns H, et al. Accelerated telomere attrition is associated with relative household income, diet and inflammation in the pSoBid cohort. PLoS One. 2011;6(7):e22521. Epub 20110727.
- Aviv A, Valdes A, Gardner JP, Swaminathan R, Kimura M, Spector TD. Menopause modifies the association of leukocyte telomere length with insulin resistance and inflammation. J Clin Endocrinol Metab. 2006;91(2):635-40. Epub 20051122.
- Houben JM, Moonen HJ, van Schooten FJ, Hageman GJ. Telomere length assessment: biomarker of chronic oxidative stress? Free Radic Biol Med. 2008;44(3):235-46. Epub 20071010.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



