Submitted:
08 October 2024
Posted:
09 October 2024
You are already at the latest version
Abstract
Keywords:
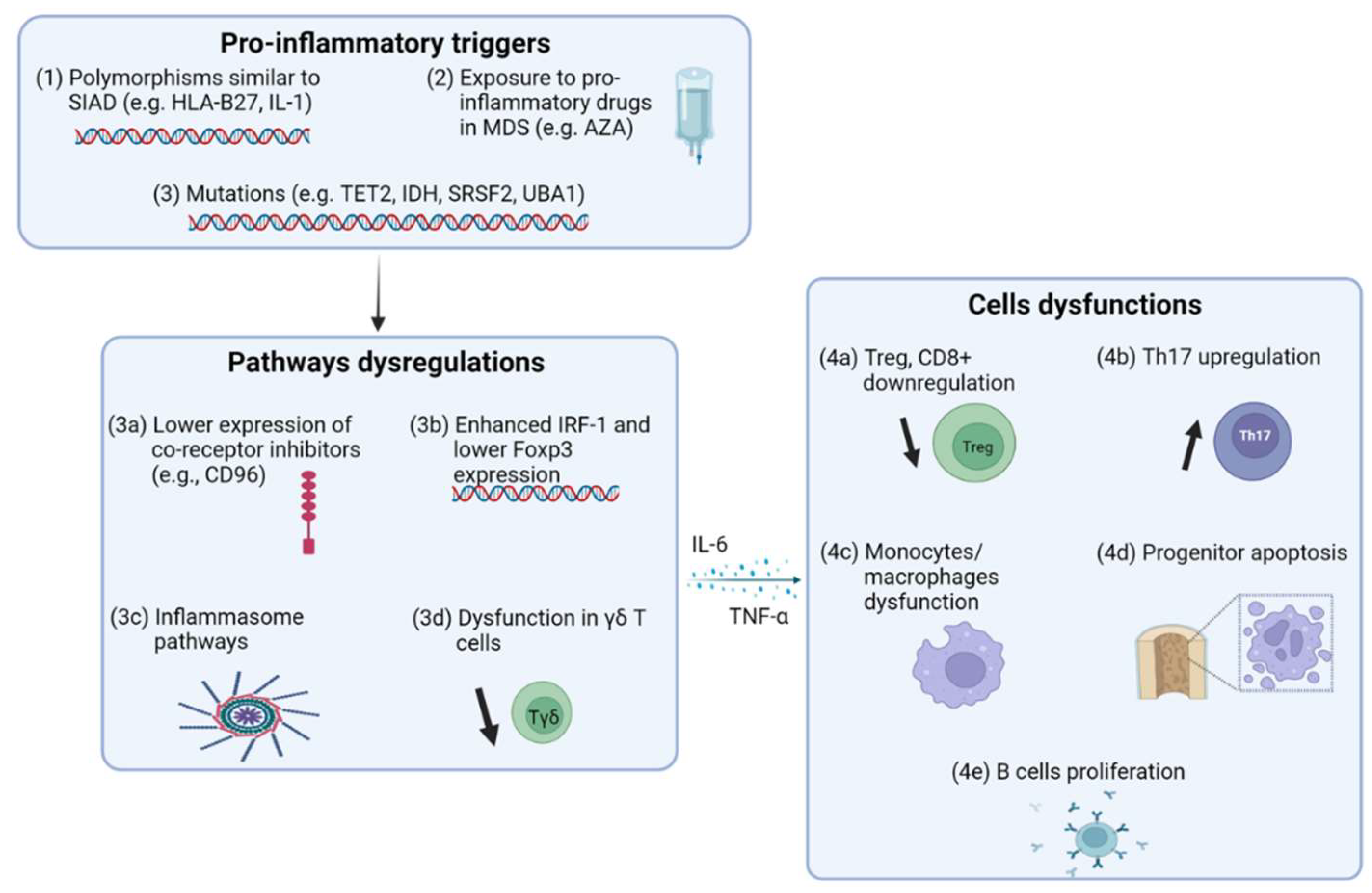
1. Introduction
2. Pro-Inflammatory State in Myelodysplastic Syndrome
3. Pro-Myelodysplastic State in Inflammation
4. Comparison between MDS with and without SIAD
3. VEXAS Syndrome
3.1. Is the MDS Homeostasis in VEXAS Similar to Other MDS?
3.2. Therapeutic Approach
3.2. Is there a Place to Treat the Clone in VEXAS without MDS?
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Beck DB, Ferrada MA, Sikora KA, Ombrello AK, Collins JC, Pei W, et al. Somatic Mutations in UBA1 and Severe Adult-Onset Autoinflammatory Disease. N Engl J Med 2020;383:2628–38. [CrossRef]
- Braun T, Fenaux P. Myelodysplastic Syndromes (MDS) and autoimmune disorders (AD): cause or consequence? Best Pract Res Clin Haematol 2013;26:327–36. [CrossRef]
- Haferlach T, Nagata Y, Grossmann V, Okuno Y, Bacher U, Nagae G, et al. Landscape of genetic lesions in 944 patients with myelodysplastic syndromes. Leukemia 2014;28:241–7. [CrossRef]
- Garcia-Manero G. Myelodysplastic syndromes: 2023 update on diagnosis, risk-stratification, and management. Am J Hematol 2023;98:1307–25. [CrossRef]
- Stalder G, Ribi C, Alberio L. Syndromes myélodysplasiques et auto-immunité. Rev Med Suisse 2015;469:820–5.
- Au WY, Hawkins BR, Cheng N, Lie AK, Liang R, Kwong YL. Risk of haematological malignancies in HLA-B27 carriers. Br J Haematol 2001;115:320–2. [CrossRef]
- Cai L, Zhang J, Xue X, Wang Z, Wang J, Tang S, et al. Meta-analysis of associations of IL1 receptor antagonist and estrogen receptor gene polymorphisms with systemic lupus erythematosus susceptibility. PloS One 2014;9:e109712. [CrossRef]
- Bodis G, Toth V, Schwarting A. Role of Human Leukocyte Antigens (HLA) in Autoimmune Diseases. Rheumatol Ther 2018;5:5–20. [CrossRef]
- Demeter J, Messer G, Rämisch S, Mee JB, di Giovine FS, Schmid M, et al. Polymorphism within the second intron of the IL-1 receptor antagonist gene in patients with hematopoietic malignancies. Cytokines Mol Ther 1996;2:239–42.
- Huang D, Pirskanen R, Hjelmström P, Lefvert AK. Polymorphisms in IL-1beta and IL-1 receptor antagonist genes are associated with myasthenia gravis. J Neuroimmunol 1998;81:76–81. [CrossRef]
- Kordasti SY, Afzali B, Lim Z, Ingram W, Hayden J, Barber L, et al. IL-17-producing CD4(+) T cells, pro-inflammatory cytokines and apoptosis are increased in low risk myelodysplastic syndrome. Br J Haematol 2009;145:64–72. [CrossRef]
- Kordasti SY, Ingram W, Hayden J, Darling D, Barber L, Afzali B, et al. CD4+CD25high Foxp3+ regulatory T cells in myelodysplastic syndrome (MDS). Blood 2007;110:847–50. [CrossRef]
- Maciejewski JP, Selleri C, Sato T, Anderson S, Young NS. Increased expression of Fas antigen on bone marrow CD34+ cells of patients with aplastic anaemia. Br J Haematol 1995;91:245–52. [CrossRef]
- Zeng W, Miyazato A, Chen G, Kajigaya S, Young NS, Maciejewski JP. Interferon-γ-induced gene expression in CD34 cells: identification of pathologic cytokine-specific signature profiles. Blood 2006;107:167–75. [CrossRef]
- Kitagawa M, Saito I, Kuwata T, Yoshida S, Yamaguchi S, Takahashi M, et al. Overexpression of tumor necrosis factor (TNF)-alpha and interferon (IFN)-gamma by bone marrow cells from patients with myelodysplastic syndromes. Leukemia 1997;11:2049–54. [CrossRef]
- Gañán-Gómez I, Wei Y, Starczynowski DT, Colla S, Yang H, Cabrero-Calvo M, et al. Deregulation of innate immune and inflammatory signaling in myelodysplastic syndromes. Leukemia 2015;29:1458–69. [CrossRef]
- Zhang Q, Zhao K, Shen Q, Han Y, Gu Y, Li X, et al. Tet2 is required to resolve inflammation by recruiting Hdac2 to specifically repress IL-6. Nature 2015;525:389–93. [CrossRef]
- Shiromizu CM, Jancic CC. γδ T Lymphocytes: An Effector Cell in Autoimmunity and Infection. Front Immunol 2018;9:2389. [CrossRef]
- Jachiet V, Fenaux P, Sevoyan A, Hakobyan Y, Ades L, Fain O, et al. Inflammatory and Immune Disorders Associated with Myelodysplastic Syndromes. Hemato 2021;2:329–46. [CrossRef]
- Yue X, Trifari S, Äijö T, Tsagaratou A, Pastor WA, Zepeda-Martínez JA, et al. Control of Foxp3 stability through modulation of TET activity. J Exp Med 2016;213:377–97. [CrossRef]
- Yue X, Rao A. TET family dioxygenases and the TET activator vitamin C in immune responses and cancer. Blood 2020;136:1394–401. [CrossRef]
- Kiladjian J-J, Visentin G, Viey E, Chevret S, Eclache V, Stirnemann J, et al. Activation of cytotoxic T-cell receptor γδ T lymphocytes in response to specific stimulation in myelodysplastic syndromes. Haematologica 2008;93:381–9. [CrossRef]
- Chan, C. J. et al. The receptors CD96 and CD226 oppose each other in the regulation of natural killer cell functions. Nature immunology - Recherche Google n.d. https://www.google.com/search?client=firefox-b-d&q=Chan%2C+C.+J.+et+al.+The+receptors+CD96+and+CD226+oppose+each+other+in+the+regulation+of+natural+killer+cell+functions.+Nature+immunology (accessed June 10, 2024).
- Vallelonga V, Gandolfi F, Ficara F, Della Porta MG, Ghisletti S. Emerging Insights into Molecular Mechanisms of Inflammation in Myelodysplastic Syndromes. Biomedicines 2023;11:2613. [CrossRef]
- Anderson LA, Pfeiffer RM, Landgren O, Gadalla S, Berndt SI, Engels EA. Risks of myeloid malignancies in patients with autoimmune conditions. Br J Cancer 2009;100:822–8. [CrossRef]
- Kristinsson SY, Björkholm M, Hultcrantz M, Derolf ÅR, Landgren O, Goldin LR. Chronic immune stimulation might act as a trigger for the development of acute myeloid leukemia or myelodysplastic syndromes. J Clin Oncol Off J Am Soc Clin Oncol 2011;29:2897–903. [CrossRef]
- Zhao L-P, Boy M, Azoulay C, Clappier E, Sébert M, Amable L, et al. Genomic landscape of MDS/CMML associated with systemic inflammatory and autoimmune disease. Leukemia 2021;35:2720–4. [CrossRef]
- Fraison J-B, Grignano E, Braun T, Adès L, Chollet-Martin S, Roland-Nicaise P, et al. Autoantibodies in myelodysplastic syndromes and chronic myelomonocytic leukemia. Leuk Lymphoma 2019;60:2594–6. [CrossRef]
- Hamidou MA, Derenne S, Audrain MA, Berthelot JM, Boumalassa A, Grolleau JY. Prevalence of rheumatic manifestations and antineutrophil cytoplasmic antibodies in haematological malignancies. A prospective study. Rheumatol Oxf Engl 2000;39:417–20. [CrossRef]
- Lin Y-C, Liang T-H, Chang H-N, Lin J-S, Lin H-Y. Behçet disease associated with myelodysplastic syndrome. J Clin Rheumatol Pract Rep Rheum Musculoskelet Dis 2008;14:169–74. [CrossRef]
- Oh Y-J, Shin D-Y, Hwang SM, Kim S-M, Im K, Park HS, et al. Mutation of ten-eleven translocation-2 is associated with increased risk of autoimmune disease in patients with myelodysplastic syndrome. Korean J Intern Med 2020;35:457–64. [CrossRef]
- Beck DB, Werner A, Kastner DL, Aksentijevich I. Disorders of ubiquitylation: unchained inflammation. Nat Rev Rheumatol 2022;18:435–47. [CrossRef]
- Gutierrez-Rodrigues F, Kusne Y, Fernandez J, Lasho T, Shalhoub R, Ma X, et al. Spectrum of clonal hematopoiesis in VEXAS syndrome. Blood 2023;142:244–59. [CrossRef]
- Ferrada MA, Savic S, Cardona DO, Collins JC, Alessi H, Gutierrez-Rodrigues F, et al. Translation of cytoplasmic UBA1 contributes to VEXAS syndrome pathogenesis. Blood 2022;140:1496–506. [CrossRef]
- Roupie AL, Guedon A, Terrier B, Lahuna C, Jachiet V, Regent A, et al. Vasculitis associated with myelodysplastic syndrome and chronic myelomonocytic leukemia: French multicenter case-control study. Semin Arthritis Rheum 2020;50:879–84. [CrossRef]
- Caratsch L, Wolff L, Comte D, Lötscher F, Seitz L, Seitz P, et al. VEXAS syndrome: a Swiss national retrospective cohort study 2024. [CrossRef]
- Georgin-Lavialle S, Terrier B, Guedon AF, Heiblig M, Comont T, Lazaro E, et al. Further characterization of clinical and laboratory features in VEXAS syndrome: large-scale analysis of a multicentre case series of 116 French patients. Br J Dermatol 2022;186:564–74. [CrossRef]
- Fraison J-B, Mekinian A, Grignano E, Kahn J-E, Arlet J-B, Decaux O, et al. Efficacy of Azacitidine in autoimmune and inflammatory disorders associated with myelodysplastic syndromes and chronic myelomonocytic leukemia. Leuk Res 2016;43:13–7. [CrossRef]
- Costantini B, Kordasti SY, Kulasekararaj AG, Jiang J, Seidl T, Abellan PP, et al. The effects of 5-azacytidine on the function and number of regulatory T cells and T-effectors in myelodysplastic syndrome. Haematologica 2013;98:1196–205. [CrossRef]
- Heiblig M, Ferrada MA, Koster MJ, Barba T, Gerfaud-Valentin M, Mékinian A, et al. Ruxolitinib is more effective than other JAK Inhibitors to treat VEXAS Syndrome: a retrospective multi center study. Blood 2022:blood.2022016642. [CrossRef]
- Cerchione C, Romano A, Daver N, DiNardo C, Jabbour EJ, Konopleva M, et al. IDH1/IDH2 Inhibition in Acute Myeloid Leukemia. Front Oncol 2021;11:639387. [CrossRef]
- Jachiet V, Kosmider O, Heiblig M, Terrier B, Le Guenno G, Outh R, et al. Efficacité et tolérance de l’azacitidine au cours du syndrome VEXAS avec et sans syndrome myélodysplasique : données du registre français. Rev Médecine Interne 2023;44:A356–7. [CrossRef]
- Maeda A, Tsuchida N, Uchiyama Y, Horita N, Kobayashi S, Kishimoto M, et al. Efficient detection of somatic UBA1 variants and clinical scoring system predicting patients with variants in VEXAS syndrome. Rheumatol Oxf Engl 2023:kead425. [CrossRef]
- Mascaro JM et al. Spanish cohort of VEXAS syndrome: clinical manifestations, outcome of treatments and novel evidences about UBA1 mosaicism. Ann Rheum Dis. 2023 Dec;82(12):1594–605 - Recherche Google n.d. https://www.google.com/search?client=firefox-b-d&q=Mascaro+JM+et+al.+Spanish+cohort+of+VEXAS+syndrome%3A+clinical+manifestations%2C+outcome+of+treatments+and+novel+evidences+about+UBA1+mosaicism.+Ann+Rheum+Dis.+2023+Dec%3B82%2812%29%3A1594%E2%80%93605 (accessed June 12, 2024).
- A M, E G, T B, O D, E L, N C-C, et al. Systemic inflammatory and autoimmune manifestations associated with myelodysplastic syndromes and chronic myelomonocytic leukaemia: a French multicentre retrospective study. Rheumatol Oxf Engl 2016;55. [CrossRef]
- Giannouli S, Voulgarelis M, Zintzaras E, Tzioufas AG, Moutsopoulos HM. Autoimmune phenomena in myelodysplastic syndromes: a 4-yr prospective study. Rheumatol Oxf Engl 2004;43:626–32. [CrossRef]
- Hollanda A de, Beucher A, Henrion D, Ghali A, Lavigne C, Lévesque H, et al. Systemic and immune manifestations in myelodysplasia: A multicenter retrospective study. Arthritis Care Res 2011;63:1188–94. [CrossRef]
- Komrokji RS, Kulasekararaj A, Al Ali NH, Kordasti S, Bart-Smith E, Craig BM, et al. Autoimmune diseases and myelodysplastic syndromes. Am J Hematol 2016;91:E280-283. [CrossRef]
| Factors contributing to pro-inflammatory state in MDS | Factors contributing to MDS in pro-inflammatory state | |
|---|---|---|
| Temporality | MDS may precede the diagnosis of AI | |
| Genetic environment | Both AI and MDS share a similar genetic background (e.g., HLA-B27 or IL-1 polymorphisms) | |
| Homeostasis | MDS environment is pro-inflammatory, with elevated levels of pro-inflammatory cytokines (e.g., IL-1, Th17) and mutations predisposing to tolerance dysfunction (e.g., TET2/IDH mutations with less Treg and immune checkpoint inhibitors) | Inflammation, even caused by infection, may increase the risk for MDS |
| Treatment induced | Drugs used to treat MDS may trigger AI manifestations (e.g., Azacytidine) | Drugs used to treat inflammation may predispose to MDS. |
| Improvement under treatments | In MDS with AI features, treatment of MDS may alleviate AI manifestations | |
| Impaired Immune Function | MDS is associated with immune dysfunction, resulting in a higher susceptibility to infections. Chronic infections can contribute to inflammation | |
| Authors | Number of patients (MDS vs MDS/AI) | Country | Study | Outcome |
|---|---|---|---|---|
| Giannouli et al. | 57 vs 13 | Greece | Prospective | No difference |
| Hollanda et al. | 189 vs 46 | France | Retrospective | No difference overall but vasculitis subgroup with reduced OS |
| Mekinian et aL | 660 vs 123 | France | Retrospective | No difference |
| Komrokji et al. | 1408 vs 391 | UK, USA | Retrospective | OS were 60 months (95% CI, 50–70) for patients with SIAD vs 45 months (95% CI, 40–49) without (P = 0,006) |
| Manifestations | VEXAS*%£^ | MDS/CMML with SIAD# |
|---|---|---|
| Median age, years, IQR | 64-74 | 70 |
| Male sex, % | 96-100 | 67 |
| Fever, % | 64-92 | 35 |
| Skin, % | 72-84 | 55 |
| Pulmonary, % | 46-67 | 17 |
| Chondritis, % | 10-54 | - |
| Adenopathy, % | 35-47 | - |
| Joint, % | 47-73 | 70 |
| Ocular, % | 23-57 | 16 |
| Peripheral nervous system, % | 6-15 | 12 |
| Heart, % | 11-12 | - |
| Kidney involvement, % | 6-10 | 10 |
| Venous thromboembolism, % | 35-60 | - |
| Myelodysplastic syndrome, % | 31-71 | 100 |
| Macrocytic anemia, % | 71-97 | - |
| Vacuoles, % | 73-100 | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




