Submitted:
07 October 2024
Posted:
08 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
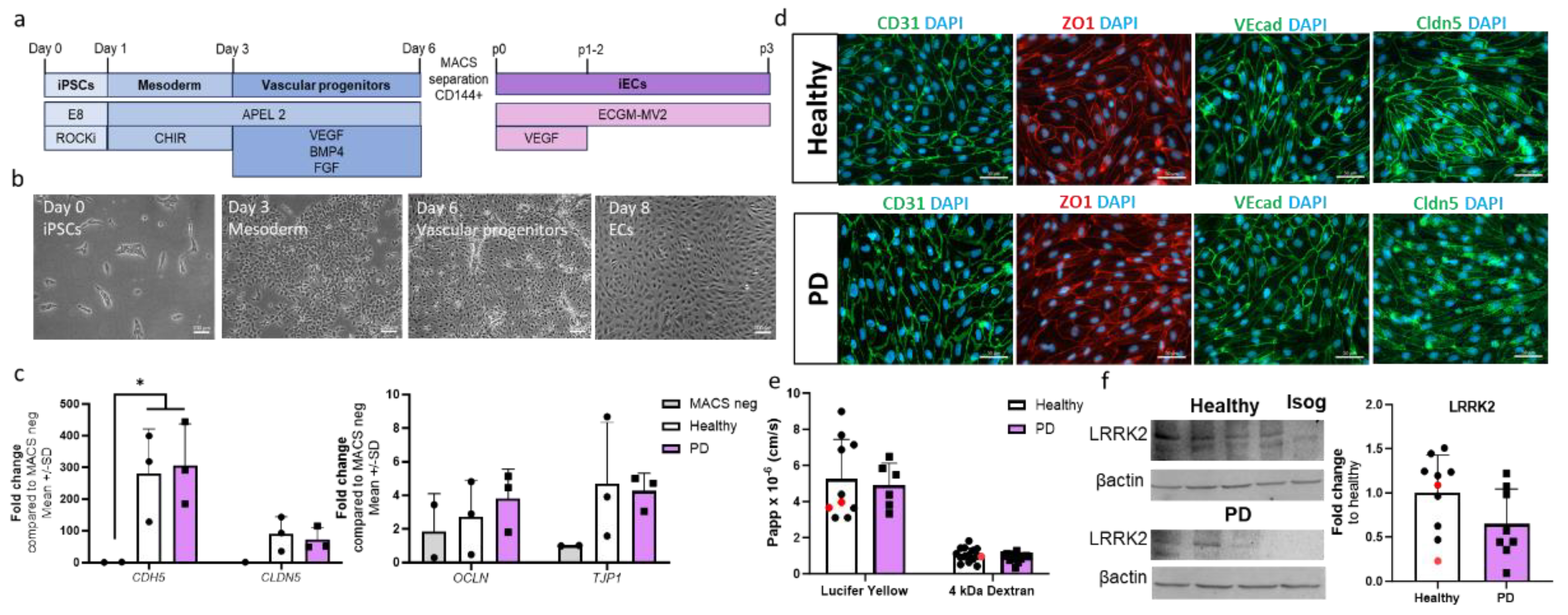
2.1. Differentiation and Characterization of hiPSC-Derived Endothelial Cells
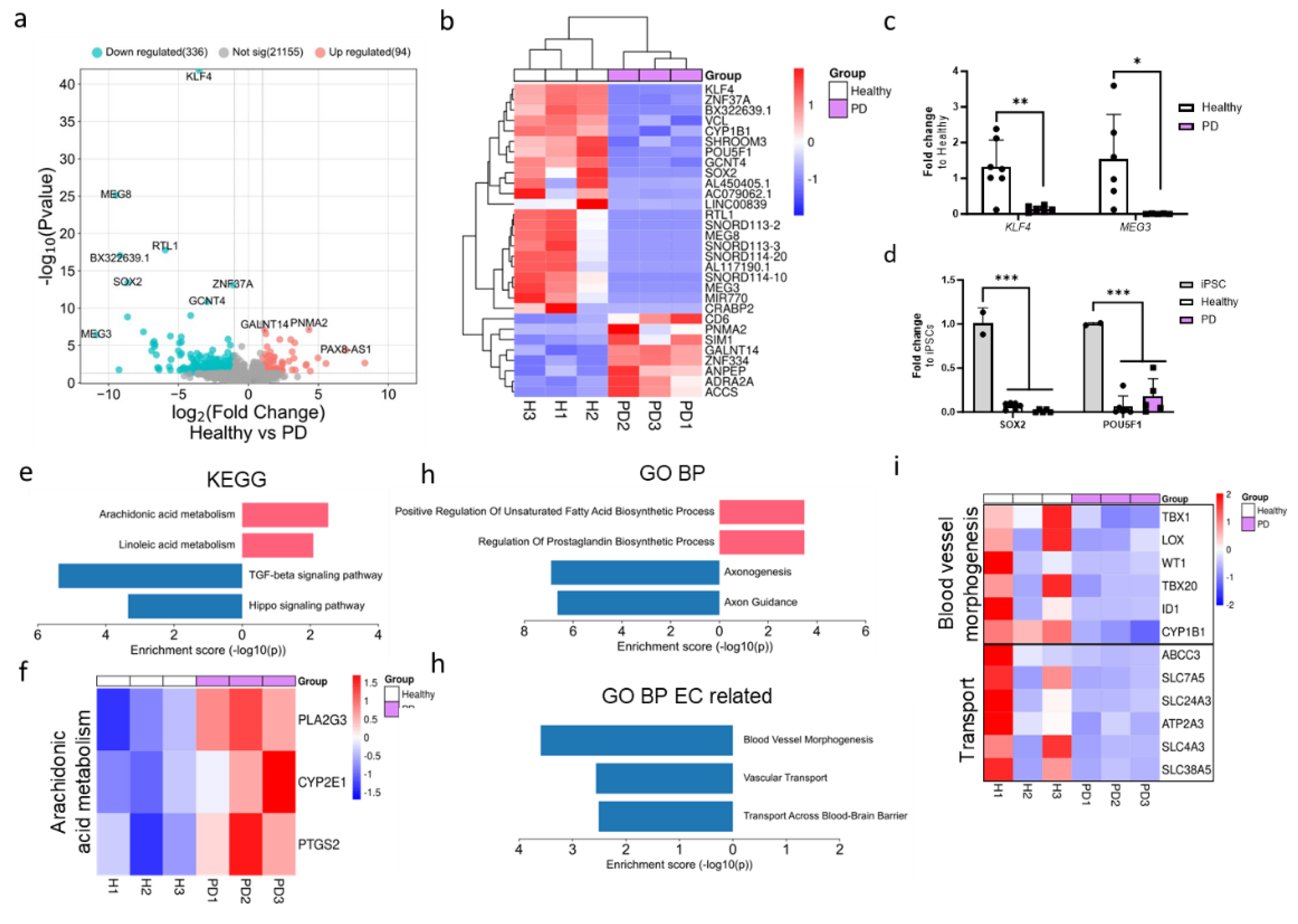
2.2. LRRK2 Mutation induces Moderate Changes in Transcriptomics of Endothelial Cells
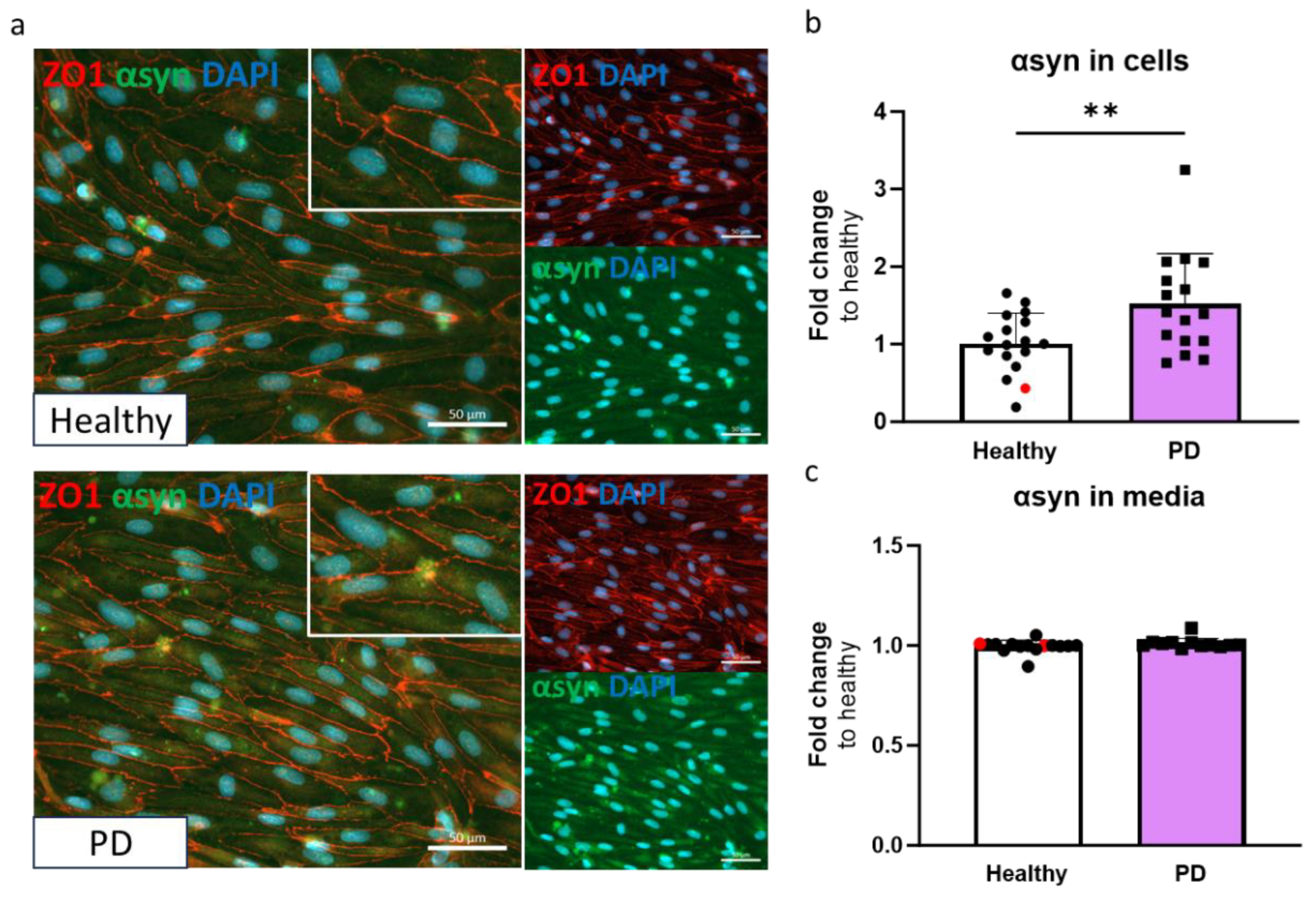
2.3. LRRK2 Mutant Endothelial Cells Have Higher Levels of A-Synuclein
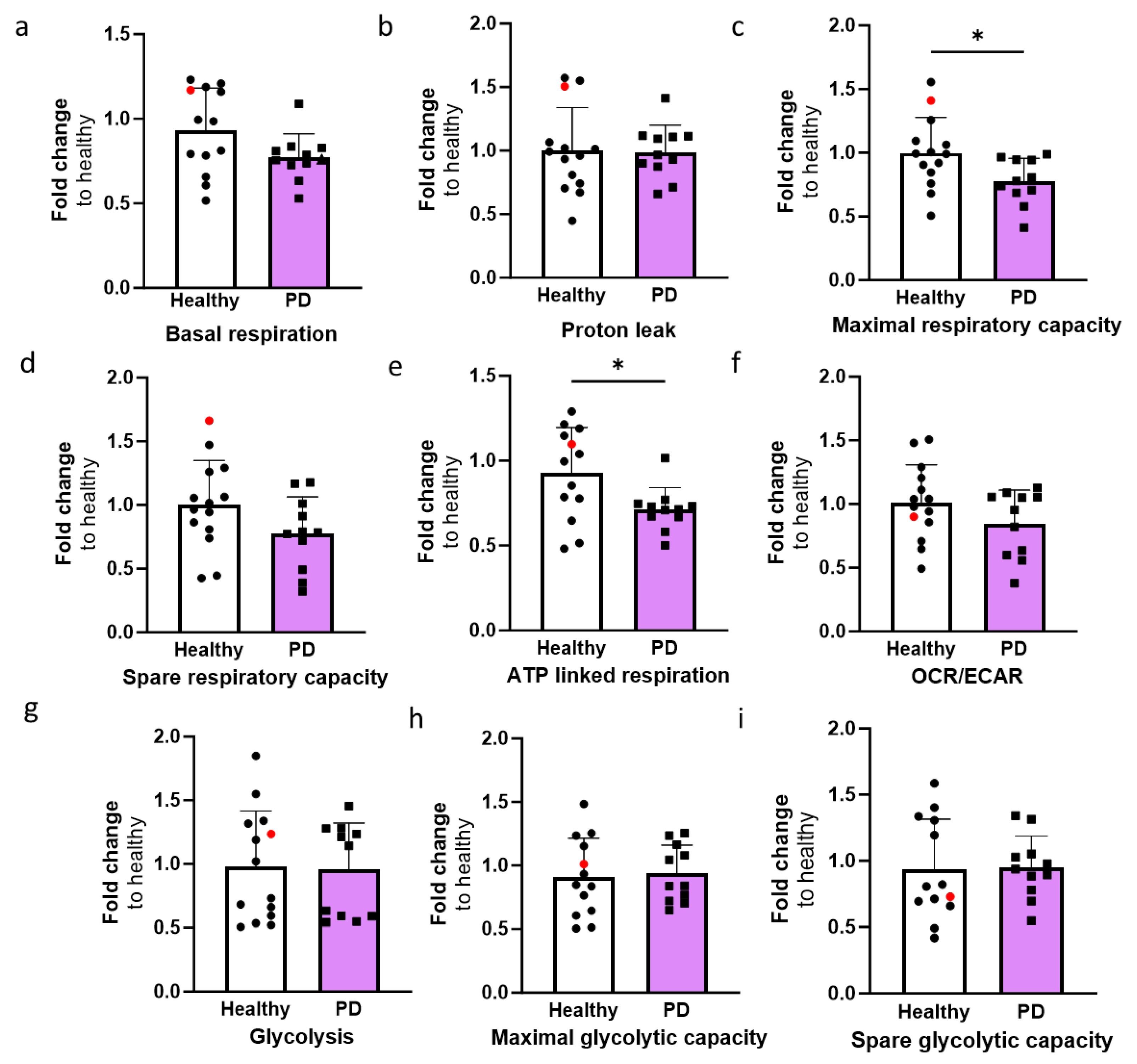
2.4. LRRK2 Mutant Endothelial Cells Have Altered Mitochondrial Respiration
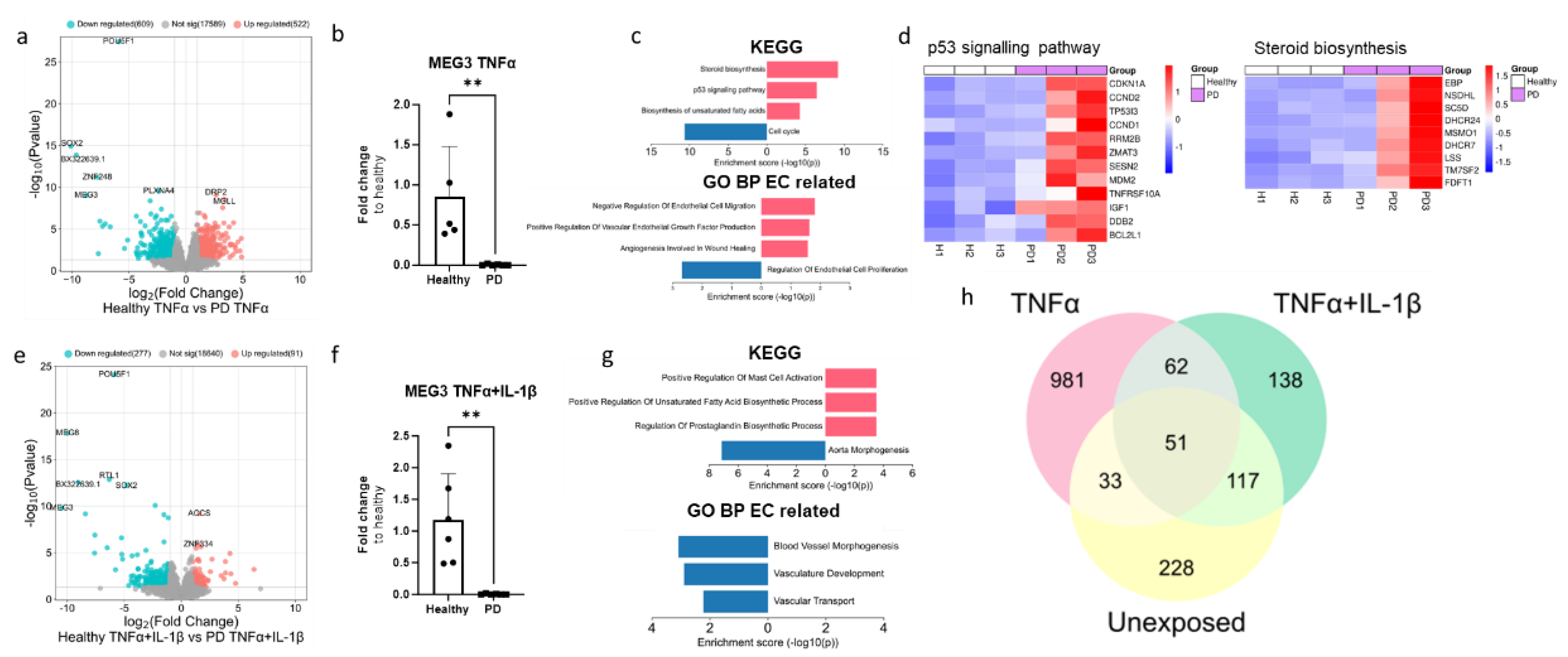
2.4. RNA Transcriptomics Reveal More Prominent effect in PD Endothelial Cells After Tnfα Exposure
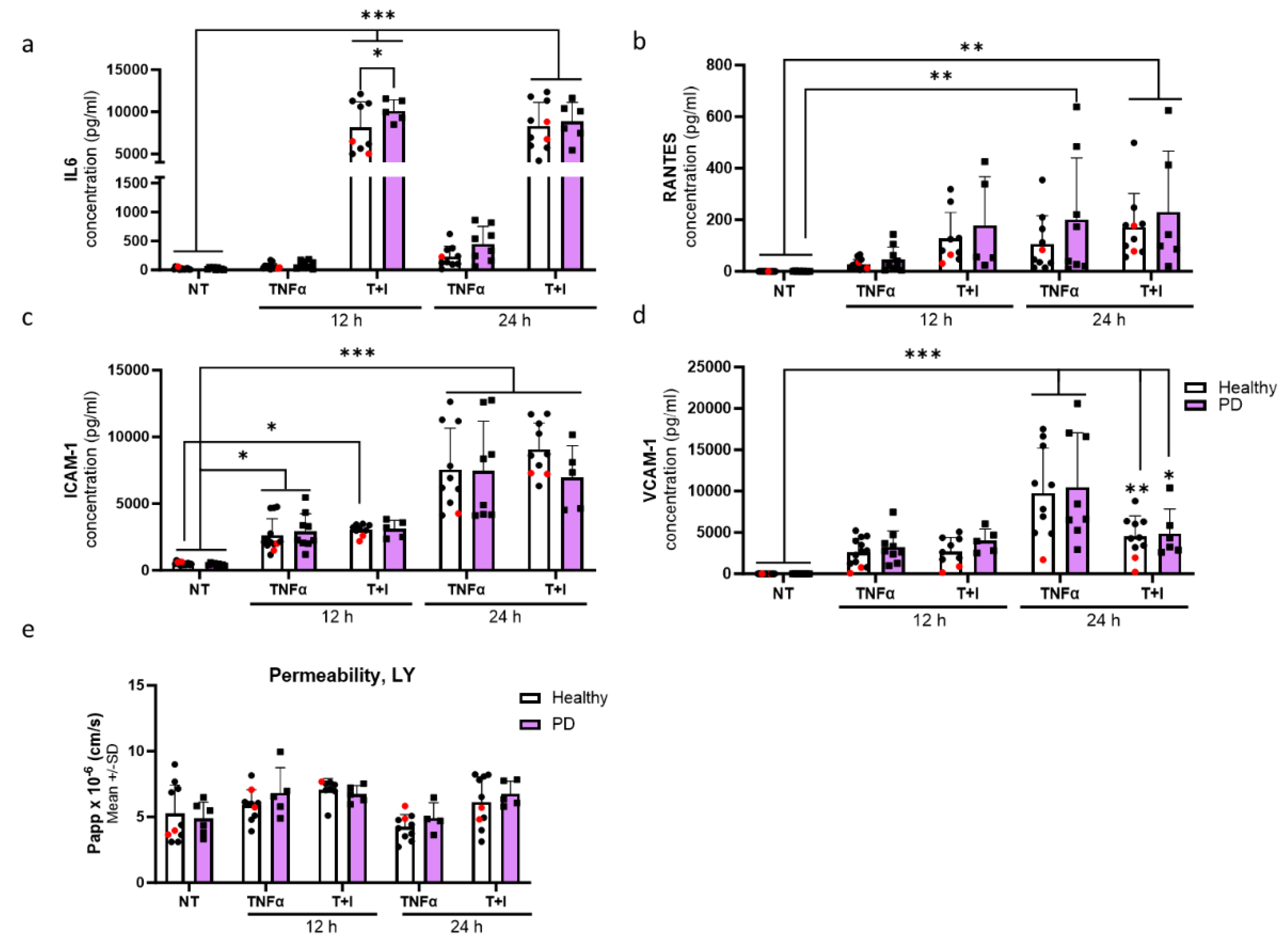
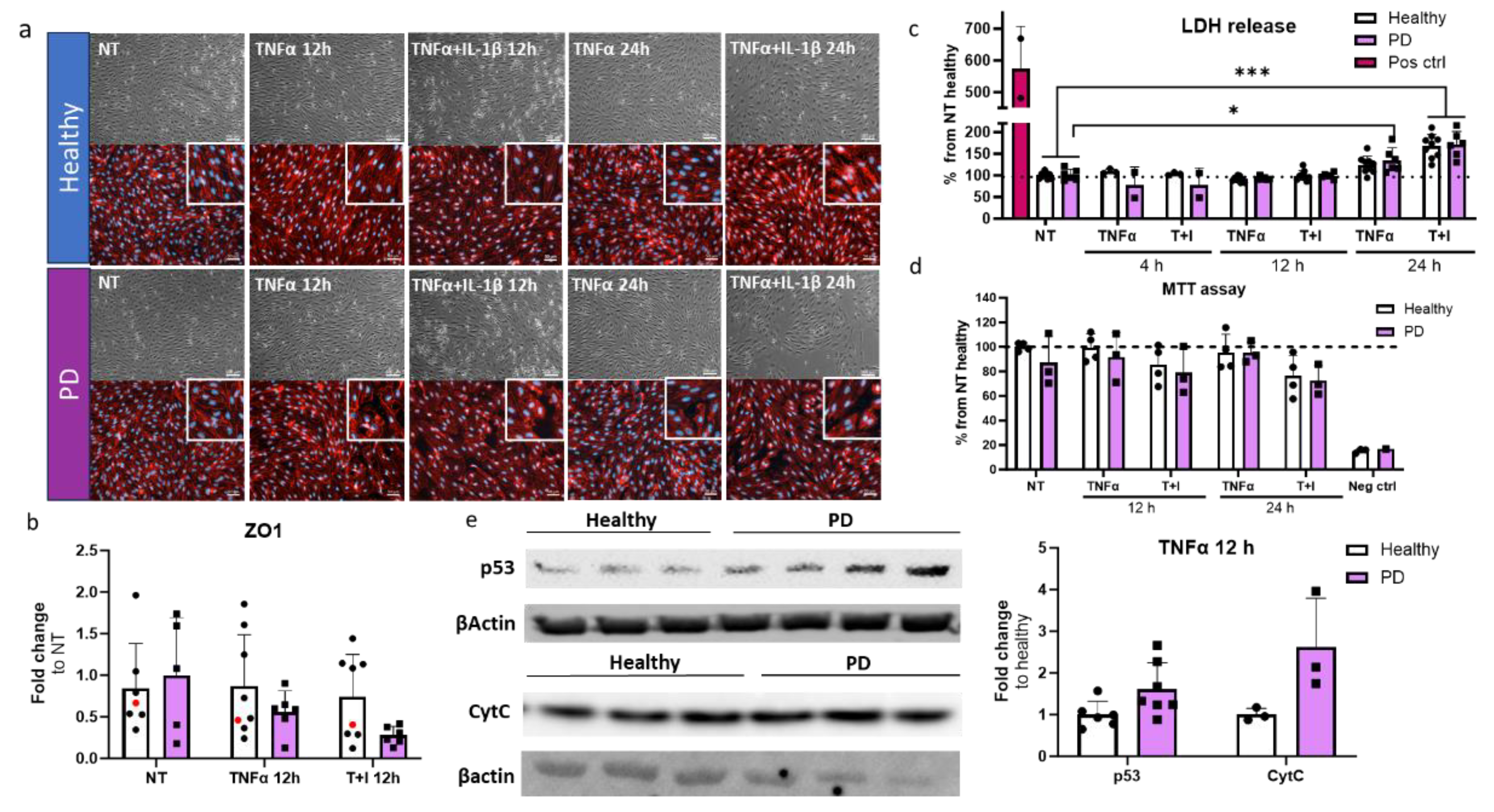
2.5. Prolonged Inflammatory Environment Affects Endothelial Cells Viability
2.6. Endothelial cells Show Aberrant Phenotype In Pro-Inflammatory Conditions
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Endothelial Cell Differentiation
4.3. Immunocytochemistry
4.4. RT-qPCR
4.5. α-Synuclein ELISA
4.6. Seahorse
4.7. Inflmammatory Exposures
4.8. Total RNA Sequencing
4.9. LDH Cytotoxicity Test
4.10. MTT Assay
4.11. Western Blotting
4.12. Quantification of Immunofluorescence
4.13. Cytometric Bead Assay
4.14. Permeability Assay
4.15. Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dorsey, E.R.; Sherer, T.; Okun, M.S.; Bloem, B.R. The Emerging Evidence of the Parkinson Pandemic. J Parkinsons Dis 2018, 8, S3–S8. [Google Scholar] [CrossRef] [PubMed]
- Tysnes, O.-B.; Storstein, A. Epidemiology of Parkinson’s Disease. J Neural Transm (Vienna) 2017, 124, 901–905. [Google Scholar] [CrossRef] [PubMed]
- Selvaraj, S.; Piramanayagam, S. Impact of Gene Mutation in the Development of Parkinson’s Disease. Genes Dis 2019, 6, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Rui, Q.; Ni, H.; Li, D.; Gao, R.; Chen, G. The Role of LRRK2 in Neurodegeneration of Parkinson Disease. Curr Neuropharmacol 2018, 16, 1348–1357. [Google Scholar] [CrossRef] [PubMed]
- Maiti, P.; Manna, J.; Dunbar, G.L. Current Understanding of the Molecular Mechanisms in Parkinson’s Disease: Targets for Potential Treatments. Transl Neurodegener 2017, 6, 28. [Google Scholar] [CrossRef]
- Wu, Y.-C.; Sonninen, T.-M.; Peltonen, S.; Koistinaho, J.; Lehtonen, Š. Blood–Brain Barrier and Neurodegenerative Diseases—Modeling with IPSC-Derived Brain Cells. Int J Mol Sci 2021, 22, 7710. [Google Scholar] [CrossRef]
- Bartels, A.L.; Willemsen, A.T.M.; Kortekaas, R.; de Jong, B.M.; de Vries, R.; de Klerk, O.; van Oostrom, J.C.H.; Portman, A.; Leenders, K.L. Decreased Blood-Brain Barrier P-Glycoprotein Function in the Progression of Parkinson’s Disease, PSP and MSA. J Neural Transm (Vienna) 2008, 115, 1001–1009. [Google Scholar] [CrossRef]
- Kortekaas, R.; Leenders, K.L.; van Oostrom, J.C.H.; Vaalburg, W.; Bart, J.; Willemsen, A.T.M.; Hendrikse, N.H. Blood-Brain Barrier Dysfunction in Parkinsonian Midbrain in Vivo. Ann Neurol 2005, 57, 176–179. [Google Scholar] [CrossRef]
- Kim, H.; Shin, J.-Y.; Lee, Y.-S.; Yun, S.P.; Maeng, H.-J.; Lee, Y. Brain Endothelial P-Glycoprotein Level Is Reduced in Parkinson’s Disease via a Vitamin D Receptor-Dependent Pathway. Int J Mol Sci 2020, 21, 8538. [Google Scholar] [CrossRef]
- Ham, J.H.; Yi, H.; Sunwoo, M.K.; Hong, J.Y.; Sohn, Y.H.; Lee, P.H. Cerebral Microbleeds in Patients with Parkinson’s Disease. J Neurol 2014, 261, 1628–1635. [Google Scholar] [CrossRef]
- Al-Bachari, S.; Vidyasagar, R.; Emsley, H.; Parkes, L. PO071 Mri Assessment of Neurovascular Changes in Idiopathic Parkinson’s Disease. J Neurol Neurosurg Psychiatry 2017, 88, A30–A30. [Google Scholar] [CrossRef]
- Gray, M.T.; Woulfe, J.M. Striatal Blood-Brain Barrier Permeability in Parkinson’s Disease. J Cereb Blood Flow Metab 2015, 35, 747–750. [Google Scholar] [CrossRef] [PubMed]
- Pienaar, I.S.; Lee, C.H.; Elson, J.L.; McGuinness, L.; Gentleman, S.M.; Kalaria, R.N.; Dexter, D.T. Deep-Brain Stimulation Associates with Improved Microvascular Integrity in the Subthalamic Nucleus in Parkinson’s Disease. Neurobiol Dis 2015, 74, 392–405. [Google Scholar] [CrossRef] [PubMed]
- Carvey, P.M.; Zhao, C.H.; Hendey, B.; Lum, H.; Trachtenberg, J.; Desai, B.S.; Snyder, J.; Zhu, Y.G.; Ling, Z.D. 6-Hydroxydopamine-Induced Alterations in Blood-Brain Barrier Permeability. Eur J Neurosci 2005, 22, 1158–1168. [Google Scholar] [CrossRef]
- Olmedo-Díaz, S.; Estévez-Silva, H.; Orädd, G.; Af Bjerkén, S.; Marcellino, D.; Virel, A. An Altered Blood-Brain Barrier Contributes to Brain Iron Accumulation and Neuroinflammation in the 6-OHDA Rat Model of Parkinson’s Disease. Neuroscience 2017, 362, 141–151. [Google Scholar] [CrossRef]
- Chung, Y.C.; Kim, Y.-S.; Bok, E.; Yune, T.Y.; Maeng, S.; Jin, B.K. MMP-3 Contributes to Nigrostriatal Dopaminergic Neuronal Loss, BBB Damage, and Neuroinflammation in an MPTP Mouse Model of Parkinson’s Disease. Mediators Inflamm 2013, 2013, 370526. [Google Scholar] [CrossRef]
- Zhao, C.; Ling, Z.; Newman, M.B.; Bhatia, A.; Carvey, P.M. TNF-Alpha Knockout and Minocycline Treatment Attenuates Blood-Brain Barrier Leakage in MPTP-Treated Mice. Neurobiol Dis 2007, 26, 36–46. [Google Scholar] [CrossRef]
- Wong, D.; Dorovini-Zis, K.; Vincent, S.R. Cytokines, Nitric Oxide, and CGMP Modulate the Permeability of an in Vitro Model of the Human Blood-Brain Barrier. Exp Neurol 2004, 190, 446–455. [Google Scholar] [CrossRef]
- Harding, A.; Cortez-Toledo, E.; Magner, N.L.; Beegle, J.R.; Coleal-Bergum, D.P.; Hao, D.; Wang, A.; Nolta, J.A.; Zhou, P. Highly Efficient Differentiation of Endothelial Cells from Pluripotent Stem Cells Requires the MAPK and the PI3K Pathways. Stem Cells 2017, 35, 909–919. [Google Scholar] [CrossRef]
- Sonninen, T.-M.; Hämäläinen, R.H.; Koskuvi, M.; Oksanen, M.; Shakirzyanova, A.; Wojciechowski, S.; Puttonen, K.; Naumenko, N.; Goldsteins, G.; Laham-Karam, N.; et al. Metabolic Alterations in Parkinson’s Disease Astrocytes. Sci Rep 2020, 10, 14474. [Google Scholar] [CrossRef]
- di Domenico, A.; Carola, G.; Calatayud, C.; Pons-Espinal, M.; Muñoz, J.P.; Richaud-Patin, Y.; Fernandez-Carasa, I.; Gut, M.; Faella, A.; Parameswaran, J.; et al. Patient-Specific IPSC-Derived Astrocytes Contribute to Non-Cell-Autonomous Neurodegeneration in Parkinson’s Disease. Stem Cell Reports 2019, 12, 213–229. [Google Scholar] [CrossRef] [PubMed]
- Ohtonen, S.; Giudice, L.; Jäntti, H.; Fazaludeen, M.F.; Shakirzyanova, A.; Gómez-Budia, M.; Välimäki, N.-N.; Niskanen, J.; Korvenlaita, N.; Fagerlund, I.; et al. Human IPSC-Derived Microglia Carrying the LRRK2-G2019S Mutation Show a Parkinson’s Disease Related Transcriptional Profile and Function. Sci Rep 2023, 13, 22118. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Liu, L.; Qin, L.; Huang, H.; Li, X. Single-Cell Transcriptomics Uncovers Cellular Heterogeneity, Mechanisms, and Therapeutic Targets for Parkinson’s Disease. Front Genet 2022, 13, 686739. [Google Scholar] [CrossRef] [PubMed]
- Zamanian, M.Y.; Golmohammadi, M.; Amin, R.S.; Bustani, G.S.; Romero-Parra, R.M.; Zabibah, R.S.; Oz, T.; Jalil, A.T.; Soltani, A.; Kujawska, M. Therapeutic Targeting of Krüppel-Like Factor 4 and Its Pharmacological Potential in Parkinson’s Disease: A Comprehensive Review. Mol Neurobiol 2023. [Google Scholar] [CrossRef]
- Tugal, D.; Jain, M.K.; Simon, D.I. Endothelial KLF4: Crippling Vascular Injury? Journal of the American Heart Association: Cardiovascular and Cerebrovascular Disease 2014, 3. [Google Scholar] [CrossRef]
- Huang, T.; Yin, J.; Ren, S.; Zhang, X. Protective Effects of KLF4 on Blood-Brain Barrier and Oxidative Stress after Cerebral Ischemia-Reperfusion in Rats through the Nrf2/Trx1 Pathway. Cytokine 2023, 169, 156288. [Google Scholar] [CrossRef]
- Yang, H.; Xi, X.; Zhao, B.; Su, Z.; Wang, Z. KLF4 Protects Brain Microvascular Endothelial Cells from Ischemic Stroke Induced Apoptosis by Transcriptionally Activating MALAT1. Biochemical and Biophysical Research Communications 2018, 495, 2376–2382. [Google Scholar] [CrossRef]
- Hamik, A.; Lin, Z.; Kumar, A.; Balcells, M.; Sinha, S.; Katz, J.; Feinberg, M.W.; Gerszten, R.E.; Edelman, E.R.; Jain, M.K. Kruppel-like Factor 4 Regulates Endothelial Inflammation*. Journal of Biological Chemistry 2007, 282, 13769–13779. [Google Scholar] [CrossRef]
- Huang, H.; Zheng, S.; Lu, M. Downregulation of LncRNA MEG3 Is Involved in Parkinson’s Disease. Metab Brain Dis 2021, 36, 2323–2328. [Google Scholar] [CrossRef]
- Quan, Y.; Wang, J.; Wang, S.; Zhao, J. Association of the Plasma Long Non-Coding RNA MEG3 With Parkinson’s Disease. Front Neurol 2020, 11, 532891. [Google Scholar] [CrossRef]
- Honarmand Tamizkar, K.; Gorji, P.; Gholipour, M.; Hussen, B.M.; Mazdeh, M.; Eslami, S.; Taheri, M.; Ghafouri-Fard, S. Parkinson’s Disease Is Associated With Dysregulation of Circulatory Levels of LncRNAs. Front Immunol 2021, 12, 763323. [Google Scholar] [CrossRef] [PubMed]
- Lan, Y.; Li, Y.-J.; Li, D.-J.; Li, P.; Wang, J.-Y.; Diao, Y.-P.; Ye, G.-D.; Li, Y.-F. Long Noncoding RNA MEG3 Prevents Vascular Endothelial Cell Senescence by Impairing MiR-128-Dependent Girdin Downregulation. American Journal of Physiology-Cell Physiology 2019, 316, C830–C843. [Google Scholar] [CrossRef] [PubMed]
- Ruan, W.; Zhao, F.; Zhao, S.; Zhang, L.; Shi, L.; Pang, T. Knockdown of Long Noncoding RNA MEG3 Impairs VEGF-Stimulated Endothelial Sprouting Angiogenesis via Modulating VEGFR2 Expression in Human Umbilical Vein Endothelial Cells. Gene 2018, 649, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Qiu, G.-Z.; Tian, W.; Fu, H.-T.; Li, C.-P.; Liu, B. Long Noncoding RNA-MEG3 Is Involved in Diabetes Mellitus-Related Microvascular Dysfunction. Biochem Biophys Res Commun 2016, 471, 135–141. [Google Scholar] [CrossRef]
- Song, J.; Huang, S.; Wang, K.; Li, W.; Pao, L.; Chen, F.; Zhao, X. Long Non-Coding RNA MEG3 Attenuates the Angiotensin II-Induced Injury of Human Umbilical Vein Endothelial Cells by Interacting With P53. Front Genet 2019, 10, 78. [Google Scholar] [CrossRef]
- Cheng, X.; Shihabudeen Haider Ali, M.S.; Moran, M.; Viana, M.P.; Schlichte, S.L.; Zimmerman, M.C.; Khalimonchuk, O.; Feinberg, M.W.; Sun, X. Long Non-Coding RNA Meg3 Deficiency Impairs Glucose Homeostasis and Insulin Signaling by Inducing Cellular Senescence of Hepatic Endothelium in Obesity. Redox Biol 2021, 40, 101863. [Google Scholar] [CrossRef]
- Yang, P.; Min, X.-L.; Mohammadi, M.; Turner, C.; Faull, R.; Waldvogel, H.; Dragunow, M.; Guan, J. Endothelial Degeneration of Parkinson’s Disease Is Related to Alpha-Synuclein Aggregation. J Alzheimers Dis Parkinsonism 2017, 7. [Google Scholar] [CrossRef]
- Shi, M.; Liu, C.; Cook, T.J.; Bullock, K.M.; Zhao, Y.; Ginghina, C.; Li, Y.; Aro, P.; Dator, R.; He, C.; et al. Plasma Exosomal α-Synuclein Is Likely CNS-Derived and Increased in Parkinson’s Disease. Acta Neuropathol 2014, 128, 639–650. [Google Scholar] [CrossRef]
- Kawahata, I.; Fukunaga, K. Pathogenic Impact of Fatty Acid-Binding Proteins in Parkinson’s Disease—Potential Biomarkers and Therapeutic Targets. International Journal of Molecular Sciences 2023, 24, 17037. [Google Scholar] [CrossRef]
- Perrin, R.J.; Woods, W.S.; Clayton, D.F.; George, J.M. Exposure to Long Chain Polyunsaturated Fatty Acids Triggers Rapid Multimerization of Synucleins. J Biol Chem 2001, 276, 41958–41962. [Google Scholar] [CrossRef]
- Sharon, R.; Goldberg, M.S.; Bar-Josef, I.; Betensky, R.A.; Shen, J.; Selkoe, D.J. Alpha-Synuclein Occurs in Lipid-Rich High Molecular Weight Complexes, Binds Fatty Acids, and Shows Homology to the Fatty Acid-Binding Proteins. Proc Natl Acad Sci U S A 2001, 98, 9110–9115. [Google Scholar] [CrossRef] [PubMed]
- Sharon, R.; Bar-Joseph, I.; Frosch, M.P.; Walsh, D.M.; Hamilton, J.A.; Selkoe, D.J. The Formation of Highly Soluble Oligomers of Alpha-Synuclein Is Regulated by Fatty Acids and Enhanced in Parkinson’s Disease. Neuron 2003, 37, 583–595. [Google Scholar] [CrossRef] [PubMed]
- Miyake, Y.; Sasaki, S.; Tanaka, K.; Fukushima, W.; Kiyohara, C.; Tsuboi, Y.; Yamada, T.; Oeda, T.; Miki, T.; Kawamura, N.; et al. Dietary Fat Intake and Risk of Parkinson’s Disease: A Case-Control Study in Japan. J Neurol Sci 2010, 288, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Zhi, L.; Zhang, H. LRRK2 and Mitochondria: Recent Advances and Current Views. Brain research 2019, 1702, 96. [Google Scholar] [CrossRef] [PubMed]
- Cooper, O.; Seo, H.; Andrabi, S.; Guardia-Laguarta, C.; Graziotto, J.; Sundberg, M.; McLean, J.R.; Carrillo-Reid, L.; Xie, Z.; Osborn, T.; et al. Familial Parkinson’s Disease IPSCs Show Cellular Deficits in Mitochondrial Responses That Can Be Pharmacologically Rescued. Sci Transl Med 2012, 4, 141ra90. [Google Scholar] [CrossRef]
- Diaz, K.; Kohut, M.L.; Russell, D.W.; Stegemöller, E.L. Peripheral Inflammatory Cytokines and Motor Symptoms in Persons with Parkinson’s Disease. Brain, Behavior, & Immunity - Health 2022, 21, 100442. [Google Scholar] [CrossRef]
- Hofmann, K.W.; Schuh, A.F.S.; Saute, J.; Townsend, R.; Fricke, D.; Leke, R.; Souza, D.O.; Portela, L.V.; Chaves, M.L.F.; Rieder, C.R.M. Interleukin-6 Serum Levels in Patients with Parkinson’s Disease. Neurochem Res 2009, 34, 1401–1404. [Google Scholar] [CrossRef]
- Dufek, M.; Rektorova, I.; Thon, V.; Lokaj, J.; Rektor, I. Interleukin-6 May Contribute to Mortality in Parkinson’s Disease Patients: A 4-Year Prospective Study. Parkinsons Dis 2015, 2015, 898192. [Google Scholar] [CrossRef]
- Scalzo, P.; Kümmer, A.; Cardoso, F.; Teixeira, A.L. Serum Levels of Interleukin-6 Are Elevated in Patients with Parkinson’s Disease and Correlate with Physical Performance. Neuroscience Letters 2010, 468, 56–58. [Google Scholar] [CrossRef]
- Li, H.; Zhang, Z.; Li, H.; Pan, X.; Wang, Y. New Insights into the Roles of P53 in Central Nervous System Diseases. International Journal of Neuropsychopharmacology 2023, 26, 465–473. [Google Scholar] [CrossRef]
- Li, D.-W.; Li, G.-R.; Zhang, B.-L.; Feng, J.-J.; Zhao, H. Damage to Dopaminergic Neurons Is Mediated by Proliferating Cell Nuclear Antigen through the P53 Pathway under Conditions of Oxidative Stress in a Cell Model of Parkinson’s Disease. Int J Mol Med 2016, 37, 429–435. [Google Scholar] [CrossRef] [PubMed]
- Mogi, M.; Kondo, T.; Mizuno, Y.; Nagatsu, T. P53 Protein, Interferon-Gamma, and NF-KappaB Levels Are Elevated in the Parkinsonian Brain. Neurosci Lett 2007, 414, 94–97. [Google Scholar] [CrossRef] [PubMed]
- Sekar, S.; Taghibiglou, C. Nuclear Accumulation of GAPDH, GluA2 and P53 in Post-Mortem Substantia Nigral Region of Patients with Parkinson’s Disease. Neurosci Lett 2020, 716, 134641. [Google Scholar] [CrossRef] [PubMed]
- Schuler, M.; Bossy-Wetzel, E.; Goldstein, J.C.; Fitzgerald, P.; Green, D.R. P53 Induces Apoptosis by Caspase Activation through Mitochondrial Cytochrome c Release. J Biol Chem 2000, 275, 7337–7342. [Google Scholar] [CrossRef]
- Yan, H.; Yuan, J.; Gao, L.; Rao, J.; Hu, J. Long Noncoding RNA MEG3 Activation of P53 Mediates Ischemic Neuronal Death in Stroke. Neuroscience 2016, 337, 191–199. [Google Scholar] [CrossRef]
- Piccoli, M.-T.; Gupta, S.K.; Viereck, J.; Foinquinos, A.; Samolovac, S.; Kramer, F.L.; Garg, A.; Remke, J.; Zimmer, K.; Batkai, S.; et al. Inhibition of the Cardiac Fibroblast-Enriched LncRNA Meg3 Prevents Cardiac Fibrosis and Diastolic Dysfunction. Circ Res 2017, 121, 575–583. [Google Scholar] [CrossRef]
- Shihabudeen Haider Ali, M.S.; Cheng, X.; Moran, M.; Haemmig, S.; Naldrett, M.J.; Alvarez, S.; Feinberg, M.W.; Sun, X. LncRNA Meg3 Protects Endothelial Function by Regulating the DNA Damage Response. Nucleic Acids Res 2019, 47, 1505–1522. [Google Scholar] [CrossRef]
- Nishihara, H.; Gastfriend, B.D.; Soldati, S.; Perriot, S.; Mathias, A.; Sano, Y.; Shimizu, F.; Gosselet, F.; Kanda, T.; Palecek, S.P.; et al. Advancing Human Induced Pluripotent Stem Cell-Derived Blood-Brain Barrier Models for Studying Immune Cell Interactions. FASEB J 2020, 34, 16693–16715. [Google Scholar] [CrossRef]
- Delsing, L.; Dönnes, P.; Sánchez, J.; Clausen, M.; Voulgaris, D.; Falk, A.; Herland, A.; Brolén, G.; Zetterberg, H.; Hicks, R.; et al. Barrier Properties and Transcriptome Expression in Human IPSC-Derived Models of the Blood-Brain Barrier. Stem Cells 2018, 36, 1816–1827. [Google Scholar] [CrossRef]
- Lu, T.M.; Houghton, S.; Magdeldin, T.; Durán, J.G.B.; Minotti, A.P.; Snead, A.; Sproul, A.; Nguyen, D.-H.T.; Xiang, J.; Fine, H.A.; et al. Pluripotent Stem Cell-Derived Epithelium Misidentified as Brain Microvascular Endothelium Requires ETS Factors to Acquire Vascular Fate. Proc Natl Acad Sci U S A 2021, 118. [Google Scholar] [CrossRef]
- Ozgür, B.; Puris, E.; Brachner, A.; Appelt-Menzel, A.; Oerter, S.; Balzer, V.; Holst, M.R.; Christiansen, R.F.; Hyldig, K.; Buckley, S.T.; et al. Characterization of an IPSC-Based Barrier Model for Blood-Brain Barrier Investigations Using the SBAD0201 Stem Cell Line. Fluids and Barriers of the CNS 2023, 20, 96. [Google Scholar] [CrossRef] [PubMed]
- Holmqvist, S.; Lehtonen, Š.; Chumarina, M.; Puttonen, K.A.; Azevedo, C.; Lebedeva, O.; Ruponen, M.; Oksanen, M.; Djelloul, M.; Collin, A.; et al. Creation of a Library of Induced Pluripotent Stem Cells from Parkinsonian Patients. npj Parkinson’s Disease 2016, 2, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Patsch, C.; Challet-Meylan, L.; Thoma, E.C.; Urich, E.; Heckel, T.; O’Sullivan, J.F.; Grainger, S.J.; Kapp, F.G.; Sun, L.; Christensen, K.; et al. Generation of Vascular Endothelial and Smooth Muscle Cells from Human Pluripotent Stem Cells. Nat Cell Biol 2015, 17, 994–1003. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



