3.2.1. Optical Microscope Analyses
The mineralogical analysis of mine tailings is crucial to understand the composition and distribution of the actual minerals, as well as to assess the environmental and economic impacts of mining activities. The following section presents the symbols used to identify the minerals and their overall mineralogical composition by sample. This information is fundamental for the characterization of the tailings and the identification of valuable or potentially toxic minerals.
Table 3 provides the abbreviations used for the identified minerals, facilitating the interpretation of the results.
Table 4 summarizes the overall mineralogical composition of each sample, expressed in percentages. The differences in the presence of primary minerals such as pyrite, rutile, magnetite, and goethite are highlighted, as well as the identification of secondary minerals of interest such as galena, chalcopyrite, arsenopyrite, hematite, sphalerite, and gray copper. These variations suggest the diversity of mineral contents and the potential exploitation of different resources.
Figure 3 show photographs of these minerals.
Samples T1, M1, P1, C2, S1, and M2 exhibit a high concentration of pyrite as the predominant mineral. Differences are highlighted with the additional content of goethite in T1, as well as traces of hematite, chalcopyrite, covellite, chalcocite, and marcasite. For M1, traces of minerals such as hematite, goethite, magnetite, and chalcopyrite. For P1, percentages of goethite, rutile, chalcopyrite, galena, and arsenopyrite. For C2, goethite, rutile, and chalcopyrite. For S1, chalcopyrite along with minerals containing bismuth, lead, zinc, copper, gold, and silver. And for M2, which complementarily contains rutile, chalcopyrite, and arsenopyrite.
On the other hand, sample K1 contains hematite and goethite as the main minerals, and additionally magnetite, pyrite, and rutile, as well as the presence of native gold. Similarly, samples K2 and O1 contain magnetite and pyrite as the main minerals. K2 is notable for having additional rutile, suggesting the presence of titanium. While O1 presents traces of hematite, goethite, and chalcopyrite.
Regarding samples C1, they contain goethite as the main mineral and also present small amounts of pyrite.
Figure 3 illustrates various samples of environmental liabilities and active tailings.
Figure 3(a) shows a photograph of the environmental liability K1, where free grains of GGs and native gold are observed.
Figure 3(b) corresponds to K2, with free grains of cp, rt, and GGs.
Figure 3(c) pertains to the environmental liability C1, where free grains of goe, hm, and GGs are visible.
Figure 3(d) presents T1, which displays a lateral intergrowth of goe and hm, along with free grains of cp.
Figure 3(e) provides a low-magnification view of sample M1, where py can be observed.
Figure 3(f) corresponds to the environmental liability O1, where the primary metallic mineral is mt, with free grains of py and GGs.
Figure 3(g) shows the photograph of the active tailings P1, which features apy included in cp, and free grains of GGs, apy, and py.
Figure 3(h) corresponds to C2, showing hm with included goe, as well as py laterally associated with goe, rt, and free GGs.
Figure 3(i) presents a photograph of M2, where the primary metallic mineral is free py, along with inclusions of GGs. Finally,
Figure 3(j) corresponds to sample S1, which displays an intergrowth of pyrite laterally associated with a silver-gold solid solution (Ag, Au), as well as an intergrowth of cp with inclusions of copper-silver sulfide (SULs_AgCu), cv, and goe at its edges
3.2.2. Scanning Electron Microscope Analyses and EDS X-ray Energy Dispersive Spectroscopy
Scanning electron microscopy (SEM) detects mineral-bearing elements in the examined samples. The comparative analysis of results obtained through scanning electron microscopy across various mineral samples has facilitated the identification of significant similarities and differences in their overall mineralogical composition.
The combined analysis of scanning electron microscopy (SEM) images and energy dispersive spectroscopy (EDS) spectra was essential in this study for the comprehensive characterization of the composition and structure of elements present in the tailings.
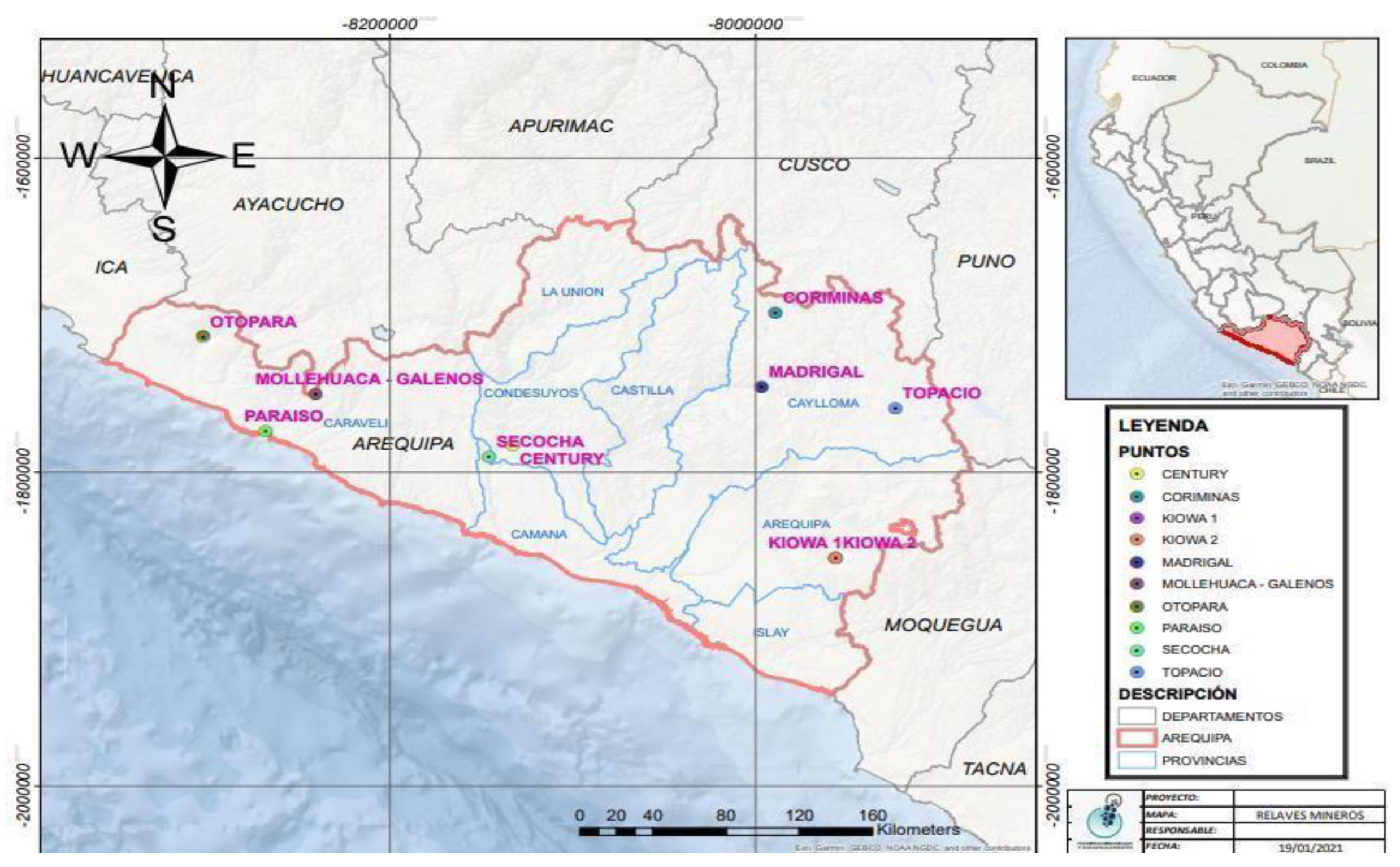
Table 5 presents the main metallic minerals and the elemental distribution across the 10 samples under study. The analysis reveals that lead (Pb) is the most prevalent element in the samples K1, K2, T1, C1, S1, M2, and P1, as shown in
Table 5. Arsenic (As) is present in the samples C1, M2, and P1; zinc (Zn) is found in the samples S1 and P1; mercury (Hg) is detected in sample M2; and no presence of cadmium (Cd) is observed in any of the samples.
Regarding valuable elements such as Au, it is present in the K1 and S1 samples; Silver (Ag) is found in the K1, C1, and S1 samples; and Cu is present in the K1, K2, T1, C1, M1, S1, C2, P1, and O1 samples, which can be recovered with further in-depth study. On the other hand, Uranium (U) was found in the O1 sample, which is a radioactive element that should be taken into consideration due to its unstable nucleus, easy disintegration, and energy emission. It is also known to be a health hazard.
Figure 4 and
Figure 6 present the SEM micrographs and EDS spectra of the environmental liabilities. In K1, the presence of native gold (Figures 4(a) and 6(a)); and in K2, chalcopyrite as a source of copper (Figures 4(b) and 6(b)). Galena is found in both samples, indicating a potential source of lead and other associated elements that can be highly toxic in Arequipa. Arsenopyrite (Figures 4(c) and 6(c)), contains arsenic, a highly toxic metalloid for the environment and human health. Covellite (Figures 4(d) and 6(d)) is found in C1 and T1 in Caylloma, suggesting oxidation and sulfidation processes that may influence the mobility of heavy metals. The identification of pyrite and chalcopyrite (Figures 4(e) and 6(e)) in Madrigal is significant due to their role in the generation of acid mine drainage (AMD), a phenomenon that can release toxic metals into the environment. The presence of tetrahedrite (Figures 4(f) and 6(f)) in Paraíso indicates the potential release of zinc, a highly toxic metal for the environment and human health. These are illustrated in
Figure 4 and
Figure 6.
In addition,
Figure 5 and
Figure 7 show the SEM micrographs and EDS spectra obtained for assets C2, M2, S1 and O1. Goethite (Figures 5(a) and 7(a)), identified in Century is indicative of oxidation processes that may affect the mobility of heavy metals present in the tailings. The presence of cinnabar (Figures 5(b) and 7(b)) in Mollehuaca suggests the presence of mercury, a metal with significant implications for public health and the environment. Conversely, the detection of argentocuproite (Figures 5(c) and 7(c)) in Secocha indicates the presence of economically valuable minerals. Finally, (Figures 5(d) and 7(d)) in Otapara features the mineral uraninite, which contains uranium radioactive element that requires special handling due to its unstable nucleus.
Figure 4 displays SEM micrographs. Photograph (a) of sample K1 at a magnification of 3850x shows free Au particles. Photograph (b) of sample K2 at a magnification of 2240x displays free cp grains, a mineral containing Cu. Photograph (c) of sample C1 at a magnification of 1050x shows free apy grains. Photograph (d) of sample T1 at a magnification of 1030x presents free cv grains. Photograph (e) of sample M1 at a magnification of 853x shows free cp grains. Finally, Photograph (f) of sample P1 at a magnification of 972x displays free particles of py, goe, cp, apy, and td.
Figure 5 displays SEM micrographs. Photograph (a) of sample C2 at a magnification of 313x shows free particles of goe and py. Photograph (b) of sample M2 at a magnification of 4195x reveals free particles of cnn. Photograph (c) of sample S1 at a magnification of 1480x shows intergrowths of goe, bn, cv, and cp; free hm particles; and free AgCu_cos particles. Finally, Photograph (d) of sample O1 at a magnification of 1130x presents lateral intergrowths of mt and urt.
The importance of SEM lies in its ability to detect trace minerals not visible with optical microscopy, such as small grains of barite, wolframite, phosphates, and uraninite, providing greater accuracy in identifying metallic minerals and revealing additional crucial details about the composition of the samples. This method complemented and validated the results obtained from optical microscopy, offering a more comprehensive and precise view of the mineralogical composition of the samples, which is essential for assessing their economic potential and developing exploitation strategies.
In summary, SEM not only confirmed the previous findings of optical microscopy but also significantly enhanced the precision and depth of mineralogical analysis, revealing additional important details for the evaluation and exploitation of mineral resources.
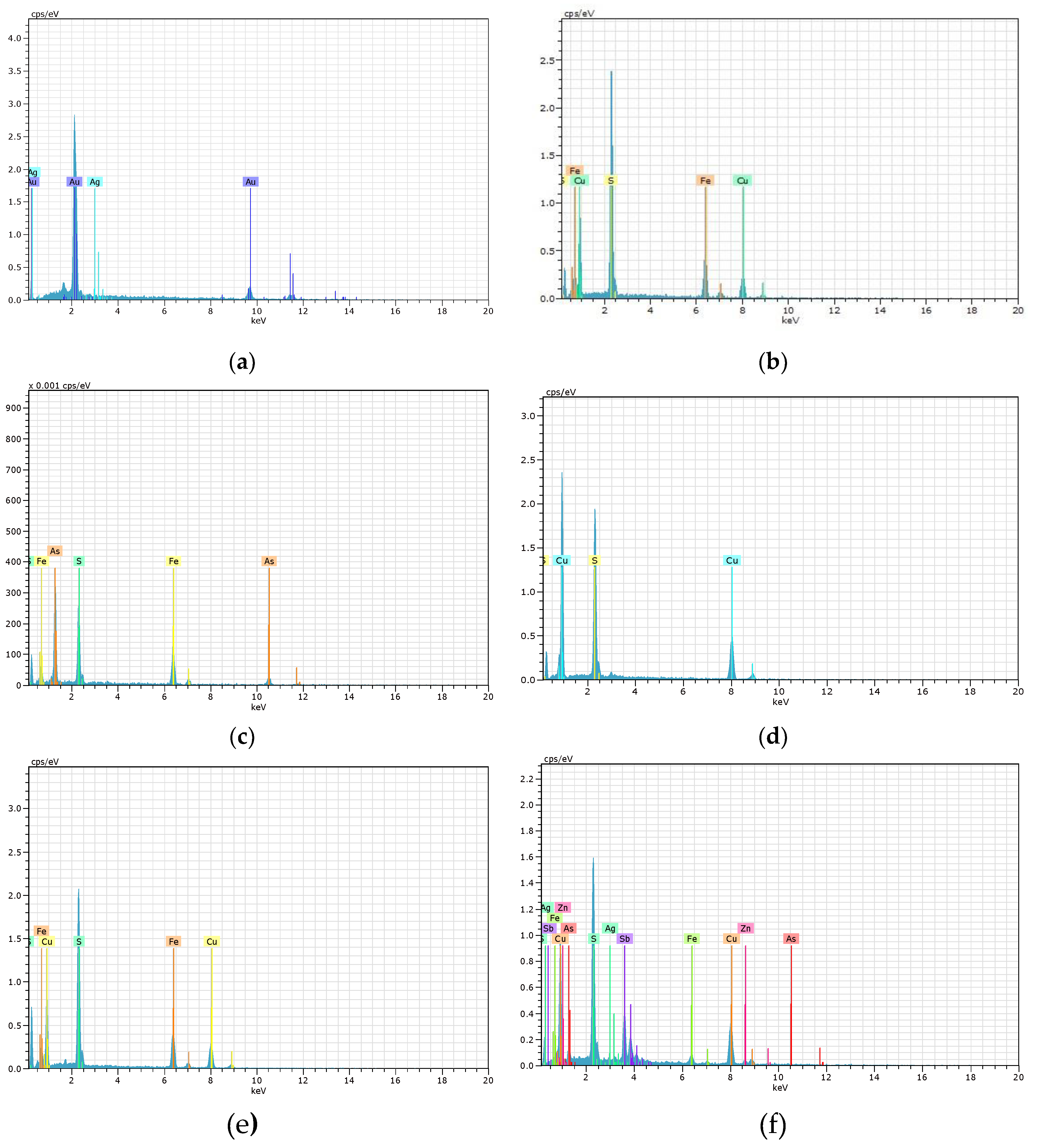
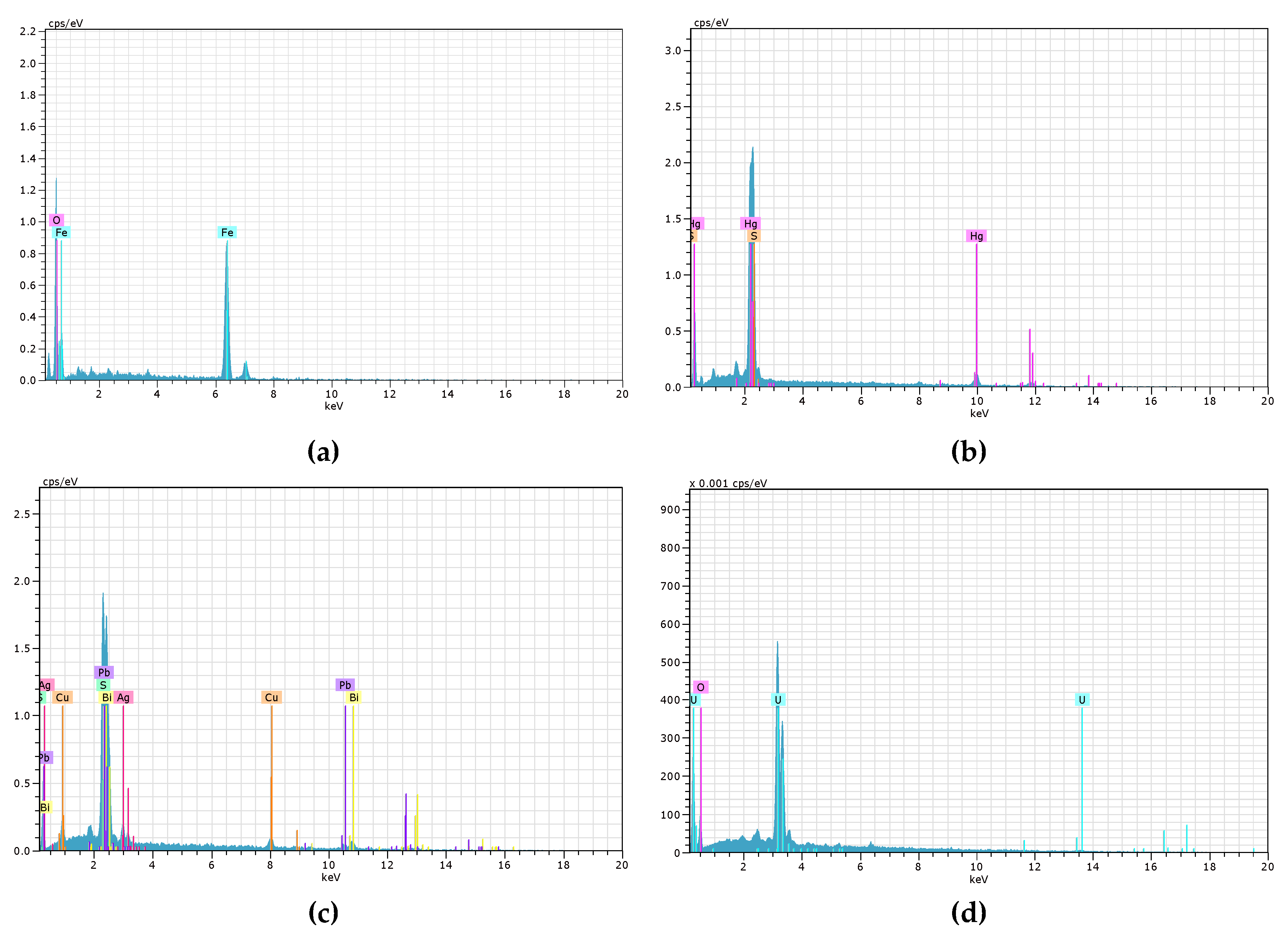
Figure 6 and
Figure 7 display the EDS spectra for the 6 environmental liability samples (K1, K2, C1, T1, M1, and P1) and 4 active tailings samples (C2, M2, S1, and O1). The position of the peaks in the spectra identifies the elements present in the material under study, while the intensity of the peaks corresponds to the concentration of each element. Both
Figure 6 and
Figure 7 corroborate the values tabulated in
Table 5 regarding the valuable elements.
Figure 6.
EDS spectra of various minerals studied in the environmental liabilities: (a) Native gold (K1), (b) Chalcopyrite (K2), (c) Arsenopyrite (C1), (d) Covellite (T1), (e) Chalcopyrite (M1), (f) Tetrahedrite (P1).
Figure 6.
EDS spectra of various minerals studied in the environmental liabilities: (a) Native gold (K1), (b) Chalcopyrite (K2), (c) Arsenopyrite (C1), (d) Covellite (T1), (e) Chalcopyrite (M1), (f) Tetrahedrite (P1).
Figure 7.
EDS spectra of various minerals studied in the active tailings: (a) Goethite (C2), (b) Cinnabar (M2), (c) Argentocuproite (S1), and (d) Uraninite (O1).
Figure 7.
EDS spectra of various minerals studied in the active tailings: (a) Goethite (C2), (b) Cinnabar (M2), (c) Argentocuproite (S1), and (d) Uraninite (O1).
The application of SEM combined with EDS in the analysis of mine tailings provides a detailed characterization of the elemental composition of the minerals present. This approach enables a comprehensive evaluation of the mineralogy of the tailings and helps to understand the geochemical processes that may affect the mobility of contaminants in the environment.